In Vitro Antimicrobial Activity of Plant-Derived Diterpenes against Bovine Mastitis Bacteria
Abstract
:1. Introduction
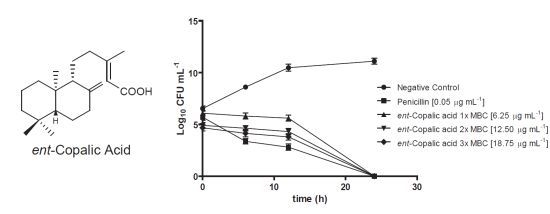
2. Results and Discussion

| Bacteria | MIC in µg mL−1 (MIC in µM) | |||
|---|---|---|---|---|
| Positive control 1 | MO | KA | CA 2 | |
| S. aureus (ATCC 6538) | 0.05 (0.05) | * | * | 6.25 (20.54) |
| S. aureus (clinical isolate) | >5.89 (>6.48) | 200.00 (689.03) | * | 6.25 (20.54) |
| E. coli (ATCC 14948) | >1.50 (>1.65) | * | * | * |
| E. coli (clinical isolate) | >2.95 (>3.25) | * | * | * |
| S. epidermidis (ATCC 12228) | 0.05 (0.05) | 100.00 (344.51) | 25.00 (82.78) | 6.25 (20.54) |
| S. agalactiae (ATCC 12386) | 0.05 (0.05) | 6.25 (21.53) | 3.12 (10.33) | 1.56 (5.13) |
| S. dysgalactiae (ATCC 12238) | 0.05 (0.05) | 6.25 (21.53) | 3.12 (10.33) | 1.56 (5.13) |

3. Experimental
3.1. Compound Isolation and Identification
3.2. Bacterial Strains and Antimicrobial Testing
3.3. Kill Kinetics
3.4. Cytotoxicity Assay
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Hillerton, J.E.; Berry, E.A. Treating mastitis in the cow—A tradition or an archaism. J. Appl. Microbiol. 2005, 98, 1250–1255. [Google Scholar] [CrossRef]
- Baskaran, S.A.; Kazmer, G.W.; Hinckley, L.; Andrew, S.M.; Venkitanarayanan, K. Antibacterial effect of plant-derived antimicrobials on major bacterial mastitis pathogens in vitro. J. Dairy Sci. 2009, 92, 1423–1429. [Google Scholar] [CrossRef]
- Laport, M.S.; Marinho, P.R.; da Silva Santos, O.C.; de Almeida, P.; Villela Romanos, M.T.; Muricy, G.; Vasconcelos Paiva Brito, M.A.; Giambiagi-deMarval, M. Antimicrobial activity of marine sponges against coagulase-negative staphylococci isolated from bovine mastitis. Vet. Microbiol. 2012, 155, 362–368. [Google Scholar] [CrossRef]
- Newman, D.J. Natural products as leads to potential drugs: An old process or the new hope for drug discovery? J. Med. Chem. 2008, 51, 2589–2599. [Google Scholar] [CrossRef]
- Almeida, L.S.B.; Murata, R.M.; Yatsuda, R.; Dos Santos, M.H.; Nagem, T.J.; Alencar, S.M.; Koo, H.; Rosalen, P.L. Antimicrobial activity of Rheedia brasiliensis and 7-epiclusianone against Streptococcus mutans. Phytomedicine 2008, 15, 886–891. [Google Scholar] [CrossRef]
- Kuzma, L.; Rozalski, M.; Walencka, E.; Rozalska, B.; Wysokinska, H. Antimicrobial activity of diterpenoids from hairy roots of Salvia sclarea L.: Salvipisone as a potential anti-biofilm agent active against antibiotic resistant Staphylococci. Phytomedicine 2007, 14, 31–35. [Google Scholar]
- Porto, T.S.; Furtado, N.A.J.C.; Heleno, V.C.G.; Martins, C.H.G.; Da Costa, F.B.; Severiano, M.E.; Silva, A.N.; Veneziani, R.C.S.; Ambrosio, S.R. Antimicrobial ent-pimarane diterpenes from Viguiera arenaria against Gram-positive bacteria. Fitoterapia 2009, 80, 432–436. [Google Scholar] [CrossRef]
- Porto, T.S.; Rangel, R.; Furtado, N.; De Carvalho, T.C.; Martins, C.H.G.; Veneziani, R.C.S.; Da Costa, F.B.; Vinholis, A.H.C.; Cunha, W.R.; Heleno, V.C.G.; et al. Pimarane-type diterpenes: antimicrobial activity against oral pathogens. Molecules 2009, 14, 191–199. [Google Scholar] [CrossRef]
- Habtemariam, S. In vitro antileishmanial effects of antibacterial diterpenes from two Ethiopian Premna species: P. schimperi and P. oligotricha. BMC Pharmacol. 2003, 3, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Kalpoutzakis, E.; Chinou, I.; Mitaku, S.; Skaitsounis, A.L.; Hervala, C. Antibacterial labdane-type diterpenes from the resin "Ladano" of Cistus creticus Subsp. creticus. Nat. Prod. Lett. 1998, 11, 173–179. [Google Scholar] [CrossRef]
- Radulovic, N.; Denic, M.; Stojanovic-Radic, Z. Antimicrobial phenolic abietane diterpene from Lycopus europaeus L. (Lamiaceae). Bioorg Med. Chem. Lett. 2010, 20, 4988–4991. [Google Scholar] [CrossRef]
- Stavri, M.; Paton, A.; Skelton, B.W.; Gibbons, S. Antibacterial diterpenes from Plectranthus ernstii. J. Nat. Prod. 2009, 72, 1191–1194. [Google Scholar]
- Wiart, C.; Au, T.S.; Mohd, Y.; Hamimah, H.; Sulaiman, M. 16 Alpha hydroxy-(-)-kauran-19-oic: An antibacterial diterpene from sweet apple (Annona squamosa L., Annonaceae). Int. J. Pharmacol. 2005, 1, 296–298. [Google Scholar] [CrossRef]
- Woldemichael, G.M.; Wächter, G.; Singh, M.P.; Maiese, W.M.; Timmermann, B.N. Antibacterial diterpenes from Calceolaria pinifolia. J. Nat. Prod. 2003, 66, 242–246. [Google Scholar] [CrossRef]
- Ambrosio, S.R.; Furtado, N.A.J.C.; De Oliveira, D.C.R.; Da Costa, F.B.; Martins, C.H.G.; De Carvalho, T.C.; Porto, T.S.; Veneziani, R.C.S. Antimicrobial activity of kaurane diterpenes against oral pathogens. Z. Naturforsch. C 2008, 63c, 326–330. [Google Scholar]
- De Andrade, B.B.; Moreira, M.R.; Ambrosio, S.R.; Furtado, N.A.J.C.; Cunha, W.R.; Martins, C.H.G.; Veneziani, R.C.S. Evaluation of ent-kaurenoic acid derivatives for their anticariogenic activity. Nat. Prod. Commun. 2011, 6, 777–780. [Google Scholar]
- Souza, A.B.; De Souza, M.G.M.; Moreira, M.A.; Moreira, M.R.; Furtado, N.A.J.C.; Martins, C.H.G.; Bastos, J.K.; Santos, R.A.; Heleno, V.C.G.; Ambrosio, S.R.; et al. Antimicrobial evaluation of diterpenes from Copaifera langsdorffii oleoresin against periodontal anaerobic bacteria. Molecules 2011, 16, 9611–9619. [Google Scholar] [CrossRef]
- Souza, A.B.; Martins, C.H.G.; Souza, M.G.M.; Furtado, N.A.J.C.; Heleno, V.C.G.; Sousa, J.P.B.; Rocha, E.M.P.; Bastos, J.K.; Cunha, W.R.; Veneziani, R.C.S.; et al. Antimicrobial activity of terpenoids from Copaifera langsdorffii Desf. against cariogenic bacteria. Phytother. Res. 2011, 25, 215–220. [Google Scholar]
- Bastard, J.; Duc, D.K.; Fetizon, M.; Francis, M.J.; Grant, P.K.; Weavers, R.T.; Kaneko, C.; Baddeley, G.V.; Bernassau, J.M.; Burfitt, I.R.; et al. C-13 Nuclear Magnetic-Resonance Spectroscopy of Naturally-Occurring Substances .81. Cmr Spectroscopy of Labdanic Diterpenes and Related Substances. J. Nat. Prod. 1984, 47, 592–599. [Google Scholar] [CrossRef]
- Scheffran, R.H.; HSu, R.C.; Su, N.Y.; Huffman, J.B.; Midland, S.L.; Sims, J.J. Allelochemical resistence of Bald cypress Taxodium distichum, heartwood to the subterranean termite, Coptotermes formosanu. J. Chem. Ecol. 1988, 14, 765–776. [Google Scholar] [CrossRef]
- Do Nascimento, A.M.; De Oliveira, D.C.R. Kaurene Diterpenes and Other Chemical Constituents from Mikania stipulacea (M. Vahl) Willd. J. Brazil. Chem. Soc. 2001, 12, 552–555. [Google Scholar] [CrossRef]
- Ohsaki, A.; Yan, L.T.; Ito, S.; Edatsugi, H.; Iwata, D.; Komoda, Y. The Isolation and in-Vivo Potent Antitumor-Activity of Clerodane Diterpenoid from the Oleoresin of the Brazilian Medicinal Plant, Copaifera langsdorfii Desfon. Bioorg. Med. Chem. 1994, 4, 2889–2892. [Google Scholar] [CrossRef]
- Gibbons, S. Phytochemicals for bacterial resistance—Strengths, weaknesses and opportunities. Planta. Med. 2008, 74, 594–602. [Google Scholar] [CrossRef]
- Rios, J.L.; Recio, M.C. Medicinal plants and antimicrobial activity. J. Ethnopharmacol 2005, 100, 80–84. [Google Scholar] [CrossRef]
- Gibbons, S. Anti-staphylococcal plant natural products. Nat. Prod. Rep. 2004, 21, 263–277. [Google Scholar] [CrossRef]
- Santana, H.F.; Barbosa, A.A.T.; Ferreira, S.O.; Mantovani, H.C. Bactericidal activity of ethanolic extracts of propolis against Staphylococcus aureus isolated from mastitic cows. World J. Microbiol. Biotechnol. 2012, 28, 485–491. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI). Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically, In Approved Standard—Eighth Edition, CLSI Document M7-A8; CLSI: Wayne, PA, USA, 2009. [Google Scholar]
- D'Arrigo, M.; Ginestra, G.; Mandalari, G.; Furneri, P.M.; Bisignano, G. Synergism and postantibiotic effect of tobramycin and Melaleuca alternifolia (tea tree) oil against Staphylococcus aureus and Escherichia coli. Phytomedicine 2010, 17, 317–322. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fonseca, A.P.; Estrela, F.T.; Moraes, T.S.; Carneiro, L.J.; Bastos, J.K.; Santos, R.A.d.; Ambrósio, S.R.; Martins, C.H.G.; Veneziani, R.C.S. In Vitro Antimicrobial Activity of Plant-Derived Diterpenes against Bovine Mastitis Bacteria. Molecules 2013, 18, 7865-7872. https://doi.org/10.3390/molecules18077865
Fonseca AP, Estrela FT, Moraes TS, Carneiro LJ, Bastos JK, Santos RAd, Ambrósio SR, Martins CHG, Veneziani RCS. In Vitro Antimicrobial Activity of Plant-Derived Diterpenes against Bovine Mastitis Bacteria. Molecules. 2013; 18(7):7865-7872. https://doi.org/10.3390/molecules18077865
Chicago/Turabian StyleFonseca, Ariana P., Fernanda T. Estrela, Thaís S. Moraes, Luiza J. Carneiro, Jairo K. Bastos, Raquel A. dos Santos, Sérgio R. Ambrósio, Carlos H. G. Martins, and Rodrigo C. S. Veneziani. 2013. "In Vitro Antimicrobial Activity of Plant-Derived Diterpenes against Bovine Mastitis Bacteria" Molecules 18, no. 7: 7865-7872. https://doi.org/10.3390/molecules18077865