In Vitro and In Vivo Antiangiogenic Activity of Caged Polyprenylated Xanthones Isolated from Garcinia hanburyi Hook. f.
Abstract
:1. Introduction
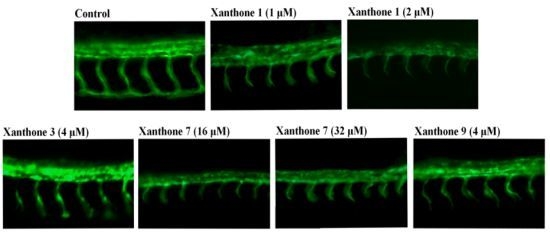
2. Results and Discussion

| Xanthone | IC50 (μM) | ||||
|---|---|---|---|---|---|
| HeLa | A549 | HCT-116 | HepG-2 | HUVEC | |
| 1 | 1.59 | 1.55 | 0.64 | 0.78 | 0.72 |
| 2 | 11.50 | 4.76 | 4.76 | 6.35 | 4.12 |
| 3 | 13.78 | 14.23 | 8.89 | 13.34 | 6.14 |
| 4 | 7.58 | 11.30 | 4.49 | 10.21 | 2.48 |
| 5 | 5.33 | 6.43 | 2.76 | 5.97 | 2.39 |
| 6 | 4.07 | 4.88 | 1.47 | 2.44 | 2.12 |
| 7 | 3.26 | 4.88 | 1.30 | 6.51 | 1.04 |
| 8 | 4.77 | 2.86 | 1.59 | 6.37 | 0.91 |
| 9 | 6.35 | 12.69 | 11.74 | 6.35 | 1.73 |
| 10 | 2.60 | 2.05 | 1.20 | 1.00 | 0.90 |
| 11 | 2.95 | 1.80 | 1.35 | 1.15 | 1.60 |
| Xanthone | Antiangiogenic Phenotype | |||||
|---|---|---|---|---|---|---|
| 1 μM | 2 μM | 4 μM | 8 μM | 16 μM | 32 μM | |
| 1 | √ | √ | ||||
| 2 | × | × | ||||
| 3 | × | × | √ | |||
| 4 | × | × | × | × | ||
| 5 | × | × | × | |||
| 6 | × | × | × | × | × | × |
| 7 | × | × | × | × | √ | √ |
| 8 | × | × | × | × | ||
| 9 | × | × | √ | |||
| 10 | × | × | × | × | ||
| 11 | × | × | × | × | ||

| Xanthone | Death Rate | |||||
|---|---|---|---|---|---|---|
| 1 μM | 2 μM | 4 μM | 8 μM | 16 μM | 32 μM | |
| 1 | 0 | 1/8 | 8/8 | |||
| 2 | 0 | 0 | 8/8 | |||
| 3 | 0 | 0 | 8/8 | |||
| 4 | 0 | 0 | 0 | |||
| 5 | 0 | 0 | 8/8 | |||
| 6 | 0 | 1/8 | 4/8 | |||
| 7 | 0 | 0 | 0 | |||
| 8 | 0 | 1/8 | 6/8 | |||
| 9 | 0 | 0 | 8/8 | |||
| 10 | 0 | 0 | 0 | 0 | ||
| 11 | 0 | 0 | 0 | 0 | ||
| Xanthone | Heart Rate (per 10 s) | |||||
|---|---|---|---|---|---|---|
| 1 μM | 2 μM | 4 μM | 8 μM | 16 μM | 32 μM | |
| 1 | 8 | 3 | 0 | |||
| 2 | 21 | 18 | 0 | |||
| 3 | 8 | 6 | 0 | |||
| 4 | 21 | 17 | 13 | |||
| 5 | 13 | 10 | 8 | |||
| 6 | 17 | 14 | 8 | |||
| 7 | 6 | 6 | 5 | |||
| 8 | 17 | 13 | 6 | |||
| 9 | 8 | 5 | 0 | |||
| 10 | 20 | 17 | 16 | 16 | ||
| 11 | 21 | 21 | 21 | 20 | ||

3. Experimental
3.1. Plant Material
3.2. Isolation
3.3. MTT Assay
3.4. Antiangiogenic Activity Assay on Blood Vessel Formation in Zebrafish Embryos
3.5. HUVEC Wound Migration Assay
4. Conclusion
Acknowledgments
Conflicts of Interest
References
- Folkman, J.; Hanahan, D. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell 1996, 86, 353–364. [Google Scholar] [CrossRef]
- Folkman, J. What is the evidence that tumors are angiogenesis dependent? J. Natl. Cancer Inst. 1990, 82, 4–6. [Google Scholar] [CrossRef]
- Wang, C.Y.; Tao, W.Y.; Wang, Y.D.; Jennifer, B.; Lu, B.X.; Armand, K.; Subodh, V.; Thomas, G.P.; Han, R.F.; Wen, X.Y. Rosuvastatin, identified from a zebrafish chemical genetic screen for antiangiogenic compounds, Suppresses the growth of prostate cancer. Eur. Urol. 2010, 58, 418–426. [Google Scholar] [CrossRef]
- Sukpondma, Y.; Rukachaisirikui, V.; Phongpaichit, S. Antibacterial caged-tetraprenylated xanthones from the fruits of Garcinia hanburyi. Chem. Pharm. Bull. 2005, 53, 850–852. [Google Scholar] [CrossRef]
- Han, Q.B.; Wang, Y.L.; Yang, L.; Tso, T.F.; Qiao, C.F.; Song, J.Z.; Xu, L.J.; Chen, S.L.; Yang, D.J.; Xu, H.X. Cytotoxic polyprenylated xanthones from the resin of Garcinia hanburyi. Chem. Pharm. Bull. 2006, 54, 265–267. [Google Scholar] [CrossRef]
- Zhang, H.Z.; Kasibhatla, S.; Wang, Y.; Herich, J.; Guastella, J.; Tseng, B.; John, B.; Cai, S.X. Discovery, characterization and SAR of gambogic acid as a potent apoptosis inducer by a HTS assay. Bioorg. Med. Chem. 2004, 12, 309–317. [Google Scholar] [CrossRef]
- Wu, Z.Q.; Guo, Q.L.; You, Q.D.; Zhao, L.; Gu, H.Y. Gambogic acid inhibits proliferation of human lung carcinoma SPC-A1 cells in vivo and in vitro and represses telomerase activity and telomerase reverse transcriptase mRNA expression in the cells. Biol. Pharm. Bull. 2004, 27, 1769–1774. [Google Scholar] [CrossRef]
- Zhao, L.; Guo, Q.L.; You, Q.D.; Wu, Z.Q.; Gu, H.Y. Gambogic acid Induces apoptosis and regulates expressions of Bax and Bcl-2 protein in human gastric carcinoma MGC-803 cells. Bio. Pharm. Bull. 2004, 27, 998–1003. [Google Scholar] [CrossRef]
- Guo, Q.L.; Lin, S.S.; You, Q.D.; Gu, H.Y.; Yu, J.; Zhao, L.; Qi, Q.; Liang, F.; Tan, Z.; Wang, X. Inhibition of human telomerase reverse Transcriptase gene expression by gambogic acid in human hepatoma SMMC-7721 cells. Life Sci. 2006, 78, 1238–1245. [Google Scholar] [CrossRef]
- Jun, Y.; Guo, Q.L.; You, Q.D.; Zhao, L.; Gu, H.Y.; Yang, Y.; Zhang, H.W.; Tan, Z.; Wang, X. Gambogic acid induced G2/M phase cell cycle arrest via disturbing CDK7 mediated phosphorylation of CDC2/P34 in human gastric carcinoma BGC-823 cells. Carcinogenesis 2007, 28, 632–638. [Google Scholar]
- Yu, J.; Guo, Q.L.; You, Q.D.; Lin, S.S.; Li, Z.; Gu, H.Y.; Zhang, H.W.; Tan, Z.; Wang, X. Repression of telomerase reverse transcriptase mRNA and hTERT promoter by gambogic acid in human gastric carcinoma cells. Cancer Chemoth. Pharm. 2006, 58, 434–443. [Google Scholar] [CrossRef]
- Lu, N.; Yang, Y.; You, Q.D.; Ling, Y.; Gao, Y.; Gu, H.Y.; Zhao, L.; Wang, X.T.; Guo, Q.L. Gambogic Acid Inhibits angiogenesis through suppressing vascular endothelial growth factor-induced tyrosine phosphorylation of KDR/Flk-1. Cancer Lett. 2007, 258, 80–89. [Google Scholar] [CrossRef]
- Yi, T.F.; Yi, Z.F.; Sung, G.C.; Luo, J.; Manoj, K.P.; Bharat, B.A.; Liu, M.Y. Gambogic acid inhibits angiogenesis and prostate tumor growth by suppressing vascular endothelial growth factor receptor2 signaling. Cancer Res. 2008, 68, 1843–1850. [Google Scholar] [CrossRef]
- Annelii, N.; Monica, A.; Peter, C. Zebrafish and Xenopus tadpoles: Small animal models to study angiogenesis and lymphangiogenesis. Exp. Cell. Res. 2006, 312, 684–685. [Google Scholar] [CrossRef]
- Norrby, K. In vivo models of angiogenesis. J. Cell. Mol. Med. 2006, 10, 588–612. [Google Scholar] [CrossRef]
- Long, Q.; Meng, A.; Wang, H.; Jessen, J.R.; Farrell, M.J.; Lin, S. GATA-1 expression pattern can be recapitulated in living transgenic zebrafish using GFP reporter gene. Development 1997, 124, 4105–4111. [Google Scholar]
- Isogai, S.; Horiguchi, M. The vascular anatomy of the developing zebrafish: An atlas of embryonic and early larval development. Dev. Biol. 2001, 230, 278–301. [Google Scholar]
- Fan, Y.; Peng, A.H.; He, S.C.; Shao, X.; Nie, C.L.; Chen, L.J. Rho GTPases and vascular endothelial growth factor receptor 2 signalling pathway. J. Chemother. 2013, 25, 298–308. [Google Scholar] [CrossRef]
- Lin, L.J.; Lin, L.Z.; Pezzuto, J.M.; Cordell, G.A. Isogambogic acid and isomorellinol from Garcinia hanburyi. Magn. Reson. Chem. 1993, 31, 340–347. [Google Scholar] [CrossRef]
- Asano, J.; Chiba, K.; Tada, M.; Yoshi, T. Cytotoxic xanthones from Garcinia hanburyi. Phytochemistry 1996, 41, 815–820. [Google Scholar] [CrossRef]
- Karanjgaokar, C.G.; Nair, P.M.; Venkataraman, K. Coloring matters of Garcinia morella-(VI): Morellic, Isomorellic, and gambogic acids. Tetrahedron Lett. 1996, 7, 687–691. [Google Scholar]
- Tao, S.J.; Guan, S.H.; Wang, W.; Lu, Z.Q.; Chen, G.T.; Sha, N.; Yue, Q.X.; Liu, X.; Guo, D.A. Cytotoxic polyprenylated xanthones from the resin of Garcinia hanburyi. J. Nat. Prod. 2009, 72, 117–124. [Google Scholar] [CrossRef]
- Rajagopal, R.D.; Gurudutt, K.N.; Mamatha, S.; Mohan Rao, L.J. Guttiferic acid, A novel rearrangement product from minor chromenoxanthone pigments of Garcinia morella Desr. Magn. Reson. Chem. 2007, 45, 578–582. [Google Scholar] [CrossRef]
- Tisdale, E.J.; Slobodov, I.; Theodorakis, E.A. Biomimetic total synthesis of forbesione and desoxymorellin utilizing a tandem Claisen/Diels–Alder/Claisen rearrangement. Org. Biomol. Chem. 2003, 1, 4418–4422. [Google Scholar] [CrossRef]
- Alley, M.C.; Scudiero, D.A.; Monks, A.; Hursey, M.L.; Czerwinski, M.J.; Fine, D.L.; Abbott, B.J.; Mayo, J.G.; Shoemaker, R.H.; Boyd, M.R. Feasibility of drug screening with panels of human tumor cell lines using a microculture tetrazolium assay. Cancer Res. 1998, 48, 589–601. [Google Scholar]
- Zhao, C.J.; Yang, H.S.; Shi, H.S.; Wang, X.F.; Chen, X.C.; Yuan, Y.K. Distinct contributions of angiogenesis and vascular co-option during the initiation of primary microtumors and micrometastases. Carcinogenesis 2011, 32, 1143–1150. [Google Scholar] [CrossRef]
- Hu, J.; Ye, H.Y.; Fu, A.F.; Chen, X.; Wang, Y.S.; Chen, X.C.; Ye, X.; Xiao, W.J.; Duan, X.M.; Wei, Y.Q.; et al. Deguelin-an inhibitor to tumor lymphangiogenesis and lymphatic metastasis by downregulation of vascular endothelial cell growth factor-D in lung tumor model. Inter. J. Cancer 2010, 127, 2455–2466. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 1,3,7,9 are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, J.; He, S.; Li, S.; Zhang, R.; Peng, A.; Chen, L. In Vitro and In Vivo Antiangiogenic Activity of Caged Polyprenylated Xanthones Isolated from Garcinia hanburyi Hook. f. Molecules 2013, 18, 15305-15313. https://doi.org/10.3390/molecules181215305
Yang J, He S, Li S, Zhang R, Peng A, Chen L. In Vitro and In Vivo Antiangiogenic Activity of Caged Polyprenylated Xanthones Isolated from Garcinia hanburyi Hook. f. Molecules. 2013; 18(12):15305-15313. https://doi.org/10.3390/molecules181215305
Chicago/Turabian StyleYang, Jianhong, Shichao He, Shucai Li, Ronghong Zhang, Aihua Peng, and Lijuan Chen. 2013. "In Vitro and In Vivo Antiangiogenic Activity of Caged Polyprenylated Xanthones Isolated from Garcinia hanburyi Hook. f." Molecules 18, no. 12: 15305-15313. https://doi.org/10.3390/molecules181215305