Investigating the Hydration, Mechanical Properties, and Pozzolanic Activity of Cement Paste Containing Co-Combustion Fly Ash
Abstract
:1. Introduction
2. Materials and Test Methods
2.1. Raw Materials
2.2. Sample Preparation
2.3. Test Methods
2.3.1. Isothermal Calorimetry
2.3.2. Mechanical Properties and PEC
2.3.3. SEM-EDS
2.3.4. XRD
2.3.5. TG-DTG
3. Results and Discussion
3.1. Basic Properties and Variability of CCFA
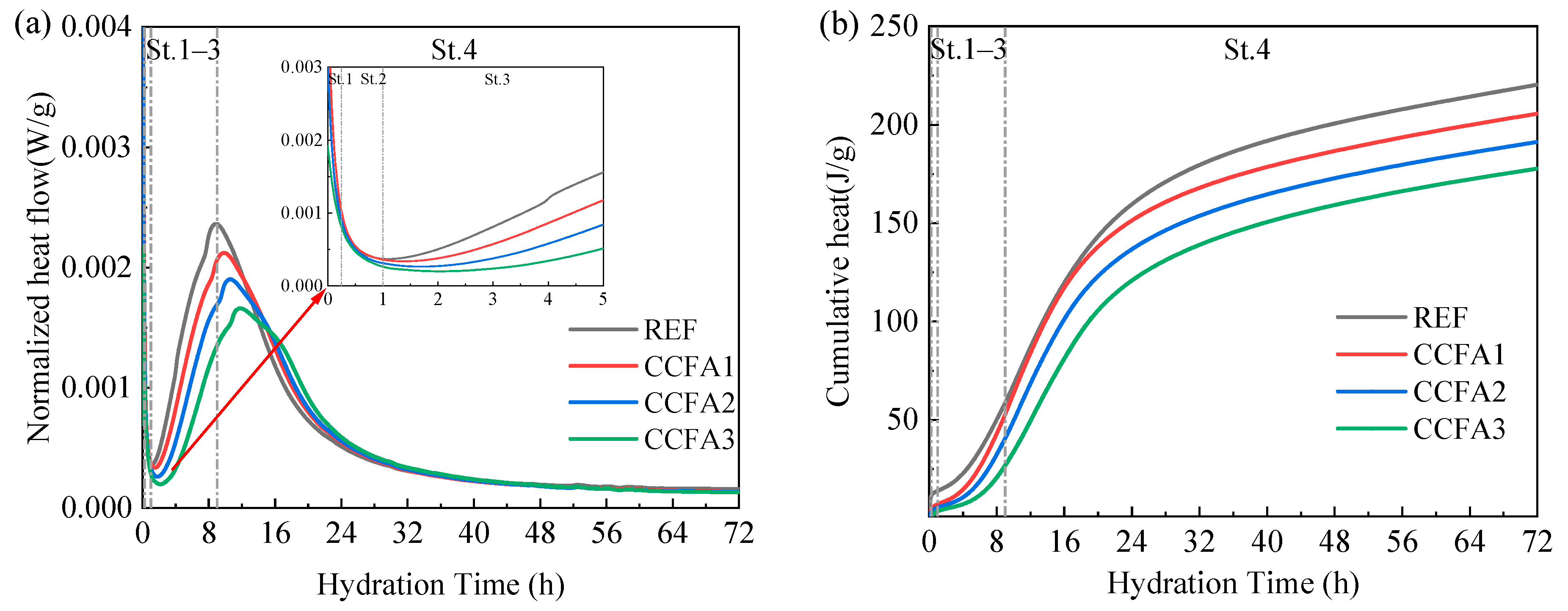
3.2. Hydration Performance of SCMs
3.3. Mechanical Properties and Pozzolanic Activity of SCMs
3.4. Microscopic Characteristics of SCMs
3.4.1. SEM-EDS Analysis
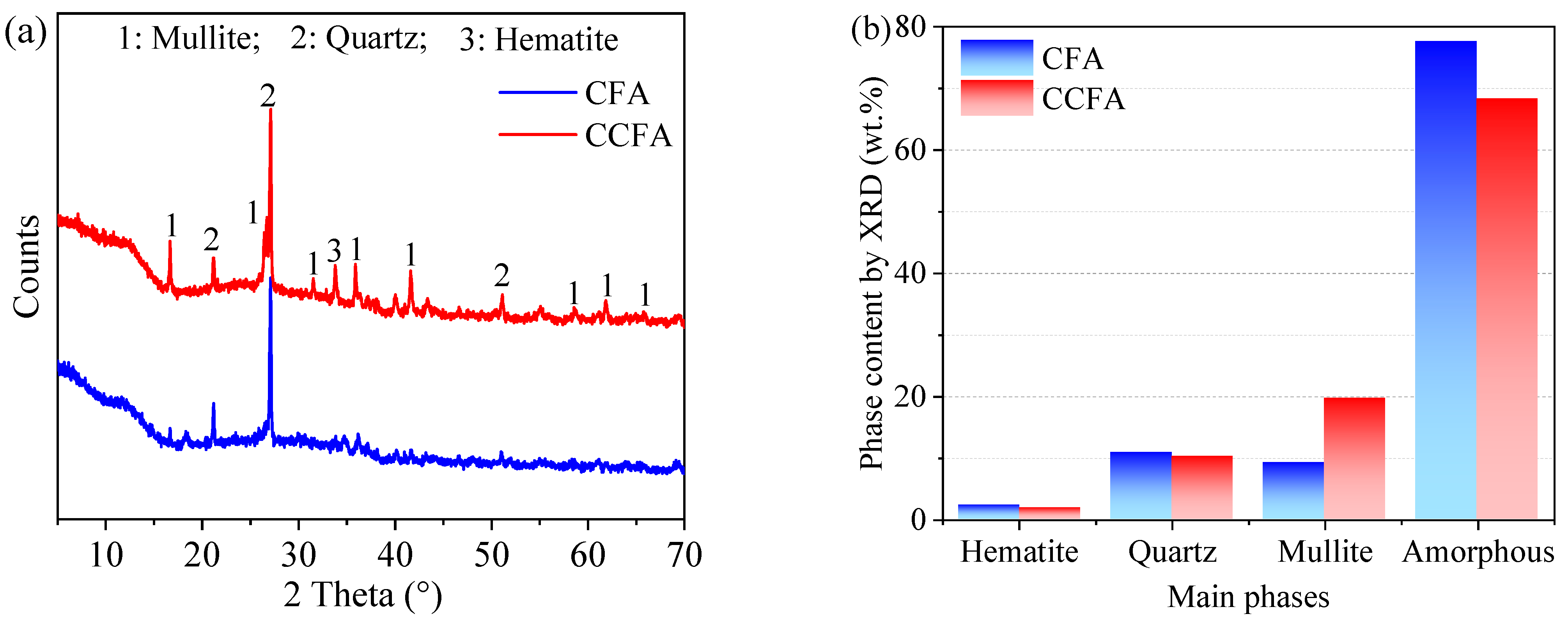
3.4.2. XRD Analysis
3.4.3. TG-DTG Analysis
4. Conclusions
- (1)
- CCFA and CFA are similar in terms of major mineral composition. Compared with CFA, CCFA has more mullite and less amorphous phase. The physical and chemical properties of all batches of CCFA comply with the standard limits.
- (2)
- The heat of hydration displays a declining trend with an increase in fly ash blending. Although there are differences in the early heat release rate between CFA and CCFA samples, the final cumulative heat release demonstrates substantial similarity.
- (3)
- All mortars containing blended fly ash had PEC < 0 at 3 days; CFA and CCFA behaved as inert fillers at this stage. After 180 days of hydration, all fly ash-blended samples exhibited considerable pozzolanic activity (PEC > 0.5). Incorporating CCFA into the cement improved the long-term mechanical properties.
- (4)
- At 180 d, both CFA and CCFA samples produced the secondary hydration product, C-A-S-H. The consumption of CH by two kinds of fly ash is comparable, demonstrating that CCFA and CFA have similar pozzolanic activity.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, S.; Wang, F.; Mei, Z.; Lv, L.; Chi, Y. Status and development of sludge incineration in China. Waste Biomass Valorization 2021, 12, 3541–3574. [Google Scholar] [CrossRef]
- Jin, L.; Zhang, G.; Tian, H. Current state of sewage treatment in China. Water Res. 2014, 66, 85–98. [Google Scholar] [CrossRef]
- Global and Chinese Sludge Treatment and Disposal Industry Development Research Report 2018. Available online: http://www.gepresearch.com/77/view-767588-1.html (accessed on 21 January 2019).
- Sludge Treatment Discharge Should Develop in the Direction of Green and Low Carbon. 2022. Available online: http://epaper.cenews.com.cn/html/2022-07/12/content_78019.htm (accessed on 12 July 2022).
- Disposal of Factory Sludge from Water Purification Plants in Shenzhen. 2022. Available online: http://swj.sz.gov.cn/sjfb/psglxgtjsj/content/post_10510005.html (accessed on 28 March 2023).
- Yu, Q.; Lei, H.; Li, Z.; Li, H.; Chen, K.; Zhang, X.; Liang, R. Physical and chemical properties of waste-activated sludge after microwave treatment. Water Res. 2010, 44, 2841–2849. [Google Scholar] [CrossRef] [PubMed]
- Wahaab, R.A.; Mahmoud, M.; van Lier, J.B. Toward achieving sustainable management of municipal wastewater sludge in Egypt: The current status and future prospective. Renew. Sustain. Energy Rev. 2020, 127, 109880. [Google Scholar] [CrossRef]
- Meer, I.; Nazir, R. Removal techniques for heavy metals from fly ash. J. Mater. Cycles Waste Manag. 2017, 20, 703–722. [Google Scholar] [CrossRef]
- Jambhulkar, H.P.; Shaikh, S.M.S.; Kumar, M.S. Fly ash toxicity, emerging issues and possible implications for its exploitation in agriculture; Indian scenario: A review. Chemosphere 2018, 213, 333–344. [Google Scholar] [CrossRef]
- Stelmach, S.; Wasielewski, R. Co-combustion of dried sewage sludge and coal in a pulverized coal boiler. J. Mater. Cycles Waste Manag. 2008, 10, 110–115. [Google Scholar] [CrossRef]
- Tang, Y.; Pan, J.; Li, B.; Zhao, S.; Zhang, L. Residual and ecological risk assessment of heavy metals in fly ash from co-combustion of excess sludge and coal. Sci. Rep. 2021, 11, 2499. [Google Scholar] [CrossRef]
- Chen, J.; Sun, Y.; Zhang, Z. Evolution of trace elements and polluting gases toward clean co-combustion of coal and sewage sludge. Fuel 2020, 280, 118685. [Google Scholar] [CrossRef]
- Zhang, G.; Hai, J.; Ren, M.; Zhang, S.; Cheng, J.; Yang, Z. Emission, mass balance, and distribution characteristics of PCDD/Fs and heavy metals during cocombustion of sewage sludge and coal in power plants. Environ. Sci. Technol. 2013, 47, 2123–2130. [Google Scholar] [CrossRef]
- Sarabèr, A.J. Co-combustion and its impact on fly ash quality; full-scale experiments. Fuel Process. Technol. 2014, 128, 68–82. [Google Scholar] [CrossRef]
- Danish, A.; Ozbakkaloglu, T. Greener cementitious composites incorporating sewage sludge ash as cement replacement: A review of progress, potentials, and future prospects. J. Clean. Prod. 2022, 371, 133364. [Google Scholar] [CrossRef]
- Murakami, T.; Suzuki, Y.; Nagasawa, H.; Yamamoto, T.; Koseki, T.; Hirose, H.; Okamoto, S. Combustion characteristics of sewage sludge in an incineration plant for energy recovery. Fuel Process. Technol. 2009, 90, 778–783. [Google Scholar] [CrossRef]
- DB 31/1291-2021; Emission Standard of Air Pollutants for Coal and Sludge Co-Fired Power Plant. Shanghai Municipal Bureau of Ecology and Environment: Shanghai, China, 2021.
- Li, J.S.; Xue, Q.; Fang, L.; Poon, C.S. Characteristics and metal leachability of incinerated sewage sludge ash and air pollution control residues from Hong Kong evaluated by different methods. Waste Manag. 2017, 64, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Lynn, C.J.; Dhir, R.K.; Ghataora, G.S. Environmental impacts of sewage sludge ash in construction: Leaching assessment, Resources. Conserv. Recycl. 2018, 136, 306–314. [Google Scholar] [CrossRef]
- Wu, Z.; Jiang, Y.; Guo, W.; Jin, J.; Wu, M.; Shen, D.; Long, Y. The long-term performance of concrete amended with municipal sewage sludge incineration ash. Environ. Technol. Innov. 2021, 23, 101574. [Google Scholar] [CrossRef]
- Nilimaa, J. Smart materials and technologies for sustainable concrete construction. Dev. Built Environ. 2023, 15, 100177. [Google Scholar] [CrossRef]
- Zhang, L.; Ito, M.; Sato, A.; Ninomiya, Y.; Sakano, T.; Kanaoka, C.; Masui, M. Combustibility of dried sewage sludge and its mineral transformation at different oxygen content in drop tube furnace. Fuel Process. Technol. 2004, 85, 983–1011. [Google Scholar] [CrossRef]
- Sarabèr, A. Co-combustion and its impact on fly ash quality; pilot-scale experiments. Fuel Process. Technol. 2012, 104, 105–114. [Google Scholar] [CrossRef]
- Kijo-Kleczkowska, A.; Środa, K.; Kosowska-Golachowska, M.; Musiał, T.; Wolski, K. Experimental research of sewage sludge with coal and biomass co-combustion, in pellet form. Waste Manag. 2016, 53, 165–181. [Google Scholar] [CrossRef]
- Kijo-Kleczkowska, A.; Środa, K.; Kosowska-Golachowska, M.; Musiał, T.; Wolski, K. Combustion of pelleted sewage sludge with reference to coal and biomass. Fuel 2016, 170, 141–160. [Google Scholar] [CrossRef]
- Gao, F.; Zhou, C.; Du, J.; Zhang, Y.; Wu, W.; Liu, G.; Li, D. Effect of gaseous agents on co-combustion characteristics of sewage sludge and coal. J. Environ. Chem. Eng. 2021, 9, 106227. [Google Scholar] [CrossRef]
- Ma, M.; Liang, Y.; Xu, D.; Sun, S.; Zhao, J.; Wang, S. Gas emission characteristics of sewage sludge co-combustion with coal: Effect of oxygen atmosphere and feedstock mixing ratio. Fuel 2022, 322, 124102. [Google Scholar] [CrossRef]
- Xu, T.; Wang, C.; Hong, D.; Li, S.; Yue, S. The synergistic effect during co-combustion of municipal sludge and coal: Experimental and ReaxFF molecular dynamic study. Energy 2023, 262, 125553. [Google Scholar] [CrossRef]
- Stasta, P.; Boran, J.; Bebar, L.; Stehlik, P.; Oral, J. Thermal processing of sewage sludge. Appl. Therm. Eng. 2006, 26, 1420–1426. [Google Scholar] [CrossRef]
- Rao, M.; Ye, J.; Chen, C.; Kuang, C.; Chen, G.; Zou, X.; Qin, S.; Chen, L.; Liang, S. Construction of iterative algorithm and economic optimization analysis of co-combustion of sewage sludge with coal. Int. J. Green Energy 2022, 20, 925–933. [Google Scholar] [CrossRef]
- Tan, P.; Ma, L.; Xia, J.; Fang, Q.; Zhang, C.; Chen, G. Co-firing sludge in a pulverized coal-fired utility boiler: Combustion characteristics and economic impacts. Energy 2017, 119, 392–399. [Google Scholar] [CrossRef]
- Tong, C. Study on the Formation Mechanism and Control Technology of Dioxins in the Process of Municipal Solid Waste Incineration; Zhejiang University: Zhejiang, China, 2006. [Google Scholar]
- Huang, H.; Buekens, A. On the mechanisms of dioxin formation in combustion processes. Chemosphere 1995, 31, 4099–4117. [Google Scholar] [CrossRef]
- Nadziakiewicz, J.; Kozioł, M. Co-combustion of sludge with coal. Appl. Energy 2003, 75, 239–248. [Google Scholar] [CrossRef]
- Cenni, R.; Janisch, B.; Spliethoff, H.; Hein, K. Legislative and environmental issues on the use of ash from coal and municipal sewage sludge co-firing as construction material. Waste Manag. 2001, 21, 17–31. [Google Scholar] [CrossRef]
- Al-Ayish, N.; During, O.; Malaga, K.; Silva, N.; Gudmundsson, K. The influence of supplementary cementitious materials on climate impact of concrete bridges exposed to chlorides. Constr. Build. Mater. 2018, 188, 391–398. [Google Scholar] [CrossRef]
- Cretescu, I.; Harja, M.; Teodosiu, C.; Isopescu, D.N.; Chok, M.F.; Sluser, B.M.; Salleh, M.A.M. Synthesis and characterisation of a binder cement replacement based on alkali activation of fly ash waste. Process Saf. Environ. Prot. 2018, 119, 23–35. [Google Scholar] [CrossRef]
- Tan, H.; Nie, K.; He, X.; Deng, X.; Zhang, X.; Su, Y.; Yang, J. Compressive strength and hydration of high-volume wet-grinded coal fly ash cementitious materials. Constr. Build. Mater. 2019, 206, 248–260. [Google Scholar] [CrossRef]
- Goñi, S.; Guerrero, A.; Luxán, M.P.; Macías, A. Activation of the fly ash pozzolanic reaction by hydrothermal conditions. Cem. Concr. Res. 2003, 33, 1399–1405. [Google Scholar] [CrossRef]
- GB 8076-2008; Concrete Admixtures. Standards Press of China: Beijing, China, 2008.
- Schöler, A.; Lothenbach, B.; Winnefeld, F.; Zajac, M. Hydration of quaternary Portland cement blends containing blast-furnace slag, siliceous fly ash and limestone powder. Cem. Concr. Compos. 2015, 55, 374–382. [Google Scholar] [CrossRef]
- GB/T 17671-2021; Test Method of Cement Mortar Strength (ISO Method). Standards Press of China: Beijing, China, 2021.
- Záleská, M.; Pavlíková, M.; Pavlík, Z.; Jankovský, O.; Pokorný, J.; Tydlitát, V.; Svora, P.; Černý, R. Physical and chemical characterization of technogenic pozzolans for the application in blended cements. Constr. Build. Mater. 2018, 160, 106–116. [Google Scholar] [CrossRef]
- Piasta, W.; Lukawska, M. The effect of sewage sludge ash on properties of cement composites. Procedia Eng. 2016, 161, 1018–1024. [Google Scholar] [CrossRef]
- GB/T 1596-2017; Fly Ash Used for Cement and Concrete. Standards Press of China: Beijing, China, 2017.
- Qian, J. Fly Ash Properties and Fly Ash Concrete; Science Press: Beijing, China, 2000. (In Chinese) [Google Scholar]
- Taylor, H.F.W. Cement Chemistry; Thomas Telford: London, UK, 1997. [Google Scholar]
- Jansen, D.; Neunhoeffer, F.G.; Lothenbach, B.; Neubauer, J. The early hydration of ordinary Portland cement (OPC): An approach comparing measured heat flow with calculated heat flow from QXRD. Cem. Concr. Res. 2012, 42, 134–138. [Google Scholar] [CrossRef]
- Bensted, J. Some applications of conduction calorimetry to cement hydration. Adv. Cem. Res. 1987, 1, 35–44. [Google Scholar] [CrossRef]
- Langan, B.W.; Weng, K.; Ward, M.A. Effect of silica fume and fly ash on heat of hydration of Portland cement. Cem. Concr. Res. 2002, 32, 1045–1051. [Google Scholar] [CrossRef]
- Snelson, D.G.; Wild, S.; O’Farrell, M. Heat of hydration of Portland Cement–Metakaolin–Fly ash (PC–MK–PFA) blends. Cem. Concr. Res. 2008, 38, 832–840. [Google Scholar] [CrossRef]
- Tkaczewska, E.; Małolepszy, J. Hydration of coal–biomass fly ash cement. Constr. Build. Mater. 2009, 23, 2694–2700. [Google Scholar] [CrossRef]
- Kaminskas, R.; Cesnauskas, V.; Kubiliute, R. Influence of different artificial additives on Portland cement hydration and hardening. Constr. Build. Mater. 2015, 95, 537–544. [Google Scholar] [CrossRef]
- Deschner, F.; Winnefeld, F.; Lothenbach, B.; Seufert, S.; Schwesig, P.; Dittrich, S.; Goetz-Neunhoeffer, F.; Neubauer, J. Hydration of Portland cement with high replacement by siliceous fly ash. Cem. Concr. Res. 2012, 42, 1389–1400. [Google Scholar] [CrossRef]
- Fořt, J.; Šál, J.; Ševčík, R.; Doleželová, M.; Keppert, M.; Jerman, M.; Záleská, M.; Stehel, V.; Černý, R. Biomass fly ash as an alternative to coal fly ash in blended cements: Functional aspects. Constr. Build. Mater. 2021, 271, 121544. [Google Scholar] [CrossRef]
- El Fami, N.; Ez-zaki, H.; Sassi, O.; Boukhari, A.; Diouri, A. Rheology, calorimetry and electrical conductivity related-properties for monitoring the dissolution and precipitation process of cement-fly ash mixtures. Powder Technol. 2022, 411, 117937. [Google Scholar] [CrossRef]
- Nilimaa, J.; Hösthagen, A.; Emborg, M. Thermal crack risk of concrete structures: Evaluation of theoretical models for tunnels and bridges. Nord. Concr. Res. 2017, 56, 55–69. [Google Scholar]
- Papadakis, V.G. Effect of fly ash on Portland cement systems. Cem. Concr. Res. 1999, 29, 1727–1736. [Google Scholar] [CrossRef]
- Ram, S.; Tare, M.S.; Aswath, P.B.; Ralegaonkar, R.V. Potential of co-fired fly ashes as a construction material—A review. Encycl. Renew. Sustain. Mater. 2020, 1, 674–685. [Google Scholar]
- Kamali, M.; Ghahremaninezhad, A. Effect of glass powders on the mechanical and durability properties of cementitious materials. Constr. Build. Mater. 2015, 98, 407–416. [Google Scholar] [CrossRef]
- Cho, Y.K.; Jung, S.H.; Choi, Y.C. Effects of chemical composition of fly ash on compressive strength of fly ash cement mortar. Constr. Build. Mater. 2019, 204, 255–264. [Google Scholar] [CrossRef]
- Geng, Z.; Tang, S.; Wang, Y.; He, Z.; Wu, K.; Wang, L. Stress relaxation properties of calcium silicate hydrate: A molecular dynamics study. J. Zhejiang Univ. -SCIENCE A 2024, 25, 97–115. [Google Scholar] [CrossRef]
- Tang, S.; Wang, Y.; Geng, Z.; Xu, X.; Yu, W.; Chen, J. Structure, fractality, mechanics and durability of calcium silicate hydrates. Fractal Fract. 2021, 5, 47. [Google Scholar] [CrossRef]
- Tang, S.; Hubao, A.; Chen, J.; Yu, W.; Yu, P.; Chen, E.; Deng, H.; He, Z. The interactions between water molecules and CSH surfaces in loads-induced nanopores: A molecular dynamics study. Appl. Surf. Sci. 2019, 496, 143744. [Google Scholar] [CrossRef]
- Rossen, J.E.; Scrivener, K.L. Optimization of SEM-EDS to determine the C–A–S–H composition in matured cement paste samples. Mater. Charact. 2017, 123, 294–306. [Google Scholar] [CrossRef]
- Rodger, S.A.; Groves, G.W. The microstructure of tricalcium silicate/pulverized-fuel ash blended cement pastes. Adv. Cem. Res. 1988, 1, 84–91. [Google Scholar] [CrossRef]
- Zhang, Z.; Angst, U. Microstructure and moisture transport in carbonated cement-based materials incorporating cellulose nanofibrils. Cem. Concr. Res. 2022, 162, 106990. [Google Scholar] [CrossRef]
- Lin, X.; Mao, T.; Chen, Z.; Chen, J.; Zhang, S.; Li, X.; Yan, J. Thermal cotreatment of municipal solid waste incineration fly ash with sewage sludge: Phases transformation, kinetics and fusion characteristics, and heavy metals solidification. J. Clean. Prod. 2021, 317, 128429. [Google Scholar] [CrossRef]
- Ogawa, K.; Uchikawa, H.; Takemoto, K.; Yasui, I. The mechanism of the hydration in the system C3S-pozzolana. Cem. Concr. Res. 1980, 10, 683–696. [Google Scholar] [CrossRef]
- Park, B.; Choi, Y.C. Hydration and pore-structure characteristics of high-volume fly ash cement pastes. Constr. Build. Mater. 2021, 278, 122390. [Google Scholar] [CrossRef]














| Characterization | Value | Standard Limit |
|---|---|---|
| High/low calorific value of dry basis (MJ/kg) | 7.91/7.2 | None |
| High/low calorific value on wet basis (MJ/kg) | 6.28/5.3 | |
| Received base volatile (wt. %) | 27.19 | |
| Received base fixed carbon (wt. %) | 3.86 | |
| Received base ash (wt. %) | 48.36 | |
| Organic matter (wt. %) | 42.41 | |
| Full moisture (wt. %) | 20.59 | |
| Total number of bacteria (count /g) | 1.7 × 104 | |
| Ascaris egg mortality (%) | 100 | ≥95 [38] |
| Fecal coliform value | >11.1 | ≥0.01 [38] |
| Name | SiO2 | Al2O3 | Fe2O3 | CaO | P2O5 | K2O | MgO | SO3 | Na2O | Others |
|---|---|---|---|---|---|---|---|---|---|---|
| SS | 48.18 | 24.08 | 15.22 | 1.08 | 6.17 | 2.35 | 0.90 | 0.05 | 0.48 | 1.49 |
| OPC | 22.37 | 4.36 | 3.38 | 61.08 | - | - | 2.43 | 2.45 | - | 3.93 |
| CFA | 44.31 | 20.50 | 9.16 | 15.31 | 0.32 | 1.21 | 1.99 | 1.59 | 3.81 | 1.80 |
| CCFA | 46.56 | 30.66 | 7.50 | 7.46 | 1.54 | 1.32 | 1.23 | 0.99 | 1.02 | 1.72 |
| Density/(g/cm3) | Specific Area/(m2/kg) | Compressive Strength/MPa | Flexural Strength/MPa | ||
|---|---|---|---|---|---|
| 3 d | 28 d | 3 d | 28 d | ||
| 3.15 | 340 | 28.4 | 48.2 | 6.0 | 8.7 |
| Name | w/c | Cement (g) | CFA (g) | CCFA (g) | Water (g) |
|---|---|---|---|---|---|
| REF | 0.5 | 450 | 0 | 0 | 225 |
| CFA1 | 0.5 | 405 | 45 | 0 | 225 |
| CFA2 | 0.5 | 360 | 90 | 0 | 225 |
| CFA3 | 0.5 | 315 | 135 | 0 | 225 |
| CCFA1 | 0.5 | 405 | 0 | 45 | 225 |
| CCFA2 | 0.5 | 360 | 0 | 90 | 225 |
| CCFA3 | 0.5 | 315 | 0 | 135 | 225 |
| Physical and Chemical Properties | Range of Fluctuations | Standard Limit [44] |
|---|---|---|
| Water content (%) | 0.1–0.2 | ≤1.0 |
| Water demand ratio (%) | 90–98 | ≤105 |
| Fineness (45 μm) (%) | 3.1–25.6 | ≤30.0 |
| Stability (mm) | 0–1.5 | ≤5.0 |
| Ignition loss (%) | 0.61–2.15 | ≤8.0 |
| Mass fraction of SO3 (%) | 0.28–1.07 | ≤3.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ding, X.; Du, H.; Wu, E.; Yi, P.; Li, Y.; Luo, Y.; Liu, W. Investigating the Hydration, Mechanical Properties, and Pozzolanic Activity of Cement Paste Containing Co-Combustion Fly Ash. Buildings 2024, 14, 1305. https://doi.org/10.3390/buildings14051305
Ding X, Du H, Wu E, Yi P, Li Y, Luo Y, Liu W. Investigating the Hydration, Mechanical Properties, and Pozzolanic Activity of Cement Paste Containing Co-Combustion Fly Ash. Buildings. 2024; 14(5):1305. https://doi.org/10.3390/buildings14051305
Chicago/Turabian StyleDing, Xiaobo, Hangyu Du, Enfeng Wu, Peng Yi, Yongqiang Li, Yaoming Luo, and Wei Liu. 2024. "Investigating the Hydration, Mechanical Properties, and Pozzolanic Activity of Cement Paste Containing Co-Combustion Fly Ash" Buildings 14, no. 5: 1305. https://doi.org/10.3390/buildings14051305





