Antioxidant Interactions between Major Phenolic Compounds Found in ‘Ataulfo’ Mango Pulp: Chlorogenic, Gallic, Protocatechuic and Vanillic Acids
Abstract
:1. Introduction
2. Results and Discussion
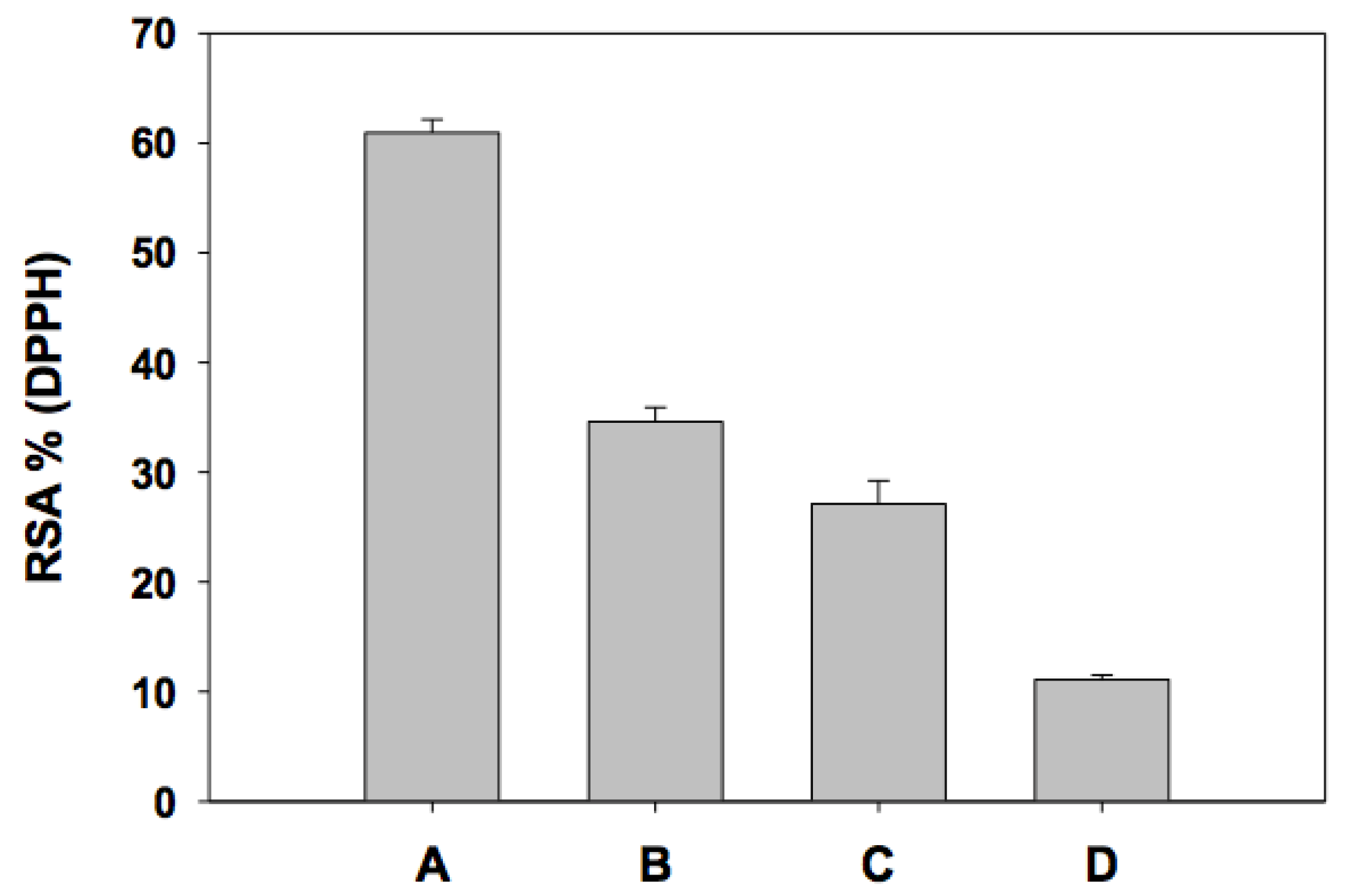
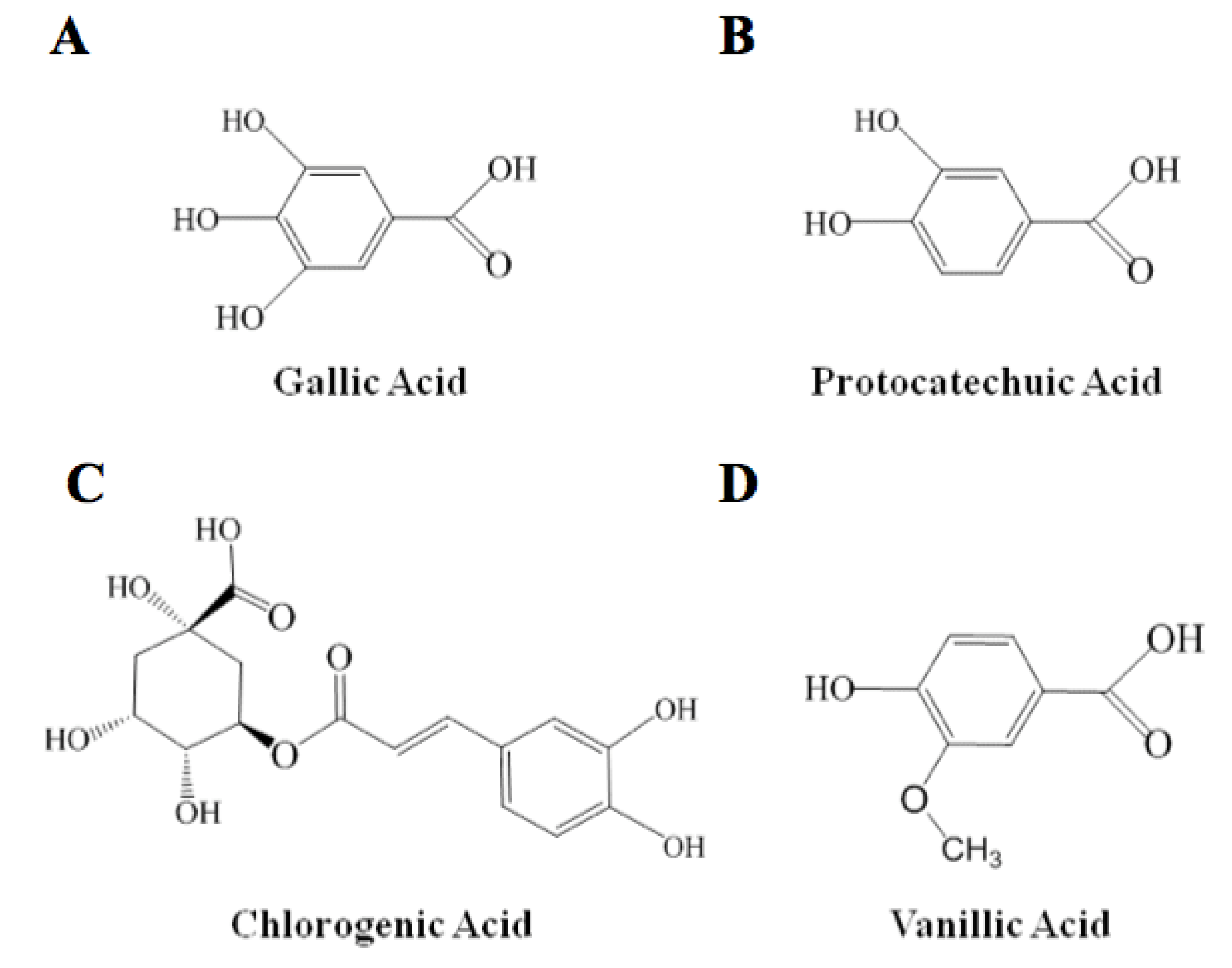
| I. | Individual (0.1 mM) | % RSA Real | % RSA Theoretical (Sum) | Type of Interaction |
| A | 30.47 ± 1.8 | |||
| B | 17.30 ± 1.3 | |||
| C | 13.56 ± 0.8 | |||
| D | 5.55 ± 0.2 | |||
| Combination | ||||
| AB | 67.58µa | 47.77µb | Synergic | |
| AC | 44.96µa | 44.03µb | Synergic | |
| BC | 34.83µa | 30.86µb | Synergic | |
| AD | 33.03µa | 36.02µb | Antagonist | |
| BD | 23.66µa | 22.85µa | ||
| CD | 20.46µa | 19.11µa | ||
| II. | Individual (0.066 mM) | % RSA Real | % RSA Theoretical (Sum) | Type of Interaction |
| A | 20.11 ± 1.3 | |||
| B | 11.42 ± 0.7 | |||
| C | 8.95 ± 0.4 | |||
| D | 3.66 ± 0.5 | |||
| Combination | ||||
| ACD | 58.10µa | 32.72µb | Synergic | |
| ABC | 43.03µa | 40.48µb | Synergic | |
| ABD | 42.52µa | 35.19µb | Synergic | |
| BCD | 19.70µa | 24.03µb | Antagonist | |
| III. | Individual (0.05 mM) | % RSA Real | % RSA Theoretical (Sum) | Type of Interaction |
| A | 15.23 ± 1.1 | |||
| B | 8.65 ± 0.8 | |||
| C | 6.78 ± 0.6 | |||
| D | 2.77 ± 0.2 | |||
| Combination | ||||
| ABCD | 39.76µa | 33.44µb | Synergic |
3. Experimental
4. Conclusions
Acknowledgments
References
- Kelsey, N.A.; Wilkins, H.M.; Linseman, D.A. Nutraceutical antioxidants as novel neuroprotective agents. Molecules 2010, 15, 7792–7814. [Google Scholar] [CrossRef]
- González-Aguilar, G.A.; Celis, J.; Sotelo-Mundo, R.R.; De La Rosa, L.A.; Rodrigo-Garcia, J.; Alvarez-Parrilla, E. Physiological and biochemical changes of different fresh-cut mango cultivars stored at 5 °C. Int. J. Food Sci. Technol. 2008, 43, 91–101. [Google Scholar] [CrossRef]
- Severi, J.A.; Lima, Z.P.; Kushima, H.; Brito, A.R.; Santos, L.C.; Vilegas, W.; Hiruma-Lima, C.A. Polyphenols with antiulcerogenic action from aqueous decoction of mango leaves (Mangifera indica L.). Molecules 2009, 14, 1098–1110. [Google Scholar] [CrossRef]
- Yahia, E.M. The contribution of fruit and vegetable consumption to human health. In Fruit and Vegetable Phytochemicals; De La Rosa, L.A., Alvarez-Parrilla, E., González-Aguilar, G.A., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2010; pp. 3–51. [Google Scholar]
- Leontowicz, H.; Leontowicz, M.; Jesion, I.; Bielecki, W.; Poovarodom, S.; Vearasilp, S.; González-Aguilar, G.; Robles-Sánchez, M.; Trakhtenberg, S.; Gorinstein, S. Positive effects of durian fruit at different stages of ripening on the hearts and livers of rats fed diets high in cholesterol. Eur. J. Integr. Med. 2011, 3, e169–e181. [Google Scholar] [CrossRef]
- Ma, X.; Wu, H.; Liu, L.; Yao, Q.; Wang, S.; Zhan, R.; Xing, S.; Zhou, Y. Polyphenolic compounds and antioxidant properties in mango fruits. Sci. Hortic. 2011, 129, 102–107. [Google Scholar]
- Kim, Y.; Brecht, J.K.; Talcott, S.T. Antioxidant phytochemical and fruit quality changes in mango (Mangifera indica L.) following hot water immersion and controlled atmosphere storage. Food Chem. 2007, 105, 1327–1334. [Google Scholar] [CrossRef]
- Robles-Sánchez, M.; Astiazaran-García, H.; Martín-Belloso, O.; Gorinstein, S.; Alvarez-Parrilla, E.; De la Rosa, L.A.; Yepiz-Plascencia, G.; González-Aguilar, G.A. Influence of whole and fresh-cut mango intake on plasma lipids and antioxidant capacity of healthy adults. Food Res. Int. 2011, 44, 1386–1391. [Google Scholar] [CrossRef]
- Manthey, J.; Perkins-Veazie, P. Influences of harvest date and location on the levels of b-carotene, ascorbic acid, total phenols, the in vitro antioxidant capacity, and phenolic profiles of five commercial varieties of mango (Mangifera indica L.). J. Agric. Food Chem. 2009, 57, 10825–10830. [Google Scholar] [CrossRef]
- Palafox-Carlos, H.; Yahia, E.; González-Aguilar, G.A. Identification and Quantification of Major Phenolic Compounds from Mango (Mangifera indica cv. Ataulfo) Fruit by HPLC-DAD-MS/MS-ESI and Their Individual Contribution to the Antioxidant Activity during Ripening. Food Chem. 2012, 135, 105–111. [Google Scholar] [CrossRef]
- Palafox Carlos, H.; Ayala Zavala, J.F.; González Aguilar, G.A. The Role of Dietary Fiber in the Bioaccessibility and Bioavailability of Fruit and Vegetable Antioxidants. J. Food Sci. 2011, 76, R6–R15. [Google Scholar]
- Balasundram, N.; Sundram, K.; Samman, S. Phenolic compounds in plants and agri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chem. 2006, 99, 191–203. [Google Scholar] [CrossRef]
- Heo, H.; Kim, Y.; Chung, D.; Kim, D. Antioxidant capacities of individual and combined phenolics in a model system. Food Chem. 2007, 104, 87–92. [Google Scholar] [CrossRef]
- Corral-Aguayo, R.D.; Yahia, E.M.; Carrillo-Lopez, A.; Gonzalez-Aguilar, G. Correlation between some nutritional components and the total antioxidant capacity measured with six different assays in eight horticultural crops. J. Agric. Food Chem. 2008, 56, 10498–10504. [Google Scholar]
- Rice-Evans, C.A.; Miller, N.J.; Paganga, G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic. Biol. Med. 1996, 20, 933–956. [Google Scholar] [CrossRef]
- Leopoldini, M.; Marino, T.; Russo, N.; Toscano, M. Antioxidant properties of phenolic compounds: H-atom versus electron transfer mechanism. J. Phys. Chem. A 2004, 108, 4916–4922. [Google Scholar]
- Gonthier, M.P.; Verny, M.A.; Besson, C.; Rémésy, C.; Scalbert, A. Chlorogenic acid bioavailability largely depends on its metabolism by the gut microflora in rats. J. Nutr. 2003, 133, 1853–1859. [Google Scholar]
- Garcia-Alonso, M.; Rimbach, G.; Sasai, M.; Nakahara, M.; Matsugo, S.; Uchida, Y.; Rivas-Gonzalo, J.C.; De Pascual-Teresa, S. Electron spin resonance spectroscopy studies on the free radical scavenging activity of wine anthocyanins and pyranoanthocyanins. Mol. Nutr. Food Res. 2005, 49, 1112–1119. [Google Scholar] [CrossRef]
- Pinelo, M.; Manzocco, L.; Nuñez, M.J.; Nicoli, M.C. Interaction among phenols in food fortification: Negative synergism on antioxidant capacity. J. Agric. Food Chem. 2004, 52, 1177–1180. [Google Scholar] [CrossRef]
- Rossetto, M.; Vanzani, P.; Mattivi, F.; Lunelli, M.; Scarpa, M.; Rigo, A. Synergistic antioxidant effect of catechin and malvidin 3-glucoside on free radical-initiated peroxidation of linoleic acid in micelles. Arch. Biochem. Biophys. 2002, 408, 239–245. [Google Scholar] [CrossRef]
- Pignatelli, P.; Pulcinelli, F.M.; Celestini, A.; Lenti, L.; Ghiselli, A.; Gazzaniga, P.P.; Violi, F. The flavonoids quercetin and catechin synergistically inhibit platelet function by antagonizing the intracellular production of hydrogen peroxide. Am. J. Clin. Nutr. 2000, 72, 1150–1155. [Google Scholar]
- Reber, J.D.; Eggett, D.L.; Parker, T.L. Antioxidant capacity interactions and a chemical/structural model of phenolic compounds found in strawberries. Int. J. Food Sci. Nutr. 2011, 62, 445–452. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Palafox-Carlos, H.; Gil-Chávez, J.; Sotelo-Mundo, R.R.; Namiesnik, J.; Gorinstein, S.; González-Aguilar, G.A. Antioxidant Interactions between Major Phenolic Compounds Found in ‘Ataulfo’ Mango Pulp: Chlorogenic, Gallic, Protocatechuic and Vanillic Acids. Molecules 2012, 17, 12657-12664. https://doi.org/10.3390/molecules171112657
Palafox-Carlos H, Gil-Chávez J, Sotelo-Mundo RR, Namiesnik J, Gorinstein S, González-Aguilar GA. Antioxidant Interactions between Major Phenolic Compounds Found in ‘Ataulfo’ Mango Pulp: Chlorogenic, Gallic, Protocatechuic and Vanillic Acids. Molecules. 2012; 17(11):12657-12664. https://doi.org/10.3390/molecules171112657
Chicago/Turabian StylePalafox-Carlos, Hugo, Joana Gil-Chávez, Rogerio R. Sotelo-Mundo, Jacek Namiesnik, Shela Gorinstein, and Gustavo A. González-Aguilar. 2012. "Antioxidant Interactions between Major Phenolic Compounds Found in ‘Ataulfo’ Mango Pulp: Chlorogenic, Gallic, Protocatechuic and Vanillic Acids" Molecules 17, no. 11: 12657-12664. https://doi.org/10.3390/molecules171112657





