Plant Nucleotide Binding Site–Leucine-Rich Repeat (NBS-LRR) Genes: Active Guardians in Host Defense Responses
Abstract
:1. Introduction
2. Genomic Organization and Evolution of NBS-LRR Genes in the Plant Genome
2.1. Genomic Organization and Evolution of NBS-LRR Genes
2.2. Pseudogenes
2.3. Evolution of NBS-LRR Genes in Plants
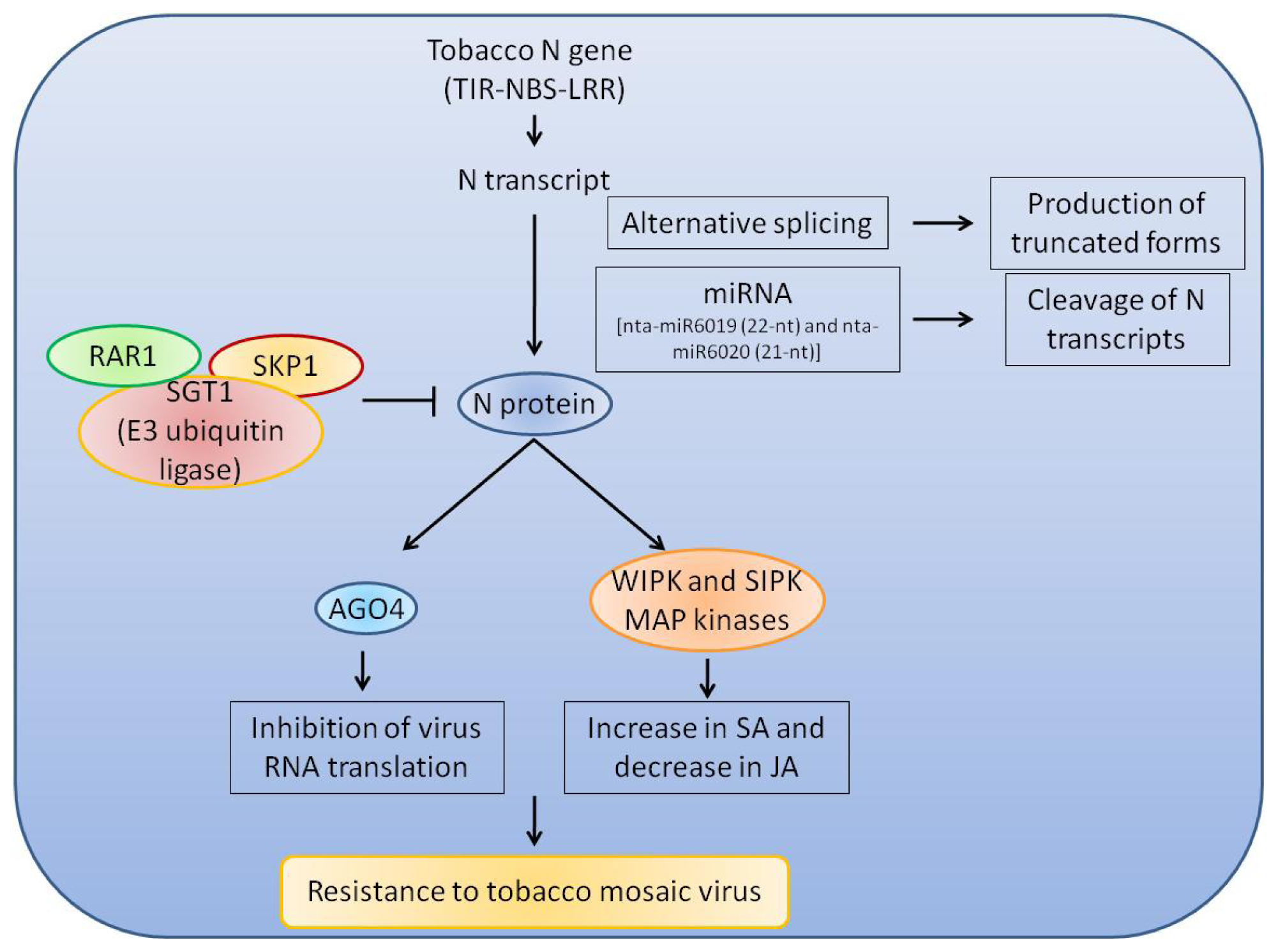
3. Regulation and Function of NBS-LRR Genes
3.1. Molecular Basis of Resistance Responses Induced by NBS-LRR Proteins
3.2. Regulation of Expression and Activity of NBS-LRR Genes
4. NBS-LRR Genes: A Resource for Plant Breeding
4.1. Co-Localization of NBS-LRR Genes with R Loci
4.2. Breeding for More Resistant Crops
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Albersheim, P.; Anderson-Prouty, A.J. Carbohydrates, proteins, cell surfaces and biochemistry of pathogenesis. Annu. Rev. Plant Physiol 1975, 26, 31–52. [Google Scholar]
- Jones, D.A.; Takemoto, D. Plant innate immunity—Direct and indirect recognition of general and specific pathogen-associated molecules. Curr. Opin. Immunol 2004, 16, 48–62. [Google Scholar]
- Bent, A.F.; Mackey, D. Elicitors, effectors, and R genes: The new paradigm and a lifetime supply of questions. Annu. Rev. Phytopathol 2007, 45, 399–436. [Google Scholar]
- Flor, H.H. Current status of the gene-for-gene concept. Annu. Rev. Phytopathol 1971, 9, 275–296. [Google Scholar]
- Dodds, P.N.; Rathjen, J.P. Plant immunity: Towards an integrated view of plant–pathogen interactions. Nat. Rev. Genet 2010, 11, 539–548. [Google Scholar]
- Ade, J.; de Young, B.J.; Golstein, C.; Innes, R.W. Indirect activation of a plant nucleotide binding site-leucine-rich repeat protein by a bacterial protease. Proc. Natl. Acad. Sci. USA 2007, 104, 2531–2536. [Google Scholar]
- Van Ooijen, G.; van den Burg, H.A.; Cornelissen, B.J.; Takken, F.L. Structure and function of resistance proteins in solanaceous plants. Annu. Rev. Phytopathol 2007, 45, 43–72. [Google Scholar]
- Dangl, J.L.; Jones, J.D.G. Plant pathogens and integrated defence responses to infection. Nature 2001, 411, 826–833. [Google Scholar]
- Tan, S.; Wu, S. Genome wide analysis of nucleotide-binding site disease resistance genes inBrachypodium distachyon. Comp. Funct. Genomics 2012. [Google Scholar] [CrossRef]
- Ellis, J.; Dodds, P.; Pryor, T. Structure, function and evolution of plant disease resistance genes. Curr. Opin. Plant Biol 2000, 3, 278–84. [Google Scholar]
- Jones, J.D.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar]
- Porter, B.W.; Paidi, M.; Ming, R.; Alam, M.; Nishijima, W.T.; Zhu, Y.J. Genome-wide analysis of Carica papaya reveals a small NBS resistance gene family. Mol. Genet. Genomics 2009, 281, 609–626. [Google Scholar]
- Wan, H.; Yuan, W.; Bo, K.; Shen, J.; Pang, X.; Chen, J. Genome-wide analysis of NBS-encoding disease resistance in Cucumis sativus and phylogenetic study of NBS-encoding genes in Cucurbitaceae crops. BMC Genomics 2013, 14, 109. [Google Scholar]
- Shang, J.; Tao, Y.; Chen, X.; Zou, Y.; Lei, C.; Wang, J.; Li, X.; Zhao, X.; Zhang, M.; Lu, Z.; et al. Identification of a new rice blast resistance gene, Pid3, by genome wide comparison of paired nucleotide-binding site leucine-rich repeat genes and their pseudogene alleles between the two sequenced rice genomes. Genetics 2009, 182, 1303–1311. [Google Scholar]
- Jupe, F.; Pritchard, L.; Etherington, G.J.; MacKenzie, K.; Cock, P.J.A.; Wright, F.; Sharma, S.K.; Bolser, D.; Bryan, G.J.; Jones, J.D.G.; et al. Identification and localisation of the NB-LRR gene family within the potato genome. BMC Genomics 2012, 13, 75. [Google Scholar]
- Lozano, R.; Ponce, O.; Ramirez, M.; Mostajo, N.; Orjeda, G. Genome-wide identification and mapping of NBS-encoding resistance genes in Solanum tuberosum group Phureja. PLoS One 2012, 7, e34775. [Google Scholar]
- Mun, J.H.; Yu, H.J.; Park, S.; Park, B.S. Genome-wide identification of NBS-encoding resistance genes in Brassica rapa. Mol. Genet. Genomics 2009, 282, 617–631. [Google Scholar]
- Li, X.; Cheng, Y.; Ma, W.; Zhao, Y.; Jiang, H.; Zhang, M. Identification and characterization of NBS-encoding disease resistance genes in Lotus japonicus. Plant Syst. Evol 2010, 289, 101–110. [Google Scholar]
- Ameline-Torregrosa, C.; Wang, B.B.; O’Bleness, M.S. Identification and characterization of nucleotide-binding site-leucine-rich repeat genes in the model plant Medicago truncatula. Plant Physiol 2008, 146, 5–21. [Google Scholar]
- Meyers, B.C.; Kozik, A.; Griego, A.; Kuang, H.; Michelmore, R.W. Genome-wide analysis of NBS-LRR-encoding genes in Arabidopsis. Plant Cell 2003, 15, 809–834. [Google Scholar]
- Guo, Y.L.; Fitz, J.; Schneeberger, K.; Ossowski, S.; Cao, J.; Weigel, D. Genome-wide comparison of nucleotide-binding site-leucine-rich-repeat-encoding genes in Arabidopsis. Plant Physiol 2011, 157, 757–769. [Google Scholar]
- Kohler, A.; Rinaldi, C.; Duplessis, S.; Baucher, M.; Geelen, D.; Duchaussoy, F.; Meyers, B.C.; Boerjan, W.; Martin, F. Genome-wide identification of NBS resistance genes in Populus trichocarpa. Plant Mol. Biol 2008, 66, 619–636. [Google Scholar]
- Yang, S.; Zhang, X.; Yue, J.X.; Tian, D.; Chen, J.Q. Recent duplications dominate NBS-encoding gene expansion in two woody species. Mol. Genet. Genomics 2008, 280, 187–198. [Google Scholar]
- Kang, Y.J.; Kim, K.H.; Shim, S.; Yoon, M.Y.; Sun, S.; Kim, M.Y.; Van, K.; Lee, S.H. Genome-wide mapping of NBS-LRR genes and their association with disease resistance in soybean. BMC Plant Biol 2012, 12, 139. [Google Scholar]
- Hulbert, S.H.; Webb, C.A.; Smith, S.M.; Sun, Q. Resistance gene complexes: Evolution and utilization. Annu. Rev. Phytopathol 2001, 39, 285–312. [Google Scholar]
- McDowell, J.M.; Simon, S.A. Recent insights into R gene evolution. Mol. Plant Pathol 2006, 7, 437–448. [Google Scholar]
- Leister, D. Tandem and segmental gene duplication and recombination in the evolution of plant disease resistance gene. Trends Genet 2004, 20, 116–122. [Google Scholar]
- Yang, S.; Feng, Z.; Zhang, X.; Jiang, K.; Jin, X.; Hang, Y.; Chen, J.Q.; Tian, D. Genome-wide investigation on the genetic variations of rice disease resistance genes. Plant Mol. Biol 2006, 62, 181–193. [Google Scholar]
- Chandrasekaran, C.; Betrán, E. Origins of new genes and pseudogenes. Nat. Educ 2008, 1, 1. [Google Scholar]
- He, L.; Du, C.; Covaleda, L.; Xu, Z.; Robinson, A.F.; Yu, J.Z.; Kohel, R.J.; Zhang, H.B. Cloning, characterization, and evolution of the NBS-LRR-encoding resistance gene analogue family in polyploid cotton (Gossypium hirsutum L.). Mol. Plant Microbe Interact 2004, 17, 1234–1241. [Google Scholar]
- Luo, S.; Zhang, Y.; Hu, Q.; Chen, J.; Li, K.; Lu, C.; Liu, H.; Wang, W.; Kuang, H. Dynamic nucleotide-binding-site and leucine-rich-repeat-encoding genes in the grass family. Plant Physiol 2012, 159, 197–210. [Google Scholar]
- Meyers, B.C.; Dickerman, A.W.; Michelmore, R.W.; Sivaramakrishnan, S.; Sobral, B.W.; Young, N.D. Plant disease resistance genes encode members of an ancient and diverse protein family within the nucleotide-binding superfamily. Plant J 1999, 20, 317–332. [Google Scholar] [Green Version]
- Monosi, B.; Wisser, R.J.; Pennill, L.; Hulbert, S.H. Full-genome analysis of resistance gene homologues in rice. Theor. Appl. Genet 2004, 109, 1434, –1447.. [Google Scholar]
- Liu, J.J.; Ekramoddoullah, A.K. Isolation, genetic variation and expression of TIR-NBS-LRR resistance gene analogs from western white pine (Pinus monticola Dougl. ex. D. Don.). Mol. Genet. Genomics 2003, 270, 432–441. [Google Scholar]
- Hirotsune, S.; Yoshida, N.; Chen, A.; Garrett, L.; Sugiyama, F.; Takahashi, S.; Yagami, K.; Wynshaw-Boris, A.; Yoshiki, A. An expressed pseudogene regulates the messenger-RNA stability of its homologous coding gene. Nature 2003, 423, 91–96. [Google Scholar]
- Mastrangelo, A.M.; Marone, D.; Laidò, G.; De Leonardis, A.M.; de Vita, P. Alternative splicing: Enhancing ability to cope with stress via transcriptome plasticity. Plant Sci. 2012, 185–186, 40–49. [Google Scholar]
- Michelmore, R.W.; Meyers, B.C. Clusters of resistance genes in plants evolve by divergent selection and a birth-and-death process. Genome Res 1998, 8, 1113–1130. [Google Scholar]
- Young, N.D. The genetic architecture of resistance. Curr. Opin. Plant Biol 2000, 3, 285–290. [Google Scholar]
- Sun, Q.; Collins, N.C.; Ayliffe, M.; Smith, S.M.; Drake, J.; Pryor, T.; Hulbert, S.H. Recombination between paralogues at the Rp1 rust resistance locus in maize. Genetics 2001, 158, 423–438. [Google Scholar]
- Kuang, H.; Woo, S.S.; Meyers, B.C.; Nevo, E.; Michelmore, R.W. Multiple genetic processes result in heterogeneous rates of evolution within the major cluster disease resistance genes in lettuce. Plant Cell 2004, 16, 2870–2894. [Google Scholar]
- Mondragon-Palomino, M.; Meyers, B.C.; Michelmore, R.W.; Gaut, B.S. Patterns of positive selection in the complete NBS-LRR gene family of Arabidopsis thaliana. Genome Res 2002, 12, 1305–1315. [Google Scholar]
- McHale, L.; Tan, X.; Koehl, P.; Michelmore, R.W. Plant NBS-LRR proteins: Adaptable guards. Genome Biol 2006, 7, 212. [Google Scholar]
- Yue, J.X.; Meyers, B.C.; Chen, J.Q.; Tian, D.; Yang, S. Tracing the origin and evolutionary history of plant NBS-LRR genes. New Phytol 2012, 193, 1049–1063. [Google Scholar]
- Xue, J.-Y.; Wang, Y.; Wu, P.; Wang, Q.; Yang, L.-T.; Pan, X.-H.; Wang, B.; Chen, J.-Q. A primary survey on bryophyte species reveals two novel classes of nucleotide-binding site (NBS) genes. PLoS One 2012, 7, e36700. [Google Scholar]
- Pan, Q.; Wendel, J.; Fluhr, R. Divergent evolution of plant NBS-LRR resistance gene homologues in dicot and cereal genomes. J. Mol. Evol 2000, 50, 203–213. [Google Scholar]
- Gou, M.; Su, N.; Zheng, J.; Huai, J.; Wu, G.; Zhao, J.; He, J.; Tang, D.; Yang, S.; Wang, G. An F-box gene, CPR30, functions as a negative regulator of the defense response in Arabidopsis. Plant J 2009, 60, 757–770. [Google Scholar]
- Głowacki, S.; Macioszek, V.K.; Kononowicz, A.K. R proteins as fundamentals of plant innate immunity. Cell. Mol. Biol. Lett 2011, 16, 1–24. [Google Scholar]
- Hull, R. Matthew’s Plant Virology; Academic Press: New York NY, USA, 2002. [Google Scholar]
- Sekine, K.-T.; Kawakami, S.; Hase, S.; Kubota, M.; Ichinose, Y.; Shah, J.; Kang, H.-G.; Klessig, D.F.; Takahashi, H. High level expression of a virus resistance gene, RCY1, confers extreme resistance to cucumber mosaic virus in Arabidopsis thaliana. Mol. Plant Microbe Interact 2008, 21, 1398–1407. [Google Scholar]
- McDowell, J.M.; Dhandaydham, M.; Long, T.A.; Aarts, M.G.; Goff, S.; Holub, E.B.; Dangl, J.L. Intragenic recombination and diversifying selection contribute to the evolution of downy mildew resistance at the RPP8 locus of Arabidopsis. Plant Cell 1998, 10, 1861–1874. [Google Scholar]
- Cooley, M.B.; Pathirana, S.; Wu, H.J.; Kachroo, P.; Klessig, D.F. Members of the Arabidopsis HRT/RPP8 family of resistance genes confer resistance to both viral and oomycete pathogens. Plant Cell 2000, 12, 663–676. [Google Scholar]
- Takahashi, H.; Miller, J.; Nozaki, Y.; Takeda, M.; Shah, J.; Hase, S.; Ikegami, M.; Ehara, Y.; Dinesh-Kumar, S.P. RCY1, an Arabidopsis thaliana RPP8/HRT family resistance gene, conferring resistance to cucumber mosaic virus requires salicylic acid, ethylene and a novel signal transduction mechanism. Plant J 2002, 32, 655–667. [Google Scholar]
- Bendahmane, A.; Querci, M.; Kanyuka, K.; Baulcombe, D.C. Agrobacterium transient expression system as a tool for the isolation of disease resistance genes: Application to the Rx2 locus in potato. Plant J 2000, 21, 73–81. [Google Scholar]
- Van der Vossen, E.; Rouppe van der Voort, J.; Kanyuka, K.; Bendahmane, A.; Sandbrink, H.; Baulcombe, D.; Bakker, J.; Stiekema, W.; Klein-Lankhorst, R. Homologs of a single resistance-gene cluster in potato confer resistance to distinct pathogens: A virus and a nematode. Plant J 2000, 23, 567–576. [Google Scholar]
- Qi, D.; de Young, B.J.; Innes, R.W. Structure-function analysis of the coiled-coil and leucine-rich repeat domains of the RPS5 disease resistance protein. Plant Physiol 2012, 158, 1819–1832. [Google Scholar]
- Williams, S.J.; Sornaraj, P.; deCourcy-Ireland, E.; Menz, R.I.; Kobe, B.; Ellis, J.G.; Dodds, P.N.; Anderson, P.A. An autoactive mutant of the M flax rust resistance protein has a preference for binding ATP, whereas wild-type M protein binds ADP. Mol. Plant Microbe Interact 2011, 24, 897–906. [Google Scholar]
- Van Ooijen, G.; Mayr, G.; Kasiem, M.M.A.; Albrecht, M.; Cornelissen, B.J.C.; Takken, F.L.W. Structure-function analysis of the NB-ARC domain of plant disease resistance proteins. J. Exp. Bot 2008, 59, 1383–1397. [Google Scholar]
- Takken, F.L.; Albrecht, M.; Tameling, W.I. Resistance proteins: Molecular switches of plant defense. Curr. Opin. Plant Biol 2006, 9, 383–390. [Google Scholar]
- Moffett, P.; Farnham, G.; Peart, J.; Baulcombe, D.C. Interaction between domains of a plant NBS-LRR protein in disease resistance-related cell death. EMBO J 2002, 21, 4511–4519. [Google Scholar]
- Dodds, P.N.; Lawrence, G.J.; Catanzariti, A.M.; The, T.; Wang, C.I.; Ayliffe, M.A.; Kobe, B.; Ellis, J.G. Direct protein interaction underlies gene-for-gene specificity and coevolution of the flax resistance genes and flax rust avirulence genes. Proc. Natl. Acad. Sci. USA 2006, 103, 8888–8893. [Google Scholar]
- Hwang, C.F.; Williamson, V.M. Leucine-rich repeat-mediated intramolecular interactions in nematode recognition and cell death signaling by the tomato resistance protein Mi. Plant J 2003, 34, 585–593. [Google Scholar]
- Rairdan, G.J.; Moffett, P. Distinct domains in the ARC region of the potato resistance protein Rx mediate LRR binding and inhibition of activation. Plant Cell 2006, 18, 2082–2093. [Google Scholar]
- Van Ooijen, G.; Mayr, G.; Albrecht, M.; Cornelissen, B.J.; Takken, F.L. Transcomplementation, but not physical association of the CC-NB-ARC and LRR domains of tomato R protein Mi-1.2 is altered by mutations in the ARC2 subdomain. Mol. Plant 2008, 1, 401–410. [Google Scholar]
- Mestre, P.; Baulcombe, D.C. Elicitor-mediated oligomerization of the tobacco N disease resistance protein. Plant Cell 2006, 18, 491–501. [Google Scholar]
- Tao, Y.; Yuan, F.; Leister, RT.; Ausubel, F.M.; Katagiri, F. Mutational analysis of the Arabidopsis nucleotide binding site-leucine-rich repeat resistance gene RPS2. Plant Cell 2000, 12, 2541–2554. [Google Scholar]
- Collier, S.M.; Hamel, L.P.; Moffett, P. Cell death mediated by the N-terminal domains of a unique and highly conserved class of NB-LRR protein. Mol. Plant Microbe Interact 2011, 24, 918–931. [Google Scholar]
- Gao, Z.; Chung, E.-H.; Eitas, T.K.; Dangl, J.L. Plant intracellular innate immune receptor Resistance to Pseudomonas syringae pv. maculicola 1 (RPM1) is activated at, and functions on, the plasma membrane. Proc. Natl. Acad. Sci. USA 2011, 108, 7619–7624. [Google Scholar]
- Maekawa, T.; Cheng, W.; Spiridon, L.N.; Töller, A.; Lukasik, E.; Saijo, Y.; Liu, P.; Shen, Q.H.; Micluta, M.A.; Somssich, I.E.; et al. Coiled-coil domain-dependent homodimerization of intracellular barley immune receptors defines a minimal functional module for triggering cell death. Cell Host Microbe 2011, 9, 187–199. [Google Scholar]
- Rairdan, G.J.; Collier, S.M.; Sacco, M.A.; Baldwin, T.T.; Boettrich, T.; Moffett, P. The coiled-coil and nucleotide binding domains of the potato Rx disease resistance protein function in pathogen recognition and signaling. Plant Cell 2008, 20, 739–751. [Google Scholar]
- Shen, Q.H.; Saijo, Y.; Mauch, S.; Biskup, C.; Bieri, S.; Keller, B.; Seki1, H.; Ülker, B.; Somssich, I.E.; Schulze-Lefert, P. Nuclear activity of MLA immune receptors links isolate-specific and basal disease-resistance responses. Science 2007, 315, 1098–1103. [Google Scholar]
- Meier, I.; Somers, D.E. Regulation of nucleocytoplasmic trafficking in plants. Curr. Opin. Plant Biol 2011, 14, 538–546. [Google Scholar]
- Burch-Smith, T.M.; Schiff, M.; Caplan, J.L.; Tsao, J.; Czymmek, K.; Dinesh-Kumar, S.P. A novel role for the TIR domain in association with pathogen-derived elicitors. PLoS Biol 2007, 5, e68. [Google Scholar]
- Deslandes, L.; Olivier, J.; Peeters, N.; Feng, D.X.; Khounlotham, M.; Boucher, C.; Somssich, I.; Genin, S.; Marco, Y. Physical interaction between RRS1-R, a protein conferring resistance to bacterial wilt, and PopP2, a type III effector targeted to the plant nucleus. Proc. Natl. Acad. Sci. USA 2003, 100, 8024–8029. [Google Scholar]
- Wirthmueller, L.; Zhang, Y.; Jones, J.D.G.; Parker, J.E. Nuclear accumulation of the Arabidopsis immune receptor RPS4 is necessary for triggering EDS1-dependent defense. Curr. Biol 2007, 17, 2023–2029. [Google Scholar]
- Cheng, Y.T.; Germain, H.; Wiermer, M.; Bi, D.; Xu, F.; Garcíae, A.V.; Wirthmuellere, L.; Desprésf, C.; Parkere, J.E.; Zhang, Y.; et al. Nuclear pore complex component MOS7/Nup88 is required for innate immunity and nuclear accumulation of defense regulators in Arabidopsis. Plant Cell 2009, 21, 2503–2516. [Google Scholar]
- Bai, S.; Liu, J.; Chang, C.; Zhang, L.; Maekawa, T.; Wang, Q.; Xiao, W.; Liu, Y.; Chai, J.; Takken, F.L.W.; et al. Structure-function analysis of barley NLR immune receptor MLA10 reveals its cell compartment specific activity in cell death and disease resistance. PLoS Pathog 2012, 8, e1002752. [Google Scholar]
- Bhattacharjee, S.; Zamora, A.; Tehseen Azhar, M.; Sacco, M.A.; Lambert, L.H.; Moffett, P. Virus resistance induced by NB–LRR proteins involves Argonaute4-dependent translational control. Plant J 2009, 58, 940–951. [Google Scholar]
- Kobayashi, M.; Seo, S.; Hirai, K.; Yamamoto-Katou, A.; Katou, S.; Seto, H.; Meshi, T.; Mitsuhara, I.; Ohashi, Y. Silencing of WIPK and SIPK mitogen-activated protein kinases reduces tobacco mosaic virus accumulation but permits systemic viral movement in tobacco possessing the N resistance gene. Mol. Plant Microbe Interact 2010, 23, 1032–1041. [Google Scholar]
- Kim, S.H.; Kwon, S.I.; Saha, D.; Anyanwu, N.C.; Gassmann, W. Resistance to the Pseudomonas syringae effector HopA1 is governed by the TIR-NBS-LRR protein RPS6 and is enhanced by mutations in SRFR11. Plant Physiol 2009, 150, 1723–1732. [Google Scholar]
- Staal, J.; Dixelius, C. RLM3, a potential adaptor between specific TIR-NB-LRR receptors and DZC proteins. Commun. Integr. Biol 2008, 1, 59–61. [Google Scholar]
- Dinesh-Kumar, S.P.; Baker, B. Alternatively spliced N resistance gene transcripts: Their possible role in tobacco mosaic virus resistance. Proc. Natl. Acad. Sci. USA 2000, 97, 1908–1913. [Google Scholar]
- Weaver, L.M.; Swiderski, M.R.; Li, Y.; Jones, J.D.G. The Arabidopsis thaliana TIR-NB-LRR R-protein, RPP1A; protein localization and constitutive activation of defence by truncated alleles in tobacco and Arabidopsis. Plant J 2006, 47, 829–840. [Google Scholar]
- Zhang, X.C.; Gassmann, W. RPS4-mediated disease resistance requires the combined presence of RPS4 transcripts with full-length and truncated open reading frames. Plant Cell 2003, 15, 2333–2342. [Google Scholar]
- Zhang, Y.; Goritschnig, S.; Dong, X.; Li, X. A gain-of-function mutation in a plant disease resistance gene leads to constitutive activation of downstream signal transduction pathways in suppressor of npr1-1, constitutive 1. Plant Cell 2003, 15, 2636–2646. [Google Scholar]
- Duque, P. A role for SR proteins in plant stress responses. Plant Signal. Behav 2011, 6, 49–54. [Google Scholar]
- Xu, S.; Zhang, Z.; Jing, B.; Gannon, P.; Ding, J.; Xu, F.; Li, X.; Zhang, Y. Transportin-SR is required for proper splicing of resistance genes and plant immunity. PLoS Genet 2011, 7, e1002159. [Google Scholar]
- Mallory, A.C.; Bouché, N. MicroRNA-directed regulation: To cleave or not to cleave. Trends Plant Sci 2008, 13, 359–367. [Google Scholar]
- Baulcombe, D. RNA silencing in plants. Nature 2004, 431, 356–363. [Google Scholar]
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar]
- Voinnet, O. Origin, biogenesis, and activity of plant microRNAs. Cell 2009, 136, 669–687. [Google Scholar]
- Bari, R.; Datt Pant, B.; Stitt, M.; Scheible, W.-R. PHO2, microRNA399, and PHR1 define a phosphate-signaling pathway in plants. Plant Physiol 2006, 141, 988–999. [Google Scholar]
- Pant, B.D.; Buhtz, A.; Kehr, J.; Scheible, W.R. Micro-RNA399 is a long-distance signal for the regulation of plant phosphate homeostasis. Plant J 2008, 53, 731–738. [Google Scholar]
- Yi, H.; Richards, E.J. A cluster of disease resistance genes in Arabidopsis is coordinately regulated by transcriptional activation and RNA silencing. Plant Cell 2007, 19, 2929–2939. [Google Scholar]
- Li, F.; Pignatta, D.; Bendix, C.; Brunkard, J.O.; Cohn, M.M.; Tung, J.; Sun, H.; Kumar, P.; Baker, B. MicroRNA regulation of plant innate immune receptors. Proc. Natl. Acad. Sci. USA 2012, 109, 1790–1795. [Google Scholar]
- Shivaprasad, P.V.; Chen, H.-M.; Patel, K.; Bond, D.M.; Santos, B.A.C.M.; Baulcombe, D.C. A microRNA superfamily regulates nucleotide binding site leucine-rich repeats and other mRNAs. Plant Cell 2012, 24, 859–874. [Google Scholar]
- Li, X.; Clarke, J.D.; Zhang, Y.; Dong, X. Activation of an EDS1-mediated R-gene pathway in the snc1 mutant leads to constitutive, NPR1-independent pathogen resistance. Mol. Plant Microbe Interact 2001, 14, 1131–1139. [Google Scholar]
- Tornero, P.; Merritt, P.; Sadanandom, A.; Shirasu, K.; Innes, R.W.; Dangl, J.L. RAR1 and NDR1 contribute quantitatively to disease resistance in Arabidopsis, and their relative contributions are dependent on the R gene assayed. Plant Cell 2002, 14, 1005–1015. [Google Scholar]
- Bieri, S.; Mauch, S.; Shen, Q.H.; Peart, J.; Devoto, A.; Casais, C.; Ceron, F.; Schulze, S.; Steinbiß, H.-H.; Shirasu, K.; Schulze-Lefert, P. RAR1 positively controls steady state levels of barley MLA resistance proteins and enables sufficient MLA6 accumulation for effective resistance. Plant Cell 2004, 16, 3480–3495. [Google Scholar]
- Holt, B.F.; Belkhadir, Y.; Dangl, J.L. Antagonistic control of disease resistance protein stability in the plant immune system. Science 2005, 309, 929–932. [Google Scholar]
- Hubert, D.A.; Tornero, P.; Belkhadir, Y.; Krishna, P.; Takahashi, A.; Shirasu, K.; Dangl, J.L. Cytosolic HSP90 associates with and modulates the Arabidopsis RPM1 disease resistance protein. EMBO J 2003, 22, 5679–5689. [Google Scholar]
- Lu, R.; Malcuit, I.; Moffett, P.; Ruiz, M.T.; Peart, J.; Wu, A.-J.; Rathjen, J.P.; Bendahmane, A.; Day, L.; Baulcombe, D.C. High throughput virus-induced gene silencing implicates heat shock protein 90 in plant disease resistance. EMBO J 2003, 22, 5690–5699. [Google Scholar]
- Li, Y.; Li, S.; Bi, D.; Cheng, Y.T.; Li, X.; Zhang, Y. SRFR1 Negatively regulates plant NB-LRR resistance protein accumulation to prevent autoimmunity. PLoS Pathog 2010, 6, e1001111. [Google Scholar]
- Austin, M.J.; Muskett, P.; Kahn, K.; Feys, B.J.; Jones, J.D.; Parker, J.E. Regulatory role of SGT1 in early R gene-mediated plant defenses. Science 2002, 295, 2077–2080. [Google Scholar]
- Tör, M.; Gordon, P.; Cuzick, A.; Eulgem, T.; Sinapidou, E.; Mert, F.; Can, C.; Dangl, J.L.; Holub, E.B. Arabidopsis SGT1b is required for defense signaling conferred by several downy mildew (Peronospora parasitica) resistance genes. Plant Cell 2002, 14, 993–1003. [Google Scholar]
- Dielen, A.S.; Badaoui, S.; Candresse, T.; German-Retana, S. The ubiquitin/26S proteasome system in plant-pathogen interactions: A never-ending hide-and-seek game. Mol. Plant Pathol 2010, 11, 293–308. [Google Scholar]
- Wiermer, M.; Feys, B.J.; Parker, J.E. Plant immunity: The EDS1 regulatory node. Curr. Opin. Plant Biol 2005, 8, 383–389. [Google Scholar]
- Marone, D.; Panio, G.; Ficco, D.B.M.; Russo, M.A.; de Vita, P.; Papa, R.; Rubiales, D.; Cattivelli, L.; Mastrangelo, A. Characterization of wheat DArT markers: Genetic and functional features. Mol. Genet. Genomics 2012, 287, 741–753. [Google Scholar]
- Marone, D.; Laidò, G.; Gadaleta, A.; Colasuonno, P.; Ficco, D.B.M.; Giancaspro, A.; Giove, S.; Panio, G.; Russo, M.A.; de Vita, P.; et al. A high-density consensus map of A and B wheat genomes. Theor. Appl. Genet 2012, 125, 1619–1638. [Google Scholar]
- Maccaferri, M.; Ratti, C.; Rubies-Autonell, C.; Vallega, V.; Demontis, A.; Stefanelli, S.; Tuberosa, R.; Sanguineti, M.C. Resistance to soil-borne cereal mosaic virus in durum wheat is controlled by a major QTL on chromosome arm 2BS and minor loci. Theor. Appl. Genet 2011, 123, 527–544. [Google Scholar]
- Russo, M.A.; Ficco, D.B.M.; Marone, D.; de Vita, P.; Vallega, V.; Rubies-Autonell, C.; Ratti, C.; Ferragonio, P.; Giovanniello, V.; Pecchioni, N.; et al. A major QTL for resistance to soil-borne cereal mosaic virus derived from an old Italian durum wheat cultivar. J. Plant Int 2012, 7, 290–300. [Google Scholar]
- Yu, L.X.; Lorenz, A.; Rutkoski, J.; Singh, R.P.; Bhavani, S.; Huerta-Espino, J.; Sorrells, M.E. Association mapping and gene–gene interaction for stem rust resistance in CIMMYT spring wheat germplasm. Theor. Appl. Genet 2011, 123, 1257–1268. [Google Scholar]
- Ashfield, T.; Bocian, A.; Held, D.; Henk, A.D.; Marek, L.F.; Danesh, D.; Peñuela, S.; Meksem, K.; Lightfoot, D.A.; Young, N.D.; et al. Genetic and physical localization of the soybean Rpg1-b disease resistance gene reveals a complex locus containing several tightly linked families of NBS-LRR genes. Mol. Plant Microbe Interact 2003, 16, 817–826. [Google Scholar]
- Bakker, E.; Borm, T.; Prins, P.; van der Vossen, E.; Uenk, G.; Arens, M.; de Boer, J.; van Eck, H.; Muskens, M.; Vossen, J.; et al. A genome-wide genetic map of NB-LRR disease resistance loci in potato. Theor. Appl. Genet 2011, 123, 493–508. [Google Scholar]
- Van der Vossen, E.A.; Gros, J.; Sikkema, A.; Muskens, M.; Wouters, D.; Wolters, P.; Pereira, A.; Allefs, S. The Rpi-blb2 gene from Solanum bulbocastanum is an Mi-1 gene homolog conferring broad-spectrum late blight resistance in potato. Plant J 2005, 44, 208–222. [Google Scholar]
- Gòmez, P.; Rodrìguez-Hernàndez, A.M.; Moury, B.; Aranda, M.A. Genetic resistance for the sustainable control of plant virus disease: Breeding, mechanisms and durability. Eur. J. Plant Pathol 2009, 125, 1–22. [Google Scholar]
- Todorovska, E.; Christov, N.; Slavov, S.; Christova, P.; Vassilev, D. Biotic stress resistance in wheat—Breeding and genomic selection implications. Biotechnol. Biotechnol. Equip 2009, 23, 1417–1426. [Google Scholar]
- Collinge, D.B.; Jørgensen, H.J.L.; Lund, O.S.; Lyngkjær, M.F. Engineering pathogen resistance in crop plants: Current trends and future prospects. Annu. Rev. Phytopathol 2010, 48, 269–291. [Google Scholar]
- Parlevliet, J.E. Durability of resistance against fungal, bacterial and viral pathogens; present situation. Euphytica 2002, 124, 147–156. [Google Scholar]
- Poland, J.A.; Balint-Kurti, P.J.; Wisser, R.J.; Pratt, R.C.; Nelson, R.J. Shades of gray: The world of quantitative disease resistance. Trends Plant Sci 2008, 14, 21–29. [Google Scholar]
- Bhullar, N.K.; Zhang, Z.; Wicker, T.; Keller, B. Wheat gene bank accessions as a source of new alleles of the powdery mildew resistance gene Pm3: A large scale allele mining project. BMC Plant Biol 2010, 10, 88. [Google Scholar]
- Brunner, S.; Stirnweis, D.; Diaz Quijano, C.; Buesing, G.; Herren, G.; Parlange, F.; Barret, P.; Tassy, C.; Sautter, C.; Winzeler, M.; et al. Transgenic Pm3 multilines of wheat show increased powdery mildew resistance in the field. Plant Biotechnol. J 2012, 10, 398–409. [Google Scholar]
- Miedaner, T.; Korzun, V. Marker-assisted selection for disease resistance in wheat and barley breeding. Phytopathology 2012, 102, 560–566. [Google Scholar]
- Okuyama, Y.; Kanzaki, H.; Abe, A.; Yoshida, K.; Tamiru, M.; Saitoh, H.; Fujibe, T.; Matsumura, H.; Shenton, M.; Galam, D.C.; et al. A multifaceted genomics approach allows the isolation of the rice Pia-blast resistance gene consisting of two adjacent NBS-LRR protein genes. Plant J 2011, 66, 467–479. [Google Scholar]
- Liu, J.; Wang, X.; Mitchell, T.; Hu, Y.; Liu, X.; Dai, L.; Wang, G.L. Recent progress and understanding of the molecular mechanisms of the rice–Magnaporthe oryzae interaction. Mol. Plant Pathol 2010, 11, 419–427. [Google Scholar]
- Lee, S.K.; Song, M.Y.; Seo, Y.S. Rice Pi5 mediated rsistance to Magnaporthe oryzae requires the presence of two coiled-coil-nucleotide-binding-leucine-rich repeat genes. Genetics 2009, 181, 1627–1638. [Google Scholar]
- Jeung, J.U.; Kim, B.R.; Cho, Y.C.; Han, S.S.; Moon, H.P.; Lee, Y.T.; Jena, K.K. A novel gene, Pi40(t), linked to the DNA markers derived from NBS-LRR motifs confers broad spectrum of blast resistance in rice. Theor. Appl. Genet 2007, 115, 1163–1177. [Google Scholar]
- Narayanan, N.N.; Baisakh, N.; Oliva, N.P.; VeraCruz, C.M.; Gnanamanickam, S.S.; Datta, K.; Datta, S.K. Molecular breeding: Marker-assisted selection combined with biolistic transformation for blast and bacterial blight resistance in Indica rice (cv. CO39). Mol. Breed 2004, 14, 61–71. [Google Scholar]
- Borrelli, G.M.; de Vita, P.; Mastrangelo, A.M.; Cattivelli, L. Integrated Views in Plant Breeding: Modern Approaches for An Old Topic. In Applied Crop Physiology: Boundaries with Genetic Improvement and Agronomy. Part 3—Crop Physiology, Genetic Improvement, and Agronomy; Sadras, V.O., Calderini, D.F., Eds.; Elsevier: New York, NY, USA, 2009. [Google Scholar]
- Cloutier, S.; McCallum, B.D.; Loutre, C.; Banks, T.W.; Wicker, T.; Feuillet, C.; Keller, B.; Jordan, M.C. Leaf rust resistance gene Lr1, isolated from bread wheat (Triticum aestivum L.) is a member of the large psr567 gene family. Plant Mol. Biol 2007, 65, 93–106. [Google Scholar]
- Feuillet, C.; Travella, S.; Stein, N.; Albar, L.; Nublat, A.; Keller, B. Map-based isolation of the leaf rust disease resistance gene Lr10 from the hexaploid wheat (Triticum aestivum L.) genome. Proc. Natl. Acad. Sci. USA 2003, 100, 15253–15258. [Google Scholar]
- Huang, L.; Brooks, S.A.; Li, W.; Fellers, J.P.; Trick, H.N.; Gill, B.S. Map-based cloning of leaf rust resistance gene Lr21 from the large and polyploid genome of bread wheat. Genetics 2003, 164, 655–664. [Google Scholar]
| Plant species | Total number of NBS-LRR genes | TNL * | CNL * | Number of pseudogenes | References |
|---|---|---|---|---|---|
| Arabidopsis thaliana | 149 | 94 | 55 | 10 | [20] |
| 159 | 98 | 50 | - | [21] | |
| Populus trichocarpa | 402 | 91 | 119 | 161 | [22] |
| Medicago truncatula | 333 | 156 | 177 | 49 | [19] |
| Vitis vinifera | 459 | 97 | 203 | - | [23] |
| Oryza sativa L. spp. indica | 653 | - | - | 184 | [14] |
| Oryza sativa L. spp. japonica | 553 | - | - | 150 | |
| Carica papaya | 54 | 7 | 6 | - | [12] |
| Cucumis sativus | 57 | 13 | 18 | - | [13] |
| Brassica rapa | 92 | 62 | 30 | - | [17] |
| Lotus japonicus | 158 | 32 | 28 | 62 | [18] |
| Arabidopsis lyrata | 185 | 123 | 38 | - | [21] |
| Glycine max (soybean) | 319 | - | - | - | [24] |
| Brachypodium distachyon | 126 | 0 | 113 | - | [9] |
| Solanum tuberosum | 438 | 77 | 361 | - | [15] |
| 435 | 65 | 370 | 179 | [16] |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Marone, D.; Russo, M.A.; Laidò, G.; De Leonardis, A.M.; Mastrangelo, A.M. Plant Nucleotide Binding Site–Leucine-Rich Repeat (NBS-LRR) Genes: Active Guardians in Host Defense Responses. Int. J. Mol. Sci. 2013, 14, 7302-7326. https://doi.org/10.3390/ijms14047302
Marone D, Russo MA, Laidò G, De Leonardis AM, Mastrangelo AM. Plant Nucleotide Binding Site–Leucine-Rich Repeat (NBS-LRR) Genes: Active Guardians in Host Defense Responses. International Journal of Molecular Sciences. 2013; 14(4):7302-7326. https://doi.org/10.3390/ijms14047302
Chicago/Turabian StyleMarone, Daniela, Maria A. Russo, Giovanni Laidò, Anna M. De Leonardis, and Anna M. Mastrangelo. 2013. "Plant Nucleotide Binding Site–Leucine-Rich Repeat (NBS-LRR) Genes: Active Guardians in Host Defense Responses" International Journal of Molecular Sciences 14, no. 4: 7302-7326. https://doi.org/10.3390/ijms14047302





