Comparison of In Silico Signal Sequence-Phospholipid Results with Described In Vitro and In Vivo Protein Translocation Studies Seems to Underscore the Significance of Phospholipids
Abstract
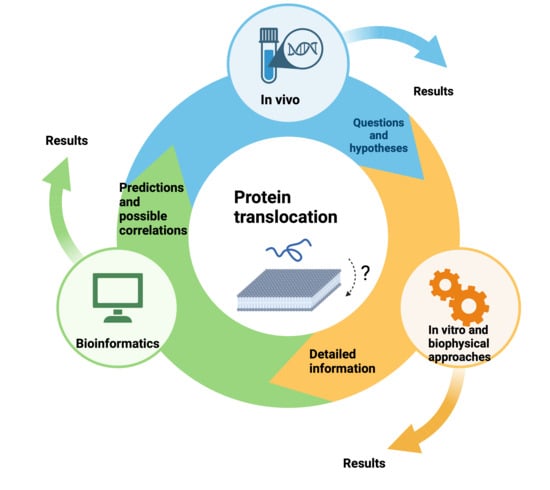
:1. Introduction
2. Results
2.1. Signal Sequence Experimental Results of prePhoE Correlate with In Silico Data
2.2. In Silico Results Suggest a Possible In Silico Protein Translocation Assay
2.3. In Silico Analysis Correlations with In Vitro and In Vivo Findings of Protein Translocation Effects of Signal Sequence Mutations
2.4. In Silico Analysis of Signal Peptide–Lipid Interactions Correlate with In Vitro and In Vivo Findings That Increasing Hydrophobicity in Signal Sequences Changes the Degree of Anionic Phospholipid Dependency in Protein Translocation
2.5. In Vivo Proven Signal Sequence Lipid Contacts in ER Translocation
2.6. An Example of a Closely Related Transmembrane Insertion Experiment
3. Discussion
4. Methods
4.1. Primary and Secondary Structure
4.2. Determination Lipid-Binding Potential and Eisenberg Plot Approach
4.3. Heliquest-Based Eisenberg Plot Approach
4.4. Monte Carlo Simulations Using MCPep
4.5. Structural Modelling
4.6. In Silico Protein Translocation Assay
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- White, S.H. Fifty Years of Biophysics at the Membrane Frontier. Annu. Rev. Biophys. 2023, 52, 21–67. [Google Scholar] [CrossRef]
- Rapoport, T.A.; Li, L.; Park, E. Structural and Mechanistic Insights into Protein Translocation. Annu. Rev. Cell Dev. Biol. 2017, 33, 369–390. [Google Scholar] [CrossRef]
- Chatzi, K.E.; Sardis, M.F.; Economou, A.; Karamanou, S. SecA-mediated targeting and translocation of secretory proteins. Biochim. Biophys. Acta Mol. Cell Res. 2014, 1843, 1466–1474. [Google Scholar] [CrossRef]
- Driessen, A.J.; Nouwen, N. Protein translocation across the bacterial cytoplasmic membrane. Annu. Rev. Biochem. 2008, 77, 643–667. [Google Scholar] [CrossRef]
- De Keyzer, J.; Van Der Does, C.; Driessen, A.J.M. The bacterial translocase: A dynamic protein channel complex. Cell. Mol. Life Sci. 2003, 60, 2034–2052. [Google Scholar] [CrossRef]
- Dowhan, W. Molecular basis for membrane phospholipid diversity: Why are there so many lipids? Annu. Rev. Biochem. 1997, 66, 199–232. [Google Scholar] [CrossRef]
- Von Heijne, G.V. Membrane proteins: From sequence to structure. Annu. Rev. Biophys. Biomol. Struct. 1994, 23, 167–192. [Google Scholar] [CrossRef]
- Izard, J.W.; Kendall, D.A. Signal peptides: Exquisitely designed transport promoters. Mol. Microbiol. 1994, 13, 765–773. [Google Scholar] [CrossRef]
- Arkowitz, R.A.; Bassilana, M. Protein translocation in Escherichia coli. Biochim. Biophys. Acta Rev. Biomembr. 1994, 1197, 311–343. [Google Scholar] [CrossRef]
- Lill, R.; Dowhan, W.; Wickner, W. The ATPase activity of SecA is regulated by acidic phospholipids, SecY, and the leader and mature domains of precursor proteins. Cell 1990, 60, 271–280. [Google Scholar] [CrossRef]
- Akimaru, J.; Matsuyama, S.L.; Tokuda, H.; Mizushima, S. Reconstitution of a protein translocation system containing purified SecY, SecE, and SecA from Escherichia coli. Proc. Natl. Acad. Sci. USA 1991, 88, 6545–6549. [Google Scholar] [CrossRef]
- Brundage, L.; Hendrick, J.P.; Schiebel, E.; Driessen, A.J.; Wickner, W. The purified E. coli integral membrane protein SecYE is sufficient for reconstitution of SecA-dependent precursor protein translocation. Cell 1990, 62, 649–657. [Google Scholar] [CrossRef]
- Emr, S.D.; Hanley-Way, S.; Silhavy, T.J. Suppressor mutations that restore export of a protein with a defective signal sequence. Cell 1981, 23, 79–88. [Google Scholar] [CrossRef]
- Emanuelsson, O.; Nielsen, H.; Brunak, S.; Von Heijne, G. Predicting subcellular localization of proteins based on their N-terminal amino acid sequence. J. Mol. Biol. 2000, 300, 1005–1016. [Google Scholar] [CrossRef]
- Von Heijne, G. The signal peptide. J. Membr. Biol. 1990, 115, 195–201. [Google Scholar] [CrossRef]
- Von Heijne, G.; Abrahmsen, L. Species-specific variation in signal peptide design. Implications for secretion in foreign hosts. FEBS Lett. 1989, 244, 439–446. [Google Scholar] [CrossRef]
- Von Heijne, G. Analysis of the distribution of charged residues in the N-terminal region of signal sequences: Implications for protein export in prokaryotic and eukaryotic cells. EMBO J. 1984, 3, 2315–2318. [Google Scholar] [CrossRef]
- De Vrije, T.; De Swart, R.L.; Dowhan, W.; Tommassen, J.; de Kruijff, B. Phosphatidylglycerol is involved in protein translocation across Escherichia coli inner membranes. Nature 1988, 334, 173–175. [Google Scholar] [CrossRef]
- Zimmer, J.; Nam, Y.; Rapoport, T.A. Structure of a complex of the ATPase SecA and the protein-translocation channel. Nature 2008, 455, 936–943. [Google Scholar] [CrossRef]
- You, Z.; Liao, M.; Zhang, H.; Yang, H.; Pan, X.; Houghton, J.E.; Sui, S.-F.; Tai, P.C. Phospholipids induce conformational changes of SecA to form membrane-specific domains: AFM structures and implication on protein-conducting channels. PLoS ONE 2013, 8, e72560. [Google Scholar] [CrossRef]
- Lin, B.-R.; Hsieh, Y.-H.; Jiang, C.; Tai, P.C. Escherichia coli membranes depleted of SecYEG elicit SecA-dependent ion-channel activity but lose signal peptide specificity. J. Membr. Biol. 2012, 245, 747–757. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.H.; Huang, Y.J.; Zhang, H.; Liu, Q.; Lu, Y.; Yang, H.; Houghton, J.; Jiang, C.; Sui, S.F.; Tai, P.C. Dissecting structures and functions of SecA-only protein-conducting channels: ATPase, pore structure, ion channel activity, protein translocation, and interaction with SecYEG/SecDF•YajC. PLoS ONE 2017, 12, e0178307. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.H.; Zhang, H.; Lin, B.R.; Cui, N.; Na, B.; Yang, H.; Jiang, H.; Sui, S.-F.; Tai, P.C. SecA alone can promote protein translocation and ion channel activity SecYEG increases efficiency and signal peptide specificity. J. Biol. Chem. 2011, 286, 44702–44709. [Google Scholar] [CrossRef] [PubMed]
- Phoenix, D.A.; Kusters, R.; Hikita, C.; Mizushima, S.; De Kruijff, B. OmpF-Lpp signal sequence mutants with varying charge hydrophobicity ratios provide evidence for a phosphatidylglycerol-signal sequence interaction during protein translocation across the Escherichia coli inner membrane. J. Biol. Chem. 1993, 268, 17069–17073. [Google Scholar] [CrossRef] [PubMed]
- Keller, R.C.A.; Killian, J.A.; de Kruijff, B. Anionic phospholipids are essential for α-helix formation of the signal peptide of prePhoE upon interaction with phospholipid vesicles. Biochemistry 1992, 31, 1672–1677. [Google Scholar] [CrossRef] [PubMed]
- Roussel, G.; Lindner, E.; White, S.H. Topology of the SecA ATPase bound to large unilamellar vesicles. J. Mol. Biol. 2022, 434, 167607. [Google Scholar] [CrossRef] [PubMed]
- Keller, R.C.A. The prediction of novel multiple lipid-binding regions in protein translocation motor proteins: A possible general feature. Cell. Mol. Biol. Lett. 2011, 16, 40–54. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.W.; Chen, Y.; Yang, H.; Chen, X.; Duan, M.X.; Tai, P.C.; Sui, S.F. Ring-like pore structures of SecA: Implication for bacterial protein-conducting channels. Proc. Natl. Acad. Sci. USA 2003, 100, 4221–4226. [Google Scholar] [CrossRef]
- Keller, R.C.A.; Snel, M.M.E.; de Kruijff, B.; Marsh, D. SecA restricts in a nucleotide-dependent manner acyl chain mobility up to the center of a phospholipid bilayer. FEBS Lett. 1995, 358, 251–254. [Google Scholar] [CrossRef]
- Breukink, E.; Demel, R.A.; De Korte-Kool, G.; De Kruijff, B. SecA insertion into phospholipids is stimulated by negatively charged lipids and inhibited by ATP: A monolayer study. Biochemistry 1992, 31, 1119–1124. [Google Scholar] [CrossRef]
- Breukink, E.; Keller, R.C.A.; de Kruijff, B. Nucleotide and negatively charged lipid-dependent vesicle aggregation caused by SecA. Evidence that SecA contains two lipid-binding sites. FEBS Lett. 1993, 33, 19–24. [Google Scholar] [CrossRef]
- Keller, R.C.A. New user-friendly approach to obtain an Eisenberg plot and its use as a practical tool in protein sequence analysis. Int. J. Mol. Sci. 2011, 12, 5577–5591. [Google Scholar] [CrossRef]
- Keller, R.C.A. Identification and in silico analysis of helical lipid-binding regions in proteins belonging to the amphitropic protein family. J. Biosci. 2014, 39, 771–783. [Google Scholar] [CrossRef]
- Jarrin, M.; Kalligeraki, A.A.; Uwineza, A.; Cawood, C.S.; Brown, A.P.; Ward, E.N.; Le, K.; Freitag-Pohl, S.; Pohl, E.; Kiss, B.; et al. Independent Membrane Binding Properties of the Caspase Generated Fragments of the Beaded Filament Structural Protein 1 (BFSP1) Involves an Amphipathic Helix. Cells 2023, 12, 1580. [Google Scholar] [CrossRef]
- Ehlers, L.; Kuppe, A.; Damerau, A.; Wilantri, S.; Kirchner, M.; Mertins, P.; Strehl, C.; Buttgereit, F.; Gaber, T. Surface AMP deaminase 2 as a novel regulator modifying extracellular adenine nucleotide metabolism. FASEB J. 2021, 35, e21684. [Google Scholar] [CrossRef]
- Phoenix, D.A.; Harris, F.; Mura, M.; Dennison, S.R. The increasing role of phosphatidylethanolamine as a lipid receptor in the action of host defence peptides. Prog. Lipid Res. 2015, 59, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Saikia, K.; Sravani, Y.D.; Ramakrishnan, V.; Chaudhary, N. Highly potent antimicrobial peptides from N-terminal membrane-binding region of E. coli MreB. Sci. Rep. 2017, 7, 42994. [Google Scholar] [CrossRef]
- Gorbenko, G.; Trusova, V.; Girych, M.; Adachi, E.; Mizuguchi, C.; Akaji, K.; Saito, H. FRET evidence for untwisting of amyloid fibrils on the surface of model membranes. Soft Matter 2015, 11, 6223–6234. [Google Scholar] [CrossRef] [PubMed]
- Deffieu, M.S.; Alayi, T.D.; Slomianny, C.; Tomavo, S. The Toxoplasma gondii dense granule protein TgGRA3 interacts with host Golgi and dysregulates anterograde transport. Biol. Open 2019, 8, bio039818. [Google Scholar] [CrossRef]
- Wang, C.; Piao, J.; Li, Y.; Tian, X.; Dong, Y.; Liu, D. Construction of Liposomes Mimicking Cell Membrane Structure through Frame-Guided Assembly. Angew. Chem. Int. Ed. 2020, 59, 15176–15180. [Google Scholar] [CrossRef]
- Killian, J.A.; Keller, R.C.A.; Struyvé, M.; de Kroon, A.I.P.M.; Tommassen, J.; de Kruijff, B. Tryptophan fluorescence study on the interaction of the signal peptide of the Escherichia coli outer membrane protein PhoE with model membranes. Biochemistry 1990, 29, 8131–8137. [Google Scholar] [CrossRef] [PubMed]
- Keller, R.C.A.; ten Berge, D.; Nouwen, N.; Snel, M.M.E.; Tommassen, J.; Marsh, D.; de Kruijff, B. Mode of insertion of the signal sequence of a bacterial precursor protein into phospholipid bilayers as revealed by cysteine-based site directed spectroscopy. Biochemistry 1996, 35, 3063–3071. [Google Scholar] [CrossRef] [PubMed]
- Van Raalte, A.L.J.; Demel, R.A.; Verberkmoes, G.; Breukink, E.; Keller, R.C.A.; de Kruijff, B. Influence of the signal sequence and chaperone SecB on the interaction between precursor protein prePhoE and phospholipids. Eur. J. Biochem. 1996, 235, 207–214. [Google Scholar] [CrossRef] [PubMed]
- De Wolf, F.A.; Keller, R.C.A. Characterization of helical structures in gelatin networks and model polypeptides by circular dichroism. In Gels; Progress in Colloid & Polymer Science; Steinkopff: Heidelberg, Germany, 1996; Volume 102, pp. 9–14. [Google Scholar]
- Gautier, R.; Douguet, D.; Antonny, B.; Drin, G. HELIQUEST: A web server to screen sequences with specific alpha-helical properties. Bioinformatics 2008, 24, 2101–2102. [Google Scholar] [CrossRef] [PubMed]
- Gofman, Y.; Haliloglu, T.; Ben-Tal, N. Monte Carlo simulations of peptide-membrane interactions with the MCPep web server. Nucleic Acids Res. 2012, 40, W358–W363. [Google Scholar] [CrossRef]
- Keller, R.C.A. Identification of Possible Lipid Binding Regions in Food Proteins and Peptides and Additional In Silico Analysis. Food Biophys. 2018, 13, 139–146. [Google Scholar] [CrossRef]
- Van Dalen, A.; Killian, J.A.; de Kruijff, B. Δψ stimulates membrane translocation of the C-terminal part of a signal sequence. J. Biol. Chem. 1999, 274, 19913–19918. [Google Scholar] [CrossRef]
- McKnight, C.J.; Briggs, M.S.; Gierasch, L.M. Functional and nonfunctional LamB signal sequences can be distinguished by their biophysical properties. J. Biol. Chem. 1989, 264, 17293–17297. [Google Scholar] [CrossRef]
- Gallusser, A.; Kuhn, A. Initial steps in protein membrane insertion. Bacteriophage M13 procoat protein binds to the membrane surface by electrostatic interaction. EMBO J. 1990, 9, 2723–2729. [Google Scholar] [CrossRef] [PubMed]
- Petersen, T.N.; Brunak, S.; von Heijne, G.; Nielsen, H. SignalP 4.0: Discriminating signal peptides from transmembrane regions. Nat. Methods 2011, 8, 785–786. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, Y.; Morioka, K.; Shirai, J.; Yokomizo, Y.; Yoshida, K. Gene design of signal sequence for effective secretion of protein. Nucleic Acids Symp. Ser. 2003, 3, 261–262. [Google Scholar] [CrossRef]
- Goldstein, J.; Lehnhardt, S.; Inouye, M. In vivo effect of asparagine in the hydrophobic region of the signal sequence. J. Biol. Chem. 1991, 266, 14413–14417. [Google Scholar] [CrossRef] [PubMed]
- Hikita, C.; Mizushima, S. The requirement of a positive charge at the amino terminus can be compensated for by a longer central hydrophobic stretch in the functioning of signal peptides. J. Biol. Chem. 1992, 267, 12375–12379. [Google Scholar] [CrossRef]
- Martoglio, B.; Hofmann, M.W.; Brunner, J.; Dobberstein, B. The protein-conducting channel in the membrane of the endoplasmic reticulum is open laterally toward the lipid bilayer. Cell 1995, 81, 207–214. [Google Scholar] [CrossRef]
- Mothes, W.; Jungnickel, B.; Brunner, J.; Rapoport, T.A. Signal sequence recognition in cotranslational translocation by protein components of the endoplasmic reticulum membrane. J. Cell Biol. 1998, 142, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Brambillasca, S.; Yabal, M.; Makarow, M.; Borgese, N. Unassisted translocation of large polypeptide domains across phospholipid bilayers. J. Cell Biol. 2006, 175, 767–777. [Google Scholar] [CrossRef]
- Brambillasca, S.M.; Yabal, M.; Soffientini, P.; Stefanovic, S.; Makarow, M.; Hegde, R.S.; Borgese, N. Transmembrane topogenesis of a tail-anchored protein is modulated by membrane lipid composition. EMBO J. 2005, 24, 2533–2542. [Google Scholar] [CrossRef] [PubMed]
- Cymer, F.; von Heijne, G.; White, S.H. Mechanisms of Integral Membrane Protein Insertion and Folding. J. Mol. Biol. 2015, 427, 999–1022. [Google Scholar] [CrossRef]
- Crane, J.M.; Randall, L.L. The Sec system: Protein export in Escherichia coli. EcoSal Plus 2017, 7. [Google Scholar] [CrossRef]
- Den Uijl, M.J.; Driessen, A.J. Phospholipid dependency of membrane protein insertion by the Sec translocon. Biochim. Biophys. Acta Biomembr. 2024, 1866, 184232. [Google Scholar] [CrossRef]
- Spencer, C.; Bensing, B.A.; Mishra, N.N.; Sullam, P.M. Membrane trafficking of the bacterial adhesin GspB and the accessory Sec transport machinery. J. Biol. Chem. 2019, 294, 1502–1515. [Google Scholar] [CrossRef]
- Adam, G.; Delbrück, M. Reduction of dimensionality in biological diffusion processes. In Structural Chemistry and Molecular Biology; Rich, A., Davidson, N., Eds.; WH Freeman: San Francisco, CA, USA, 1968; pp. 198–215. [Google Scholar]
- Koch, S.; Exterkate, M.; López, C.A.; Patro, M.; Marrink, S.J.; Driessen, A.J. Two distinct anionic phospholipid-dependent events involved in SecA-mediated protein translocation. Biochim. Biophys. Acta 2019, 1861, 183035. [Google Scholar] [CrossRef] [PubMed]
- Kater, L.; Frieg, B.; Berninghausen, O.; Gohlke, H.; Beckmann, R.; Kedrov, A. Partially inserted nascent chain unzips the lateral gate of the Sec translocon. EMBO Rep. 2019, 20, e48191. [Google Scholar] [CrossRef] [PubMed]
- Ulmschneider, M.B.; Ulmschneider, J.P.; Schiller, N.; Wallace, B.A.; Von Heijne, G.; White, S.H. Spontaneous transmembrane helix insertion thermodynamically mimics translocon-guided insertion. Nat. Commun. 2014, 5, 4863. [Google Scholar] [CrossRef] [PubMed]
- Singer, S.J.; Nicolson, G.L. The fluid mosaic model of the structure of cell membranes. Science 1972, 175, 720–731. [Google Scholar] [CrossRef] [PubMed]
- Sezgin, E.; Levental, I.; Mayor, S.; Eggeling, C. The mystery of membrane organization: Composition, regulation and roles of lipid rafts. Nat. Rev. Mol. Cell Biol. 2017, 18, 361–374. [Google Scholar] [CrossRef]
- Bernardino de la Serna, B.J.; Schütz, G.J.; Eggeling, C.; Cebecauer, M. There is no simple model of the plasma membrane organization. Front. Cell Dev. Biol. 2016, 4, 106. [Google Scholar] [CrossRef]
- The UniProt Consortium. UniProt: The universal protein knowledge base. Nucleic Acids Res. 2017, 45, D158–D169. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, Y. I-TASSER server: New development for protein structure and function predictions. Nucleic Acids Res. 2015, 43, W174–W181. [Google Scholar] [CrossRef]
- Zheng, W.; Zhang, C.; Li, Y.; Pearce, R.; Bell, E.W.; Zhang, Y. Folding non-homologous proteins by coupling deep-learning contact maps with I-TASSER assembly simulations. Cell Rep. Methods. 2021, 1, 100014. [Google Scholar] [CrossRef]
- Hessa, T.; Meindl-Beinker, N.; Bernsel, A.; Kim, J.; Sato, Y.; Lerch, M.; Lundin, C.; Nilsson, I.; White, S.H.; von Heijne, G. Molecular code for transmembrane-helix recognition by the Sec61 translocon. Nature 2007, 450, 1026–1030. [Google Scholar] [CrossRef] [PubMed]
- Hessa, T.; Kim, H.; Bihlmaier, K.; Lundin, C.; Boekel, J.; Andersson, H.; Nilsson, I.; White, S.H.; von Heijne, G. Recognition of transmembrane helices by the endoplasmic reticulum translocon. Nature 2005, 433, 377–381. [Google Scholar] [CrossRef] [PubMed]



| Name | Sequence | z | <µH> | <H> | LBR a |
|---|---|---|---|---|---|
| PhoE SP | MKKSTLALVVMGIVASASVQA | 2 | 0.027 | 0.578 | D |
| PhoE SP G10L | MKKSTLALVVMLIVASASVQA | 2 | 0.105 | 0.673 | D |
| LamB SP | MMITLRKLPLAVAVAAGVMSAQAMA | 2 | 0.222 | 0.582 | D |
| LamB SP A13D | MMITLRKLPLAVDVAAGVMSAQAMA | 1 | 0.262 | 0.522 | No |
| LamB ∆78 | MMITLRKLP----VAAGVMSAQAMA | 2 | 0.178 | 0.476 | D |
| LamB ∆78r1 | MMITLRKLP----VAACVMSAQAMA | 2 | 0.176 | 0.562 | D |
| LamB ∆78r2 | MMITLRKLL----VAAGVMSAQAMA | 2 | 0.206 | 0.531 | D |
| 2K7L SP | MMKKNNLLLLLLLGTANAAS | 2 | 0.165 | 0.637 | D |
| 2K8L SP | MMKKNNLLLLLLLLGTANAAS | 2 | 0.086 | 0.714 | D |
| 2K9L SP | MMKKNNLLLLLLLLLGTANAAS | 2 | 0.095 | 0.842 | TM/D |
| 2K8V SP | MMKKNNVVVVVVVVGTANAAS | 2 | 0.107 | 0.501 | D |
| 2K9V SP | MMKKNNVVVVVVVVVGTANAAS | 2 | 0.080 | 0.602 | D |
| 2K10V SP | MMKKNNVVVVVVVVVVGTANAAS | 2 | 0.136 | 0.652 | D |
| CLY SP | MRSLLILVLCFLPLAALG | 1 | 0.081 | 1.098 | TM |
| CLY R3 | MRRRSLLILVLCFLPLAALG | 3 | 0.100 | 0.918 | TM/D |
| CLY R17D | MDSLLILVLCFLPLAALG | −1 | 0.067 | 1.112 | TM |
| M13 coat SP | MKKSLVLKASVAVATLVPMLSFA b | 3 | 0.072 | 0.559 | D |
| M13 coat SP | MKKSLVLKASVAVATLVPMLSFA b | 1 | 0.109 | 0.863 | TM |
| M13 coat SP ∆N | M--------NSVAVATLVPMLSFA | 0 | 0.164 | 0.784 | TM |
| M13 coat SP ∆N + 3 | M--------NSLRRRNSVAVATLVPMLSFA | 3 | 0.250 | 0.440 | D |
| OmpA SP | MKKTAIAIAVALAGFATVAQA | 2 | 0.251 | 0.620 | D |
| OmpA SP I8N | MKKTAIANAVALAGFATVAQA | 2 | 0.118 | 0.487 | D |
| OmpA SP L12N | MKKTAIAIAVANAGFATVAQA | 2 | 0.174 | 0.492 | D |
| Signal Peptide | Peptide Sequence | Add. AA a |
|---|---|---|
| PhoE | MKKSTLALVVMGIVASASVQAAEIYNKDGNKLDVYGKV | +17 |
| PhoE G10L | MKKSTLALVVMLIVASASVQAAEIYNKDGNKLDVYGKV | +17 |
| LamB | MMITLRKLPLAVAVAAGVMSAQAMAVDFHGYARSG | +10 |
| LamB A13D | MMITLRKLPLAVDVAAGVMSAQAMA | 0 |
| LamB∆78 | MMITLRKLP----VAAGVMSAQAMA | 0 |
| LamB∆78r1 | MMITLRKLP----VAACVMSAQAMAVDFH | +4 |
| LamB∆78r2 | MMITLRKLL----VAAGVMSAQAMAVDFHGYARSG | +10 |
| OmpA | MKKTAIAIAVALAGFATVAQAATSTKKLHKEPATLIKAIDGT | +21 |
| OmpA I8N | MKKTAIANAVALAGFATVAQAATSTKKLHKEPATLIKAI | +18 |
| OmpA I8D | MKKTAIADAVALAGFATVAQAATSTKKLHKEP | +11 |
| OmpA L12N | MKKTAIAIAVANAGFATVAQAATSTKKLHKE | +11 |
| OmpA L12D | MKKTAIAIAVADAGFATVAQA | 0 |
| 2K7L | MMKKNNLLLLLLLGTANAASEIYNKDGNKVDLYGKAV | +17 |
| 2K8L | MMKKNNLLLLLLLLGTANAASEIYNKDGNKVDLYGKAVGL | +19 |
| 2K9L | MMKKNNLLLLLLLLLGTANAASEIYNKDGNKVDLYGKAVGL | +19 |
| 2K8V | MMKKNNVVVVVVVVGTANAASEIYNKDGNKVDLYGK | +15 |
| 2K9V | MMKKNNVVVVVVVVVGTANAASEIYNKDGNKVDLYGK | +15 |
| 2K10V | MMKKNNVVVVVVVVVVGTANAASEIYNKDGNKVDLYGKAV | +17 |
| CLY | MRSLLILVLCFLPLAALGKVFERCELARTLKRLGMDGYRGIS | +24 |
| CLY R3 | MRRRSLLILVLCFLPLAALGKVFERCELARTLKRLGMDGYRGIS | +24 |
| CLY R17D | MDSLLILVLCFLPLAALGKVFERCELARTLKRLGMDGYRGIS | +24 |
| M13 coat SP | MKKSLVLKASVAVATLVPMLSFAAEGDDPAKAAFNSLQASATEYI | +22 |
| M13 coat SP ∆N | M--------NSVAVATLVPMLSFA | 0 |
| M13 coat SP ∆N + 3 | M--------NSLRRRNSVAVATLVPMLSFAAEGDDPAKAAFNSL | +14 |
| Name | Translocated Mature | ∆Gapp | MCPeppred/ ∆Gapp c | In Vivo/In Vitro Activity |
|---|---|---|---|---|
| PhoE SP WT | +7 AA (TM) | 1.972 | 100/+ | 100 (+pmf) |
| PhoE SP WT | +17 AA (TM) | 1.972 | 100/+ | ND a |
| PhoE SP G10L | +7 AA (TM) | 0.958 | 100/++ | 100 (−pmf) |
| PhoE SP G10L | +17 AA (TM) | 0.958 | 100/++ | ND |
| LamB WT | +10 AA (TM) | 1.633 | 100/+ | 100 |
| LamB A13D | +1 AA (Surf.) | 3.818 | 0/− | 10 |
| LamB ∆78 | +5 AA (Surf.) b | No pred. | 0/− | 0 |
| LamB ∆78r1 | +4 AA (TM) b | 3.798 | 40/− | 50 |
| LamB ∆78r2 | +10 AA (TM) b | 2.533 | 100/+ | 90 |
| OmpA SP WT | +21 AA (TM) | 1.382 | 100/+ | 100 |
| OmpA SP I8N | +18 AA (TM) | 2.581 | 86/− | 0 |
| OmpA SP I8D | +11 AA (TM) | 2.933 | 52/− | ND |
| OmpA SP L12N | +11 AA (TM) | 2.961 | 52/− | 0 |
| OmpA SP L12D | +2 AA (Surf.) | 3.468 | 0/− | ND |
| CLY WT | +24 AA | −1.184 | 100/++ | 100 |
| CLY R3 | +24 AA | −1.569 | 100/++ | 216 |
| CLY R17D | +24 AA | −0.536 | 100/++ | 17 |
| M13 SP WT | +22 AA (TM) | 2.806 | 100/+ | 100 |
| M13 coat SP ∆N | +3 AA (Surf.) | 3.610 | 0/− | 0 |
| M13 coat SP ∆N + 3 | +14 AA (TM) | 4.562 | 64/− | 90 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Keller, R.C.A. Comparison of In Silico Signal Sequence-Phospholipid Results with Described In Vitro and In Vivo Protein Translocation Studies Seems to Underscore the Significance of Phospholipids. Lipidology 2024, 1, 3-17. https://doi.org/10.3390/lipidology1010002
Keller RCA. Comparison of In Silico Signal Sequence-Phospholipid Results with Described In Vitro and In Vivo Protein Translocation Studies Seems to Underscore the Significance of Phospholipids. Lipidology. 2024; 1(1):3-17. https://doi.org/10.3390/lipidology1010002
Chicago/Turabian StyleKeller, Rob C. A. 2024. "Comparison of In Silico Signal Sequence-Phospholipid Results with Described In Vitro and In Vivo Protein Translocation Studies Seems to Underscore the Significance of Phospholipids" Lipidology 1, no. 1: 3-17. https://doi.org/10.3390/lipidology1010002