-
 Methylene Blue Metabolic Therapy Restrains In Vivo Ovarian Tumor Growth
Methylene Blue Metabolic Therapy Restrains In Vivo Ovarian Tumor Growth -
 Gender-Specific Genetic Predisposition to Breast Cancer: BRCA Genes and Beyond
Gender-Specific Genetic Predisposition to Breast Cancer: BRCA Genes and Beyond -
 The Role of the Complement in Clear Cell Renal Carcinoma (ccRCC)—What Future Prospects Are There for Its Use in Clinical Practice?
The Role of the Complement in Clear Cell Renal Carcinoma (ccRCC)—What Future Prospects Are There for Its Use in Clinical Practice? -
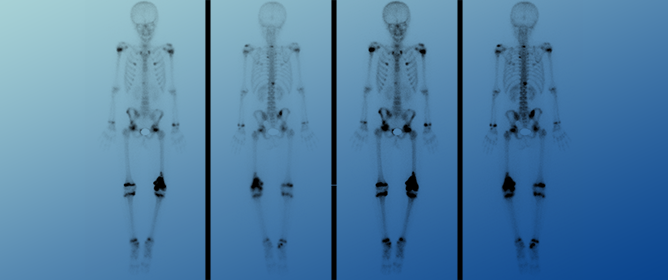 Primary Multi-Systemic Metastases in Osteosarcoma: Presentation, Treatment, and Survival of 83 Patients of the Cooperative Osteosarcoma Study Group
Primary Multi-Systemic Metastases in Osteosarcoma: Presentation, Treatment, and Survival of 83 Patients of the Cooperative Osteosarcoma Study Group -
 Addressing Genetic Tumor Heterogeneity, Post-Therapy Metastatic Spread, Cancer Repopulation, and Development of Acquired Tumor Cell Resistance
Addressing Genetic Tumor Heterogeneity, Post-Therapy Metastatic Spread, Cancer Repopulation, and Development of Acquired Tumor Cell Resistance
Journal Description
Cancers
Cancers
is a peer-reviewed, open access journal of oncology, published semimonthly online by MDPI. The Irish Association for Cancer Research (IACR), Spanish Association for Cancer Research (ASEICA), Biomedical Research Centre (CIBM), British Neuro-Oncology Society (BNOS) and Spanish Group for Cancer Immuno-Biotherapy (GÉTICA) are affiliated with Cancers and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Oncology) / CiteScore - Q1 (Oncology)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 17.9 days after submission; acceptance to publication is undertaken in 2.8 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Sections: published in 18 topical sections.
- Companion journals for Cancers include: Radiation and Onco.
Impact Factor:
5.2 (2022);
5-Year Impact Factor:
5.6 (2022)
Latest Articles
Relapse Prevention in Acute Myeloid Leukemia: The Role of Immunotherapy with Histamine Dihydrochloride and Low-Dose Interleukin-2
Cancers 2024, 16(10), 1824; https://doi.org/10.3390/cancers16101824 (registering DOI) - 10 May 2024
Abstract
The treatment and management of acute myeloid leukemia (AML) has improved in recent decennia by targeted therapy for subgroups of patients, expanded indications for allogeneic stem cell transplantation (allo-SCT) and surveillance of residual or arising leukemia. However, hematological relapse among patients who have
[...] Read more.
The treatment and management of acute myeloid leukemia (AML) has improved in recent decennia by targeted therapy for subgroups of patients, expanded indications for allogeneic stem cell transplantation (allo-SCT) and surveillance of residual or arising leukemia. However, hematological relapse among patients who have attained complete remission (CR) after the initial courses of chemotherapy remains a significant cause of morbidity and mortality. Here, we review an immunotherapeutic option using histamine dihydrochloride and low-dose interleukin-2 (HDC/LD-IL-2) for remission maintenance in AML. The treatment is approved in Europe in the post-consolidation phase to avoid relapse among patients in CR who are not candidates for upfront allo-SCT. We present aspects of the purported anti-leukemic mechanism of this regimen, including translation of preclinical results into the clinical setting, along with relapse prevention in subgroups of patients. We consider that HDC/LD-IL-2 is a conceivable option for younger adults, in particular patients with AML of normal karyotype and those with favorable responses to the initial chemotherapy. HDC/LD-IL-2 may form an emerging landscape of remission maintenance in AML.
Full article
(This article belongs to the Topic Myeloma and Leukemia-Challenges and Current Treatment Options)
Open AccessArticle
Frequency of Common and Uncommon BRAF Alterations among Colorectal and Non-Colorectal Gastrointestinal Malignancies
by
Amit Mahipal, Michael H. Storandt, Emily A. Teslow, Ellen Jaeger, Melissa C. Stoppler, Zhaohui Jin and Sakti Chakrabarti
Cancers 2024, 16(10), 1823; https://doi.org/10.3390/cancers16101823 (registering DOI) - 10 May 2024
Abstract
Background: The predictive and prognostic role of BRAF alterations has been evaluated in colorectal cancer (CRC); however, BRAF alterations have not been fully characterized in non-CRC gastrointestinal (GI) malignancies. In the present study, we report the frequency and spectrum of BRAF alterations among
[...] Read more.
Background: The predictive and prognostic role of BRAF alterations has been evaluated in colorectal cancer (CRC); however, BRAF alterations have not been fully characterized in non-CRC gastrointestinal (GI) malignancies. In the present study, we report the frequency and spectrum of BRAF alterations among patients with non-CRC GI malignancies. Methods: Patients with CRC and non-CRC GI malignancies who underwent somatic tumor profiling via a tissue-based or liquid-based assay were included in this study. Gain-of-function BRAF alterations were defined as pathogenic/likely pathogenic somatic short variants (SVs), copy number amplifications ≥8, or fusions (RNA or DNA). Results: Among 51,560 patients with somatic profiling, 40% had CRC and 60% had non-CRC GI malignancies. BRAF GOF alterations were seen more frequently in CRC (8.9%) compared to non-CRC GI malignancies (2.2%) (p < 0.001). Non-CRC GI malignancies with the highest prevalence of BRAF GOF alterations were bile duct cancers (4.1%) and small intestine cancers (4.0%). Among BRAF GOF alterations, class II (28% vs. 6.8%, p < 0.001) and class III (23% vs. 14%, p < 0.001) were more common in non-CRC GI malignancies. Among class II alterations, rates of BRAF amplifications (3.1% vs. 0.3%, p < 0.001) and BRAF fusions (12% vs. 2.2%, p < 0.001) were higher in non-CRC GI malignancies compared to CRC. Conclusions: Non-CRC GI malignancies demonstrate a distinct BRAF alteration profile compared to CRC, with a higher frequency of class II and III mutations, and more specifically, a higher incidence of BRAF fusions. Future studies should evaluate clinical implications for the management of non-CRC GI patients with BRAF alterations, especially BRAF fusions.
Full article
(This article belongs to the Special Issue Clinical and Pathologic Response to Therapy in Gastrointestinal Oncology)
►▼
Show Figures

Figure 1
Open AccessArticle
Autonomous Tumor Signature Extraction Applied to Spatially Registered Bi-Parametric MRI to Predict Prostate Tumor Aggressiveness: A Pilot Study
by
Rulon Mayer, Baris Turkbey and Charles B. Simone II
Cancers 2024, 16(10), 1822; https://doi.org/10.3390/cancers16101822 (registering DOI) - 10 May 2024
Abstract
Background: Accurate, reliable, non-invasive assessment of patients diagnosed with prostate cancer is essential for proper disease management. Quantitative assessment of multi-parametric MRI, such as through artificial intelligence or spectral/statistical approaches, can provide a non-invasive objective determination of the prostate tumor aggressiveness without side
[...] Read more.
Background: Accurate, reliable, non-invasive assessment of patients diagnosed with prostate cancer is essential for proper disease management. Quantitative assessment of multi-parametric MRI, such as through artificial intelligence or spectral/statistical approaches, can provide a non-invasive objective determination of the prostate tumor aggressiveness without side effects or potential poor sampling from needle biopsy or overdiagnosis from prostate serum antigen measurements. To simplify and expedite prostate tumor evaluation, this study examined the efficacy of autonomously extracting tumor spectral signatures for spectral/statistical algorithms for spatially registered bi-parametric MRI. Methods: Spatially registered hypercubes were digitally constructed by resizing, translating, and cropping from the image sequences (Apparent Diffusion Coefficient (ADC), High B-value, T2) from 42 consecutive patients in the bi-parametric MRI PI-CAI dataset. Prostate cancer blobs exceeded a threshold applied to the registered set from normalizing the registered set into an image that maximizes High B-value, but minimizes the ADC and T2 images, appearing “green” in the color composite. Clinically significant blobs were selected based on size, average normalized green value, sliding window statistics within a blob, and position within the hypercube. The center of mass and maximized sliding window statistics within the blobs identified voxels associated with tumor signatures. We used correlation coefficients (R) and p-values, to evaluate the linear regression fits of the z-score and SCR (with processed covariance matrix) to tumor aggressiveness, as well as Area Under the Curves (AUC) for Receiver Operator Curves (ROC) from logistic probability fits to clinically significant prostate cancer. Results: The highest R (R > 0.45), AUC (>0.90), and lowest p-values (<0.01) were achieved using z-score and modified registration applied to the covariance matrix and tumor signatures selected from the “greenest” parts from the selected blob. Conclusions: The first autonomous tumor signature applied to spatially registered bi-parametric MRI shows promise for determining prostate tumor aggressiveness.
Full article
(This article belongs to the Section Methods and Technologies Development)
►▼
Show Figures

Figure 1
Open AccessArticle
Poor Mobilization-Associated Factors in Autologous Hematopoietic Stem Cell Harvest
by
Won Kee Ahn, Hyun-Jun Nam, Hae Won Lee, Seungmin Hahn, Jung Woo Han, Chuhl Joo Lyu, Sinyoung Kim, Soon Sung Kwon, Haerim Chung, Jin Seok Kim, June-Won Cheong and Kyung-A Lee
Cancers 2024, 16(10), 1821; https://doi.org/10.3390/cancers16101821 - 10 May 2024
Abstract
Peripheral blood stem cell transplantation (PBSCT) is an important therapeutic measure for both hematologic and non-hematologic diseases. For PBSCT to be successful, sufficient CD34+ cells need to be mobilized and harvested. Although risk factors associated with poor mobilization in patients with hematologic
[...] Read more.
Peripheral blood stem cell transplantation (PBSCT) is an important therapeutic measure for both hematologic and non-hematologic diseases. For PBSCT to be successful, sufficient CD34+ cells need to be mobilized and harvested. Although risk factors associated with poor mobilization in patients with hematologic diseases have been reported, studies of patients with non-hematologic diseases and those receiving plerixafor are rare. To identify factors associated with poor mobilization, data from autologous PBSC harvest (PBSCH) in 491 patients were retrospectively collected and analyzed. A multivariate analysis revealed that in patients with a hematologic disease, an age older than 60 years (odds ratio [OR] 1.655, 95% confidence interval [CI] 1.049–2.611, p = 0.008), the use of myelotoxic agents (OR 4.384, 95% CI 2.681–7.168, p < 0.001), and a low platelet count (OR 2.106, 95% CI 1.205–3.682, p = 0.009) were associated with poor mobilization. In patients with non-hematologic diseases, a history of radiation on the pelvis/spine was the sole associated factor (OR 12.200, 95% CI 1.934–76.956, p = 0.008). Among the group of patients who received plerixafor, poor mobilization was observed in 19 patients (19/134, 14.2%) and a difference in the mobilization regimen was noted among the good mobilization group. These results show that the risk factors for poor mobilization in patients with non-hematologic diseases and those receiving plerixafor differ from those in patients with hematologic diseases; as such, non-hematologic patients require special consideration to enable successful PBSCH.
Full article
(This article belongs to the Section Clinical Research of Cancer)
Open AccessReview
Updates on the Management of Colorectal Cancer in Older Adults
by
Conor D. J. O’Donnell, Joleen Hubbard and Zhaohui Jin
Cancers 2024, 16(10), 1820; https://doi.org/10.3390/cancers16101820 - 10 May 2024
Abstract
Colorectal cancer (CRC) poses a significant global health challenge. Notably, the risk of CRC escalates with age, with the majority of cases occurring in those over the age of 65. Despite recent progress in tailoring treatments for early and advanced CRC, there is
[...] Read more.
Colorectal cancer (CRC) poses a significant global health challenge. Notably, the risk of CRC escalates with age, with the majority of cases occurring in those over the age of 65. Despite recent progress in tailoring treatments for early and advanced CRC, there is a lack of prospective data to guide the management of older patients, who are frequently underrepresented in clinical trials. This article reviews the contemporary landscape of managing older individuals with CRC, highlighting recent advancements and persisting challenges. The role of comprehensive geriatric assessment is explored. Opportunities for treatment escalation/de-escalation, with consideration of the older adult’s fitness level. are reviewed in the neoadjuvant, surgical, adjuvant, and metastatic settings of colon and rectal cancers. Immunotherapy is shown to be an effective treatment option in older adults who have CRC with microsatellite instability. Promising new technologies such as circulating tumor DNA and recent phase III trials adding later-line systemic therapy options are discussed. Clinical recommendations based on the data available are summarized. We conclude that deliberate efforts to include older individuals in future colorectal cancer trials are essential to better guide the management of these patients in this rapidly evolving field.
Full article
(This article belongs to the Topic Advances in Colorectal Cancer Therapy)
Open AccessReview
FLT3-Mutated Leukemic Stem Cells: Mechanisms of Resistance and New Therapeutic Targets
by
Debora Capelli
Cancers 2024, 16(10), 1819; https://doi.org/10.3390/cancers16101819 - 10 May 2024
Abstract
Despite the availability of target drugs in the first and second line, only 30% of FLT3mut AMLs are cured. Among the multiple mechanisms of resistance, those of FLT3mut LSC are the most difficult to eradicate because of their metabolic and genomic characteristics. Reactivation
[...] Read more.
Despite the availability of target drugs in the first and second line, only 30% of FLT3mut AMLs are cured. Among the multiple mechanisms of resistance, those of FLT3mut LSC are the most difficult to eradicate because of their metabolic and genomic characteristics. Reactivation of glycogen synthesis, inhibition of the RAS/MAPK pathway, and degradation of FLT3 may be potential aids to fight the resistance of LSC to FLT3i. LSC is also characterized by the expression of a CD34+/CD25+/CD123+/CD99+ immunophenotype. The receptor and ligand of FLT3, the natural killer group 2 member D ligand (NKGD2L), and CD123 are some of the targets of chimeric antigen receptor T cells (CAR-T), bispecific T-cell engager molecules (BiTEs), CAR-NK and nanoparticles recently designed and reported here. The combination of these new therapeutic options, hopefully in a minimal residual disease (MRD)-driven approach, could provide the future answer to the challenge of treating FLT3mut AML.
Full article
(This article belongs to the Special Issue The New Challenge of Curing AML: Analysis of Mechanisms of Resistance of Target Drugs and Future Perspectives)
►▼
Show Figures

Figure 1
Open AccessArticle
LKB1 Loss Correlates with STING Loss and, in Cooperation with β-Catenin Membranous Loss, Indicates Poor Prognosis in Patients with Operable Non-Small Cell Lung Cancer
by
Eleni D. Lagoudaki, Anastasios V. Koutsopoulos, Maria Sfakianaki, Chara Papadaki, Georgios C. Manikis, Alexandra Voutsina, Maria Trypaki, Eleftheria Tsakalaki, Georgia Fiolitaki, Dora Hatzidaki, Emmanuel Yiachnakis, Dimitra Koumaki, Dimitrios Mavroudis, Maria Tzardi, Efstathios N. Stathopoulos, Kostas Marias, Vassilis Georgoulias and John Souglakos
Cancers 2024, 16(10), 1818; https://doi.org/10.3390/cancers16101818 - 10 May 2024
Abstract
To investigate the incidence and prognostically significant correlations and cooperations of LKB1 loss of expression in non-small cell lung cancer (NSCLC), surgical specimens from 188 metastatic and 60 non-metastatic operable stage I-IIIA NSCLC patients were analyzed to evaluate their expression of LKB1 and
[...] Read more.
To investigate the incidence and prognostically significant correlations and cooperations of LKB1 loss of expression in non-small cell lung cancer (NSCLC), surgical specimens from 188 metastatic and 60 non-metastatic operable stage I-IIIA NSCLC patients were analyzed to evaluate their expression of LKB1 and pAMPK proteins in relation to various processes. The investigated factors included antitumor immunity response regulators STING and PD-L1; pro-angiogenic, EMT and cell cycle targets, as well as metastasis-related (VEGFC, PDGFRα, PDGFRβ, p53, p16, Cyclin D1, ZEB1, CD24) targets; and cell adhesion (β-catenin) molecules. The protein expression levels were evaluated via immunohistochemistry; the RNA levels of LKB1 and NEDD9 were evaluated via PCR, while KRAS exon 2 and BRAFV600E mutations were evaluated by Sanger sequencing. Overall, loss of LKB1 protein expression was observed in 21% (51/248) patients and correlated significantly with histotype (p < 0.001), KRAS mutations (p < 0.001), KC status (concomitant KRAS mutation and p16 downregulation) (p < 0.001), STING loss (p < 0.001), and high CD24 expression (p < 0.001). STING loss also correlated significantly with loss of LKB1 expression in the metastatic setting both overall (p = 0.014) and in lung adenocarcinomas (LUACs) (p = 0.005). Additionally, LKB1 loss correlated significantly with a lack of or low β-catenin membranous expression exclusively in LUACs, both independently of the metastatic status (p = 0.019) and in the metastatic setting (p = 0.007). Patients with tumors yielding LKB1 loss and concomitant nonexistent or low β-catenin membrane expression experienced significantly inferior median overall survival of 20.50 vs. 52.99 months; p < 0.001 as well as significantly greater risk of death (HR: 3.32, 95% c.i.: 1.71–6.43; p <0.001). Our findings underscore the impact of the synergy of LKB1 with STING and β-catenin in NSCLC, in prognosis.
Full article
(This article belongs to the Special Issue Advancements in Lung Cancer Surgical Treatment and Prognosis)
►▼
Show Figures

Figure 1
Open AccessArticle
Ki67 Tumor Expression Predicts Treatment Benefit Achieved by Macroscopic Radical Lung-Preserving Surgery in Pleural Mesothelioma—A Retrospective Multicenter Analysis
by
Sarah Hintermair, Stephanie Iser, Alexander Varga, Melanie Biesinger, Tomas Bohanes, Ali Celik, Muhammet Sayan, Aykut Kankoç, Nalan Akyurek, Betul Öğüt, Elisabeth Stubenberger and Bahil Ghanim
Cancers 2024, 16(10), 1817; https://doi.org/10.3390/cancers16101817 - 10 May 2024
Abstract
Pleural mesothelioma (PM), linked to asbestos-induced inflammation, carries a poor prognosis. Therapy ranges from therapy limitation to aggressive multimodality treatment. Given the uncertainty about treatment benefits for patients, this study aimed to assess the role of Ki67 as a prognostic and predictive parameter
[...] Read more.
Pleural mesothelioma (PM), linked to asbestos-induced inflammation, carries a poor prognosis. Therapy ranges from therapy limitation to aggressive multimodality treatment. Given the uncertainty about treatment benefits for patients, this study aimed to assess the role of Ki67 as a prognostic and predictive parameter in PM. Ki67 was measured in the specimens of 70 PM patients (17 female, 53 male) from two centers and correlated to overall survival (OS) and therapy outcome. The median OS was 16.1 months. The level of Ki67 expression was divided into low (≤15%) and high (>15%). A low value of Ki67 expression was associated with a longer OS (Ki67 ≤ 15%: 31.2 (95% CI 6.5–55.8) months vs. Ki67 > 15%: 11.1 (95% CI 7.7–14.6) months, p = 0.012). The 5-year survival represents 22% in the low Ki67 expression group, in contrast to 5% in the high Ki67 expression group. We found a significant interaction term of Ki67 with multimodality treatment (p = 0.031) translating to an OS of 48.1 months in the low expression Ki67 group compared to 24.3 months in the high Ki67 expression group when receiving surgery within multimodality therapy. Therefore, Ki67 stands out as a validated prognostic and, most importantly, novel predictive biomarker for treatment benefits, particularly regarding surgery within multimodality therapy.
Full article
(This article belongs to the Special Issue Recent Research on Mesothelioma)
►▼
Show Figures

Figure 1
Open AccessArticle
Extended Genotyping to Stratify the Risk of CIN2+ in Women with Persistent HPV Infection, Negative Cytology and Type 3 Transformation Zone
by
Maria Teresa Bruno, Gaetano Valenti, Antonino Giovanni Cavallaro, Ilenia Palermo, Tiziana Aiello, Jessica Farina, Marco Marzio Panella and Liliana Mereu
Cancers 2024, 16(10), 1816; https://doi.org/10.3390/cancers16101816 - 10 May 2024
Abstract
Persistent human papillomavirus (HPV) infection is recognized as a major risk factor for cervical cancer. Women with persistent HPV and negative cytology are at greater risk of CIN2+ than women with negative infection. The diagnosis becomes more complicated when the woman has a
[...] Read more.
Persistent human papillomavirus (HPV) infection is recognized as a major risk factor for cervical cancer. Women with persistent HPV and negative cytology are at greater risk of CIN2+ than women with negative infection. The diagnosis becomes more complicated when the woman has a type 3 transformation zone at colposcopy. The aim of this study was to determine the prevalence of CIN2+ in women with persistent HPV, negative cytology and TZ3; how to stratify the risk of CIN2+; and what the best diagnostic strategy is, given TZ3. Methods: In a multicenter retrospective cohort study, we enrolled women with negative cytology and TZ3 among the 213 women referred for colposcopy for persistent HPV. The average age of the women was 53 years; in particular, 83% were postmenopausal women. In the presence of a TZ3, the entire transformation zone cannot be explored, making colposcopy and targeted biopsy useless and inadequate, with great risks of underdiagnosis or missed diagnosis. Women with TZ3 underwent diagnostic LEEP to ensure correct diagnoses. Results: The study highlighted 19% (16/84) of CIN2+ lesions, a higher frequency of non-HPV 16/18 genotypes (76.2%), and 50% of CIN2+ lesions being due to non-HPV 16/18 genotypes. Furthermore, more than half of the women (80.9%) had normal histopathological results in the LEEP sample. Conclusion. Women with viral persistence, negative cytology, and TZ3 have a 19% risk of CIN2+; genotyping helps stratify risk, but extensive genotyping is necessary instead of partial genotyping (16/18), referring to a population of women over 50 years old in which the prevalence of genotypes 16,18 decreases and the prevalence of other genotypes increases; diagnostic LEEP is excessive (only 16 cases of CIN2+ out of 48 cases treated), even though 83% of women had viral clearance after LEEP; p16/Ki67 double staining could be a potential risk marker, which would only highlight women at risk of CIN2+ to undergo LEEP. To individualize the diagnostic workup and treatment and minimize the risk of under diagnosis and overtreatment, future studies should explore the use of extended genotyping and new biomarkers for individual risk stratification.
Full article
(This article belongs to the Section Cancer Causes, Screening and Diagnosis)
►▼
Show Figures

Figure 1
Open AccessArticle
Magnetic-Resonance-Imaging-Guided Cryoablation for Solitary-Biopsy-Proven Renal Cell Carcinoma: A Tertiary Cancer Center Experience
by
Mohamed E. Abdelsalam, Nabeel Mecci, Ahmed Awad, Roland L. Bassett, Bruno C. Odisio, Peiman Habibollahi, Thomas Lu, David Irwin, Jose A. Karam, Surena F. Matin and Kamran Ahrar
Cancers 2024, 16(10), 1815; https://doi.org/10.3390/cancers16101815 - 10 May 2024
Abstract
Background: Our purpose is to evaluate the long-term oncologic efficacy and survival rates of MRI-guided cryoablation for patients with biopsy-proven cT1a renal cell carcinoma (RCC). Materials and Methods: We retrospectively reviewed our renal ablation database between January 2007 and June 2021 and only
[...] Read more.
Background: Our purpose is to evaluate the long-term oncologic efficacy and survival rates of MRI-guided cryoablation for patients with biopsy-proven cT1a renal cell carcinoma (RCC). Materials and Methods: We retrospectively reviewed our renal ablation database between January 2007 and June 2021 and only included patients with solitary-biopsy-proven cT1a RCC (≤4 cm) who underwent MRI-guided cryoablation. We excluded patients with genetic syndromes, bilateral RCC, recurrent RCC or benign lesions, those without pathologically proven RCC lesions and patients who underwent radiofrequency ablation or CT-guided cryoablation. For each patient, we collected the following: age, sex, lesion size, right- or left-sided, pathology, ablation zone tumor recurrence, development of new tumor in the kidney other than ablation zone, development of metastatic disease, patient alive or not, date and cause of death. We used the Kaplan and Meier product limit estimator to estimate the survival outcomes. Results: Twenty-nine patients (median age 70 years) met our inclusion criteria. Twenty-nine MRI-guided cryoablation procedures were performed for twenty-nine tumor lesions with a median size of 2.2 cm. A Clavien–Dindo grade III complication developed in one patient (3.4%). Clear cell RCC was the most reported histology (n = 19). The median follow up was 4.5 years. No tumor recurrence or metastatic disease developed in any of the patients. Two patients developed new renal lesions separate from the ablation zone. The 5- and 10-year OS were 72% and 55.6%, respectively. The 5- and 10-year DFS were 90.5% and the 5-year and 10-year LRFS, MFS and CSS were all 100%. Conclusions: MRI-guided cryoablation is a safe treatment with a low complication rate. Long-term follow-up data revealed long-standing oncologic control.
Full article
(This article belongs to the Special Issue Genitourinary Malignancies)
►▼
Show Figures

Figure 1
Open AccessReview
Pediatric Diffuse Midline Glioma H3K27-Altered: From Developmental Origins to Therapeutic Challenges
by
Manuela Mandorino, Ahana Maitra, Domenico Armenise, Olga Maria Baldelli, Morena Miciaccia, Savina Ferorelli, Maria Grazia Perrone and Antonio Scilimati
Cancers 2024, 16(10), 1814; https://doi.org/10.3390/cancers16101814 - 10 May 2024
Abstract
Diffuse intrinsic pontine glioma (DIPG), now referred to as diffuse midline glioma (DMG), is a highly aggressive pediatric cancer primarily affecting children aged 4 to 9 years old. Despite the research and clinical trials conducted to identify a possible treatment for DIPG, no
[...] Read more.
Diffuse intrinsic pontine glioma (DIPG), now referred to as diffuse midline glioma (DMG), is a highly aggressive pediatric cancer primarily affecting children aged 4 to 9 years old. Despite the research and clinical trials conducted to identify a possible treatment for DIPG, no effective drug is currently available. These tumors often affect deep midline brain structures in young children, suggesting a connection to early brain development’s epigenetic regulation targets, possibly affecting neural progenitor functions and differentiation. The H3K27M mutation is a known DIPG trigger, but the exact mechanisms beyond epigenetic regulation remain unclear. After thoroughly examining the available literature, we found that over 85% of DIPG tumors contain a somatic missense mutation, K27M, in genes encoding histone H3.3 and H3.1, leading to abnormal gene expression that drives tumor growth and spread. This mutation impacts crucial brain development processes, including the epithelial–mesenchymal transition (EMT) pathway, and may explain differences between H3K27M and non-K27M pediatric gliomas. Effects on stem cells show increased proliferation and disrupted differentiation. The genomic organization of H3 gene family members in the developing brain has revealed variations in their expression patterns. All these observations suggest a need for global efforts to understand developmental origins and potential treatments.
Full article
(This article belongs to the Topic From Basic Research to a Clinical Perspective in Oncology)
►▼
Show Figures

Figure 1
Open AccessArticle
Resection Ratios and Tumor Eccentricity in Breast-Conserving Surgery Specimens for Surgical Accuracy Assessment
by
Dinusha Veluponnar, Behdad Dashtbozorg, Marcos Da Silva Guimaraes, Marie-Jeanne T. F. D. Vrancken Peeters, Lisanne L. de Boer and Theo J. M. Ruers
Cancers 2024, 16(10), 1813; https://doi.org/10.3390/cancers16101813 - 9 May 2024
Abstract
This study aims to evaluate several defined specimen parameters that would allow to determine the surgical accuracy of breast-conserving surgeries (BCS) in a representative population of patients. These specimen parameters could be used to compare surgical accuracy when using novel technologies for intra-operative
[...] Read more.
This study aims to evaluate several defined specimen parameters that would allow to determine the surgical accuracy of breast-conserving surgeries (BCS) in a representative population of patients. These specimen parameters could be used to compare surgical accuracy when using novel technologies for intra-operative BCS guidance in the future. Different specimen parameters were determined among 100 BCS patients, including the ratio of specimen volume to tumor volume (resection ratio) with different optimal margin widths (0 mm, 1 mm, 2 mm, and 10 mm). Furthermore, the tumor eccentricity [maximum tumor-margin distance − minimum tumor-margin distance] and the relative tumor eccentricity [tumor eccentricity ÷ pathological tumor diameter] were determined. Different patient subgroups were compared using Wilcoxon rank sum tests. When using a surgical margin width of 0 mm, 1 mm, 2 mm, and 10 mm, on average, 19.16 (IQR 44.36), 9.94 (IQR 18.09), 6.06 (IQR 9.69) and 1.35 (IQR 1.78) times the ideal resection volume was excised, respectively. The median tumor eccentricity among the entire patient population was 11.29 mm (SD = 3.99) and the median relative tumor eccentricity was 0.66 (SD = 2.22). Resection ratios based on different optimal margin widths (0 mm, 1 mm, 2 mm, and 10 mm) and the (relative) tumor eccentricity could be valuable outcome measures to evaluate the surgical accuracy of novel technologies for intra-operative BCS guidance.
Full article
(This article belongs to the Special Issue Recent Advances and Challenges in Breast Cancer Surgery)
►▼
Show Figures

Figure 1
Open AccessArticle
Zebrafish Avatars: Toward Functional Precision Medicine in Low-Grade Serous Ovarian Cancer
by
Charlotte Fieuws, Jan Willem Bek, Bram Parton, Elyne De Neef, Olivier De Wever, Milena Hoorne, Marta F. Estrada, Jo Van Dorpe, Hannelore Denys, Koen Van de Vijver and Kathleen B. M. Claes
Cancers 2024, 16(10), 1812; https://doi.org/10.3390/cancers16101812 - 9 May 2024
Abstract
Ovarian cancer (OC) is an umbrella term for cancerous malignancies affecting the ovaries, yet treatment options for all subtypes are predominantly derived from high-grade serous ovarian cancer, the largest subgroup. The concept of "functional precision medicine" involves gaining personalized insights on therapy choice,
[...] Read more.
Ovarian cancer (OC) is an umbrella term for cancerous malignancies affecting the ovaries, yet treatment options for all subtypes are predominantly derived from high-grade serous ovarian cancer, the largest subgroup. The concept of "functional precision medicine" involves gaining personalized insights on therapy choice, based on direct exposure of patient tissues to drugs. This especially holds promise for rare subtypes like low-grade serous ovarian cancer (LGSOC). This study aims to establish an in vivo model for LGSOC using zebrafish embryos, comparing treatment responses previously observed in mouse PDX models, cell lines and 3D tumor models. To address this goal, a well-characterized patient-derived LGSOC cell line with the KRAS mutation c.35 G > T (p.(Gly12Val)) was used. Fluorescently labeled tumor cells were injected into the perivitelline space of 2 days’ post-fertilization zebrafish embryos. At 1 day post-injection, xenografts were assessed for tumor size, followed by random allocation into treatment groups with trametinib, luminespib and trametinib + luminespib. Subsequently, xenografts were euthanized and analyzed for apoptosis and proliferation by confocal microscopy. Tumor cells formed compact tumor masses (n = 84) in vivo, with clear Ki67 staining, indicating proliferation. Zebrafish xenografts exhibited sensitivity to trametinib and luminespib, individually or combined, within a two-week period, establishing them as a rapid and complementary tool to existing in vitro and in vivo models for evaluating targeted therapies in LGSOC.
Full article
(This article belongs to the Special Issue Advances in Ovarian Cancer Research and Treatment)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Impact of Nutritional Status on Neutrophil-to-Lymphocyte Ratio as a Predictor of Efficacy and Adverse Events of Immune Check-Point Inhibitors
by
Masahiko Sue, Yasuto Takeuchi, Shoichiro Hirata, Akinobu Takaki and Motoyuki Otsuka
Cancers 2024, 16(10), 1811; https://doi.org/10.3390/cancers16101811 - 9 May 2024
Abstract
The neutrophil -to-lymphocyte ratio (NLR) is useful for predicting the effectiveness of treatment with immune checkpoint inhibitors (ICIs) and immune-related adverse events (irAEs). Because a growing body of evidence has recently shown that the number of lymphocytes that comprise NLR fluctuates according to
[...] Read more.
The neutrophil -to-lymphocyte ratio (NLR) is useful for predicting the effectiveness of treatment with immune checkpoint inhibitors (ICIs) and immune-related adverse events (irAEs). Because a growing body of evidence has recently shown that the number of lymphocytes that comprise NLR fluctuates according to nutritional status, this study examined whether the usefulness of NLR varies in ICI treatment due to changes in nutritional status. A retrospective analysis was performed on 1234 patients who received ICI treatment for malignant tumors at our hospital. Progression-free survival (PFS) was significantly prolonged in patients with NLR < 4. Multivariate analysis revealed that the factors associated with the occurrence of irAE were NLR < 4 and the use of ipilimumab. However, when limited to cases with serum albumin levels <3.8 g/dL, lymphocyte counts significantly decreased, and the associations between NLR and PFS and between NLR and irAE occurrence disappeared. In contrast, when limited to the cases with serum albumin levels ≥3.8 g/dL, the associations remained, with significantly prolonged PFS and significantly increased irAE occurrence at NLR < 4. NLR may be a good predictive tool for PFS and irAE occurrence during ICI treatment when a good nutritional status is maintained.
Full article
(This article belongs to the Special Issue Immune-Related Adverse Events in Patients with Cancer: From Bench to Bedside)
Open AccessArticle
Prediction of Mismatch Repair Status in Endometrial Cancer from Histological Slide Images Using Various Deep Learning-Based Algorithms
by
Mina Umemoto, Tasuku Mariya, Yuta Nambu, Mai Nagata, Toshihiro Horimai, Shintaro Sugita, Takayuki Kanaseki, Yuka Takenaka, Shota Shinkai, Motoki Matsuura, Masahiro Iwasaki, Yoshihiko Hirohashi, Tadashi Hasegawa, Toshihiko Torigoe, Yuichi Fujino and Tsuyoshi Saito
Cancers 2024, 16(10), 1810; https://doi.org/10.3390/cancers16101810 - 9 May 2024
Abstract
The application of deep learning algorithms to predict the molecular profiles of various cancers from digital images of hematoxylin and eosin (H&E)-stained slides has been reported in recent years, mainly for gastric and colon cancers. In this study, we investigated the potential use
[...] Read more.
The application of deep learning algorithms to predict the molecular profiles of various cancers from digital images of hematoxylin and eosin (H&E)-stained slides has been reported in recent years, mainly for gastric and colon cancers. In this study, we investigated the potential use of H&E-stained endometrial cancer slide images to predict the associated mismatch repair (MMR) status. H&E-stained slide images were collected from 127 cases of the primary lesion of endometrial cancer. After digitization using a Nanozoomer virtual slide scanner (Hamamatsu Photonics), we segmented the scanned images into 5397 tiles of 512 × 512 pixels. The MMR proteins (PMS2, MSH6) were immunohistochemically stained, classified into MMR proficient/deficient, and annotated for each case and tile. We trained several neural networks, including convolutional and attention-based networks, using tiles annotated with the MMR status. Among the tested networks, ResNet50 exhibited the highest area under the receiver operating characteristic curve (AUROC) of 0.91 for predicting the MMR status. The constructed prediction algorithm may be applicable to other molecular profiles and useful for pre-screening before implementing other, more costly genetic profiling tests.
Full article
(This article belongs to the Special Issue Novel Approaches to Machine Learning and Artificial Intelligence in Cancer Research and Care)
Open AccessReview
Catalyzing Precision Medicine: Artificial Intelligence Advancements in Prostate Cancer Diagnosis and Management
by
Ali Talyshinskii, B. M. Zeeshan Hameed, Prajwal P. Ravinder, Nithesh Naik, Princy Randhawa, Milap Shah, Bhavan Prasad Rai, Theodoros Tokas and Bhaskar K. Somani
Cancers 2024, 16(10), 1809; https://doi.org/10.3390/cancers16101809 - 9 May 2024
Abstract
Background: The aim was to analyze the current state of deep learning (DL)-based prostate cancer (PCa) diagnosis with a focus on magnetic resonance (MR) prostate reconstruction; PCa detection/stratification/reconstruction; positron emission tomography/computed tomography (PET/CT); androgen deprivation therapy (ADT); prostate biopsy; associated challenges and their
[...] Read more.
Background: The aim was to analyze the current state of deep learning (DL)-based prostate cancer (PCa) diagnosis with a focus on magnetic resonance (MR) prostate reconstruction; PCa detection/stratification/reconstruction; positron emission tomography/computed tomography (PET/CT); androgen deprivation therapy (ADT); prostate biopsy; associated challenges and their clinical implications. Methods: A search of the PubMed database was conducted based on the inclusion and exclusion criteria for the use of DL methods within the abovementioned areas. Results: A total of 784 articles were found, of which, 64 were included. Reconstruction of the prostate, the detection and stratification of prostate cancer, the reconstruction of prostate cancer, and diagnosis on PET/CT, ADT, and biopsy were analyzed in 21, 22, 6, 7, 2, and 6 studies, respectively. Among studies describing DL use for MR-based purposes, datasets with magnetic field power of 3 T, 1.5 T, and 3/1.5 T were used in 18/19/5, 0/1/0, and 3/2/1 studies, respectively, of 6/7 studies analyzing DL for PET/CT diagnosis which used data from a single institution. Among the radiotracers, [68Ga]Ga-PSMA-11, [18F]DCFPyl, and [18F]PSMA-1007 were used in 5, 1, and 1 study, respectively. Only two studies that analyzed DL in the context of DT met the inclusion criteria. Both were performed with a single-institution dataset with only manual labeling of training data. Three studies, each analyzing DL for prostate biopsy, were performed with single- and multi-institutional datasets. TeUS, TRUS, and MRI were used as input modalities in two, three, and one study, respectively. Conclusion: DL models in prostate cancer diagnosis show promise but are not yet ready for clinical use due to variability in methods, labels, and evaluation criteria. Conducting additional research while acknowledging all the limitations outlined is crucial for reinforcing the utility and effectiveness of DL-based models in clinical settings.
Full article
(This article belongs to the Section Cancer Causes, Screening and Diagnosis)
►▼
Show Figures

Figure 1
Open AccessReview
Driver Mutations in Pancreatic Cancer and Opportunities for Targeted Therapy
by
Olamide T. Olaoba, Temitope I. Adelusi, Ming Yang, Tessa Maidens, Eric T. Kimchi, Kevin F. Staveley-O’Carroll and Guangfu Li
Cancers 2024, 16(10), 1808; https://doi.org/10.3390/cancers16101808 - 9 May 2024
Abstract
Pancreatic cancer is the sixth leading cause of cancer-related mortality globally. As the most common form of pancreatic cancer, pancreatic ductal adenocarcinoma (PDAC) represents up to 95% of all pancreatic cancer cases, accounting for more than 300,000 deaths annually. Due to the lack
[...] Read more.
Pancreatic cancer is the sixth leading cause of cancer-related mortality globally. As the most common form of pancreatic cancer, pancreatic ductal adenocarcinoma (PDAC) represents up to 95% of all pancreatic cancer cases, accounting for more than 300,000 deaths annually. Due to the lack of early diagnoses and the high refractory response to the currently available treatments, PDAC has a very poor prognosis, with a 5-year overall survival rate of less than 10%. Targeted therapy and immunotherapy are highly effective and have been used for the treatment of many types of cancer; however, they offer limited benefits in pancreatic cancer patients due to tumor-intrinsic and extrinsic factors that culminate in drug resistance. The identification of key factors responsible for PDAC growth and resistance to different treatments is highly valuable in developing new effective therapeutic strategies. In this review, we discuss some molecules which promote PDAC initiation and progression, and their potential as targets for PDAC treatment. We also evaluate the challenges associated with patient outcomes in clinical trials and implications for future research.
Full article
(This article belongs to the Section Cancer Immunology and Immunotherapy)
►▼
Show Figures

Figure 1
Open AccessArticle
Systemic DNA Damage and Repair Activity Vary by Race in Breast Cancer Survivors
by
Shraddha Divekar, Ryan Kritzer, Haokai Shu, Keval Thakkar, Jennifer Hicks, Mary G. Mills, Kepher Makambi, Chiranjeev Dash and Rabindra Roy
Cancers 2024, 16(10), 1807; https://doi.org/10.3390/cancers16101807 - 9 May 2024
Abstract
Non-Hispanic Black breast cancer survivors have poorer outcomes and higher mortality rates than White survivors, but systemic biological mechanisms underlying these disparities are unclear. We used circulating leukocytes as a surrogate for measuring systemic mechanisms, which might be different from processes in the
[...] Read more.
Non-Hispanic Black breast cancer survivors have poorer outcomes and higher mortality rates than White survivors, but systemic biological mechanisms underlying these disparities are unclear. We used circulating leukocytes as a surrogate for measuring systemic mechanisms, which might be different from processes in the target tissue (e.g., breast). We investigated race-based differences in DNA damage and repair, using a novel CometChip assay, in circulating leukocytes from breast cancer survivors who had completed primary cancer therapy and were cancer free. We observed novel race-based differences in systemic DNA damage and repair activity in cancer survivors, but not in cells from healthy volunteers. Basal DNA damage in leukocytes was higher in White survivors, but Black survivors showed a much higher induction after bleomycin treatment. Double-strand break repair activity was also significantly different between the races, with cells from White survivors showing more sustained repair activity compared to Black leukocytes. These results suggest that cancer and cancer therapy might have long-lasting effects on systemic DNA damage and repair mechanisms that differ in White survivors and Black survivors. Findings from our preliminary study in non-cancer cells (circulating leukocytes) suggest systemic effects beyond the target site, with implications for accelerated aging-related cancer survivorship disparities.
Full article
(This article belongs to the Collection Advances in Cancer Disparities)
►▼
Show Figures

Figure 1
Open AccessReview
Gut Microbiota Are a Novel Source of Biomarkers for Immunotherapy in Non-Small-Cell Lung Cancer (NSCLC)
by
Teresa Del Giudice, Nicoletta Staropoli, Pierfrancesco Tassone, Pierosandro Tagliaferri and Vito Barbieri
Cancers 2024, 16(10), 1806; https://doi.org/10.3390/cancers16101806 - 9 May 2024
Abstract
Despite the recent availability of immune checkpoint inhibitors, not all patients affected by Non-Small-Cell Lung Cancer (NSCLC) benefit from immunotherapy. The reason for this variability relies on a variety of factors which may allow for the identification of novel biomarkers. Presently, a variety
[...] Read more.
Despite the recent availability of immune checkpoint inhibitors, not all patients affected by Non-Small-Cell Lung Cancer (NSCLC) benefit from immunotherapy. The reason for this variability relies on a variety of factors which may allow for the identification of novel biomarkers. Presently, a variety of biomarkers are under investigation, including the PD1/PDL1 axis, the tumor mutational burden, and the microbiota. The latter is made by all the bacteria and other microorganisms hosted in our body. The gut microbiota is the most represented and has been involved in different physiological and pathological events, including cancer. In this light, it appears that all conditions modifying the gut microbiota can influence cancer, its treatment, and its treatment-related toxicities. The aim of this review is to analyze all the conditions influencing the gut microbiota and, therefore, affecting the response to immunotherapy, iRAEs, and their management in NSCLC patients. The investigation of the landscape of these biological events can allow for novel insights into the optimal management of NSCLC immunotherapy.
Full article
(This article belongs to the Special Issue Novel Biomarkers in Non-small Cell Lung Cancer (NSCLC))
►▼
Show Figures

Figure 1
Open AccessReview
Schlafens: Emerging Therapeutic Targets
by
Ricardo E. Perez, Frank Eckerdt and Leonidas C. Platanias
Cancers 2024, 16(10), 1805; https://doi.org/10.3390/cancers16101805 - 9 May 2024
Abstract
The interferon (IFN) family of immunomodulatory cytokines has been a focus of cancer research for over 50 years with direct and indirect implications in cancer therapy due to their properties to inhibit malignant cell proliferation and modulate immune responses. Among the transcriptional targets
[...] Read more.
The interferon (IFN) family of immunomodulatory cytokines has been a focus of cancer research for over 50 years with direct and indirect implications in cancer therapy due to their properties to inhibit malignant cell proliferation and modulate immune responses. Among the transcriptional targets of the IFNs is a family of genes referred to as Schlafens. The products of these genes, Schlafen proteins, exert important roles in modulating cellular proliferation, differentiation, immune responses, viral replication, and chemosensitivity of malignant cells. Studies have demonstrated that abnormal expression of various Schlafens contributes to the pathophysiology of various cancers. Schlafens are now emerging as promising biomarkers and potentially attractive targets for drug development in cancer research. Here, we highlight research suggesting the use of Schlafens as cancer biomarkers and the rationale for the development of specific drugs targeting Schlafen proteins.
Full article
(This article belongs to the Section Cancer Therapy)
►▼
Show Figures

Figure 1

Journal Menu
► ▼ Journal Menu-
- Cancers Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Bioengineering, Biomolecules, Cancers, Diseases, Nanomaterials, Pharmaceutics
Dynamic Nano-Biomaterials in Tissue Regeneration and Cancer Therapies
Topic Editors: Ramar Thangam, Heemin Kang, Bibin G. Anand, Ramachandran Vijayan, Venugopal KrishnanDeadline: 15 May 2024
Topic in
Biomedicines, Cancers, JFB, Nanomaterials, Polymers
Advanced Functional Materials for Regenerative Medicine
Topic Editors: Antonino Morabito, Luca ValentiniDeadline: 6 June 2024
Topic in
Biology, Cancers, Current Oncology, Diseases, JCM, Pathogens
Pathogenetic, Diagnostic and Therapeutic Perspectives in Head and Neck Cancer
Topic Editors: Shun-Fa Yang, Ming-Hsien ChienDeadline: 20 June 2024
Topic in
Cancers, Cells, JCM, Radiation, Pharmaceutics, Applied Sciences, Nanomaterials, Current Oncology
Innovative Radiation Therapies
Topic Editors: Gérard Baldacchino, Eric Deutsch, Marie Dutreix, Sandrine Lacombe, Erika Porcel, Charlotte Robert, Emmanuelle Bourneuf, João Santos Sousa, Aurélien de la LandeDeadline: 30 June 2024

Conferences
Special Issues
Special Issue in
Cancers
Treatment of Hepatopancreatobiliary Cancers: Open Question, Challenge, and Future Directions
Guest Editors: Francesco Giovinazzo, Stijn van LaarhovenDeadline: 10 May 2024
Special Issue in
Cancers
Molecular and Cellular Heterogeneity in an Evolving Tumor Landscape: When Diversity Gives Rise to Aggressive and Drug Resistant Cells
Guest Editor: Michelle R. DawsonDeadline: 20 May 2024
Special Issue in
Cancers
Innovations in Soft Tissue Sarcoma Diagnosis and Treatment
Guest Editors: Erlinda M. Gordon, Sant P. Chawla, Frederick L. HallDeadline: 30 May 2024
Special Issue in
Cancers
Perioperative Care and Pain Management in Cancer Patients: From Basic Science to Clinical Practice
Guest Editors: Dario Bugada, Juan P. CataDeadline: 20 June 2024
Topical Collections
Topical Collection in
Cancers
Drug Resistance and Novel Therapies in Cancers
Collection Editor: Zhixiang Wang










