Cytotoxicity and Genotoxicity of Ceria Nanoparticles on Different Cell Lines in Vitro
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
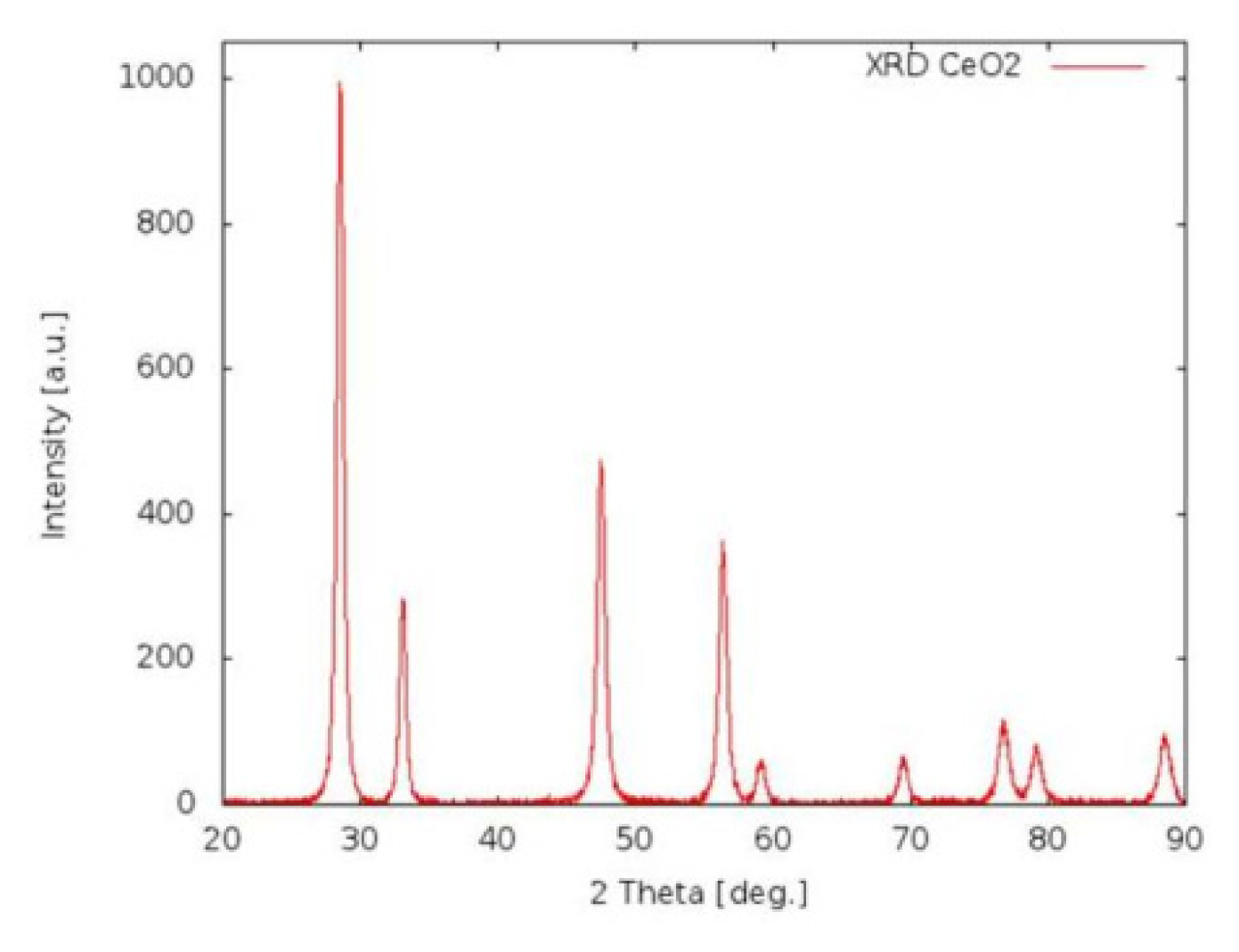
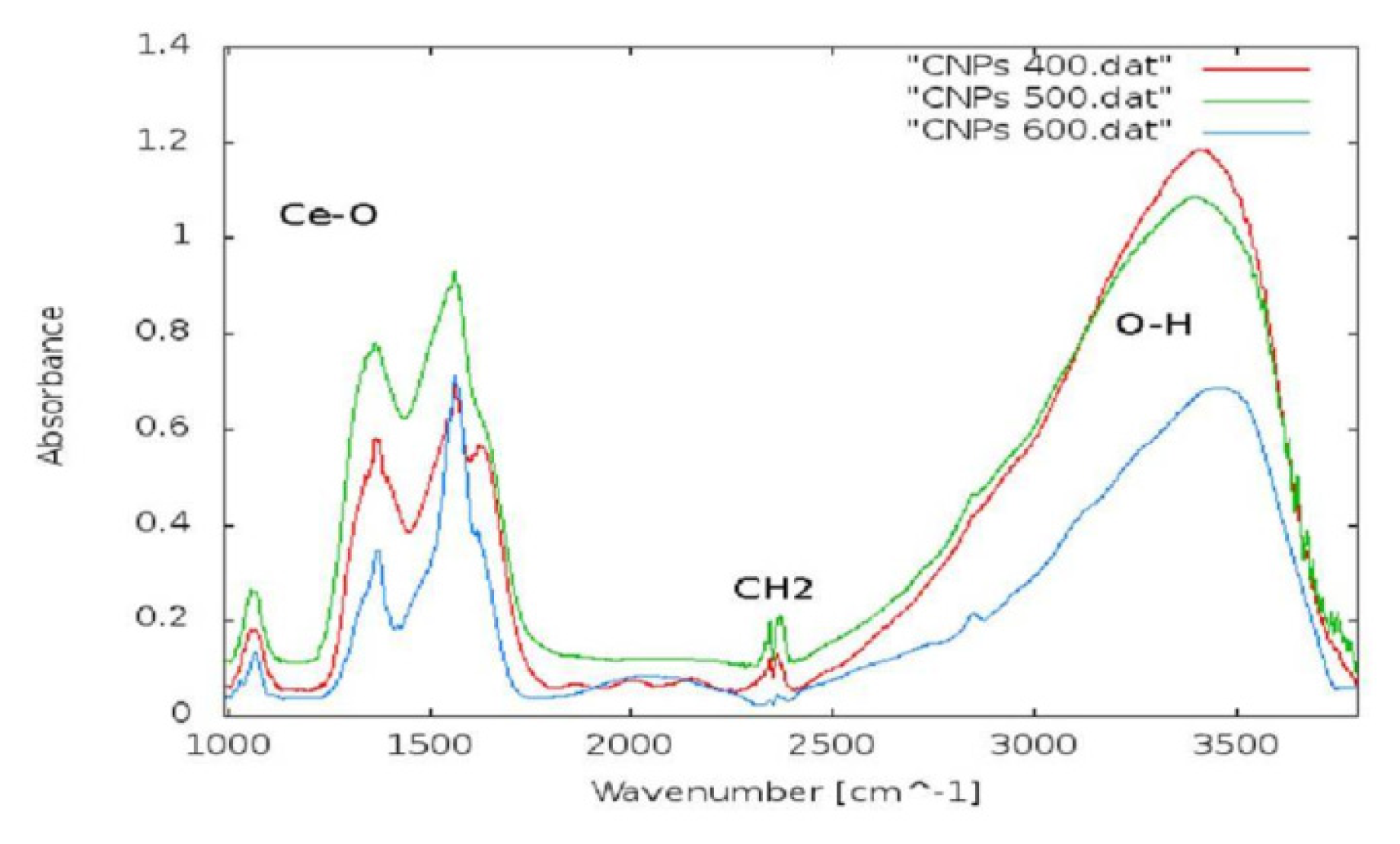
2.1.1. CeO2-NP Characterization
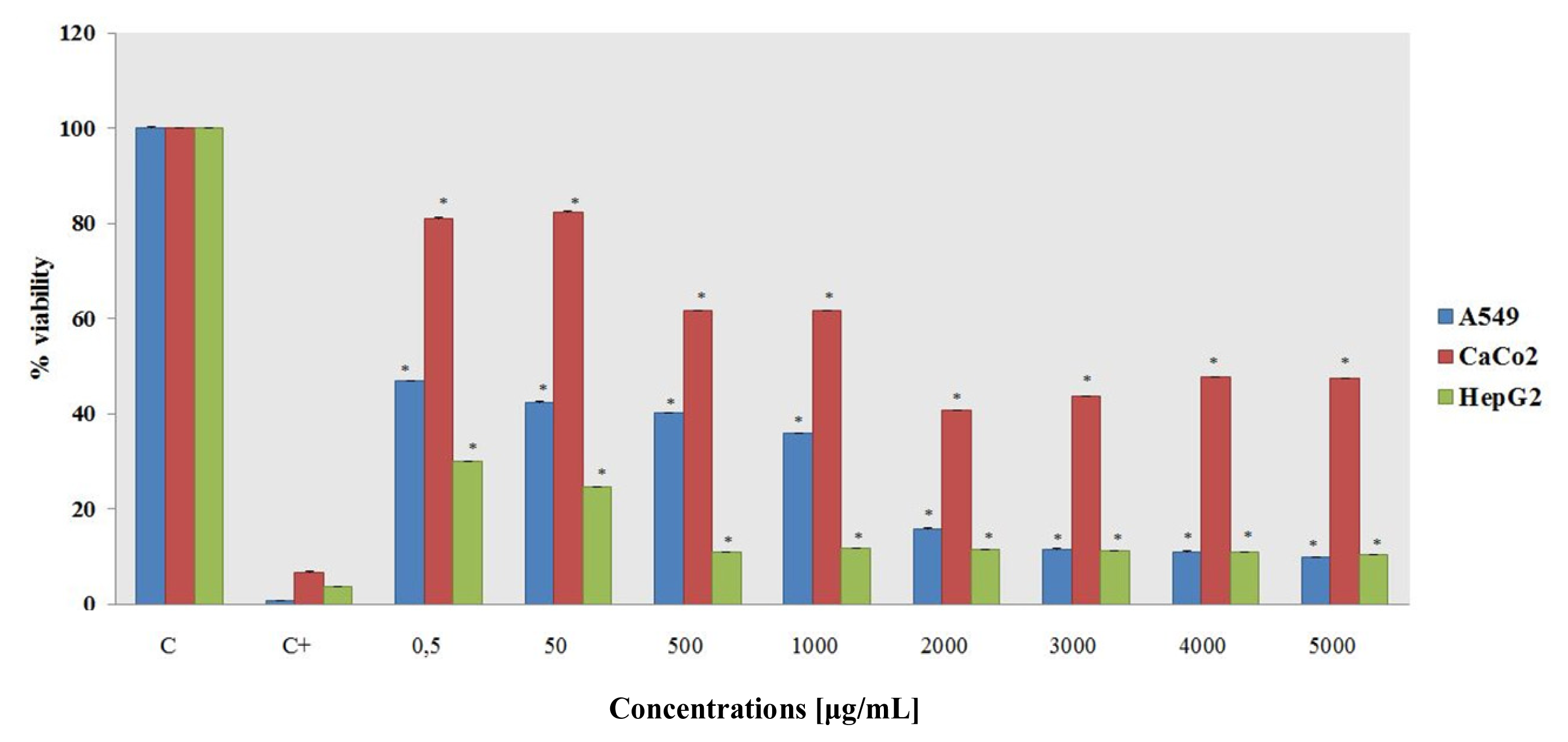
2.1.2. MTT Assay
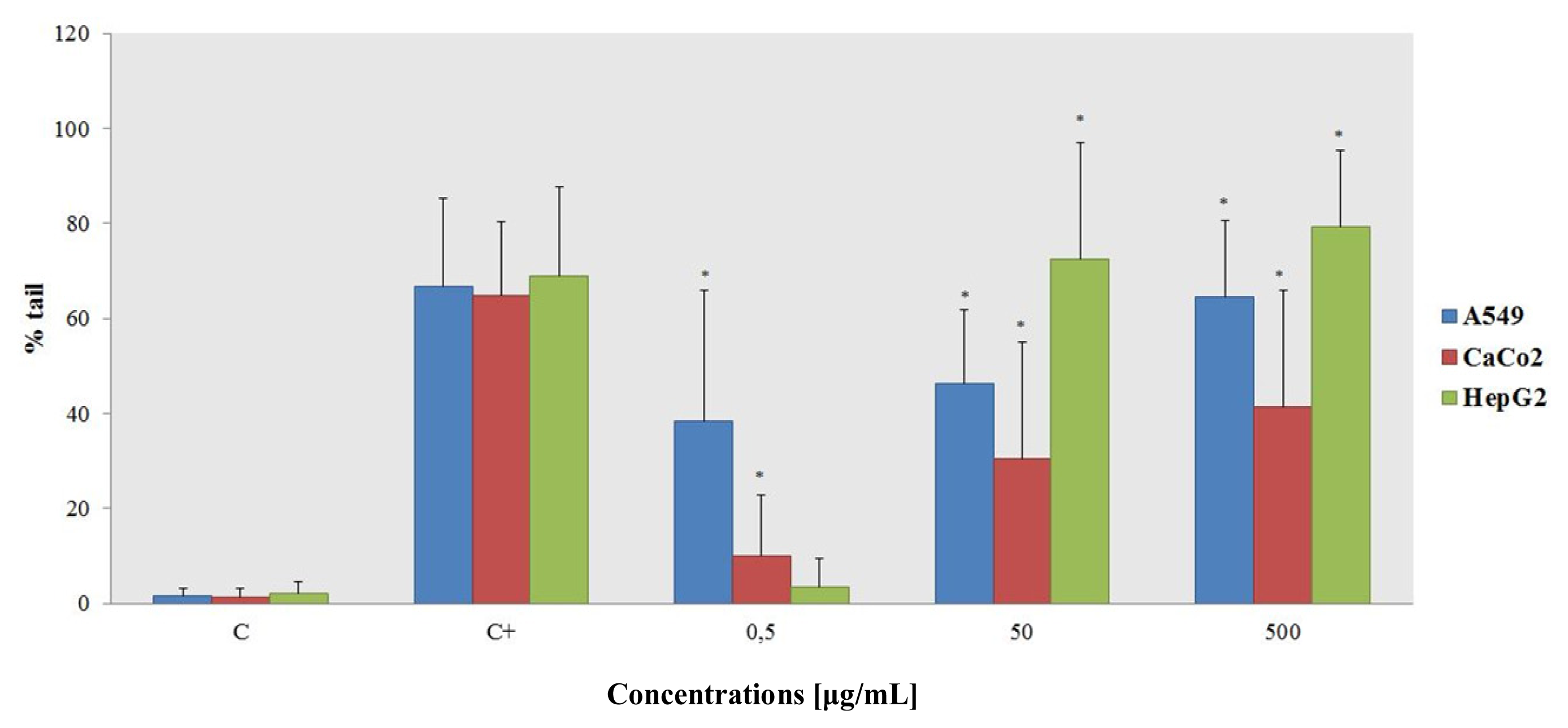
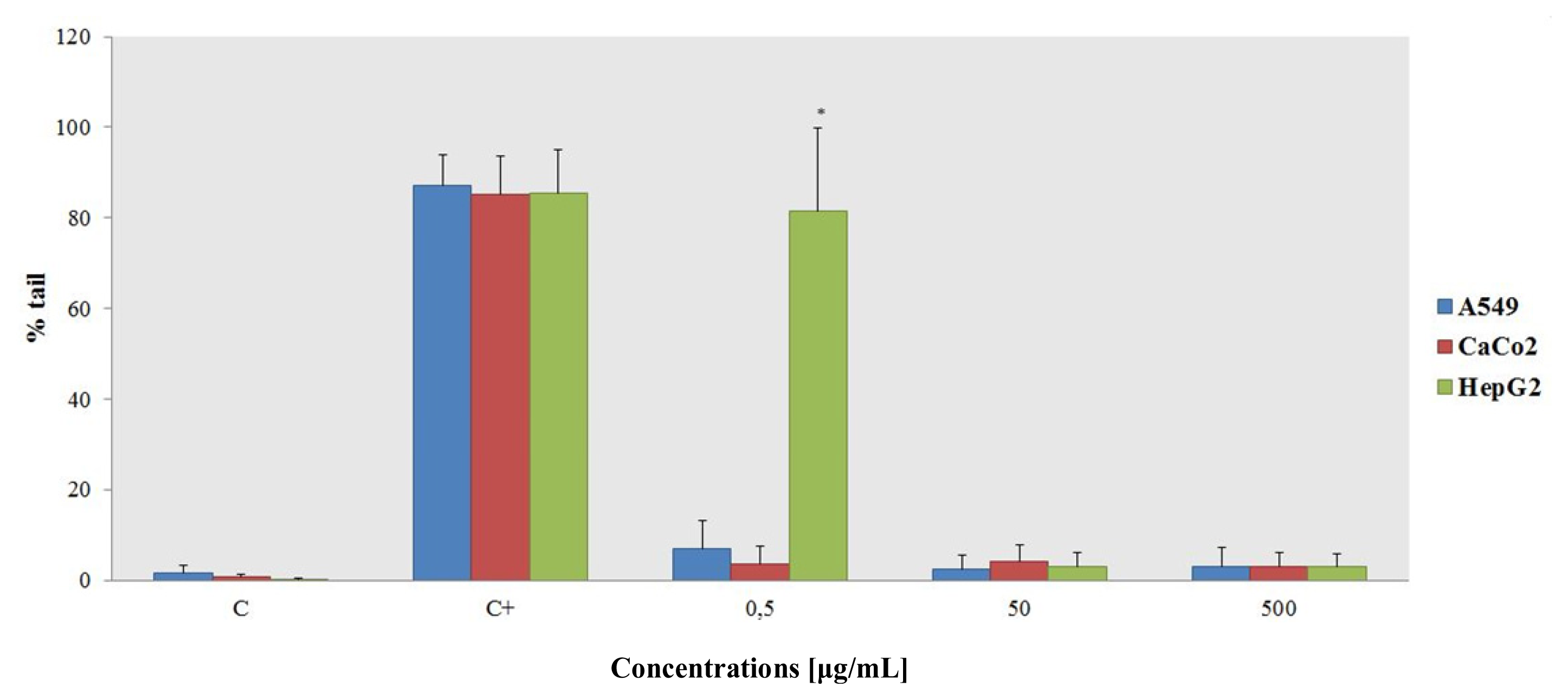
2.1.3. Alkaline Comet Assay
2.2. Discussion
3. Experimental Section
3.1. Nanoparticle Preparation and Characterization
3.2. Cell Culture Maintenance
3.3. MTT Assay
3.4. Alkaline Comet Assay
3.5. Statistical Treatment of the Data
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Organisation for Economic Co-operation and Development (OECD), Detailed Review Paper on Cell Transformation Assays for Detection of Chemical Carcinogens Series on Testing and Assessment; OECD: Paris, France, 2007.
- Organisation for Economic Co-operation and Development (OECD), In Vitro Mammalian Chromosomal Aberration Test TG473; OECD: Paris, France, 2007. [CrossRef]
- Fall, M.; Guerbet, M.; Park, B.; Gouriou, F.; Dionnet, F.; Morin, J.P. Evaluation of cerium oxide and cerium oxide based fuel additive safety on organotypic cultures of lung slices. Nanotoxicology 2007, 1, 227–234. [Google Scholar]
- Park, B.; Martin, P.; Harris, C.; Guest, R.; Whittingham, A.; Jenkinson, P.; Handley, J. Initial in vitro screening approach to investigate the potential health and environmental hazards of enviroxtrade mark—A nanoparticulate cerium oxide diesel fuel additive. Part. Fibre Toxicol 2007, 5, 4–12. [Google Scholar]
- HEI—Health Effects Institute, Evaluation of Human Health Risk from Cerium added to Diesel Fuel; Flagship Press: North Andover, MA, USA, 2001.
- Farfaletti, A.; Astorga, C.; Martini, G.; Manfredi, U.; Mueller, A.; Rey, M.; de Santi, G.; Krasenbrink, A.; Larsen, B.R. Effect of water/fuel emulsions and cerium based combustion improver additive on HD and LD diesel exhaust emissions. Environ. Sci. Technol 2005, 39, 6792–6799. [Google Scholar]
- Jung, H.; Kittelson, D.B.; Zachariah, M.R. The influence of a cerium additive on ultrafine emissions and kinetics of oxidation. Combust. Flame 2005, 142, 276–288. [Google Scholar]
- Lopez-Moreno, M.L.; de La Rosa, G.; Hernandez, J.A.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. X-ray absorption spectroscopy of CeO2 nanoparticles and assessment of their differential toxicity in four edible plants. J. Agric. Food Chem 2010, 58, 3689–3693. [Google Scholar]
- Zeyons, O.; Thill, A.; Chauvat, F.; Menguy, N.; Cassier-Chauvat, C.; Oréar, C.; Daraspe, J.; Auffan, M.; Rose, J.; Spalla, O. Direct and indirect CeO2 nanoparticles toxicity for Escherichia coli and Synechocystis. Nanotoxicology 2009, 3, 284–295. [Google Scholar]
- Flemming, R.C.; van Balen, E.C.; Singh, C. Exposure, health and ecological effects review of engineered nanoscale cerium and cerium oxide associated with its use as a fuel additive. Crit. Rev. Toxicol 2011, 41, 213–229. [Google Scholar]
- Velasco, M.V.; Sarruf, F.D.; Salgado-Santos, I.M.; Haroutiounian-Filho, C.A.; Kaneko, T.M.; Baby, A.R. Broad spectrum bioactive sunscreens. Int. J. Pharm 2008, 363, 50–57. [Google Scholar]
- Bennat, C.; Müller-Goymann, C.C. Skin penetration and stabilization of formulations containing microfine titanium dioxide as physical UV filter. Int. J. Cosmet. Sci 2000, 4, 271–283. [Google Scholar]
- Izu, N.; Shin, W.; Matsubara, I.; Murayama, N. Development of resistive oxygen sensors based on cerium oxide thick film. J. Electroceram 2004, 13, 703–706. [Google Scholar]
- Schubert, D.; Dargusch, R.; Raitano, J.; Chan, S.W. Cerium and yttrium oxide nanoparticles are neuroprotective. Biochem. Biophys. Res. Commun 2006, 342, 86–91. [Google Scholar]
- Chun, A.L. Seeing with ceria. Nat. Nanotechnol 2006. [Google Scholar] [CrossRef]
- Aukrust, P.; Gullestand, L.; Ueland, T.; Damas, J.K.; Yndestad, A. Inflammatory and anti-inflammatory cytokines in long-term exposure heart failure: Potential therapeutic implications. Ann. Med 2005, 37, 74–85. [Google Scholar]
- Grieve, D.J.; Byme, J.A.; Cave, A.C.; Shah, A.M. Role of oxidative stress in cardiac remodeling after myocardial infarction. Heart Lung Circ 2004, 13, 132–138. [Google Scholar]
- Kolattukudy, P.E.; Quach, T.; Bergese, S. Myocarditis induced by targeted expression of the MCP-1 gene in murine cardiac muscle. Am. J. Pathol 1998, 152, 101–111. [Google Scholar]
- Moldovan, N.I.; Goldschmidt-Clermont, P.J.; Parker-Thornburg, J.; Shapiro, S.D.; Kolattukudy, P.E. Contribution of monocytes/macrophages to compensatory neovascularization: the drilling of metalloelastase-positive tunnels in ischemic myocardium. Circ. Res 2000, 87, 378–384. [Google Scholar]
- Niu, J.; Azfer, A.; Rogers, L.M.; Wang, X.; Kolattukudy, P.E. Cardioprotective effects of cerium oxide nanoparticles in a transgenic murine model of cardiomyopathy. Cardiovasc. Res 2007, 73, 549–559. [Google Scholar]
- Linse, S.; Cabaleiro-Lago, C.; Xue, W.F. Nucleation of protein fibrillation by nanoparticles. Proc. Natl. Acad. Sci. USA 2007, 104, 8691–8696. [Google Scholar]
- Xia, T.; Kovochich, M.; Liong, M.; Madler, L.; Gilbert, B.; Shi, H.; Yeh, J.I.; Zink, J.I.; Nel, A.E. Comparison of the mechanism of toxicity of zinc oxide and cerium oxide nanoparticles based on dissolution and oxidative stress properties. ACS Nano 2008, 2, 2121–2134. [Google Scholar]
- Organisation for Economic Co-operation and Development (OECD), Guidance Manual for the Testing of Manufactured Nanomaterials: OECD Sponsorship Programme; OECD: Paris, France, 2009.
- Chen, J.; Patil, S.; Seal, S.; McGinnis, J.F. Nanoceria particles prevent ROI-induced blindness. Adv. Exp. Med 2008, 613, 53–59. [Google Scholar]
- ECVAM DB-ALM: INVITTOX protocols. Available online: http://researcher.nsc.gov.tw/public/woody/Attachment/97171346771.pdf accessed on 29 November 2012.
- Pierscionek, B.K.; Yuebin, L.; Yasseen, A.A.; Colhoun, L.M.; Schachar, R.A.; Chen, W. Nanoceria have no genotoxic effect on human lens epithelial cells. Nanotechnology 2010, 21, 035102. [Google Scholar]
- Singh, N.P.; McCoy, M.T.; Tice, R.R.; Schneider, E.L. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp. Cell Res 1988, 175, 184–191. [Google Scholar]
- Chigurupati, S.; Mughal, M.R.; Okun, E.; Das, S.; Kumar, A.; McCaffery, M.; Seal, S.; Mattson, M.P. Effects of cerium oxide nanoparticles on the growth of keratinocytes, fibroblasts and vascular endothelial cells in cutaneous wound healing. Biomaterials 2013, 34, 2194–2201. [Google Scholar]
- Alili, L.; Sack, M.; von Montfort, C.; Giri, S.; Das, S.; Carroll, K.; Zanger, K.; Seal, S.; Brenneisen, P. Downregulation of tumor growth and invasion by redox-active nanoparticles. Antioxid. Redox Signal. 2012. [Google Scholar] [CrossRef]
- Culcasi, M.; Benameur, L.; Mercier, A.; Lucchesi, C.; Rahmouni, H.; Asteian, A.; Casano, G.; Botta, A.; Kovacic, H.; Pietri, S. EPR spin trapping evaluation of ROS production in human fibroblasts exposed to cerium oxide nanoparticles: Evidence for NADPH oxidase and mitochondrial stimulation. Chem. Biol. Interact 2012, 199, 161–176. [Google Scholar]


© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De Marzi, L.; Monaco, A.; De Lapuente, J.; Ramos, D.; Borras, M.; Di Gioacchino, M.; Santucci, S.; Poma, A. Cytotoxicity and Genotoxicity of Ceria Nanoparticles on Different Cell Lines in Vitro. Int. J. Mol. Sci. 2013, 14, 3065-3077. https://doi.org/10.3390/ijms14023065
De Marzi L, Monaco A, De Lapuente J, Ramos D, Borras M, Di Gioacchino M, Santucci S, Poma A. Cytotoxicity and Genotoxicity of Ceria Nanoparticles on Different Cell Lines in Vitro. International Journal of Molecular Sciences. 2013; 14(2):3065-3077. https://doi.org/10.3390/ijms14023065
Chicago/Turabian StyleDe Marzi, Laura, Antonina Monaco, Joaquin De Lapuente, David Ramos, Miquel Borras, Mario Di Gioacchino, Sandro Santucci, and Anna Poma. 2013. "Cytotoxicity and Genotoxicity of Ceria Nanoparticles on Different Cell Lines in Vitro" International Journal of Molecular Sciences 14, no. 2: 3065-3077. https://doi.org/10.3390/ijms14023065




