α-Solanine Inhibits Invasion of Human Prostate Cancer Cell by Suppressing Epithelial-Mesenchymal Transition and MMPs Expression
Abstract
:1. Introduction
2. Results and Discussion
2.1. Cytotoxic Effect of α-Solanine in PC-3 Cells

2.2. Effects of α-Solanine on Inhibiting Migration and Invasion of PC-3 Cells


2.3. α-Solanine Exerts Reversion Effect on EMT Markers in PC-3 Cells

2.4. α-Solanine Decreases Expression of MMP-2, MMP-9 and EMMPRIN and Induces Expression of RECK, TIMP-1 and TIMP-2 in PC-3 Cells
| Gene | Sequence (5'–3') |
|---|---|
| MMP-2-F | CTTCCAAGTCTGGAGCGATGT |
| MMP-2-R | TACCGTCAAAGGGGTATCCAT |
| MMP-9-F | GGGACGCAGACATCGTCATC |
| MMP-9-R | TCGTCATCGTCGAAATGGGC |
| EMMPRIN-F | CTACACATTGAGAACCTGAACAT |
| EMMPRIN-R | TTCTCGTAGATGAAGATGATGGT |
| RECK-F | CCTGCATTGCTCGCTGTGTG |
| RECK-R | CCTGTGGTTTGGGTATGCACCTT |
| TIMP-1-F | CTTCTGCAATTCCGACCTCGT |
| TIMP-1-R | CCCTAAGGCTTGGAACCCTTT |
| TIMP-2-F | AAGCGGTCAGTGAGAAGGAAG |
| TIMP-2-R | CACACACTACCGAGGAGGG |
| β-actin-F | CATGTACGTTGCTATCCAGGC |
| β-actin-R | CTCCTTAATGTCACGCACGAT |
| miR-21 | CGGCGGTAGCTTATCAGACTG A |
| miR-138 | AACGGAGCTGGTGTTGTGAAT C |
| RNU6B | TTCCTCCGCAAGGATGACACG C |

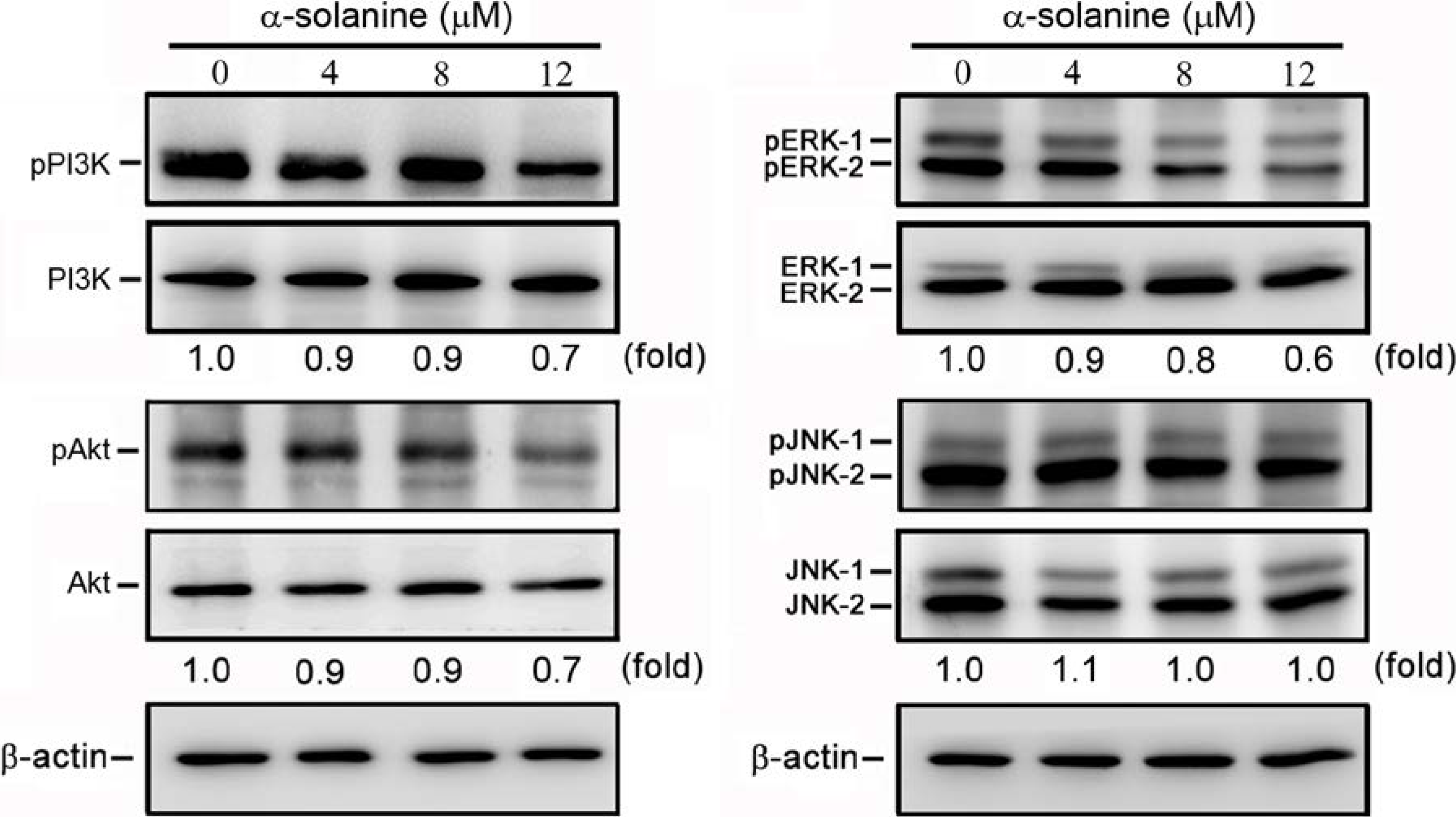
2.5. α-Solanine Inhibits Phosphorylation of PI3K, Akt and ERK


2.6. α-Solanine Down-regulates Expression of miR-21 and Up-regulates Expression of miR-138

2.7. Discussion
3. Experimental
3.1. Reagents and Cell Culture
3.2. Cell Viability Assay
3.3. Wound Healing Migration Assay
3.4. Boyden Chamber Invasion Assay
3.5. RNA Extraction and Reverse Transcription PCR and Quantitative Real-time PCR
3.6. Western Blotting
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Son, Y.O.; Kim, J.; Lim, J.C.; Chung, Y.; Chung, G.H.; Lee, J.C. Ripe fruit of Solanum nigrum L. inhibits cell growth and induces apoptosis in MCF-7 cells. Food Chem. Toxicol. 2003, 41, 1421–1428. [Google Scholar] [CrossRef]
- Ji, Y.B.; Gao, S.Y.; Ji, C.F.; Zou, X. Induction of apoptosis in HepG2 cells by solanine and Bcl-2 protein. J. Ethnopharmacol. 2008, 115, 194–202. [Google Scholar]
- Lin, H.M.; Tseng, H.C.; Wang, C.J.; Chyau, C.C.; Liao, K.K.; Peng, P.L.; Chou, F.P. Induction of autophagy and apoptosis by the extract of Solanum nigrum Linn in HepG2 cells. J. Agric. Food Chem. 2007, 55, 3620–3628. [Google Scholar]
- Jiang, L.; Liu, X.; Kolokythas, A.; Yu, J.; Wang, A.; Heidbreder, C.E.; Shi, F.; Zhou, X. Downregulation of the Rho GTPase signaling pathway is involved in the microRNA-138-mediated inhibition of cell migration and invasion in tongue squamous cell carcinoma. Int. J. Cancer 2010, 127, 505–512. [Google Scholar]
- Friedman, M. Potato glycoalkaloids and metabolites: Roles in the plant and in the diet. J. Agric. Food Chem. 2006, 54, 8655–8681. [Google Scholar]
- Lee, K.R.; Kozukue, N.; Han, J.S.; Park, J.H.; Chang, E.Y.; Baek, E.J.; Chang, J.S.; Friedman, M. Glycoalkaloids and metabolites inhibit the growth of human colon (HT29) and liver (HepG2) cancer cells. J. Agric. Food Chem. 2004, 52, 2832–2839. [Google Scholar]
- Friedman, M.; Lee, K.R.; Kim, H.J.; Lee, I.S.; Kozukue, N. Anticarcinogenic effects of glycoalkaloids from potatoes against human cervical, liver, lymphoma, and stomach cancer cells. J. Agric. Food Chem. 2005, 53, 6162–6169. [Google Scholar] [CrossRef]
- Yang, S.A.; Paek, S.H.; Kozukue, N.; Lee, K.R.; Kim, J.A. Alpha-chaconine, a potato glycoalkaloid, induces apoptosis of HT-29 human colon cancer cells through caspase-3 activation and inhibition of ERK 1/2 phosphorylation. Food Chem. Toxicol. 2006, 44, 839–846. [Google Scholar] [CrossRef]
- Mohsenikia, M.; Alizadeh, A.M.; Khodayari, S.; Khodayari, H.; Kouhpayeh, S.A.; Karimi, A.; Zamani, M.; Azizian, S.; Mohagheghi, M.A. The protective and therapeutic effects of alpha-solanine on mice breast cancer. Eur. J. Pharmacol. 2013, 718, 1–9. [Google Scholar]
- Lu, M.K.; Shih, Y.W.; Chang Chien, T.T.; Fang, L.H.; Huang, H.C.; Chen, P.S. alpha-Solanine inhibits human melanoma cell migration and invasion by reducing matrix metalloproteinase-2/9 activities. Biol. Pharm. Bull. 2010, 33, 1685–1691. [Google Scholar]
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Thun, M.J. Cancer statistics, 2009. CA-Cancer J. Clin. 2009, 59, 225–249. [Google Scholar] [CrossRef]
- Salesi, N.; Carlini, P.; Ruggeri, E.M.; Ferretti, G.; Bria, E.; Cognetti, F. Prostate cancer: The role of hormonal therapy. J. Exp. Clin. Cancer Res. 2005, 24, 175–180. [Google Scholar]
- Arnold, J.T.; Isaacs, J.T. Mechanisms involved in the progression of androgen-independent prostate cancers: It is not only the cancer cell’s fault. Endocr.-Relat. Cancer 2002, 9, 61–73. [Google Scholar] [CrossRef]
- Gupta, G.P.; Massague, J. Cancer metastasis: Building a framework. Cell 2006, 127, 679–695. [Google Scholar]
- Meyer, T.; Hart, I.R. Mechanisms of tumour metastasis. Eur. J. Cancer 1998, 34, 214–221. [Google Scholar]
- Tsai, J.H.; Yang, J. Epithelial-mesenchymal plasticity in carcinoma metastasis. Gene. Dev. 2013, 27, 2192–2206. [Google Scholar]
- Wells, A.; Chao, Y.L.; Grahovac, J.; Wu, Q.; Lauffenburger, D.A. Epithelial and mesenchymal phenotypic switchings modulate cell motility in metastasis. Front. Biosci. 2011, 16, 815–837. [Google Scholar]
- Zeisberg, M.; Neilson, E.G. Biomarkers for epithelial-mesenchymal transitions. J. Clin. Invest. 2009, 119, 1429–1437. [Google Scholar]
- Itoh, Y.; Nagase, H. Matrix metalloproteinases in cancer. Essays Biochem. 2002, 38, 21–36. [Google Scholar]
- Bernhard, E.J.; Gruber, S.B.; Muschel, R.J. Direct evidence linking expression of matrix metalloproteinase 9 (92-kDa gelatinase/collagenase) to the metastatic phenotype in transformed rat embryo cells. Proc. Natl. Acad. Sci. USA 1994, 91, 4293–4297. [Google Scholar]
- Emmert-Buck, M.R.; Roth, M.J.; Zhuang, Z.; Campo, E.; Rozhin, J.; Sloane, B.F.; Liotta, L.A.; Stetler-Stevenson, W.G. Increased gelatinase A (MMP-2) and cathepsin B activity in invasive tumor regions of human colon cancer samples. Am. J. Pathol. 1994, 145, 1285–1290. [Google Scholar]
- Mook, O.R.; Frederiks, W.M.; van Noorden, C.J. The role of gelatinases in colorectal cancer progression and metastasis. BBA-Rev. Cancer 2004, 1705, 69–89. [Google Scholar]
- Hao, J.L.; Cozzi, P.J.; Khatri, A.; Power, C.A.; Li, Y. CD147/EMMPRIN and CD44 are potential therapeutic targets for metastatic prostate cancer. Curr. Cancer Drug Targets 2010, 10, 287–306. [Google Scholar]
- Clark, J.C.; Thomas, D.M.; Choong, P.F.; Dass, C.R. RECK—A newly discovered inhibitor of metastasis with prognostic significance in multiple forms of cancer. Cancer Metast. Rev. 2007, 26, 675–683. [Google Scholar]
- Takenaka, K.; Ishikawa, S.; Kawano, Y.; Yanagihara, K.; Miyahara, R.; Otake, Y.; Morioka, Y.; Takahashi, C.; Noda, M.; Wada, H.; et al. Expression of a novel matrix metalloproteinase regulator, RECK, and its clinical significance in resected non-small cell lung cancer. Eur. J. Cancer 2004, 40, 1617–1623. [Google Scholar]
- Sun, J. Matrix metalloproteinases and tissue inhibitor of metalloproteinases are essential for the inflammatory response in cancer cells. J. Signal Transd. 2010, 2010. [Google Scholar] [CrossRef]
- Zhao, Y.; Srivastava, D. A developmental view of microRNA function. Trends Biochem. Sci. 2007, 32, 189–197. [Google Scholar]
- Kumar, M.S.; Lu, J.; Mercer, K.L.; Golub, T.R.; Jacks, T. Impaired microRNA processing enhances cellular transformation and tumorigenesis. Nat. Genet. 2007, 39, 673–677. [Google Scholar]
- Esquela-Kerscher, A.; Slack, F.J. Oncomirs-microRNAs with a role in cancer. Nat. Rev. Cancer 2006, 6, 259–269. [Google Scholar]
- Manikandan, J.; Aarthi, J.J.; Kumar, S.D.; Pushparaj, P.N. Oncomirs: The potential role of non-coding microRNAs in understanding cancer. Bioinformation 2008, 2, 330–334. [Google Scholar]
- Krichevsky, A.M.; Gabriely, G. miR-21: A small multi-faceted RNA. J. Cell. Mol. Med. 2009, 13, 39–53. [Google Scholar]
- Asangani, I.A.; Rasheed, S.A.; Nikolova, D.A.; Leupold, J.H.; Colburn, N.H.; Post, S.; Allgayer, H. MicroRNA-21 (miR-21) post-transcriptionally downregulates tumor suppressor Pdcd4 and stimulates invasion, intravasation and metastasis in colorectal cancer. Oncogene 2008, 27, 2128–2136. [Google Scholar]
- Meng, F.; Henson, R.; Wehbe-Janek, H.; Ghoshal, K.; Jacob, S.T.; Patel, T. MicroRNA-21 regulates expression of the PTEN tumor suppressor gene in human hepatocellular cancer. Gastroenterology 2007, 133, 647–658. [Google Scholar]
- Reis, S.T.; Pontes-Junior, J.; Antunes, A.A.; Dall’Oglio, M.F.; Dip, N.; Passerotti, C.C.; Rossini, G.A.; Morais, D.R.; Nesrallah, A.J.; Piantino, C.; et al. miR-21 may acts as an oncomir by targeting RECK, a matrix metalloproteinase regulator, in prostate cancer. BMC Urol. 2012, 12, 14. [Google Scholar] [CrossRef] [Green Version]
- Han, M.; Wang, Y.; Liu, M.; Bi, X.; Bao, J.; Zeng, N.; Zhu, Z.; Mo, Z.; Wu, C.; Chen, X. MiR-21 regulates epithelial-mesenchymal transition phenotype and hypoxia-inducible factor-1alpha expression in third-sphere forming breast cancer stem cell-like cells. Cancer Sci. 2012, 103, 1058–1064. [Google Scholar]
- Mitomo, S.; Maesawa, C.; Ogasawara, S.; Iwaya, T.; Shibazaki, M.; Yashima-Abo, A.; Kotani, K.; Oikawa, H.; Sakurai, E.; Izutsu, N.; et al. Downregulation of miR-138 is associated with overexpression of human telomerase reverse transcriptase protein in human anaplastic thyroid carcinoma cell lines. Cancer Sci. 2008, 99, 280–286. [Google Scholar] [CrossRef]
- Jin, Y.; Chen, D.; Cabay, R.J.; Wang, A.; Crowe, D.L.; Zhou, X. Role of microRNA-138 as a potential tumor suppressor in head and neck squamous cell carcinoma. Int. Rev. Cell Mol. Biol. 2013, 303, 357–385. [Google Scholar]
- Yeh, Y.M.; Chuang, C.M.; Chao, K.C.; Wang, L.H. MicroRNA-138 suppresses ovarian cancer cell invasion and metastasis by targeting SOX4 and HIF-1alpha. Int. J.Cancer 2013, 133, 867–878. [Google Scholar]
- Liu, X.; Wang, C.; Chen, Z.; Jin, Y.; Wang, Y.; Kolokythas, A.; Dai, Y.; Zhou, X. MicroRNA-138 suppresses epithelial-mesenchymal transition in squamous cell carcinoma cell lines. Biochem. J. 2011, 440, 23–31. [Google Scholar]
- Chan-Hui, P.Y.; Weaver, R. Human mitogen-activated protein kinase kinase kinase mediates the stress-induced activation of mitogen-activated protein kinase cascades. Biochem. J. 1998, 336, 599–609. [Google Scholar]
- Reddy, K.B.; Nabha, S.M.; Atanaskova, N. Role of MAP kinase in tumor progression and invasion. Cancer Metast. Rev. 2003, 22, 395–403. [Google Scholar]
- Chen, P.S.; Shih, Y.W.; Huang, H.C.; Cheng, H.W. Diosgenin, a steroidal saponin, inhibits migration and invasion of human prostate cancer PC-3 cells by reducing matrix metalloproteinases expression. PLoS One 2011, 6, e20164. [Google Scholar]
- Chen, P.N.; Hsieh, Y.S.; Chiou, H.L.; Chu, S.C. Silibinin inhibits cell invasion through inactivation of both PI3K-Akt and MAPK signaling pathways. Chem. Biol. Interact. 2005, 156, 141–150. [Google Scholar]
- Shih, Y.W.; Shieh, J.M.; Wu, P.F.; Lee, Y.C.; Chen, Y.Z.; Chiang, T.A. Alpha-tomatine inactivates PI3K/Akt and ERK signaling pathways in human lung adenocarcinoma A549 cells: Effect on metastasis. Food Chem. Toxicol. 2009, 47, 1985–1995. [Google Scholar]
- Kim, D.; Kim, S.; Koh, H.; Yoon, S.O.; Chung, A.S.; Cho, K.S.; Chung, J. Akt/PKB promotes cancer cell invasion via increased motility and metalloproteinase production. FASEB J. 2001, 15, 1953–1962. [Google Scholar]
- Shukla, S.; Maclennan, G.T.; Hartman, D.J.; Fu, P.; Resnick, M.I.; Gupta, S. Activation of PI3K-Akt signaling pathway promotes prostate cancer cell invasion. Int. J. Cancer 2007, 121, 1424–1432. [Google Scholar]
- Kaighn, M.E.; Narayan, K.S.; Ohnuki, Y.; Lechner, J.F.; Jones, L.W. Establishment and characterization of a human prostatic carcinoma cell line (PC-3). Invest. Urol. 1979, 17, 16–23. [Google Scholar]
- Chakraborti, S.; Mandal, M.; Das, S.; Mandal, A.; Chakraborti, T. Regulation of matrix metalloproteinases: an overview. Mol. Cell. Biochem. 2003, 253, 269–285. [Google Scholar]
- Rangaswami, H.; Bulbule, A.; Kundu, G.C. Nuclear factor-inducing kinase plays a crucial role in osteopontin-induced MAPK/IkappaBalpha kinase-dependent nuclear factor kappaB-mediated promatrix metalloproteinase-9 activation. J. Biol. Chem. 2004, 279, 38921–38935. [Google Scholar]
- Samuels, Y.; Ericson, K. Oncogenic PI3K and its role in cancer. Curr. Opin. Oncol. 2006, 18, 77–82. [Google Scholar] [CrossRef]
- Kuo, K.W.; Hsu, S.H.; Li, Y.P.; Lin, W.L.; Liu, L.F.; Chang, L.C.; Lin, C.C.; Lin, C.N.; Sheu, H.M. Anticancer activity evaluation of the solanum glycoalkaloid solamargine. Triggering apoptosis in human hepatoma cells. Biochem. Pharmacol. 2000, 60, 1865–1873. [Google Scholar] [CrossRef]
- Friedman, M.; Rayburn, J.R.; Bantle, J.A. Developmental toxicology of potato alkaloids in the frog embryo teratogenesis assay—Xenopus (FETAX). Food Chem. Toxicol. 1991, 29, 537–547. [Google Scholar]
- Friedman, M.; Henika, P.R.; Mackey, B.E. Feeding of potato, tomato and eggplant alkaloids affects food consumption and body and liver weights in mice. J. Nutr. 1996, 126, 989–999. [Google Scholar]
- Petrie, R.J.; Doyle, A.D.; Yamada, K.M. Random versus directionally persistent cell migration. Nat. Rev. Mol. Cell Biol. 2009, 10, 538–549. [Google Scholar]
- Friedl, P.; Wolf, K. Tumour-cell invasion and migration: diversity and escape mechanisms. Nat. Rev.Cancer 2003, 3, 362–374. [Google Scholar]
- Stearns, M.; Stearns, M.E. Evidence for increased activated metalloproteinase 2 (MMP-2a) expression associated with human prostate cancer progression. Oncol. Res. 1996, 8, 69–75. [Google Scholar]
- Nemeth, J.A.; Yousif, R.; Herzog, M.; Che, M.; Upadhyay, J.; Shekarriz, B.; Bhagat, S.; Mullins, C.; Fridman, R.; Cher, M.L. Matrix metalloproteinase activity, bone matrix turnover, and tumor cell proliferation in prostate cancer bone metastasis. J. Natl. Cancer I. 2002, 94, 17–25. [Google Scholar]
- Chien, C.S.; Shen, K.H.; Huang, J.S.; Ko, S.C.; Shih, Y.W. Antimetastatic potential of fisetin involves inactivation of the PI3K/Akt and JNK signaling pathways with downregulation of MMP-2/9 expressions in prostate cancer PC-3 cells. Mol. Cell. Biochem. 2010, 333, 169–180. [Google Scholar]
- Rabien, A.; Ergun, B.; Erbersdobler, A.; Jung, K.; Stephan, C. RECK overexpression decreases invasive potential in prostate cancer cells. Prostate 2012, 72, 948–954. [Google Scholar]
- Kato, K.; Long, N.K.; Makita, H.; Toida, M.; Yamashita, T.; Hatakeyama, D.; Hara, A.; Mori, H.; Shibata, T. Effects of green tea polyphenol on methylation status of RECK gene and cancer cell invasion in oral squamous cell carcinoma cells. Br. J. Cancer 2008, 99, 647–654. [Google Scholar]
- Gomez, D.E.; Alonso, D.F.; Yoshiji, H.; Thorgeirsson, U.P. Tissue inhibitors of metalloproteinases: Structure, regulation and biological functions. Eur. J. Cell Biol. 1997, 74, 111–122. [Google Scholar]
- Pulukuri, S.M.; Patibandla, S.; Patel, J.; Estes, N.; Rao, J.S. Epigenetic inactivation of the tissue inhibitor of metalloproteinase-2 (TIMP-2) gene in human prostate tumors. Oncogene 2007, 26, 5229–5237. [Google Scholar]
- Ramer, R.; Hinz, B. Inhibition of cancer cell invasion by cannabinoids via increased expression of tissue inhibitor of matrix metalloproteinases-1. J. Natl. Cancer I. 2008, 100, 59–69. [Google Scholar]
- Ramer, R.; Merkord, J.; Rohde, H.; Hinz, B. Cannabidiol inhibits cancer cell invasion via upregulation of tissue inhibitor of matrix metalloproteinases-1. Biochem. Pharmacol. 2010, 79, 955–966. [Google Scholar]
- Leite, K.R.; Tomiyama, A.; Reis, S.T.; Sousa-Canavez, J.M.; Sanudo, A.; Camara-Lopes, L.H.; Srougi, M. MicroRNA expression profiles in the progression of prostate cancer—From high-grade prostate intraepithelial neoplasia to metastasis. Urol. Oncol. 2012, 31, 796–801. [Google Scholar]
- Wang, Q.; Tang, H.; Yin, S.; Dong, C. Downregulation of microRNA-138 enhances the proliferation, migration and invasion of cholangiocarcinoma cells through the upregulation of RhoC/p-ERK/MMP-2/MMP-9. Oncol. Rep. 2013, 29, 2046–2052. [Google Scholar]
- Golubovskaya, V.M.; Sumbler, B.; Ho, B.; Yemma, M.; Cance, W.G. MiR-138 and MiR-135 directly target focal adhesion kinase, inhibit cell invasion, and increase sensitivity to chemotherapy in cancer cells. Anti-Cancer Agent. Me. 2013, 14, 18–28. [Google Scholar]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar]
- Image J — Image Processing and Analysis in Java. Available online: http://rsb.info.nih.gov/ij/ (accessed on 8 August 2014).
- Sample Availability: Samples of the compounds are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shen, K.-H.; Liao, A.C.-H.; Hung, J.-H.; Lee, W.-J.; Hu, K.-C.; Lin, P.-T.; Liao, R.-F.; Chen, P.-S. α-Solanine Inhibits Invasion of Human Prostate Cancer Cell by Suppressing Epithelial-Mesenchymal Transition and MMPs Expression. Molecules 2014, 19, 11896-11914. https://doi.org/10.3390/molecules190811896
Shen K-H, Liao AC-H, Hung J-H, Lee W-J, Hu K-C, Lin P-T, Liao R-F, Chen P-S. α-Solanine Inhibits Invasion of Human Prostate Cancer Cell by Suppressing Epithelial-Mesenchymal Transition and MMPs Expression. Molecules. 2014; 19(8):11896-11914. https://doi.org/10.3390/molecules190811896
Chicago/Turabian StyleShen, Kun-Hung, Alex Chien-Hwa Liao, Jui-Hsiang Hung, Wei-Jiunn Lee, Kai-Chieh Hu, Pin-Tsen Lin, Ruei-Fang Liao, and Pin-Shern Chen. 2014. "α-Solanine Inhibits Invasion of Human Prostate Cancer Cell by Suppressing Epithelial-Mesenchymal Transition and MMPs Expression" Molecules 19, no. 8: 11896-11914. https://doi.org/10.3390/molecules190811896




