Effects of Taraxacum officinale on Fatigue and Immunological Parameters in Mice
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of TO on the Immobility Time

2.2. Effects of TO on Blood Biochemical Parameters
| TO Treatment a | 0 mg/kg | 10 mg/kg | 100 mg/kg |
|---|---|---|---|
| BUN (mg/dL) | 23.98 ± 0.58 | 19.00 ± 0.62 * (0.007) | 17.16 ± 2.15 * (0.001) |
| LDH (IU/L) | 1625.75 ± 113.75 | 637.25 ± 70.32 * (0.000) | 640.50 ± 91.16 * (0.000) |
| CK (IU/L) | 607.75 ± 67.25 | 440.25 ± 78.23 (0.493) | 438.66 ± 48.22 (0.562) |
| Glc (mg/dL) | 121.33 ± 11.34 | 130.5 ± 4.97 (0.962) | 138.75 ± 2.83 * (0.042) |
| Alb (g/dL) | 2.9 ± 0.08 | 2.4 ± 0.11 (0.051) | 2.8 ± 0.13 (0.994) |
2.3. Effect of TO on rIFN-γ- Induced TNF-α, IL-12p70 Productions and mRNA Expressions in Mouse Peritoneal Macrophages

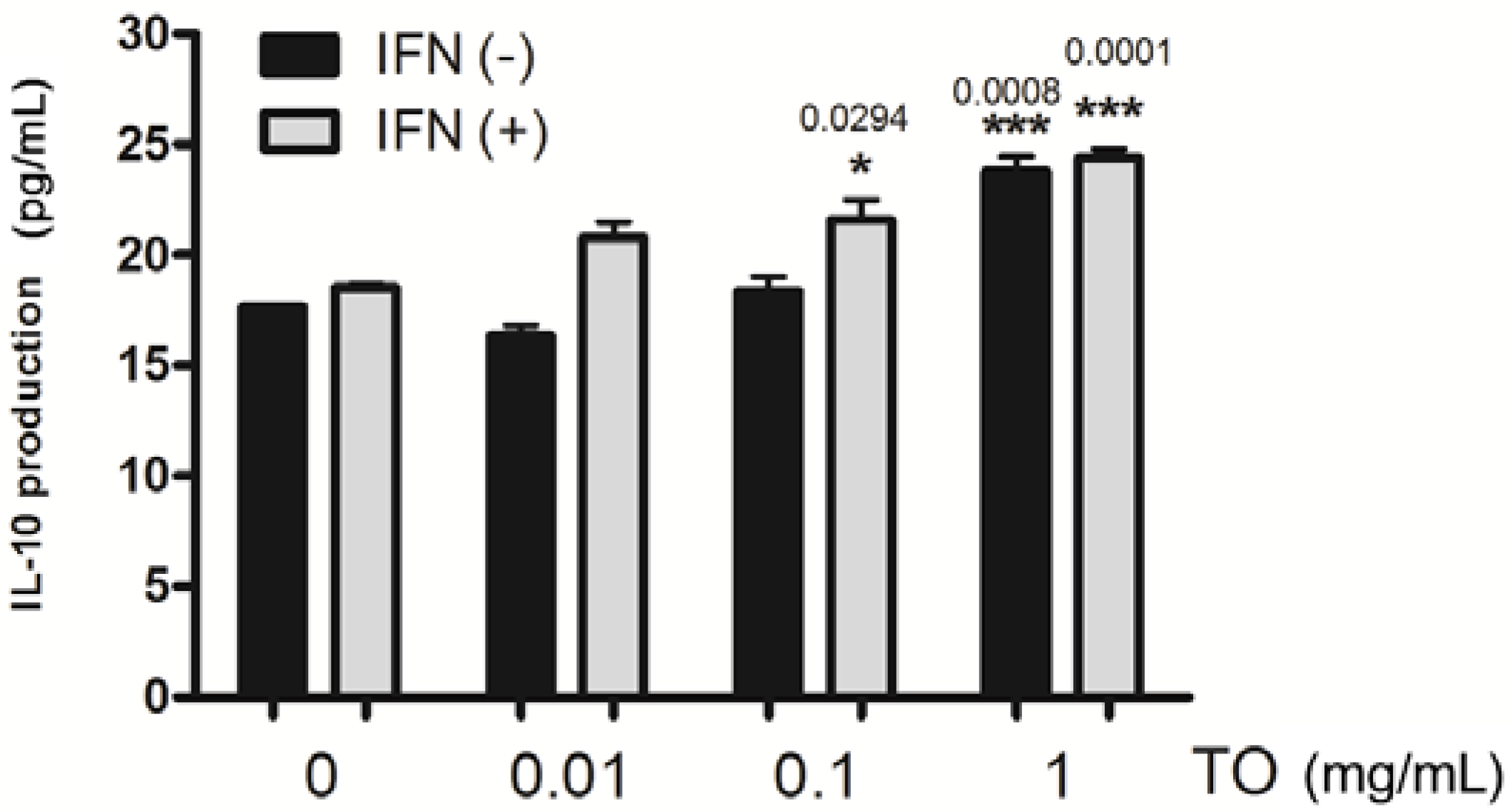
2.4. Effect of TO on rIFN-γ- Induced IL-10 Production in Mouse Peritoneal Macrophages
2.5. Effects of TO on NO Production and iNOS Protein and mRNA Expressions

3. Experimental
3.1. Reagents
3.2. Preparation of TO
3.3. Animals
3.4. FST
3.5. Preparation and Ingredient Analysis of Blood Serum
3.6. Peritoneal Macrophage Culture
3.7. Assay for Endotoxin Determination
3.8. Cytokine Assays
3.9. RNA Isolation and Real Time RT-PCR Analysis
3.10. Measurement of Nitrite Concentration
3.11. Western Blot Analysis
3.12. Statistical Analysis
4. Conclusions
Acknowledgments
- Sample Availability: Samples are available from the authors.
References
- Ahmad, V.U.; Yasmeen, S.; Ali, Z.; Khan, M.A.; Choudhary, M.I.; Akhtar, F.; Miana, G.A.; Zahid, M. Taraxacin, A newguaianolide from Taraxacum wallichii. J. Nat. Prod. 2000, 63, 1010–1011. [Google Scholar] [CrossRef]
- Kisiel, W.; Barszcz, B. Further sesquiterpenoids and phenolics from Taraxacum officinale. Fitoterapia 2000, 71, 269–273. [Google Scholar] [CrossRef]
- Schutz, K.; Carle, R.; Schieber, A. Taraxacum—A review on its phytochemical and pharmacological profile. J. Ethnopharmacol. 2006, 107, 313–323. [Google Scholar] [CrossRef]
- Seo, S.W.; Koo, H.N.; An, H.J.; Kwon, K.B.; Lim, B.C.; Seo, E.A.; Ryu, D.G.; Moon, G.; Kim, H.Y.; Kim, H.M.; et al. Taraxacum officinale protects against cholecystokinin-induced acute pancreatitis in rats. World J. Gastroentero. 2005, 11, 597–599. [Google Scholar]
- Koo, H.N.; Hong, S.H.; Song, B.K.; Kim, C.H.; Yoo, Y.H.; Kim, H.M. Taraxacum officinale induces cytotoxicity through TNF-α and IL-1β secretion in HepG2 cells. Life Sci. 2004, 74, 1149–1157. [Google Scholar] [CrossRef]
- Hu, C.; Kitts, D.D. Antioxidant, prooxidant, and cytotoxic activities of solvent-fractionated dandelion (Taraxacum officinale) flower extracts in vitro. J. Agric. Food Chem. 2003, 51, 301–310. [Google Scholar] [CrossRef]
- Kim, H.M.; Shin, H.Y.; Lim, K.H.; Ryu, S.T.; Shin, T.Y.; Chae, H.J.; Kim, H.R.; Lyu, Y.S.; An, N.H. Taraxacum officinale inhibits tumor necrosis factor-α production from rat astrocytes. Immunopharm. Immunotoxicol. 2000, 22, 519–530. [Google Scholar] [CrossRef]
- Mague, S.D.; Pliakas, A.M.; Todtenkopf, M.S. Antidepressant-like effects of kappa-opioid receptor antagonists in the forced swim test in rats. J. Pharmacol. Exp. Ther. 2003, 305, 323–330. [Google Scholar] [CrossRef]
- Deyama, T.; Nishibe, S.; Nakazawa, Y. Constituents and pharmacological effects of Eucommia and Siberian ginseng. Acta Pharmacol. Sin. 2001, 22, 1057–1070. [Google Scholar]
- Kim, M.C.; Lee, G.H.; Kim, S.J.; Chung, W.S.; Kim, S.S.; Ko, S.G.; Um, J.Y. Immune-enhancing effect of Danggwibohyeoltang, an extract from Astragali Radix and Angelicae gigantis Radix, in vitro and in vivo. Immunopharmacol. Immunotoxicol. 2011, 34, 66–73. [Google Scholar]
- Kim, K.M.; Yu, K.W.; Kang, D.H.; Suh, H.J. Anti-stress and ant-fatigue effect of fermented rice bran. Phytother. Res. 2002, 16, 700–702. [Google Scholar] [CrossRef]
- Coombes, J.S.; McNaughton, L.R. Effects of branched-chain amino acid supplementation on serum creatine kinase and lactate dehydrogenase after prolonged exercise. J. Sports Med. Phys. Fitness 2000, 40, 240–246. [Google Scholar]
- Dorchy, H. Sports and type I diabetes: personal experience. Rev. Med. Brux. 2002, 23, A211–A217. [Google Scholar]
- Adams, O.D.; Hamilton, T.A. Molecular Basis of Macrophage Activation and Its Orgins; Oxford University Press: New York, NY, USA, 1992; pp. 75–114. [Google Scholar]
- Whiteside, T.L. Immune suppression in cancer: Effects on immune cells, Mechanisms and future therapeutic intervention. Semin. Cancer Biol. 2005, 16, 3–15. [Google Scholar] [CrossRef]
- O’Farrell, A.M.; Liu, Y.; Moore, K.W.; Mui, A.L. IL-10 inhibits macrophage activation and proliferation by distinct signaling mechanisms: Evidence for Stat3-dependent and -independent pathways. EMBO J. 1998, 17, 1006–1018. [Google Scholar] [CrossRef]
- An, H.J.; Seo, M.J.; Choi, I.Y.; Park, R.K.; Jeong, S.; Lee, J.Y.; Kim, H.M.; Um, J.Y.; Hong, S.H. Induction of nitric oxide & tumour necrosis factor-α by Psoralea corylifolia. Indian J. Med. Res. 2008, 128, 752–758. [Google Scholar]
- Laires, M.J.; Alves, F.; Halpern, M.J. Changes in serum and erythrocyte magnesium and blood lipids after distance swimming. Magnes. Res. 1988, 1, 219–222. [Google Scholar]
- Brantley, E.C.; Guo, L.; Zhang, C.; Lin, Q.; Yokoi, K.; Langley, R.R.; Kruzel, E.; Maya, M.; Kim, S.W.; Kim, S.J.; et al. Nitric oxide-mediated tumoricidal activity of murine microglial cells. Transl. Oncol. 2010, 3, 380–388. [Google Scholar]
- Stuehr, D.J.; Nathan, C.F. A macrophage product responsible for cytostasis and respiratory inhibition in tumor target cells. J. Exp. Med. 1989, 169, 1543–1555. [Google Scholar] [CrossRef]
- Porsolt, R.D.; Le Pichon, M.; Jalfre, M. Depression: A new animal model sensitive to antidepressant treatments. Nature 1977, 266, 730–732. [Google Scholar] [CrossRef]
- Narumi, S.; Finke, J.H.; Hamilton, T.A. Interferon gamma and interleukin 2 synergize to induce selective monokine expression in murine peritoneal macrophages. J. Biol. Chem. 1990, 265, 7036–7041. [Google Scholar]
- An, H.J.; Lee, G.G.; Lee, K.T. Drynariae rhizoma increases immune response in mice. Nat. Prod. Commun. 2012, 7, 905–908. [Google Scholar]
- An, H.J.; Kim, I.T.; Park, H.J.; Kim, H.M.; Choi, J.H.; Lee, K.T. Tormentic acid, A triterpenoid saponin, Isolated from Rosa rugosa, Inhibited LPS-induced iNOS, COX-2, And TNF-α expression through inactivation of the nuclear factor-κb pathway in RAW 264.7 macrophages. Int. Immunopharmacol. 2011, 11, 504–510. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lee, B.-R.; Lee, J.-H.; An, H.-J. Effects of Taraxacum officinale on Fatigue and Immunological Parameters in Mice. Molecules 2012, 17, 13253-13265. https://doi.org/10.3390/molecules171113253
Lee B-R, Lee J-H, An H-J. Effects of Taraxacum officinale on Fatigue and Immunological Parameters in Mice. Molecules. 2012; 17(11):13253-13265. https://doi.org/10.3390/molecules171113253
Chicago/Turabian StyleLee, Bo-Ra, Jong-Hyun Lee, and Hyo-Jin An. 2012. "Effects of Taraxacum officinale on Fatigue and Immunological Parameters in Mice" Molecules 17, no. 11: 13253-13265. https://doi.org/10.3390/molecules171113253




