Journal Description
Viruses
Viruses
is a peer-reviewed, open access journal of virology, published monthly online by MDPI. The American Society for Virology (ASV), Spanish Society for Virology (SEV), Canadian Society for Virology (CSV), Italian Society for Virology (SIV-ISV), Australasian Virology Society (AVS) and others are affiliated with Viruses and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, MEDLINE, PMC, Embase, PubAg, AGRIS, and other databases.
- Journal Rank: JCR - Q2 (Virology) / CiteScore - Q1 (Infectious Diseases)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 13.8 days after submission; acceptance to publication is undertaken in 2.5 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Companion journals for Viruses include: COVID and Zoonotic Diseases.
Impact Factor:
4.7 (2022);
5-Year Impact Factor:
4.8 (2022)
Latest Articles
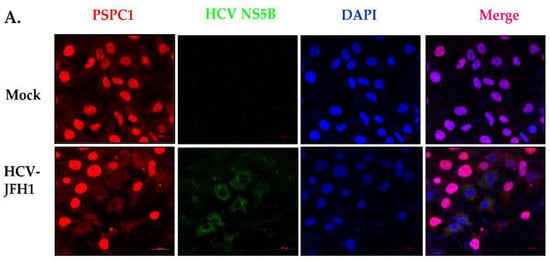
PSPC1 Binds to HCV IRES and Prevents Ribosomal Protein S5 Binding, Inhibiting Viral RNA Translation
Viruses 2024, 16(5), 738; https://doi.org/10.3390/v16050738 - 07 May 2024
Abstract
Hepatitis C virus (HCV) infects the human liver, and its chronic infection is one of the major causes of Hepatocellular carcinoma. Translation of HCV RNA is mediated by an Internal Ribosome Entry Site (IRES) element located in the 5’UTR of viral RNA. Several
[...] Read more.
Hepatitis C virus (HCV) infects the human liver, and its chronic infection is one of the major causes of Hepatocellular carcinoma. Translation of HCV RNA is mediated by an Internal Ribosome Entry Site (IRES) element located in the 5’UTR of viral RNA. Several RNA Binding proteins of the host interact with the HCV IRES and modulate its function. Here, we demonstrate that PSPC1 (Paraspeckle Component 1), an essential paraspeckle component, upon HCV infection is relocalized and interacts with HCV IRES to prevent viral RNA translation. Competition UV-crosslinking experiments showed that PSPC1 interacts explicitly with the SLIV region of the HCV IRES, which is known to play a vital role in ribosomal loading to the HCV IRES via interaction with Ribosomal protein S5 (RPS5). Partial silencing of PSPC1 increased viral RNA translation and, consequently, HCV replication, suggesting a negative regulation by PSPC1. Interestingly, the silencing of PSPC1 protein leads to an increased interaction of RPS5 at the SLIV region, leading to an overall increase in the viral RNA in polysomes. Overall, our results showed how the host counters viral infection by relocalizing nuclear protein to the cytoplasm as a survival strategy.
Full article
(This article belongs to the Special Issue Functional and Structural Features of Viral RNA Elements)
►
Show Figures
Open AccessArticle
Galectin-3-ITGB1 Signaling Mediates Interleukin 10 Production of Hepatic Conventional Natural Killer Cells in Chronic Hepatitis Virus B Transgenic Mice and Correlates with Hepatocellular Carcinoma Progression in Patients
by
Yongyan Chen, Wendi Zhang, Min Cheng, Xiaolei Hao, Haiming Wei, Rui Sun and Zhigang Tian
Viruses 2024, 16(5), 737; https://doi.org/10.3390/v16050737 - 07 May 2024
Abstract
Background and Aims: The outcomes of HBV infections are related to complex immune imbalances; however, the precise mechanisms by which HBV induces immune dysfunction are not well understood. Methods: HBV transgenic (HBs-Tg) mice were used to investigate intrahepatic NK cells in two distinct
[...] Read more.
Background and Aims: The outcomes of HBV infections are related to complex immune imbalances; however, the precise mechanisms by which HBV induces immune dysfunction are not well understood. Methods: HBV transgenic (HBs-Tg) mice were used to investigate intrahepatic NK cells in two distinct subsets: conventional NK (cNK) and liver-resident NK (LrNK) cells during a chronic HBV infection. Results: The cNK cells, but not the LrNK cells, were primarily responsible for the increase in the number of bulk NK cells in the livers of ageing HBs-Tg mice. The hepatic cNK cells showed a stronger ability to produce IL-10, coupled with a higher expression of CD69, TIGIT and PD-L1, and lower NKG2D expression in ageing HBs-Tg mice. A lower mitochondrial mass and membrane potential, and less polarized localization were observed in the hepatic cNK cells compared with the splenic cNK cells in the HBs-Tg mice. The enhanced galectin-3 (Gal-3) secreted from HBsAg+ hepatocytes accounted for the IL-10 production of hepatic cNK cells via ITGB1 signaling. For humans, LGALS3 and ITGB1 expression is positively correlated with IL-10 expression, and negatively correlated with the poor clinical progression of HCC. Conclusions: Gal-3-ITGB1 signaling shapes hepatic cNK cells but not LrNK cells during a chronic HBV infection, which may correlate with HCC progression.
Full article
(This article belongs to the Special Issue Natural Killer Cell in Viral Infection)
►▼
Show Figures

Figure 1
Open AccessArticle
Partial Alleviation of Homologous Superinfection Exclusion of SeMNPV Latently Infected Cells by G1 Phase Infection and G2/M Phase Arrest
by
Qi-Ming Fu, Zheng Fang, Lou Ren, Qing-Shan Wu, Jun-Bo Zhang, Qiu-Ping Liu, Lei-Tao Tan and Qing-Bei Weng
Viruses 2024, 16(5), 736; https://doi.org/10.3390/v16050736 - 06 May 2024
Abstract
Viral infection can regulate the cell cycle, thereby promoting viral replication. Hijacking and altering the cell cycle are important for the virus to establish and maintain a latent infection. Previously, Spodoptera exigua multiple nucleopolyhedrovirus (SeMNPV)-latently infected P8-Se301-C1 cells, which grew more slowly than
[...] Read more.
Viral infection can regulate the cell cycle, thereby promoting viral replication. Hijacking and altering the cell cycle are important for the virus to establish and maintain a latent infection. Previously, Spodoptera exigua multiple nucleopolyhedrovirus (SeMNPV)-latently infected P8-Se301-C1 cells, which grew more slowly than Se301 cells and interfered with homologous SeMNNPV superinfection, were established. However, the effects of latent and superinfection with baculoviruses on cell cycle progression remain unknown. In this study, the cell cycle profiles of P8-Se301-C1 cells and SeMNPV or Autographa californica multiple nucleopolyhedrovirus (AcMNPV)-infected P8-Se301-C1 cells were characterized by flow cytometry. The results showed that replication-related genes MCM4, PCNA, and BAF were down-regulated (p < 0.05) in P8-Se301-C1 cells, and the S phase of P8-Se301-C1 cells was longer than that of Se301 cells. P8-Se301-C1 cells infected with SeMNPV did not arrest in the G2/M phase or affect the expression of Cyclin B and cyclin-dependent kinase 1 (CDK1). Furthermore, when P8-Se301-C1 cells were infected with SeMNPV after synchronized treatment with hydroxyurea and nocodazole, light microscopy and qRT-PCR analysis showed that, compared with unsynchronized cells and S and G2/M phase cells, SeMNPV-infected P8-Se301-C1 cells in G1 phase induced G2/M phase arrest, and the amount of virus adsorption and intracellular viral DNA replication were significantly increased (p < 0.05). In addition, budded virus (BV) production and occlusion body (OB)-containing cells were both increased at 120 h post-infection (p < 0.05). The expression of Cyclin B and CDK1 was significantly down-regulated at 48 h post-infection (p < 0.05). Finally, the arrest of SeMNPV-infected G1 phase cells in the G2/M phase increased BV production (p < 0.05) and the number of OB-containing cells. In conclusion, G1 phase infection and G2/M arrest are favorable to SeMNPV proliferation in P8-Se301-C1 cells, thereby alleviating the homologous superinfection exclusion. The results contribute to a better understanding of the relationship between baculoviruses and insect cell cycle progression and regulation.
Full article
(This article belongs to the Special Issue Molecular Virus-Insect Interactions)
Open AccessArticle
Molecular Surveillance, Prevalence, and Distribution of Cacao Infecting Badnavirus Species in Côte d’Ivoire and Ghana
by
George A. Ameyaw, Koffié Kouakou, Mohammed Javed Iqbal, Luc Belé, Valentin L. F. Wolf, Cory V. Keith, Bolou A. Bolou Bi, Christophe Kouamé, Donald Livingstone, Owusu Domfeh, Ebenezer A. Gyamera, Jean-Philippe Marelli and Judith K. Brown
Viruses 2024, 16(5), 735; https://doi.org/10.3390/v16050735 - 06 May 2024
Abstract
The cacao swollen shoot disease (CSSD) caused by a complex of badnavirus species presents a major challenge for cacao production in West Africa, especially Ghana and Côte d’Ivoire. In this study, CSSD species detection efficiency, diversity, and geographic distribution patterns in cacao plantations
[...] Read more.
The cacao swollen shoot disease (CSSD) caused by a complex of badnavirus species presents a major challenge for cacao production in West Africa, especially Ghana and Côte d’Ivoire. In this study, CSSD species detection efficiency, diversity, and geographic distribution patterns in cacao plantations in Ghana and Côte d’Ivoire were investigated through field surveillance, PCR detection assays, sequencing of positive amplicons, and phylogeographic clustering. Cumulatively, the detection efficiency of the tested CSSD primer sets that were targeting the movement protein domain of the virus ranged from 0.15% (CSSD-3 primer) to 66.91% (CSSD-1 primer) on all the symptomatic cacao leaf samples assessed. The identified CSSD species differed phylogenetically and overlapped in distribution, with the cacao swollen shoot Togo B virus (CSSTBV) (n = 588 sequences) being the most prevalent and widely distributed compared to the other CSSD species that were encountered in both countries. Geographically, the cacao swollen shoot CE virus (CSSCEV) species (n = 124 sequences) that was identified was largely restricted to the bordering regions of Ghana and Côte d’Ivoire. These results provide updated knowledge of the geographic distribution of the key CSSD species and their diagnostic efficiency and, thus, provide guidance in identifying locations for structured testing of cacao germplasm and optimal diagnostics for the predominant CSSD species in Ghana and Côte d’Ivoire.
Full article
(This article belongs to the Special Issue Plant Viruses and Their Vectors: Epidemiology and Control)
►▼
Show Figures
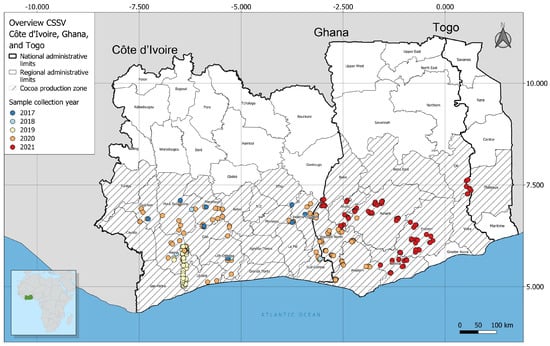
Figure 1
Open AccessReview
The Antiviral Activity of Interferon-Induced Transmembrane Proteins and Virus Evasion Strategies
by
Jingjing Wang, Yuhang Luo, Harshita Katiyar, Chen Liang and Qian Liu
Viruses 2024, 16(5), 734; https://doi.org/10.3390/v16050734 - 06 May 2024
Abstract
Interferons (IFNs) are antiviral cytokines that defend against viral infections by inducing the expression of interferon-stimulated genes (ISGs). Interferon-inducible transmembrane proteins (IFITMs) 1, 2, and 3 are crucial ISG products and members of the CD225 protein family. Compelling evidence shows that IFITMs restrict
[...] Read more.
Interferons (IFNs) are antiviral cytokines that defend against viral infections by inducing the expression of interferon-stimulated genes (ISGs). Interferon-inducible transmembrane proteins (IFITMs) 1, 2, and 3 are crucial ISG products and members of the CD225 protein family. Compelling evidence shows that IFITMs restrict the infection of many unrelated viruses by inhibiting the virus–cell membrane fusion at the virus entry step via the modulation of lipid composition and membrane properties. Meanwhile, viruses can evade IFITMs’ restrictions by either directly interacting with IFITMs via viral glycoproteins or by altering the native entry pathway. At the same time, cumulative evidence suggests context-dependent and multifaceted roles of IFITMs in modulating virus infections and cell signaling. Here, we review the diverse antiviral mechanisms of IFITMs, the viral antagonizing strategies, and the regulation of IFITM activity in host cells. The mechanisms behind the antiviral activity of IFITMs could aid the development of broad-spectrum antivirals and enhance preparedness for future pandemics.
Full article
(This article belongs to the Special Issue Interferon-Induced Transmembrane Proteins at the Intersection of Virus Infection and Immunity)
►▼
Show Figures
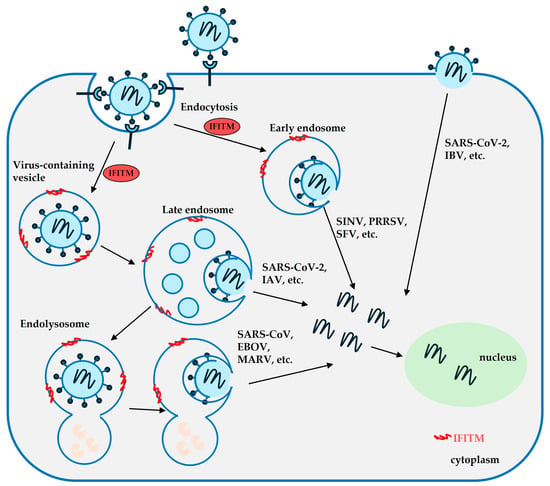
Figure 1
Open AccessEditorial
Pediatric Respiratory Viral Infection
by
Stacy L. S. Yam, Joan Marie Javillo Baguio and Renee W. Y. Chan
Viruses 2024, 16(5), 733; https://doi.org/10.3390/v16050733 - 06 May 2024
Abstract
Reflecting on this Special Issue dedicated to pediatric respiratory viruses, it is evident that the shadow cast by the global SARS-CoV-2 pandemic has profoundly impacted individuals of all ages and backgrounds, neonates and school-aged children being vulnerable cohorts resulting from the evolving immunological
[...] Read more.
Reflecting on this Special Issue dedicated to pediatric respiratory viruses, it is evident that the shadow cast by the global SARS-CoV-2 pandemic has profoundly impacted individuals of all ages and backgrounds, neonates and school-aged children being vulnerable cohorts resulting from the evolving immunological profiles and limited exposures to immunity-building experienced during this unprecedented era [...]
Full article
(This article belongs to the Special Issue Pediatric Respiratory Viral Infection)
Open AccessArticle
Regions of Bovine Adenovirus-3 Protein VII Involved in Interactions with Viral and Cellular Proteins
by
Shermila Kulanayake, Faryal Dar and Suresh K. Tikoo
Viruses 2024, 16(5), 732; https://doi.org/10.3390/v16050732 - 05 May 2024
Abstract
The L 1 region of bovine adenovirus (BAdV)-3 encodes a multifunctional protein named protein VII. Anti-protein VII sera detected a protein of 26 kDa in transfected or BAdV-3-infected cells, which localizes to nucleus and nucleolus of infected/transfected cells. Analysis of mutant protein VII
[...] Read more.
The L 1 region of bovine adenovirus (BAdV)-3 encodes a multifunctional protein named protein VII. Anti-protein VII sera detected a protein of 26 kDa in transfected or BAdV-3-infected cells, which localizes to nucleus and nucleolus of infected/transfected cells. Analysis of mutant protein VII identified four redundant overlapping nuclear/nucleolar localization signals as deletion of all four potential nuclear/nucleolar localization signals localizes protein VII predominantly to the cytoplasm. The nuclear import of protein VII appears to use importin α (α-1), importin-β (β-1) and transportin-3 nuclear transport receptors. In addition, different nuclear transport receptors also require part of protein VII outside nuclear localization sequences for efficient interaction. Proteomic analysis of protein complexes purified from recombinant BAdV-3 expressing protein VII containing Strep Tag II identified potential viral and cellular proteins interacting with protein VII. Here, we confirm that protein VII interacts with IVa2 and protein VIII in BAdV-3-infected cells. Moreover, amino acids 91–101 and 126–137, parts of non-conserved region of protein VII, are required for interaction with IVa2 and protein VIII, respectively.
Full article
(This article belongs to the Section Animal Viruses)
►▼
Show Figures

Figure 1
Open AccessBrief Report
Rapid Detection and Quick Characterization of African Swine Fever Virus Using the VolTRAX Automated Library Preparation Platform
by
Vivian O’Donnell, Jim L. Pierce, Oleg Osipenko, Lizhe Xu, Amy Berninger, Steven M. Lakin, Roger W. Barrette, Douglas P. Gladue and Bonto Faburay
Viruses 2024, 16(5), 731; https://doi.org/10.3390/v16050731 - 05 May 2024
Abstract
African swine fever virus (ASFV) is the causative agent of a severe and highly contagious viral disease affecting domestic and wild swine. The current ASFV pandemic strain has a high mortality rate, severely impacting pig production and, for countries suffering outbreaks, preventing the
[...] Read more.
African swine fever virus (ASFV) is the causative agent of a severe and highly contagious viral disease affecting domestic and wild swine. The current ASFV pandemic strain has a high mortality rate, severely impacting pig production and, for countries suffering outbreaks, preventing the export of their pig products for international trade. Early detection and diagnosis of ASFV is necessary to control new outbreaks before the disease spreads rapidly. One of the rate-limiting steps to identify ASFV by next-generation sequencing platforms is library preparation. Here, we investigated the capability of the Oxford Nanopore Technologies’ VolTRAX platform for automated DNA library preparation with downstream sequencing on Nanopore sequencing platforms as a proof-of-concept study to rapidly identify the strain of ASFV. Within minutes, DNA libraries prepared using VolTRAX generated near-full genome sequences of ASFV. Thus, our data highlight the use of the VolTRAX as a platform for automated library preparation, coupled with sequencing on the MinION Mk1C for field sequencing or GridION within a laboratory setting. These results suggest a proof-of-concept study that VolTRAX is an effective tool for library preparation that can be used for the rapid and real-time detection of ASFV.
Full article
(This article belongs to the Special Issue Early Diagnosis and Surveillance of Transboundary and Emerging Viral Diseases of Animals)
►▼
Show Figures

Figure 1
Open AccessArticle
Regional Variation of the CD4 and CD8 T Cell Epitopes Conserved in Circulating Dengue Viruses and Shared with Potential Vaccine Candidates
by
Yadya M. Chawla, Prashant Bajpai, Keshav Saini, Elluri Seetharami Reddy, Ashok Kumar Patel, Kaja Murali-Krishna and Anmol Chandele
Viruses 2024, 16(5), 730; https://doi.org/10.3390/v16050730 (registering DOI) - 05 May 2024
Abstract
As dengue expands globally and many vaccines are under trials, there is a growing recognition of the need for assessing T cell immunity in addition to assessing the functions of neutralizing antibodies during these endeavors. While several dengue-specific experimentally validated T cell epitopes
[...] Read more.
As dengue expands globally and many vaccines are under trials, there is a growing recognition of the need for assessing T cell immunity in addition to assessing the functions of neutralizing antibodies during these endeavors. While several dengue-specific experimentally validated T cell epitopes are known, less is understood about which of these epitopes are conserved among circulating dengue viruses and also shared by potential vaccine candidates. As India emerges as the epicenter of the dengue disease burden and vaccine trials commence in this region, we have here aligned known dengue specific T cell epitopes, reported from other parts of the world with published polyprotein sequences of 107 dengue virus isolates available from India. Of the 1305 CD4 and 584 CD8 epitopes, we found that 24% and 41%, respectively, were conserved universally, whereas 27% and 13% were absent in any viral isolates. With these data, we catalogued epitopes conserved in circulating dengue viruses from India and matched them with each of the six vaccine candidates under consideration (TV003, TDEN, DPIV, CYD-TDV, DENVax and TVDV). Similar analyses with viruses from Thailand, Brazil and Mexico revealed regional overlaps and variations in these patterns. Thus, our study provides detailed and nuanced insights into regional variation that should be considered for itemization of T cell responses during dengue natural infection and vaccine design, testing and evaluation.
Full article
(This article belongs to the Section Viral Immunology, Vaccines, and Antivirals)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Droplet Digital RT-PCR (dd RT-PCR) Detection of SARS-CoV-2 in Honey Bees and Honey Collected in Apiaries across the Campania Region
by
Andrea Mancusi, Yolande Thérèse Rose Proroga, Paola Maiolino, Raffaele Marrone, Claudia D’Emilio, Santa Girardi, Marica Egidio, Arianna Boni, Teresa Vicenza, Elisabetta Suffredini and Karen Power
Viruses 2024, 16(5), 729; https://doi.org/10.3390/v16050729 - 04 May 2024
Abstract
Coronaviruses (CoVs), a subfamily of Orthocoronavirinae, are viruses that sometimes present a zoonotic character. Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) is responsible for the recent outbreak of COVID-19, which, since its outbreak in 2019, has caused about 774,593,066 confirmed cases and 7,028,881 deaths.
[...] Read more.
Coronaviruses (CoVs), a subfamily of Orthocoronavirinae, are viruses that sometimes present a zoonotic character. Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) is responsible for the recent outbreak of COVID-19, which, since its outbreak in 2019, has caused about 774,593,066 confirmed cases and 7,028,881 deaths. Aereosols are the main route of transmission among people; however, viral droplets can contaminate surfaces and fomites as well as particulate matter (PM) in suspensions of natural and human origin. Honey bees are well known bioindicators of the presence of pollutants and PMs in the environment as they can collect a great variety of substances during their foraging activities. The aim of this study was to evaluate the possible role of honey bees as bioindicators of the prevalence SARS-CoV-2. In this regard, 91 samples of honey bees and 6 of honey were collected from different apiaries of Campania region (Southern Italy) in four time periods from September 2020 to June 2022 and were analyzed with Droplet Digital RT-PCR for SARS-CoV-2 target genes Orf1b and N. The screening revealed the presence of SARS-CoV-2 in 12/91 in honey bee samples and in 2/6 honey samples. These results suggest that honey bees could also be used as indicators of outbreaks of airborne pathogens such as SARS-CoV-2.
Full article
(This article belongs to the Special Issue Viruses in Mass-Reared Invertebrates 2nd Edition)
►▼
Show Figures
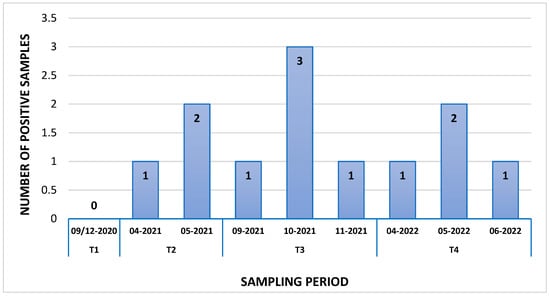
Figure 1
Open AccessArticle
The Disassociation of A3G-Related HIV-1 cDNA G-to-A Hypermutation to Viral Infectivity
by
Joanie Martin, Xin Chen, Xiangxu Jia, Qiujia Shao and Bindong Liu
Viruses 2024, 16(5), 728; https://doi.org/10.3390/v16050728 - 04 May 2024
Abstract
APOBEC3G (A3G) restricts HIV-1 replication primarily by reducing viral cDNA and inducing G-to-A hypermutations in viral cDNA. HIV-1 encodes virion infectivity factor (Vif) to counteract A3G primarily by excluding A3G viral encapsidation. Even though the Vif-induced exclusion is robust, studies suggest that A3G
[...] Read more.
APOBEC3G (A3G) restricts HIV-1 replication primarily by reducing viral cDNA and inducing G-to-A hypermutations in viral cDNA. HIV-1 encodes virion infectivity factor (Vif) to counteract A3G primarily by excluding A3G viral encapsidation. Even though the Vif-induced exclusion is robust, studies suggest that A3G is still detectable in the virion. The impact of encapsidated A3G in the HIV-1 replication is unclear. Using a highly sensitive next-generation sequencing (NGS)-based G-to-A hypermutation detecting assay, we found that wild-type HIV-1 produced from A3G-expressing T-cells induced higher G-to-A hypermutation frequency in viral cDNA than HIV-1 from non-A3G-expressing T-cells. Interestingly, although the virus produced from A3G-expressing T-cells induced higher hypermutation frequency, there was no significant difference in viral infectivity, revealing a disassociation of cDNA G-to-A hypermutation to viral infectivity. We also measured G-to-A hypermutation in the viral RNA genome. Surprisingly, our data showed that hypermutation frequency in the viral RNA genome was significantly lower than in the integrated DNA, suggesting a mechanism exists to preferentially select intact genomic RNA for viral packing. This study revealed a new insight into the mechanism of HIV-1 counteracting A3G antiviral function and might lay a foundation for new antiviral strategies.
Full article
(This article belongs to the Special Issue Cellular Mechanisms Regulating HIV Replication)
►▼
Show Figures
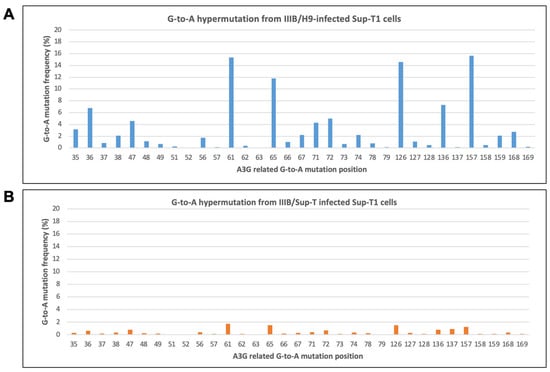
Figure 1
Open AccessReview
The Dynamic Relationship between Dengue Virus and the Human Cutaneous Innate Immune Response
by
Michelle M. Martí, Priscila M. S. Castanha and Simon M. Barratt-Boyes
Viruses 2024, 16(5), 727; https://doi.org/10.3390/v16050727 - 04 May 2024
Abstract
Dengue virus (DENV) is a continuing global threat that puts half of the world’s population at risk for infection. This mosquito-transmitted virus is endemic in over 100 countries. When a mosquito takes a bloodmeal, virus is deposited into the epidermal and dermal layers
[...] Read more.
Dengue virus (DENV) is a continuing global threat that puts half of the world’s population at risk for infection. This mosquito-transmitted virus is endemic in over 100 countries. When a mosquito takes a bloodmeal, virus is deposited into the epidermal and dermal layers of human skin, infecting a variety of permissive cells, including keratinocytes, Langerhans cells, macrophages, dermal dendritic cells, fibroblasts, and mast cells. In response to infection, the skin deploys an array of defense mechanisms to inhibit viral replication and prevent dissemination. Antimicrobial peptides, pattern recognition receptors, and cytokines induce a signaling cascade to increase transcription and translation of pro-inflammatory and antiviral genes. Paradoxically, this inflammatory environment recruits skin-resident mononuclear cells that become infected and migrate out of the skin, spreading virus throughout the host. The details of the viral–host interactions in the cutaneous microenvironment remain unclear, partly due to the limited body of research focusing on DENV in human skin. This review will summarize the functional role of human skin, the cutaneous innate immune response to DENV, the contribution of the arthropod vector, and the models used to study DENV interactions in the cutaneous environment.
Full article
(This article belongs to the Special Issue Innate and Adaptive Immunity to Cutaneous Virus Infection)
►▼
Show Figures

Figure 1
Open AccessBrief Report
Exploring Viral Genome Profile in Mpox Patients during the 2022 Outbreak, in a North-Eastern Centre of Italy
by
Michela Deiana, Denise Lavezzari, Antonio Mori, Silvia Accordini, Elena Pomari, Chiara Piubelli, Simone Malagò, Maddalena Cordioli, Niccolò Ronzoni, Andrea Angheben, Evelina Tacconelli, Maria Rosaria Capobianchi, Federico Giovanni Gobbi and Concetta Castilletti
Viruses 2024, 16(5), 726; https://doi.org/10.3390/v16050726 - 03 May 2024
Abstract
In 2022, an unprecedented outbreak of mpox raged in several nations. Sequences from the 2022 outbreak reveal a higher nucleotide substitution if compared with the estimated rate for orthopoxviruses. Recently, intra-lesion SNVs (single nucleotide variants) have been described, and these have been suggested
[...] Read more.
In 2022, an unprecedented outbreak of mpox raged in several nations. Sequences from the 2022 outbreak reveal a higher nucleotide substitution if compared with the estimated rate for orthopoxviruses. Recently, intra-lesion SNVs (single nucleotide variants) have been described, and these have been suggested as possible sources of genetic variation. Until now, it has not been clear if the presence of several SNVs could represents the result of local mutagenesis or a possible co-infection. We investigated the significance of SNVs through whole-genome sequencing analysis of four unrelated mpox cases. In addition to the known mutations harboured by the circulating strains of virus (MPXV), 7 novel mutations were identified, including SNVs located in genes that are involved in immune evasion mechanisms and/or viral fitness, six of these appeared to be APOBEC3-driven. Interestingly, three patients exhibited the coexistence of mutated and wild-type alleles for five non-synonymous variants. In addition, two patients, apparently unrelated, showed an analogous pattern for two novel mutations, albeit with divergent frequencies. The coexistence of mixed viral populations, harbouring non-synonymous mutations in patients, supports the hypothesis of possible co-infection. Additional investigations of larger clinical cohorts are essential to validating intra-patient viral genome heterogeneity and determining the possibility of co-presence events of slightly divergent MPXV strains.
Full article
(This article belongs to the Topic Human Monkeypox Research)
►▼
Show Figures
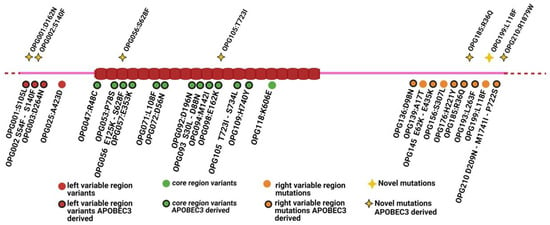
Figure 1
Open AccessArticle
CRISPR Screen Reveals PACT as a Pro-Viral Factor for Dengue Viral Replication
by
Shwetha Shivaprasad, Wenjie Qiao, Kuo-Feng Weng, Pavithra Umashankar, Jan E. Carette and Peter Sarnow
Viruses 2024, 16(5), 725; https://doi.org/10.3390/v16050725 - 03 May 2024
Abstract
The dengue virus is a single-stranded, positive-sense RNA virus that infects ~400 million people worldwide. Currently, there are no approved antivirals available. CRISPR-based screening methods have greatly accelerated the discovery of host factors that are essential for DENV infection and that can be
[...] Read more.
The dengue virus is a single-stranded, positive-sense RNA virus that infects ~400 million people worldwide. Currently, there are no approved antivirals available. CRISPR-based screening methods have greatly accelerated the discovery of host factors that are essential for DENV infection and that can be targeted in host-directed antiviral interventions. In the present study, we performed a focused CRISPR (Clustered Regularly Interspaced Palindromic Repeats) library screen to discover the key host factors that are essential for DENV infection in human Huh7 cells and identified the Protein Activator of Interferon-Induced Protein Kinase (PACT) as a novel pro-viral factor for DENV. PACT is a double-stranded RNA-binding protein generally known to activate antiviral responses in virus-infected cells and block viral replication. However, in our studies, we observed that PACT plays a pro-viral role in DENV infection and specifically promotes viral RNA replication. Knockout of PACT resulted in a significant decrease in DENV RNA and protein abundances in infected cells, which was rescued upon ectopic expression of full-length PACT. An analysis of global gene expression changes indicated that several ER-associated pro-viral genes such as ERN1, DDIT3, HERPUD1, and EIF2AK3 are not upregulated in DENV-infected PACT knockout cells as compared to infected wildtype cells. Thus, our study demonstrates a novel role for PACT in promoting DENV replication, possibly through modulating the expression of ER-associated pro-viral genes.
Full article
(This article belongs to the Special Issue Host Cell-Virus Interaction, 3rd Edition)
►▼
Show Figures
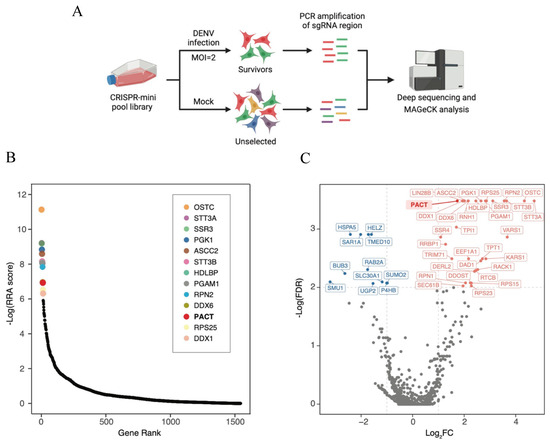
Figure 1
Open AccessReview
Hepatitis B Virus Infection: A Mini Review
by
Diana Asema Asandem, Selorm Philip Segbefia, Kwadwo Asamoah Kusi and Joseph Humphrey Kofi Bonney
Viruses 2024, 16(5), 724; https://doi.org/10.3390/v16050724 - 03 May 2024
Abstract
Hepatitis B and C viruses (HBV and HCV) are the leading causes of end-stage liver disease worldwide. Although there is a potent vaccine against HBV, many new infections are recorded annually, especially in poorly resourced places which have lax vaccination policies. Again, as
[...] Read more.
Hepatitis B and C viruses (HBV and HCV) are the leading causes of end-stage liver disease worldwide. Although there is a potent vaccine against HBV, many new infections are recorded annually, especially in poorly resourced places which have lax vaccination policies. Again, as HBV has no cure and chronic infection is lifelong, vaccines cannot help those already infected. Studies to thoroughly understand the HBV biology and pathogenesis are limited, leaving much yet to be understood about the genomic features and their role in establishing and maintaining infection. The current knowledge of the impact on disease progression and response to treatment, especially in hyperendemic regions, is inadequate. This calls for in-depth studies on viral biology, mainly for the purposes of coming up with better management strategies for infected people and more effective preventative measures for others. This information could also point us in the direction of a cure. Here, we discuss the progress made in understanding the genomic basis of viral activities leading to the complex interplay of the virus and the host, which determines the outcome of HBV infection as well as the impact of coinfections.
Full article
(This article belongs to the Special Issue Viral Hepatitis Coinfection)
Open AccessArticle
Long-Read Nanopore-Based Sequencing of Anelloviruses
by
Raghavendran Anantharam, Dylan Duchen, Andrea L. Cox, Winston Timp, David L. Thomas, Steven J. Clipman and Abraham J. Kandathil
Viruses 2024, 16(5), 723; https://doi.org/10.3390/v16050723 - 02 May 2024
Abstract
Routinely used metagenomic next-generation sequencing (mNGS) techniques often fail to detect low-level viremia (<104 copies/mL) and appear biased towards viruses with linear genomes. These limitations hinder the capacity to comprehensively characterize viral infections, such as those attributed to the Anelloviridae family. These
[...] Read more.
Routinely used metagenomic next-generation sequencing (mNGS) techniques often fail to detect low-level viremia (<104 copies/mL) and appear biased towards viruses with linear genomes. These limitations hinder the capacity to comprehensively characterize viral infections, such as those attributed to the Anelloviridae family. These near ubiquitous non-pathogenic components of the human virome have circular single-stranded DNA genomes that vary in size from 2.0 to 3.9 kb and exhibit high genetic diversity. Hence, species identification using short reads can be challenging. Here, we introduce a rolling circle amplification (RCA)-based metagenomic sequencing protocol tailored for circular single-stranded DNA genomes, utilizing the long-read Oxford Nanopore platform. The approach was assessed by sequencing anelloviruses in plasma drawn from people who inject drugs (PWID) in two geographically distinct cohorts. We detail the methodological adjustments implemented to overcome difficulties inherent in sequencing circular genomes and describe a computational pipeline focused on anellovirus detection. We assessed our protocol across various sample dilutions and successfully differentiated anellovirus sequences in conditions simulating mixed infections. This method provides a robust framework for the comprehensive characterization of circular viruses within the human virome using the Oxford Nanopore.
Full article
(This article belongs to the Special Issue Advancing Research of Anelloviruses)
►▼
Show Figures

Figure 1
Open AccessArticle
Gut Microbiome and Cytokine Profiles in Post-COVID Syndrome
by
Karakoz Mussabay, Samat Kozhakhmetov, Marat Dusmagambetov, Aitolkyn Mynzhanova, Madiyar Nurgaziyev, Zharkyn Jarmukhanov, Elizaveta Vinogradova, Aigul Dusmagambetova, Aiganym Daulbaeva, Laura Chulenbayeva, Ainur Tauekelova, Makhabbat Bekbossynova and Almagul Kushugulova
Viruses 2024, 16(5), 722; https://doi.org/10.3390/v16050722 - 02 May 2024
Abstract
Recent studies highlight the crucial role of the gut microbiome in post-infectious complications, especially in patients recovering from severe COVID-19. Our research aimed to explore the connection between gut microbiome changes and the cytokine profile of patients with post-COVID syndrome. Using 16S rRNA
[...] Read more.
Recent studies highlight the crucial role of the gut microbiome in post-infectious complications, especially in patients recovering from severe COVID-19. Our research aimed to explore the connection between gut microbiome changes and the cytokine profile of patients with post-COVID syndrome. Using 16S rRNA amplicon sequencing, we analyzed the composition of the gut microbiome in 60 COVID-19 patients over the course of one year. We also measured the levels of serum cytokines and chemokines using the Milliplex system. Our results showed that severe SARS-CoV-2 infection cases, especially those complicated by pneumonia, induce a pro-inflammatory microbial milieu with heightened presence of Bacteroides, Faecalibacterium, and Prevotella_9. Furthermore, we found that post-COVID syndrome is characterized by a cross-correlation of various cytokines and chemokines MDC, IL-1b, Fractalkine, TNFa, FGF-2, EGF, IL-1RA, IFN-a2, IL-10, sCD40L, IL-8, Eotaxin, IL-12p40, and MIP-1b as well as a shift in the gut microbiome towards a pro-inflammatory profile. At the functional level, our analysis revealed associations with post-COVID-19 in homolactic fermentation, pentose phosphate, NAD salvage, and flavin biosynthesis. These findings highlight the intricate interplay between the gut microbiota, their metabolites, and systemic cytokines in shaping post-COVID symptoms. Unraveling the gut microbiome’s role in post-infectious complications opens avenues for new treatments for those patients with prolonged symptoms.
Full article
(This article belongs to the Special Issue COVID-19 and Gastrointestinal Symptoms)
►▼
Show Figures

Figure 1
Open AccessArticle
Epigenetic Modifications of White Blood Cell DNA Caused by Transient Fetal Infection with Bovine Viral Diarrhea Virus
by
Hana Van Campen, Jeanette V. Bishop, Zella Brink, Terry E. Engle, Carolina L. Gonzalez-Berrios, Hanah M. Georges, Jessica N. Kincade, Dilyara A. Murtazina and Thomas R. Hansen
Viruses 2024, 16(5), 721; https://doi.org/10.3390/v16050721 - 01 May 2024
Abstract
Bovine viral diarrhea virus (BVDV) infections cause USD 1.5–2 billion in losses annually. Maternal BVDV after 150 days of gestation causes transient fetal infection (TI) in which the fetal immune response clears the virus. The impact of fetal TI BVDV infections on postnatal
[...] Read more.
Bovine viral diarrhea virus (BVDV) infections cause USD 1.5–2 billion in losses annually. Maternal BVDV after 150 days of gestation causes transient fetal infection (TI) in which the fetal immune response clears the virus. The impact of fetal TI BVDV infections on postnatal growth and white blood cell (WBC) methylome as an index of epigenetic modifications was examined by inoculating pregnant heifers with noncytopathic type 2 BVDV or media (sham-inoculated controls) on Day 175 of gestation to generate TI (n = 11) and control heifer calves (n = 12). Fetal infection in TI calves was confirmed by virus-neutralizing antibody titers at birth and control calves were seronegative. Both control and TI calves were negative for BVDV RNA in WBCs by RT-PCR. The mean weight of the TI calves was less than that of the controls (p < 0.05). DNA methyl seq analysis of WBC DNA demonstrated 2349 differentially methylated cytosines (p ≤ 0.05) including 1277 hypomethylated cytosines, 1072 hypermethylated cytosines, 84 differentially methylated regions based on CpGs in promoters, and 89 DMRs in islands of TI WBC DNA compared to controls. Fetal BVDV infection during late gestation resulted in epigenomic modifications predicted to affect fetal development and immune pathways, suggesting potential consequences for postnatal growth and health of TI cattle.
Full article
(This article belongs to the Special Issue Epigenetic Modifications in Viral Infections)
►▼
Show Figures
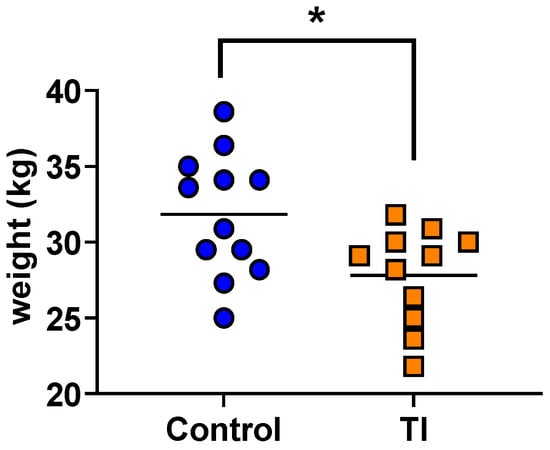
Figure 1
Open AccessArticle
Low-Level Viremia among Adults Living with HIV on Dolutegravir-Based First-Line Antiretroviral Therapy Is a Predictor of Virological Failure in Botswana
by
Ontlametse T. Bareng, Sikhulile Moyo, Mbatshi Mudanga, Kagiso Sebina, Catherine K. Koofhethile, Wonderful T. Choga, Natasha O. Moraka, Dorcas Maruapula, Irene Gobe, Modisa S. Motswaledi, Rosemary Musonda, Bornapate Nkomo, Dinah Ramaabya, Tony Chebani, Penny Makuruetsa, Joseph Makhema, Roger Shapiro, Shahin Lockman and Simani Gaseitsiwe
Viruses 2024, 16(5), 720; https://doi.org/10.3390/v16050720 - 01 May 2024
Abstract
We evaluated subsequent virologic outcomes in individuals experiencing low-level virem ia (LLV) on dolutegravir (DTG)-based first-line antiretroviral therapy (ART) in Botswana. We used a national dataset from 50,742 adults who initiated on DTG-based first-line ART from June 2016–December 2022. Individuals with at least
[...] Read more.
We evaluated subsequent virologic outcomes in individuals experiencing low-level virem ia (LLV) on dolutegravir (DTG)-based first-line antiretroviral therapy (ART) in Botswana. We used a national dataset from 50,742 adults who initiated on DTG-based first-line ART from June 2016–December 2022. Individuals with at least two viral load (VL) measurements post three months on DTG-based first-line ART were evaluated for first and subsequent episodes of LLV (VL:51–999 copies/mL). LLV was sub-categorized as low-LLV (51–200 copies/mL), medium-LLV (201–400 copies/mL) and high-LLV (401–999 copies/mL). The study outcome was virologic failure (VF) (VL ≥ 1000 copies/mL): virologic non-suppression defined as single-VF and confirmed-VF defined as two-consecutive VF measurements after an initial VL < 1000 copies/mL. Cox regression analysis identified predictive factors of subsequent VF. The prevalence of LLV was only statistically different at timepoints >6–12 (2.8%) and >12–24 (3.9%) (p-value < 0.01). LLV was strongly associated with both virologic non-suppression (adjusted hazards ratio [aHR] = 2.6; 95% CI: 2.2–3.3, p-value ≤ 0.001) and confirmed VF (aHR = 2.5; 95% CI: 2.4–2.7, p-value ≤ 0.001) compared to initially virally suppressed PLWH. High-LLV (HR = 3.3; 95% CI: 2.9–3.6) and persistent-LLV (HR = 6.6; 95% CI: 4.9–8.9) were associated with an increased hazard for virologic non-suppression than low-LLV and a single-LLV episode, respectively. In a national cohort of PLWH on DTG-based first-line ART, LLV > 400 copies/mL and persistent-LLV had a stronger association with VF. Frequent VL testing and adherence support are warranted for individuals with VL > 50 copies/mL.
Full article
(This article belongs to the Special Issue HIV Reservoirs, Latency, and the Factors Responsible)
►▼
Show Figures

Figure 1
Open AccessBrief Report
Seroprevalence of West Nile Virus in Tampa Bay Florida Patients Admitted to Hospital during 2020–2021 for Respiratory Symptoms
by
Emma C. Underwood, Iset M. Vera, Dylan Allen, Joshua Alvior, Marci O’Driscoll, Suzane Silbert, Kami Kim and Kelli L. Barr
Viruses 2024, 16(5), 719; https://doi.org/10.3390/v16050719 - 30 Apr 2024
Abstract
West Nile virus (WNV) is an arbovirus spread primarily by Culex mosquitoes, with humans being a dead-end host. WNV was introduced to Florida in 2001, with 467 confirmed cases since. It is estimated that 80 percent of cases are asymptomatic, with mild cases
[...] Read more.
West Nile virus (WNV) is an arbovirus spread primarily by Culex mosquitoes, with humans being a dead-end host. WNV was introduced to Florida in 2001, with 467 confirmed cases since. It is estimated that 80 percent of cases are asymptomatic, with mild cases presenting as a non-specific flu-like illness. Currently, detection of WNV in humans occurs primarily in healthcare settings via RT-PCR or CSF IgM when patients present with severe manifestations of disease including fever, meningitis, encephalitis, or acute flaccid paralysis. Given the short window of detectable viremia and requirement for CSF sampling, most WNV infections never receive an official diagnosis. This study utilized enzyme-linked immunosorbent assay (ELISA) to detect WNV IgG antibodies in 250 patient serum and plasma samples collected at Tampa General Hospital during 2020 and 2021. Plaque reduction neutralization tests were used to confirm ELISA results. Out of the 250 patients included in this study, 18.8% of them were IgG positive, consistent with previous WNV exposure. There was no relationship between WNV exposure and age or sex.
Full article
(This article belongs to the Section Human Virology and Viral Diseases)

Journal Menu
► ▼ Journal Menu-
- Viruses Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Diseases, Infectious Disease Reports, Pathogens, Viruses, TropicalMed
Human Monkeypox Research
Topic Editors: Shailendra K. Saxena, Ahmed Sayed Abdel-MoneimDeadline: 30 June 2024
Topic in
Biomedicines, JCM, Pathogens, Vaccines, Viruses
Discovery and Development of Monkeypox Disease Treatments
Topic Editors: Mohd Imran, Ali A. RabaanDeadline: 31 August 2024
Topic in
Brain Sciences, Clinics and Practice, COVID, Life, Vaccines, Viruses
Multifaceted Efforts from Basic Research to Clinical Practice in Controlling COVID-19 Disease
Topic Editors: Yih-Horng Shiao, Rashi OjhaDeadline: 30 September 2024

Conferences
Special Issues
Special Issue in
Viruses
Animal Coronaviruses: Infection, Prevention, and Antivirals
Guest Editor: Tomomi TakanoDeadline: 20 May 2024
Special Issue in
Viruses
Bacteriophages and Biofilms 2.0
Guest Editors: Zuzanna Drulis-Kawa, Tomasz OlszakDeadline: 31 May 2024
Special Issue in
Viruses
The Inflammasomes - Key Players in Antiviral Response
Guest Editors: Dong-Yan Jin, Tsan Sam XiaoDeadline: 15 June 2024
Special Issue in
Viruses
State-of-the-Art Virology Research in Australia
Guest Editors: Timothy Newsome, Barry SlobedmanDeadline: 30 June 2024
Topical Collections
Topical Collection in
Viruses
Poxviruses
Collection Editors: Giliane de Souza Trindade, Galileu Barbosa Costa, Flavio Guimaraes da Fonseca
Topical Collection in
Viruses
Phage Therapy
Collection Editors: Nina Chanishvili, Jean-Paul Pirnay, Mikael Skurnik
Topical Collection in
Viruses
Coronaviruses
Collection Editors: Luis Martinez-Sobrido, Fernando Almazan Toral
Topical Collection in
Viruses
SARS-CoV-2 and COVID-19
Collection Editors: Luis Martinez-Sobrido, Fernando Almazan Toral