Variation of Vitamin D in Cow’s Milk and Interaction with β-Lactoglobulin
Abstract
:1. Introduction
2. Results and Discussion
2.1. Content of Vitamin D in Commercial Milk Samples
2.2. Content of Vitamin D3 in Individual Milk Samples: Differences between Herds
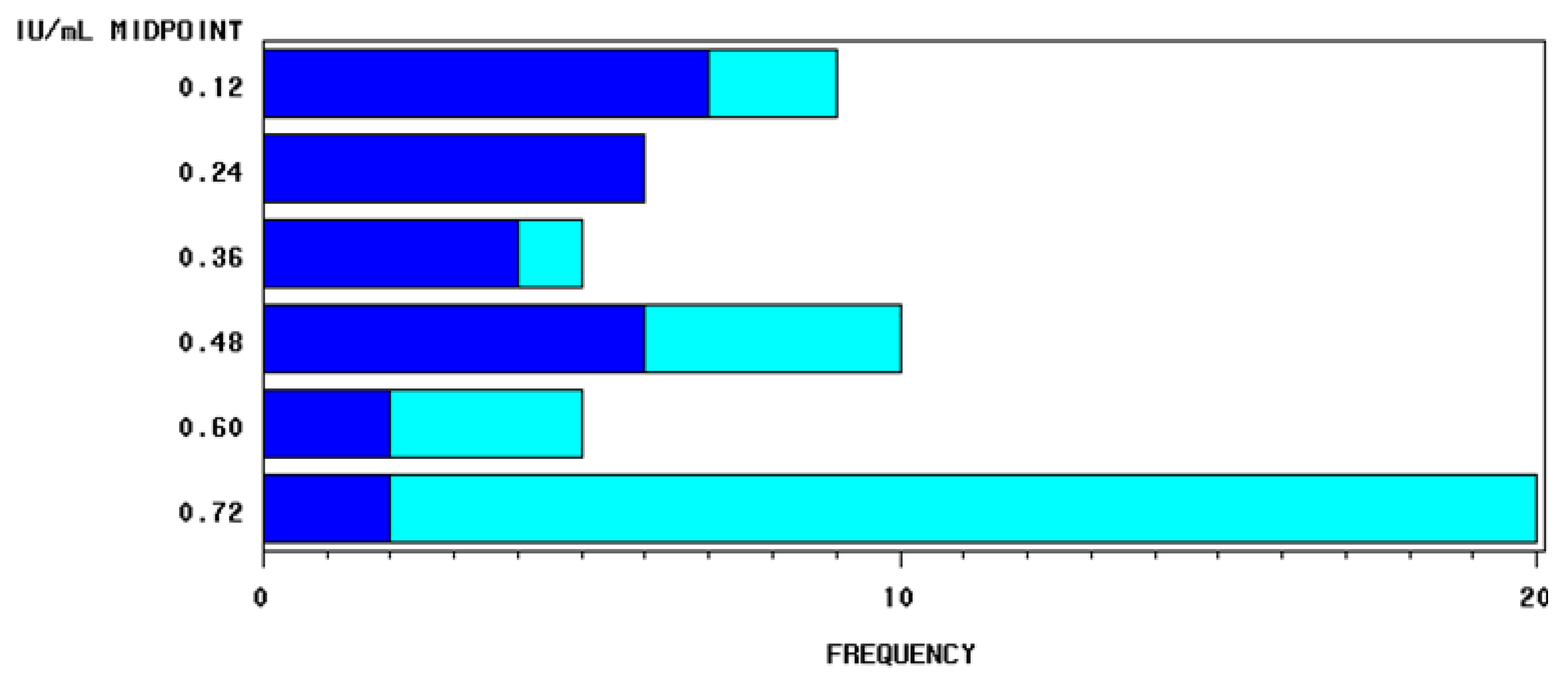
| Vitamin D3 | Herd A | Herd B | Herd A + B | |||
|---|---|---|---|---|---|---|
| n = 27 | n = 28 | n = 55 | ||||
| Measure unit | IU/mL | µg/mL | IU/mL | µg/mL | IU/mL | µg/mL |
| Mean | 0 .341 | 0 .008 | 0 .629 | 0 .016 | 0.487 | 0.012 |
| Standard Deviation | 0.192 | 0.005 | 0.184 | 0.005 | 0.237 | 0.006 |
| Median | 0 .311 | 0 .008 | 0 .750 | 0 .019 | 0.505 | 0.013 |
| Minimum value | 0.125 | 0.0031 | 0.125 | 0.0031 | 0.125 | 0.0031 |
| Maximum value | 0.750 | 0.0187 | 0.750 | 0.0187 | 0.750 | 0.0187 |
| Samples ≤ 0.125 IU/mL | 10.91% | 1.82% | 12.73% | |||
| Samples within the range of sensibility | 34.55% | 20.00% | 54.55% | |||
| Samples ≥ 0.75 IU/mL | 3.64% | 29.09% | 32.73% | |||
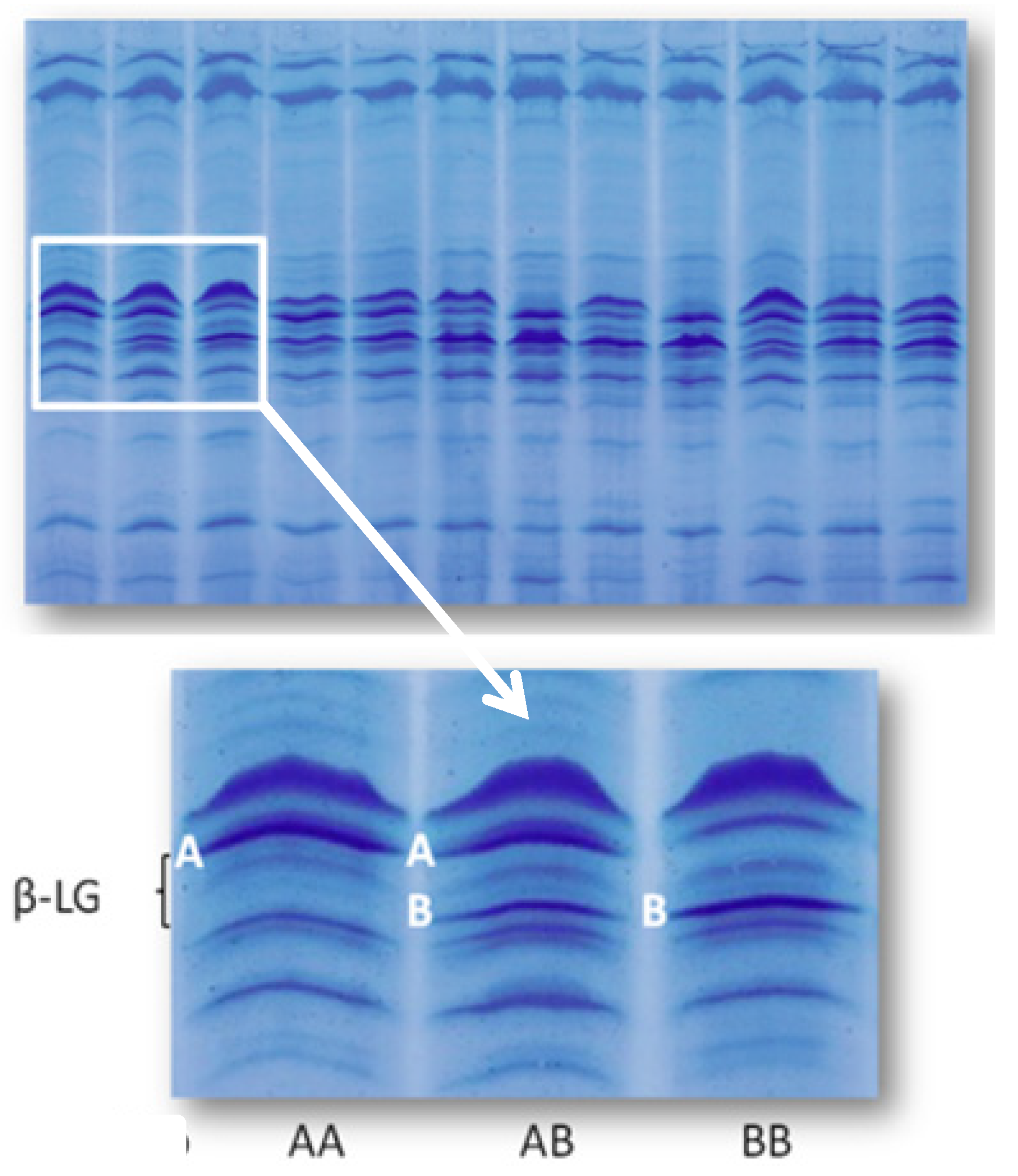
2.3. Content of Vitamin D3 in Individual Milk Samples: Physiological and Genetic Differences
| β-LG genotype | Measure unit | Number | LSM | Standard error |
|---|---|---|---|---|
| AA | μg/mL | 19 | 0.015 a | 0.001 |
| AB + BB | μg/mL | 36 | 0.011 b | 0.001 |
| AA | IU/mL | 19 | 0.602 a | 0.051 |
| AB + BB | IU/mL | 36 | 0.427 b | 0.037 |
3. Experimental
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Gombart, A.F. The vitamin D-antimicrobial peptide pathway and its role in protection against infection. Future Microbiol. 2009, 4, 1151–1165. [Google Scholar] [CrossRef]
- Webb, A.R.; Pilbeam, C.; Hanafin, N.; Holick, M.F. An evaluation of the relative contributions of exposure to sunlight and of diet to the circulating concentrations of 25-hydroxyvitamin D in an elderly nursing home population in Boston. Am. J. Clin. Nutr. 1990, 51, 1075–1081. [Google Scholar]
- Adami, S.; Romagnoli, E.; Carnevale, V.; Scillitani, A.; Giusti, A.; Rossini, M.; Gatti, D.; Nuti, R.; Minisola, S. Guidelines on prevention and treatment of vitamin D deficiency. Reumatismo 2011, 63, 129–147. [Google Scholar]
- Caroli, A.; Poli, A.; Ricotta, D.; Banfi, G.; Cocchi, D. Invited review: Dairy intake and bone health: A viewpoint from the state of the art. J. Dairy Sci. 2011, 94, 5249–5262. [Google Scholar] [CrossRef]
- DeLuca, H.F. The vitamin D story: A collaborative effort of basic science and clinical medicine. FASEB J. 1988, 2, 224–236. [Google Scholar]
- Reichel, H.; Koeffler, H.P.; Norman, A.W. The role of vitamin D endocrine system in health and disease. N. Engl. J. Med. 1989, 320, 980–991. [Google Scholar] [CrossRef]
- Nagpal, S.; Na, S.; Rathnachalam, R. Noncalcemic actions of vitamin D receptor ligands. Endocr. Rev. 2005, 26, 662–687. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Giovannucci, E.; Willett, W.C.; Dietrich, T.; Dawson-Hughes, B. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am. J. Clin. Nutr. 2006, 84, 18–28. [Google Scholar]
- Stroud, M.L.; Stilgoe, S.; Stott, V.E.; Alhabian, O.; Salman, K. Vitamin D—A review. Aust. Fam. Physician 2008, 37, 1002–1005. [Google Scholar]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Calvo, M.S.; Whiting, S.J.; Barton, C.N. Vitamin D intake: A global perspective of current status. J. Nutr. 2005, 135, 310–316. [Google Scholar]
- Chen, T.C.; Chimeh, F.; Lu, Z.; Mathieu, J.; Person, K.S.; Zhang, A.; Kohn, N.; Martinello, S.; Berkowitz, R.; Holick, M.F. Factors that influence the cutaneous synthesis and dietary sources of vitamin D. Arch. Biochem. Biophys. 2007, 460, 213–217. [Google Scholar] [CrossRef]
- Ross, A.C.; Manson, J.E.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: What clinicians need to know. J. Clin. Endocrinol. Metab. 2011, 96, 53–58. [Google Scholar] [CrossRef]
- Moore, C.; Murphy, M.M.; Keast, D.R.; Holick, M.F. Vitamin D intake in the United States. J. Am. Diet. Assoc. 2004, 104, 980–983. [Google Scholar] [CrossRef]
- Mattila, P.H.; Piironen, V.I.; Uusi-Rauva, E.J.; Koivistoinen, P.E. Contents of cholecalciferol, ergocalciferol, and their 25-hydroxylated metabolites in milk products and raw meat and liver as determined by HPLC. J. Agric. Food Chem. 1995, 43, 2394–2399. [Google Scholar] [CrossRef]
- Jakobsen, J. Bioavailability and bioactivity of vitamin D3 active compounds—Which potency should be used for 25-hydroxyvitamin D3? Int. Congr. Ser. 2007, 1297, 133–142. [Google Scholar] [CrossRef]
- FDA. PMO 2007: Appendix O: Vitamin fortification of fluid milk products. 2007. Available online: http://www.fda.gov/Food/GuidanceRegulation/GuidanceDocumentsRegulatoryInformation/Milk/ucm089032.htm (accessed on 24 June 2013). [Google Scholar]
- Fulgoni, V., III; Nicholls, J.; Reed, A.; Buckley, R.; Kafer, K.; Huth, P.; Dirienzo, D.; Miller, G. Dairy consumption and related nutrient intake in African-American adults and children in the United States: Continuing survey of food intakes by individuals 1994–1996, 1998, and the National Health and Nutrition Examination Survey 1999–2000. J. Am. Diet. Assoc. 2007, 107, 256–264. [Google Scholar]
- Reeve, L.E.; Jorgensen, N.A.; DeLuca, H.F. Vitamin D compounds in cows’ milk. J. Nutr. 1982, 112, 667–672. [Google Scholar]
- National Research Council. Nutrient Requirements of Dairy Cattle, 5th ed.; National Academy of Sciences: Washington, DC, USA, 1978. [Google Scholar]
- National Research Council. Nutrient Requirements of Dairy Cattle, 7th ed.; National Academy of Sciences: Washington, DC, USA, 2001. [Google Scholar]
- Horst, R.L.; Goff, J.P.; Reinhardt, T.A. Calcium and vitamin D metabolism in the dairy cow. J. Dairy Sci. 1994, 77, 1936–1951. [Google Scholar] [CrossRef]
- National Research Council. Nutrient Requirements of Dairy Cattle, 6th ed.; National Academy of Sciences: Washington, DC, USA, 1989. [Google Scholar]
- Littledike, E.T.; Horst, R.L. Problems with vitamin D injections for prevention of milk fever: Toxicity of large doses and increased incidence of small doses. J. Dairy Sci. 1980, 63, 89. [Google Scholar]
- Ali, A.K.A.; Shook, G.E. An optimum transformation for somatic cell concentration in milk. J. Dairy Sci. 1980, 63, 487–490. [Google Scholar] [CrossRef]
- Zecconi, A. Le cellule somatiche nel latte influenzano sanità e qualità. L’Informatore Agrario 2007, 8, 66–69. [Google Scholar]
- Farrell, H.M., Jr; Jimenez-Flores, R.; Bleck, G.T.; Brown, E.M.; Butler, J.E.; Creamer, L.K.; Hicks, C.L.; Hollar, C.M.; Ng-Kwai-Hang, F.; Swaisgood, H.E. Nomenclature of the proteins of cows’ milk-sixth edition. J. Dairy Sci. 2004, 87, 1641–1674. [Google Scholar] [CrossRef]
- Caroli, A.M.; Chessa, S.; Erhardt, G.J. Milk protein polymorphisms in cattle: Effect on animal breeding and human nutrition. J. Dairy Sci. 2009, 92, 5335–5352. [Google Scholar]
- Cerbulis, J.; Farrell, H.M., Jr. Composition of milks of dairy cattle. I. Protein, lactose, and fat contents and distribution of protein fraction. J. Dairy Sci. 1975, 58, 817–827. [Google Scholar] [CrossRef]
- Wang, Q.; Allen, J.C.; Swaisgood, H.E. Binding of vitamin D and cholesterol to beta-lactoglobulin. J. Dairy Sci. 1997, 80, 1054–1059. [Google Scholar] [CrossRef]
- Forrest, S.A.; Yada, R.Y.; Rousseau, D. Interactions of vitamin D3 with bovine β-lactoglobulin A and β-casein. J. Agric. Food Chem. 2005, 53, 8003–8009. [Google Scholar] [CrossRef]
- Erhardt, G.; Juszczak, J.; Panicke, L.; Krick-Saleck, H. Genetic polymorphism of milk proteins in Polish Red Cattle: A new genetic variant of beta-lactoglobulin. J. Anim. Breed. Genet. 1998, 115, 63–71. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bulgari, O.; Caroli, A.M.; Chessa, S.; Rizzi, R.; Gigliotti, C. Variation of Vitamin D in Cow’s Milk and Interaction with β-Lactoglobulin. Molecules 2013, 18, 10122-10131. https://doi.org/10.3390/molecules180910122
Bulgari O, Caroli AM, Chessa S, Rizzi R, Gigliotti C. Variation of Vitamin D in Cow’s Milk and Interaction with β-Lactoglobulin. Molecules. 2013; 18(9):10122-10131. https://doi.org/10.3390/molecules180910122
Chicago/Turabian StyleBulgari, Omar, Anna Maria Caroli, Stefania Chessa, Rita Rizzi, and Carmen Gigliotti. 2013. "Variation of Vitamin D in Cow’s Milk and Interaction with β-Lactoglobulin" Molecules 18, no. 9: 10122-10131. https://doi.org/10.3390/molecules180910122




