Ecology and Machine Learning-Based Classification Models of Gut Microbiota and Inflammatory Markers May Evaluate the Effects of Probiotic Supplementation in Patients Recently Recovered from COVID-19
Abstract
:1. Introduction
2. Results
2.1. Study Population Features
Clinical Outcomes and Treatment Satisfaction Assessment
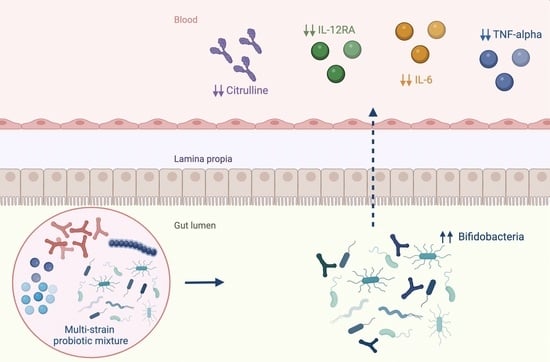
2.2. Immunological Response, Intestinal Barrier Integrity and Local and Systemic Inflammation
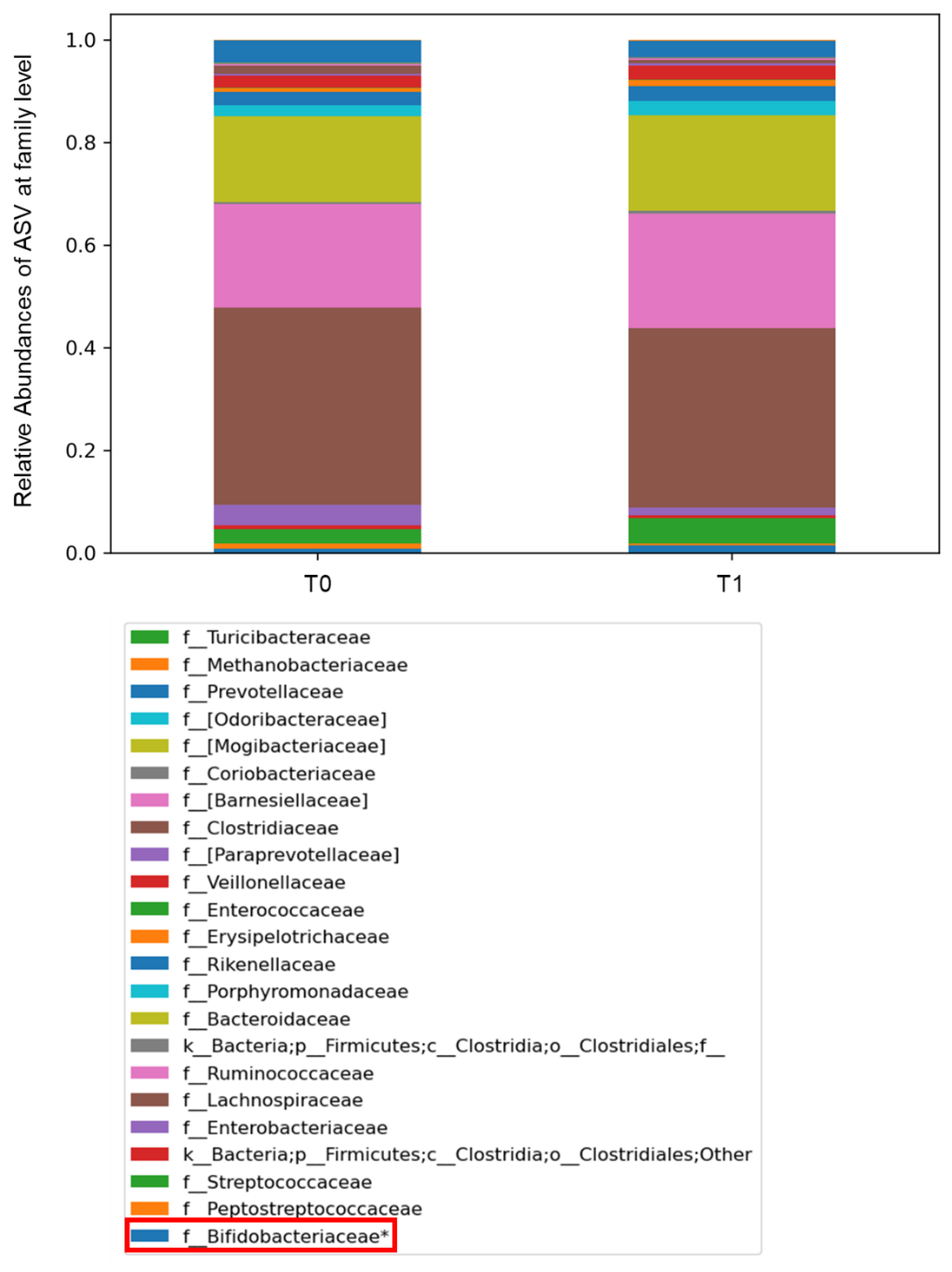
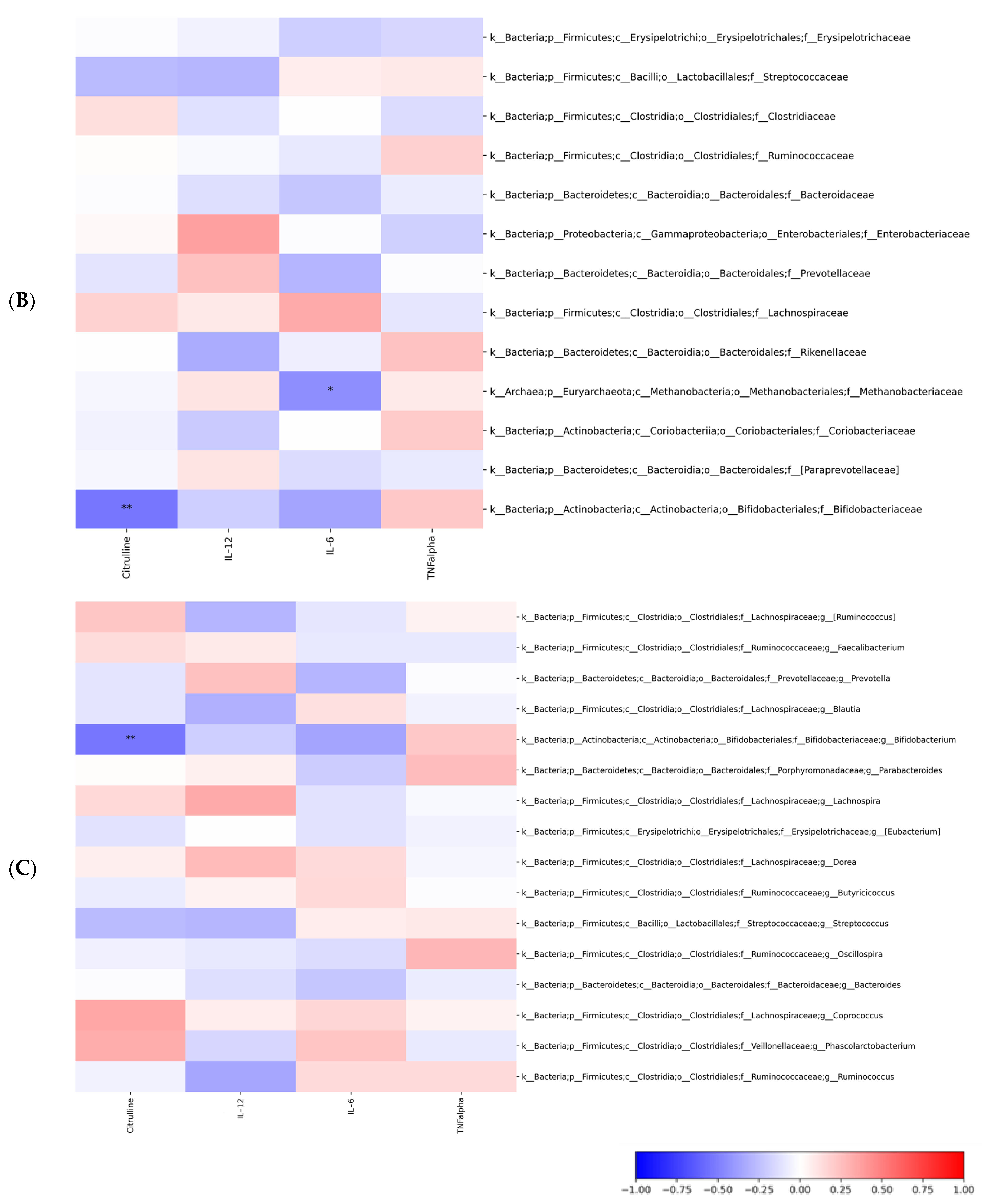
2.3. Gut Microbiota Profiling: Ecology, Predicted Functions and “Microbial Marker” Discovery
3. Discussion
4. Materials and Methods
4.1. Study Design and Aims
4.2. Measures of Clinical Outcomes
4.3. Measures of Laboratory Outcomes
4.4. Statistical Analysis of Metadata
4.5. Faecal Microbiota Analyses
4.5.1. Bacterial DNA Extraction from Stools and 16S rRNA Targeted-Metagenomics
4.5.2. Biocomputational and Statistical Analysis for GM Profile Analysis and Patients’ Metadata Correlation
4.5.3. Machine Learning Models
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- COVID-19 Was Third Leading Cause of Death in the United States in 2021, CDC Reports CNN. Available online: https://edition.cnn.com/2022/04/22/health/covid-third-leading-cause-of-death-2021/index.html (accessed on 3 July 2022).
- Rothan, H.A.; Byrareddy, S.N. The Epidemiology and Pathogenesis of Coronavirus Disease (COVID-19) Outbreak. J. Autoimmun. 2020, 109, 102433. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Han, B.; Wang, J. COVID-19: Gastrointestinal Manifestations and Potential Fecal–Oral Transmission. Gastroenterology 2020, 158, 1518. [Google Scholar] [CrossRef] [PubMed]
- Iorio, A.; Biazzo, M.; Gardini, S.; Muda, A.O.; Perno, C.F.; Dallapiccola, B.; Putignani, L. Cross-Correlation of Virome-Bacteriome-Host-Metabolome to Study Respiratory. Health Trends Microbiol. 2022, 30, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Li, S.; Wang, N.; Tan, H.Y.; Zhang, Z.; Feng, Y. The Cross-Talk between Gut Microbiota and Lungs in Common Lung Diseases. Front. Microbiol. 2020, 11, 301. [Google Scholar] [CrossRef] [PubMed]
- Dumas, A.; Bernard, L.; Poquet, Y.; Lugo-Villarino, G.; Neyrolles, O. The Role of the Lung Microbiota and the Gut-Lung Axis in Respiratory Infectious Diseases. Cell Microbiol. 2018, 20, e12966. [Google Scholar] [CrossRef] [Green Version]
- Groves, H.T.; Higham, S.L.; Moffatt, M.F.; Cox, M.J.; Tregoning, J.S. Respiratory Viral Infection Alters the Gut Microbiota by Inducing Inappetence. mBio 2020, 11, e03236-19. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Gu, S.; Chen, Y.; Lu, H.; Shi, D.; Guo, J.; Wu, W.R.; Yang, Y.; Li, Y.; Xu, K.J.; et al. Letter: Six-Month Follow-up of Gut Microbiota Richness in Patients with COVID-19. Gut 2022, 71, 222. [Google Scholar] [CrossRef]
- World Gastroenterology Organisation Global Guidelines Probiotics and Prebiotics. 2017. Available online: https://www.worldgastroenterology.org/guidelines/probiotics-and-prebiotics (accessed on 16 December 2022).
- Salminen, S. Human Studies on Probiotics: Aspects of Scientific Documentation. Microb. Ecol. Health Dis. 2016, 45, 8–12. [Google Scholar] [CrossRef]
- Sullivan, Å.; Bennet, R.; Viitanen, M.; Palmgren, A.C.; Nord, C.E. Influence of Lactobacillus F19 on Intestinal Microflora in Children and Elderly Persons and Impact on Helicobacter Pylori Infections. Microb. Ecol. Health Dis. 2009, 14, 17–21. [Google Scholar] [CrossRef]
- La Fata, G.; Weber, P.; Mohajeri, M.H. Probiotics and the Gut Immune System: Indirect Regulation. Probiotics Antimicrob. Proteins 2018, 10, 11. [Google Scholar] [CrossRef]
- Gareau, M.G.; Sherman, P.M.; Walker, W.A. Probiotics and the Gut Microbiota in Intestinal Health and Disease. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 503. [Google Scholar] [CrossRef] [Green Version]
- West, N.P.; Horn, P.L.; Pyne, D.B.; Gebski, V.J.; Lahtinen, S.J.; Fricker, P.A.; Cripps, A.W. Probiotic Supplementation for Respiratory and Gastrointestinal Illness Symptoms in Healthy Physically Active Individuals. Clin. Nutr. 2014, 33, 581–587. [Google Scholar] [CrossRef] [Green Version]
- Leyer, G.J.; Li, S.; Mubasher, M.E.; Reifer, C.; Ouwehand, A.C. Probiotic Effects on Cold and Influenza-like Symptom Incidence and Duration in Children. Pediatrics 2009, 124, e172-9. [Google Scholar] [CrossRef] [Green Version]
- Turner, R.B.; Woodfolk, J.A.; Borish, L.; Steinke, J.W.; Patrie, J.T.; Muehling, L.M.; Lahtinen, S.; Lehtinen, M.J. Effect of Probiotic on Innate Inflammatory Response and Viral Shedding in Experimental Rhinovirus Infection a Randomised Controlled Trial. Benef. Microbes 2017, 8, 207–215. [Google Scholar] [CrossRef] [Green Version]
- Paineau, D.; Carcano, D.; Leyer, G.; Darquy, S.; Alyanakian, M.A.; Simoneau, G.; Bergmann, J.F.; Brassart, D.; Bornet, F.; Ouwehand, A.C. Effects of Seven Potential Probiotic Strains on Specific Immune Responses in Healthy Adults: A Double-Blind, Randomized, Controlled Trial. FEMS. Immunol. Med. Microbiol. 2008, 53, 107–113. [Google Scholar] [CrossRef] [Green Version]
- Petito, V.; Greco, V.; Laterza, L.; Graziani, C.; Fanali, C.; Lucchetti, D.; Barbaro, M.R.; Bugli, F.; Pieroni, L.; Lopetuso, L.R.; et al. Impact of the Trophic Effects of the Secretome From a Multistrain Probiotic Preparation on the Intestinal Epithelia. Inflamm. Bowel. Dis. 2021, 27, 902–913. [Google Scholar] [CrossRef]
- Vokurka, S.; Svoboda, T.; Rajdl, D.; Sedláčková, T.; Racek, J.; Koza, V.; Trefil, L. Serum Citrulline Levels as a Marker of Enterocyte Function in Patients after Allogeneic Hematopoietic Stem Cells Transplantation a Pilot Study. Med. Sci. Monit. 2013, 19, 81. [Google Scholar] [CrossRef] [Green Version]
- Desai, A.D.; Lavelle, M.; Boursiquot, B.C.; Wan, E.Y. Long-Term Complications of COVID-19. Am. J. Physiol. Cell. Physiol. 2022, 322, C1–C11. [Google Scholar] [CrossRef]
- Aiyegbusi, O.L.; Hughes, S.E.; Turner, G.; Rivera, S.C.; McMullan, C.; Chandan, J.S.; Haroon, S.; Price, G.; Davies, E.H.; Nirantharakumar, K.; et al. Symptoms, Complications and Management of Long COVID: A Review. J. R. Soc. Med. 2021, 114, 428. [Google Scholar] [CrossRef]
- Noviello, D.; Costantino, A.; Muscatello, A.; Bandera, A.; Consonni, D.; Vecchi, M.; Basilisco, G. Functional Gastrointestinal and Somatoform Symptoms Five Months after SARS-CoV-2 Infection: A Controlled Cohort Study. Neurogastroenterol. Motil. 2022, 34, e14187. [Google Scholar] [CrossRef]
- Kamikozuru, K.; Matsumoto, T.; Miwa, H. Post-Infectious IBS. Nihon. Rinsho. 2006, 64, 1544–1548. [Google Scholar] [PubMed]
- Seok, K.K.; Loll, B.; Wai, T.C.; Shoeman, R.L.; Ngoo, L.; Chew, C.Y.; Meinhart, A. Molecular and Structural Characterization of the PezAT Chromosomal Toxin-Antitoxin System of the Human Pathogen Streptococcus Pneumoniae. J. Biol. Chem. 2007, 282, 19606–19618. [Google Scholar] [CrossRef] [Green Version]
- Pontifex, M.G.; Mushtaq, A.; Le Gall, G.; Rodriguez-Ramiro, I.; Blokker, B.A.; Hoogteijling, M.E.M.; Ricci, M.; Pellizzon, M.; Vauzour, D.; Müller, M. Differential Influence of Soluble Dietary Fibres on Intestinal and Hepatic Carbohydrate Response. Nutrients 2021, 13, 4278. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A. All Disease Begins in the (Leaky) Gut: Role of Zonulin-Mediated Gut Permeability in the Pathogenesis of Some Chronic Inflammatory Diseases. F1000Res 2020, 9, 69. [Google Scholar] [CrossRef]
- Crenn, P.; Messing, B.; Cynober, L. Citrulline as a Biomarker of Intestinal Failure Due to Enterocyte Mass Reduction. Clin. Nutr. 2008, 27, 328–339. [Google Scholar] [CrossRef]
- Cinkajzlová, A.; Lacinová, Z.; Kloučková, J.; Kaválková, P.; Kratochvílová, H.; Křížová, J.; Trachta, P.; Mráz, M.; Haluzík, M. Increased Intestinal Permeability in Patients with Short Bowel Syndrome Is Not Affected by Parenteral Nutrition. Physiol. Res. 2019, 68, 817–825. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical Features of Patients Infected with 2019 Novel Coronavirus in Wuhan, China. Lancet 2020, 395, 497. [Google Scholar] [CrossRef] [Green Version]
- Qin, C.; Zhou, L.; Hu, Z.; Zhang, S.; Yang, S.; Tao, Y.; Xie, C.; Ma, K.; Shang, K.; Wang, W.; et al. Dysregulation of Immune Response in Patients with COVID-19 in Wuhan, China. Clin. Infect. Dis. 2020, 71, 762–768. [Google Scholar] [CrossRef]
- Gao, Y.; Li, T.; Han, M.; Li, X.; Wu, D.; Xu, Y.; Zhu, Y.; Liu, Y.; Wang, X.; Wang, L. Diagnostic Utility of Clinical Laboratory Data Determinations for Patients with the Severe COVID. J. Med. Virol. 2020, 92, 791–796. [Google Scholar] [CrossRef]
- Hasanvand, A. COVID-19 and the Role of Cytokines in This Disease. Inflammopharmacology 2022, 30, 789. [Google Scholar] [CrossRef]
- Brasili, E.; Mengheri, E.; Tomassini, A.; Capuani, G.; Roselli, M.; Finamore, A.; Sciubba, F.; Marini, F.; Miccheli, A. Lactobacillus Acidophilus La5 and Bifidobacterium Lactis BB12 Induce Different Age-Related Metabolic Profiles Revealed by 1H-NMR Spectroscopy in Urine and Feces of Mice1. J. Nutr. 2013, 143, 1549–1557. [Google Scholar] [CrossRef] [Green Version]
- Warburton, P.J.; Ciric, L.; Lerner, A.; Seville, L.A.; Roberts, A.P.; Mullany, P.; Allan, E. TetAB(46), a Predicted Heterodimeric ABC Transporter Conferring Tetracycline Resistance in Streptococcus Australis Isolated from the Oral Cavity. J. Antimicrob. Chemother. 2013, 68, 17. [Google Scholar] [CrossRef] [Green Version]
- Svedlund, J.; Sjödin, I.; Dotevall, G. GSRS—A Clinical Rating Scale for Gastrointestinal Symptoms in Patients with Irritable Bowel Syndrome and Peptic Ulcer Disease. Dig. Dis. Sci. 1988, 33, 129–134. [Google Scholar] [CrossRef]
- ODonnell: Pseudo-Diarrhea in the Irritable Bowel Google Scholar. Available online: https://scholar.google.com/scholar_lookup?title=Pseudo-diarrhea+in+the+irritable+bowel+syndrome:+patients´+records+of+stool+form+reflect+transit+time+while+stool+frequency+does+not=&author=O%27Donnell+LJD&author=Heaton+KW&publication_year=1988&journal=Gut&volume=29&pages=A1455 (accessed on 11 April 2022).
- Mandel, M.; Harari, G.; Gurevich, M.; Achiron, A. Cytokine Prediction of Mortality in COVID19 Patients. Cytokine 2020, 134, 155190. [Google Scholar] [CrossRef]
- Bordoni, V.; Sacchi, A.; Cimini, E.; Notari, S.; Grassi, G.; Tartaglia, E.; Casetti, R.; Giancola, M.L.; Bevilacqua, N.; Maeurer, M.; et al. An Inflammatory Profile Correlates with Decreased Frequency of Cytotoxic Cells in Coronavirus Disease. Clin. Infect. Dis. 2020, 71, 2272–2275. [Google Scholar] [CrossRef]
- Leo, L.D.; Naviglio, S.; Vatta, S.; Benelli, E.; Stera, G.; Santon, D.; Ziberna, F.; Taddio, A.; Martelossi, S.; Giudici, F.; et al. Circulating PV-1 as a Marker of Celiac Disease-Associated Liver Injury. Biomark. Med. 2020, 14, 1675–1681. [Google Scholar] [CrossRef]
- Summerton, C.B.; Longlands, M.G.; Wiener, K.; Shreeve, D.R. Faecal Calprotectin: A Marker of Inflammation throughout the Intestinal Tract. Eur. J. Gastroenterol. Hepatol. 2002, 14, 841–845. [Google Scholar] [CrossRef]
- Posteraro, B.; De Maio, F.; Gasbarrini, A. Profiling the Gastrointestinal Microbiota. Methods Mol. Biol. 2021, 2283, 83–92. [Google Scholar] [CrossRef]
- De Pascale, G.; De Maio, F.; Carelli, S.; De Angelis, G.; Cacaci, M.; Montini, L.; Bello, G.; Cutuli, S.L.; Pintaudi, G.; Tanzarella, E.S.; et al. Staphylococcus Aureus Ventilator-Associated Pneumonia in Patients with COVID-19: Clinical Features and Potential Inference with Lung Dysbiosis. Crit. Care 2021, 25, 197. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, Interactive, Scalable and Extensible Microbiome Data Science Using QIIME. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-Resolution Sample Inference from Illumina Amplicon Data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, M.; Mathew, B.J.; Gupta, P.; Garg, G.; Khadanga, S.; Vyas, A.K.; Singh, A.K. Gut Dysbiosis and Il-21 Response in Patients with Severe COVID-19. Microorganisms 2021, 9, 1292. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Chen, S.; Chen, Y.; Tian, L.; Wu, Q.; Zheng, M.; Li, Z. Alterations of Fecal Antibiotic Resistome in COVID-19 Patients after Empirical Antibiotic Exposure. Int. J. Hyg. Env. Health 2022, 240, 113882. [Google Scholar] [CrossRef] [PubMed]
- Bokulich, N.A.; Kaehler, B.D.; Rideout, J.R.; Dillon, M.; Bolyen, E.; Knight, R.; Huttley, G.A.; Gregory Caporaso, J. Optimizing Taxonomic Classification of Marker-Gene Amplicon Sequences with QIIME 2’s Q2-Feature-Classifier Plugin. Microbiome 2018, 6, 90. [Google Scholar] [CrossRef]
- Paulson, J.N.; Colin Stine, O.; Bravo, H.C.; Pop, M. Differential Abundance Analysis for Microbial Marker-Gene Surveys. Nat. Methods 2013, 10, 1200–1202. [Google Scholar] [CrossRef] [Green Version]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B Methodol. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic Biomarker Discovery and Explanation. Genome. Biol. 2011, 12, R60. [Google Scholar] [CrossRef] [Green Version]






| Summary Statistics | Tot (n = 19) | ||
|---|---|---|---|
| Sex | Female | %, n | 31.6% (6/19) |
| Male | %, n | 68.4% (13/19) | |
| Age | n | 19 (100.0%) | |
| Mean ± SD | 55.00 ± 8.56 | ||
| Median (Q1-Q3) | 54.00 (52.00–61.00) | ||
| Min-Max | 33.0–70.0 | ||
| Missing | 0 | ||
| BMI | n | 19 (100%) | |
| Mean ± SD | 26.53 ± 4.20 | ||
| Median (Q1-Q3) | 25.56 (23.90–28.70) | ||
| Min-Max | 18.5–40.9 | ||
| Missing | 0 | ||
| Other diseases (concomitant and previous) | %, n | 68.4% (13/19) | |
| Concomitant diseases | Anxious depressive syndrome | %, n | 4.8% (1/21) |
| Benign prostatic hypertrophy | %, n | 14.3% (3/21) | |
| Diabetes | %, n | 9.5% (2/21) | |
| Gastroesophageal reflux | %, n | 14.3% (3/21) | |
| Hiatal hernia | %, n | 4.8% (1/21) | |
| Hypercholesterolemia | %, n | 4.8% (1/21) | |
| Hypertension | %, n | 28.6% (6/21) | |
| Insomnia | %, n | 4.8% (1/21) | |
| Minor beta-thalassemia | %, n | 4.8% (1/21) | |
| Osteoporosis | %, n | 4.8% (1/21) | |
| Tachycardia | %, n | 4.8% (1/21) | |
| Previous diseases | Cerebral Ischemia | %, n | 20.0% (1/5) |
| Chlamydial pneumonia | %, n | 20.0% (1/5) | |
| Previous HBV | %, n | 20.0% (1/5) | |
| Thyroiditis | %, n | 20.0% (1/5) | |
| Ulna and radius fractures | %, n | 20.0% (1/5) | |
| Concomitant medication | %, n | 78.9% (15/19) | |
| Previous surgery | %, n | 42.1% (8/19) |
| Summary Statistics | Tot (n = 19) | |
|---|---|---|
| Fever | %, n | 47.4% (9/19) |
| Cough | %, n | 21.1% (4/19) |
| Anosmia | %, n | 10.5% (2/19) |
| Diarrhea | %, n | 31.6% (6/19) |
| Other symptoms | %, n | 36.8% (7/19) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laterza, L.; Putignani, L.; Settanni, C.R.; Petito, V.; Varca, S.; De Maio, F.; Macari, G.; Guarrasi, V.; Gremese, E.; Tolusso, B.; et al. Ecology and Machine Learning-Based Classification Models of Gut Microbiota and Inflammatory Markers May Evaluate the Effects of Probiotic Supplementation in Patients Recently Recovered from COVID-19. Int. J. Mol. Sci. 2023, 24, 6623. https://doi.org/10.3390/ijms24076623
Laterza L, Putignani L, Settanni CR, Petito V, Varca S, De Maio F, Macari G, Guarrasi V, Gremese E, Tolusso B, et al. Ecology and Machine Learning-Based Classification Models of Gut Microbiota and Inflammatory Markers May Evaluate the Effects of Probiotic Supplementation in Patients Recently Recovered from COVID-19. International Journal of Molecular Sciences. 2023; 24(7):6623. https://doi.org/10.3390/ijms24076623
Chicago/Turabian StyleLaterza, Lucrezia, Lorenza Putignani, Carlo Romano Settanni, Valentina Petito, Simone Varca, Flavio De Maio, Gabriele Macari, Valerio Guarrasi, Elisa Gremese, Barbara Tolusso, and et al. 2023. "Ecology and Machine Learning-Based Classification Models of Gut Microbiota and Inflammatory Markers May Evaluate the Effects of Probiotic Supplementation in Patients Recently Recovered from COVID-19" International Journal of Molecular Sciences 24, no. 7: 6623. https://doi.org/10.3390/ijms24076623