Basic Amino Acid Residues of Human Eosinophil Derived Neurotoxin Essential for Glycosaminoglycan Binding
Abstract
:1. Introduction
2. Results and Discussion
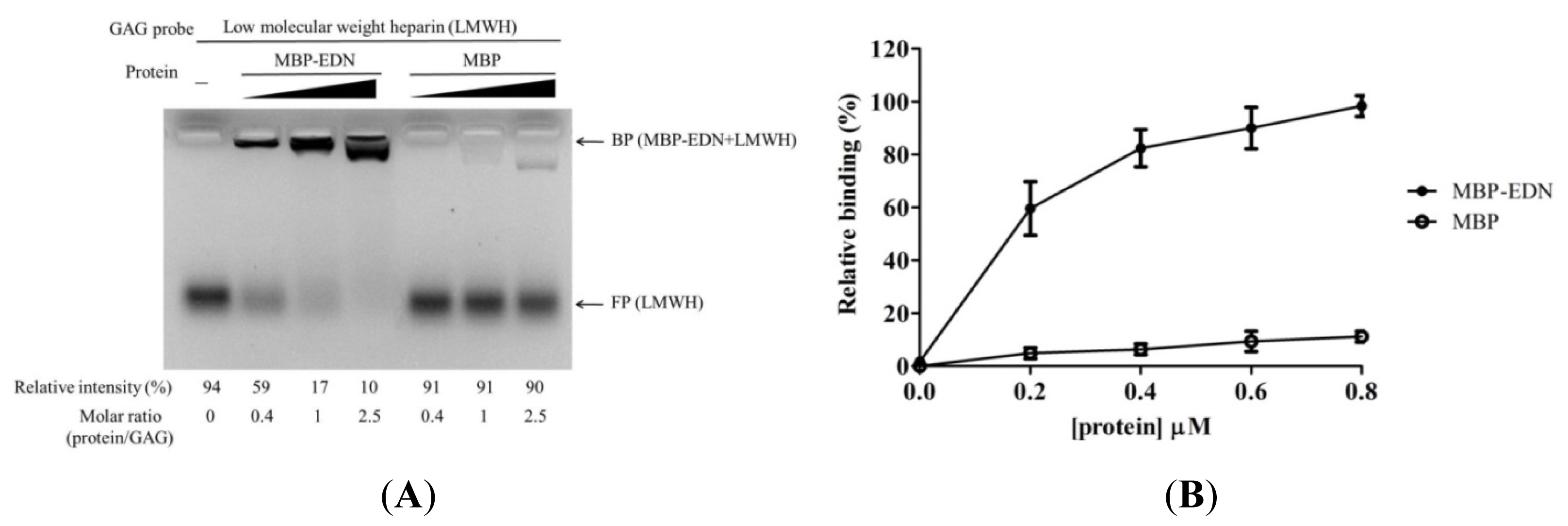
2.1. Binding of MBP-EDN to Heparin and Beas-2B Cells
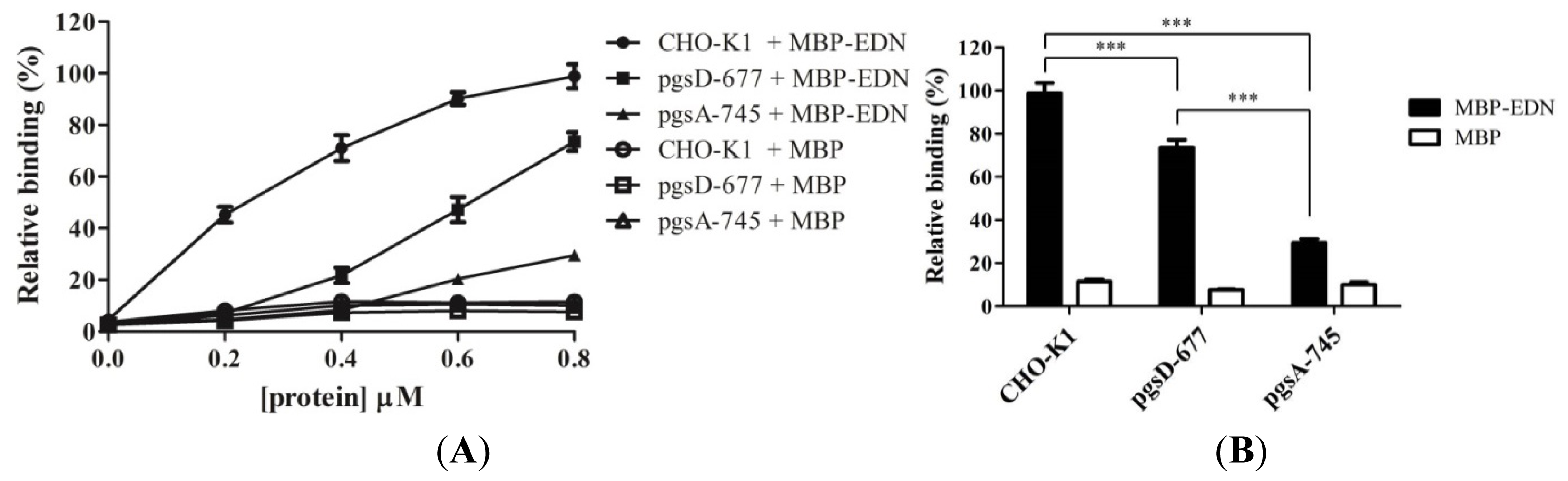
2.2. Involvement of Cell Surface GAGs in Binding to MBP-EDN
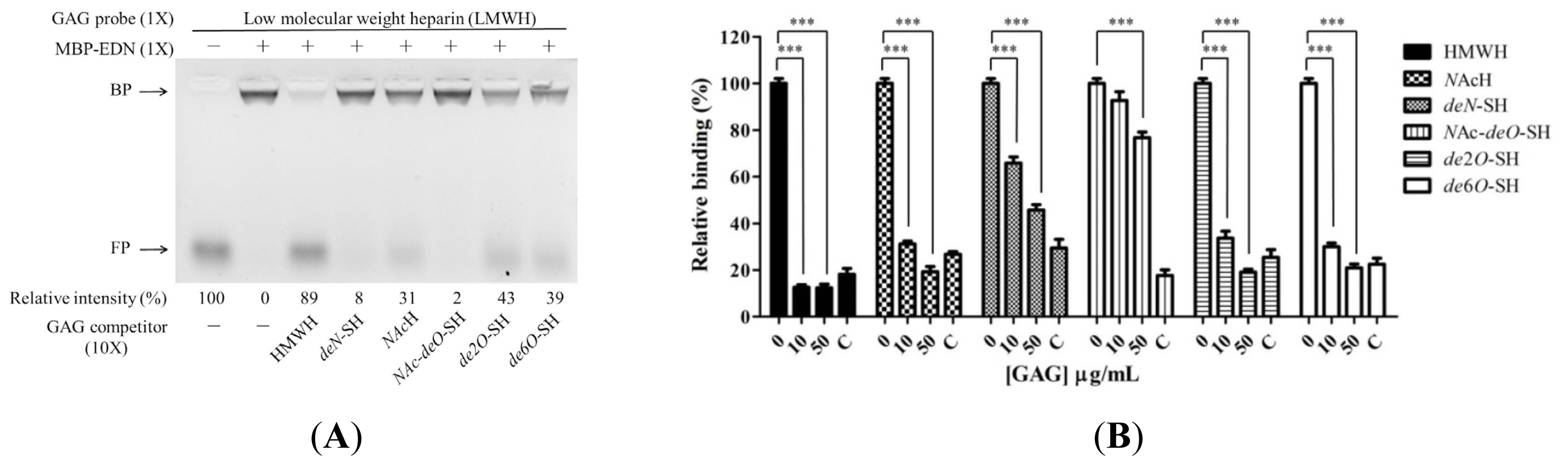
2.3. Requirement of Sulfo Groups on Heparin for Binding to EDN
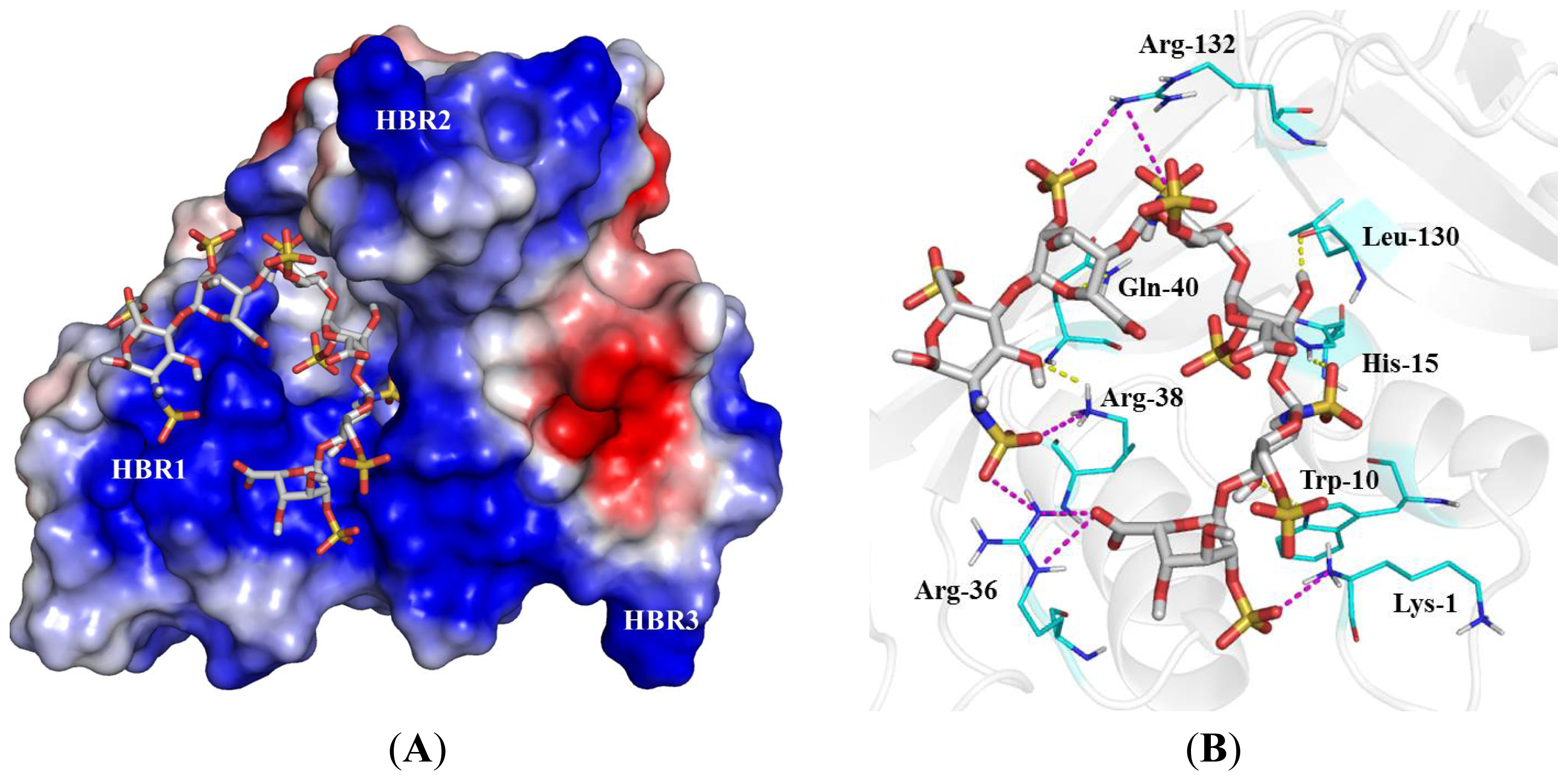
2.4. Identification of Critical Basic Residues on EDN for Interaction with Heparin and Other Cell Surface GAG
3. Experimental Section
3.1. Antibodies and Reagents
3.2. Cells and Cell Culture
3.3. Construction, Expression and Purification of MBP-EDN Mutants
3.4. Construction, Expression and Purification of Recombinant EDN Containing a C-Terminal 6His-Tag (EDN-6His)
3.5. Cell-Based Enzyme-Link Immunosorbent Assay (cELISA)
3.6. Fluorescence-Assisted Carbohydrate Electrophoresis (FACE)
3.7. Binding Competition Assay
3.8. Computer Modeling of EDN-Heparin Oligosaccharide Complex
3.9. Statistical Analysis
4. Conclusions
Supplementary Information
ijms-14-19067-s001.pdfAcknowledgments
Conflicts of Interest
Abbreviations
| AMAC | 2-aminoacridone |
| CHO | Chinese hamster ovary |
| CS | chondroitin sulfate |
| CSA | chondroitin sulfate A |
| CSB | chondroitin sulfate B |
| CSC | chondroitin sulfate C |
| DC | dendritic cell |
| DS | dermatan sulfate |
| de2O-SH | de-2-O-sulfated heparin |
| de6O-SH | de-6-O-sulfated heparin |
| deN-SH | de-N-sulfated heparin |
| ECP | eosinophil cationic protein |
| EDN | eosinophil derived neurotoxin |
| FACE | fluorescence-assisted carbohydrate electrophoresis |
| FGF | fibroblast growth factor |
| GAG | glycosaminoglycan |
| HS | heparan sulfate |
| HBR | heparin binding region |
| HMWH | high molecular weight heparin |
| HRP | horseradish peroxidase |
| HIV | human immunodeficiency virus |
| hRSV | human respiratory syncytial virus |
| HA | hyaluronic acid |
| LMWH | low molecular weight heparin |
| MBP | Maltose-binding protein |
| NAc-deO-SH | N-acetyl-de-O-sulfated heparin |
| TLR2 | toll-like receptor 2 |
| NAcH | N-acetyl heparin |
| PFA | paraformaldehyde |
| SD | standard deviation |
| mEAR | mouse eosinophil-associated RNase |
| Th2 | Type 2 helper T cell |
References
- Gleich, G.J.; Adolphson, C.R. The eosinophilic leukocyte: Structure and function. Adv. Immunol 1986, 39, 177–253. [Google Scholar]
- Rosenberg, H.F.; Tenen, D.G.; Ackerman, S.J. Molecular cloning of the human eosinophil-derived neurotoxin: A member of the ribonuclease gene family. Proc. Natl. Acad. Sci. USA 1989, 86, 4460–4464. [Google Scholar]
- Gleich, G.J. Eosinophils, basophils, and mast cells. J. Allergy Clin. Immunol 1989, 84, 1024–1027. [Google Scholar]
- Futami, J.; Tsushima, Y.; Murato, Y.; Tada, H.; Sasaki, J.; Seno, M.; Yamada, H. Tissue-specific expression of pancreatic-type rnases and rnase inhibitor in humans. DNA Cell Biol 1997, 16, 413–419. [Google Scholar]
- Durack, D.T.; Sumi, S.M.; Klebanoff, S.J. Neurotoxicity of human eosinophils. Proc. Natl. Acad. Sci. USA 1979, 76, 1443–1447. [Google Scholar]
- Durack, D.T.; Ackerman, S.J.; Loegering, D.A.; Gleich, G.J. Purification of human eosinophil-derived neurotoxin. Proc. Natl. Acad. Sci. USA 1981, 78, 5165–5169. [Google Scholar]
- Fredens, K.; Dahl, R.; Venge, P. The gordon phenomenon induced by the eosinophil cationic protein and eosinophil protein x. J. Allergy Clin. Immunol 1982, 70, 361–366. [Google Scholar]
- Domachowske, J.B.; Dyer, K.D.; Bonville, C.A.; Rosenberg, H.F. Recombinant human eosinophil-derived neurotoxin/rnase 2 functions as an effective antiviral agent against respiratory syncytial virus. J. Infect. Dis 1998, 177, 1458–1464. [Google Scholar]
- Lee-Huang, S.; Huang, P.L.; Sun, Y.; Kung, H.F.; Blithe, D.L.; Chen, H.C. Lysozyme and rnases as anti-hiv components in beta-core preparations of human chorionic gonadotropin. Proc. Natl. Acad. Sci. USA 1999, 96, 2678–2681. [Google Scholar]
- Rugeles, M.T.; Trubey, C.M.; Bedoya, V.I.; Pinto, L.A.; Oppenheim, J.J.; Rybak, S.M.; Shearer, G.M. Ribonuclease is partly responsible for the hiv-1 inhibitory effect activated by hla alloantigen recognition. AIDS 2003, 17, 481–486. [Google Scholar]
- Kephart, G.M.; Alexander, J.A.; Arora, A.S.; Romero, Y.; Smyrk, T.C.; Talley, N.J.; Kita, H. Marked deposition of eosinophil-derived neurotoxin in adult patients with eosinophilic esophagitis. Am. J. Gastroenterol 2010, 105, 298–307. [Google Scholar]
- Liu, G.T.; Hwang, C.S.; Hsieh, C.H.; Lu, C.H.; Chang, S.L.; Lee, J.C.; Huang, C.F.; Chang, H.T. Eosinophil-derived neurotoxin is elevated in patients with amyotrophic lateral sclerosis. Mediators Inflamm 2013, 2013, 421389. [Google Scholar]
- Yang, D.; Rosenberg, H.F.; Chen, Q.; Dyer, K.D.; Kurosaka, K.; Oppenheim, J.J. Eosinophil-derived neurotoxin (edn), an antimicrobial protein with chemotactic activities for dendritic cells. Blood 2003, 102, 3396–3403. [Google Scholar]
- Yang, D.; Chen, Q.; Rosenberg, H.F.; Rybak, S.M.; Newton, D.L.; Wang, Z.Y.; Fu, Q.; Tchernev, V.T.; Wang, M.; Schweitzer, B.; et al. Human ribonuclease a superfamily members, eosinophil-derived neurotoxin and pancreatic ribonuclease, induce dendritic cell maturation and activation. J. Immunol 2004, 173, 6134–6142. [Google Scholar]
- Yang, D.; Chen, Q.; Su, S.B.; Zhang, P.; Kurosaka, K.; Caspi, R.R.; Michalek, S.M.; Rosenberg, H.F.; Zhang, N.; Oppenheim, J.J. Eosinophil-derived neurotoxin acts as an alarmin to activate the tlr2-myd88 signal pathway in dendritic cells and enhances th2 immune responses. J. Exp. Med 2008, 205, 79–90. [Google Scholar]
- Mayer, A.K.; Muehmer, M.; Mages, J.; Gueinzius, K.; Hess, C.; Heeg, K.; Bals, R.; Lang, R.; Dalpke, A.H. Differential recognition of tlr-dependent microbial ligands in human bronchial epithelial cells. J. Immunol 2007, 178, 3134–3142. [Google Scholar]
- Jack, C.S.; Arbour, N.; Manusow, J.; Montgrain, V.; Blain, M.; McCrea, E.; Shapiro, A.; Antel, J.P. Tlr signaling tailors innate immune responses in human microglia and astrocytes. J. Immunol 2005, 175, 4320–4330. [Google Scholar]
- Zarember, K.A.; Godowski, P.J. Tissue expression of human toll-like receptors and differential regulation of toll-like receptor mrnas in leukocytes in response to microbes, their products, and cytokines. J. Immunol 2002, 168, 554–561. [Google Scholar]
- Hung, S.C.; Lu, X.A.; Lee, J.C.; Chang, M.D.; Fang, S.L.; Fan, T.C.; Zulueta, M.M.; Zhong, Y.Q. Synthesis of heparin oligosaccharides and their interaction with eosinophil-derived neurotoxin. Org. Biomol. Chem 2012, 10, 760–772. [Google Scholar]
- Gleich, G.J.; Loegering, D.A.; Bell, M.P.; Checkel, J.L.; Ackerman, S.J.; McKean, D.J. Biochemical and functional similarities between human eosinophil-derived neurotoxin and eosinophil cationic protein: Homology with ribonuclease. Proc. Natl. Acad. Sci. USA 1986, 83, 3146–3150. [Google Scholar]
- Prydz, K.; Dalen, K.T. Synthesis and sorting of proteoglycans. J. Cell Sci 2000, 113, 193–205. [Google Scholar]
- Capila, I.; Linhardt, R.J. Heparin-protein interactions. Angew Chem. Int. Ed. Engl 2002, 41, 391–412. [Google Scholar]
- Linhardt, R.J.; Ampofo, S.A.; Fareed, J.; Hoppensteadt, D.; Mulliken, J.B.; Folkman, J. Isolation and characterization of human heparin. Biochemistry 1992, 31, 12441–12445. [Google Scholar]
- Sasisekharan, R.; Raman, R.; Prabhakar, V. Glycomics approach to structure-function relationships of glycosaminoglycans. Annu. Rev. Biomed. Eng 2006, 8, 181–231. [Google Scholar]
- Mulloy, B.; Rider, C.C. Cytokines and proteoglycans: An introductory overview. Biochem. Soc. Trans 2006, 34, 409–413. [Google Scholar]
- Munoz, E.M.; Linhardt, R.J. Heparin-binding domains in vascular biology. Arterioscler. Thromb. Vasc. Biol 2004, 24, 1549–1557. [Google Scholar]
- Cardin, A.D.; Weintraub, H.J. Molecular modeling of protein-glycosaminoglycan interactions. Arteriosclerosis 1989, 9, 21–32. [Google Scholar]
- Fan, T.C.; Fang, S.L.; Hwang, C.S.; Hsu, C.Y.; Lu, X.A.; Hung, S.C.; Lin, S.C.; Chang, M.D. Characterization of molecular interactions between eosinophil cationic protein and heparin. J. Biol. Chem 2008, 283, 25468–25474. [Google Scholar]
- Fang, S.L.; Fan, T.C.; Fu, H.W.; Chen, C.J.; Hwang, C.S.; Hung, T.J.; Lin, L.Y.; Chang, M.D. A novel cell-penetrating peptide derived from human eosinophil cationic protein. PLoS One 2013, 8, e57318. [Google Scholar]
- Newton, D.L.; Nicholls, P.J.; Rybak, S.M.; Youle, R.J. Expression and characterization of recombinant human eosinophil-derived neurotoxin and eosinophil-derived neurotoxin-anti-transferrin receptor sfv. J. Biol. Chem 1994, 269, 26739–26745. [Google Scholar]
- Fan, T.C.; Chang, H.T.; Chen, I.W.; Wang, H.Y.; Chang, M.D. A heparan sulfate-facilitated and raft-dependent macropinocytosis of eosinophil cationic protein. Traffic 2007, 8, 1778–1795. [Google Scholar]
- Watterson, D.; Kobe, B.; Young, P.R. Residues in domain iii of the dengue virus envelope glycoprotein involved in cell-surface glycosaminoglycan binding. J. Gen. Virol 2012, 93, 72–82. [Google Scholar]
- Puck, T.T.; Kao, F.T. Genetics of somatic mammalian cells, vi, use of an antimetabolite in analysis of gene multiplicity. Proc. Natl. Acad. Sci. USA 1968, 60, 561–568. [Google Scholar]
- Lidholt, K.; Weinke, J.L.; Kiser, C.S.; Lugemwa, F.N.; Bame, K.J.; Cheifetz, S.; Massague, J.; Lindahl, U.; Esko, J.D. A single mutation affects both N-acetylglucosaminyltransferase and glucuronosyltransferase activities in a chinese hamster ovary cell mutant defective in heparan sulfate biosynthesis. Proc. Natl. Acad. Sci. USA 1992, 89, 2267–2271. [Google Scholar]
- Esko, J.D. Animal cell mutants defective in heparan sulfate polymerization. Adv. Exp. Med. Biol 1992, 313, 97–106. [Google Scholar]
- Trowbridge, J.M.; Gallo, R.L. Dermatan sulfate: New functions from an old glycosaminoglycan. Glycobiology 2002, 12, 117R–125R. [Google Scholar]
- Hamazaki, H. Ca2+-mediated association of human serum amyloid p component with heparan sulfate and dermatan sulfate. J. Biol. Chem 1987, 262, 1456–1460. [Google Scholar]
- Catlow, K.R.; Deakin, J.A.; Wei, Z.; Delehedde, M.; Fernig, D.G.; Gherardi, E.; Gallagher, J.T.; Pavao, M.S.; Lyon, M. Interactions of hepatocyte growth factor/scatter factor with various glycosaminoglycans reveal an important interplay between the presence of iduronate and sulfate density. J. Biol. Chem 2008, 283, 5235–5248. [Google Scholar]
- Hallak, L.K.; Collins, P.L.; Knudson, W.; Peeples, M.E. Iduronic acid-containing glycosaminoglycans on target cells are required for efficient respiratory syncytial virus infection. Virology 2000, 271, 264–275. [Google Scholar]
- Irie, M. structures and functions of ribonucleases. Yakugaku Zasshi 1997, 117, 561–582. [Google Scholar]
- Rosenberg, H.F. Rnase a ribonucleases and host defense: An evolving story. J. Leukoc. Biol 2008, 83, 1079–1087. [Google Scholar]
- Torrent, M.; Nogues, M.V.; Boix, E. Eosinophil cationic protein (ecp) can bind heparin and other glycosaminoglycans through its rnase active site. J. Mol. Recognit 2011, 24, 90–100. [Google Scholar]
- Ye, S.; Luo, Y.; Lu, W.; Jones, R.B.; Linhardt, R.J.; Capila, I.; Toida, T.; Kan, M.; Pelletier, H.; McKeehan, W.L. Structural basis for interaction of fgf-1, fgf-2, and fgf-7 with different heparan sulfate motifs. Biochemistry 2001, 40, 14429–14439. [Google Scholar]
- Copeland, R.; Balasubramaniam, A.; Tiwari, V.; Zhang, F.; Bridges, A.; Linhardt, R.J.; Shukla, D.; Liu, J. Using a 3-O-sulfated heparin octasaccharide to inhibit the entry of herpes simplex virus type 1. Biochemistry 2008, 47, 5774–5783. [Google Scholar]
- Seki, Y.; Mizukura, M.; Ichimiya, T.; Suda, Y.; Nishihara, S.; Masuda, M.; Takase-Yoden, S. O-sulfate groups of heparin are critical for inhibition of ecotropic murine leukemia virus infection by heparin. Virology 2012, 424, 56–66. [Google Scholar]
- Guimond, S.; Maccarana, M.; Olwin, B.B.; Lindahl, U.; Rapraeger, A.C. Activating and inhibitory heparin sequences for fgf-2 (basic fgf). Distinct requirements for fgf-1, fgf-2, and fgf-4. J. Biol. Chem 1993, 268, 23906–23914. [Google Scholar]
- Ishihara, M.; Kariya, Y.; Kikuchi, H.; Minamisawa, T.; Yoshida, K. Importance of 2-O-sulfate groups of uronate residues in heparin for activation of fgf-1 and fgf-2. J. Biochem 1997, 121, 345–349. [Google Scholar]
- Sikriwal, D.; Seth, D.; Parveen, S.; Malik, A.; Broor, S.; Batra, J.K. An insertion in loop l7 of human eosinophil derived neurotoxin is crucial for its antiviral activity. J. cell. Biochem 2012, 113, 3104–3112. [Google Scholar]
- Sakurai, H.; Williamson, R.A.; Crowe, J.E.; Beeler, J.A.; Poignard, P.; Bastidas, R.B.; Chanock, R.M.; Burton, D.R. Human antibody responses to mature and immature forms of viral envelope in respiratory syncytial virus infection: Significance for subunit vaccines. J. Virol 1999, 73, 2956–2962. [Google Scholar]
- Lien, P.C.; Kuo, P.H.; Chen, C.J.; Chang, H.H.; Fang, S.L.; Wu, W.S.; Lai, Y.K.; Pai, T.W.; Chang, M.D. In silico prediction and in vitro characterization of multifunctional human rnase3. BioMed. Res. Int 2013, 2013, 170398. [Google Scholar]
- Garcia-Mayoral, M.F.; Moussaoui, M.; de la Torre, B.G.; Andreu, D.; Boix, E.; Nogues, M.V.; Rico, M.; Laurents, D.V.; Bruix, M. Nmr structural determinants of eosinophil cationic protein binding to membrane and heparin mimetics. Biophys. J 2010, 98, 2702–2711. [Google Scholar]
- Garcia-Mayoral, M.F.; Canales, A.; Diaz, D.; Lopez-Prados, J.; Moussaoui, M.; de Paz, J.L.; Angulo, J.; Nieto, P.M.; Jimenez-Barbero, J.; Boix, E.; et al. Insights into the glycosaminoglycan-mediated cytotoxic mechanism of eosinophil cationic protein revealed by nmr. ACS Chem. Biol 2013, 8, 144–151. [Google Scholar]
- Boix, E.; Nikolovski, Z.; Moiseyev, G.P.; Rosenberg, H.F.; Cuchillo, C.M.; Nogues, M.V. Kinetic and product distribution analysis of human eosinophil cationic protein indicates a subsite arrangement that favors exonuclease-type activity. J. Biol. Chem 1999, 274, 15605–15614. [Google Scholar]
- Calabro, A.; Benavides, M.; Tammi, M.; Hascall, V.C.; Midura, R.J. Microanalysis of enzyme digests of hyaluronan and chondroitin/dermatan sulfate by fluorophore-assisted carbohydrate electrophoresis (face). Glycobiology 2000, 10, 273–281. [Google Scholar]
- Leonidas, D.D.; Boix, E.; Prill, R.; Suzuki, M.; Turton, R.; Minson, K.; Swaminathan, G.J.; Youle, R.J.; Acharya, K.R. Mapping the ribonucleolytic active site of eosinophil-derived neurotoxin (edn). High resolution crystal structures of edn complexes with adenylic nucleotide inhibitors. J. Biol. Chem 2001, 276, 15009–15017. [Google Scholar]
- Trott, O.; Olson, A.J. Autodock vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem 2010, 31, 455–461. [Google Scholar]

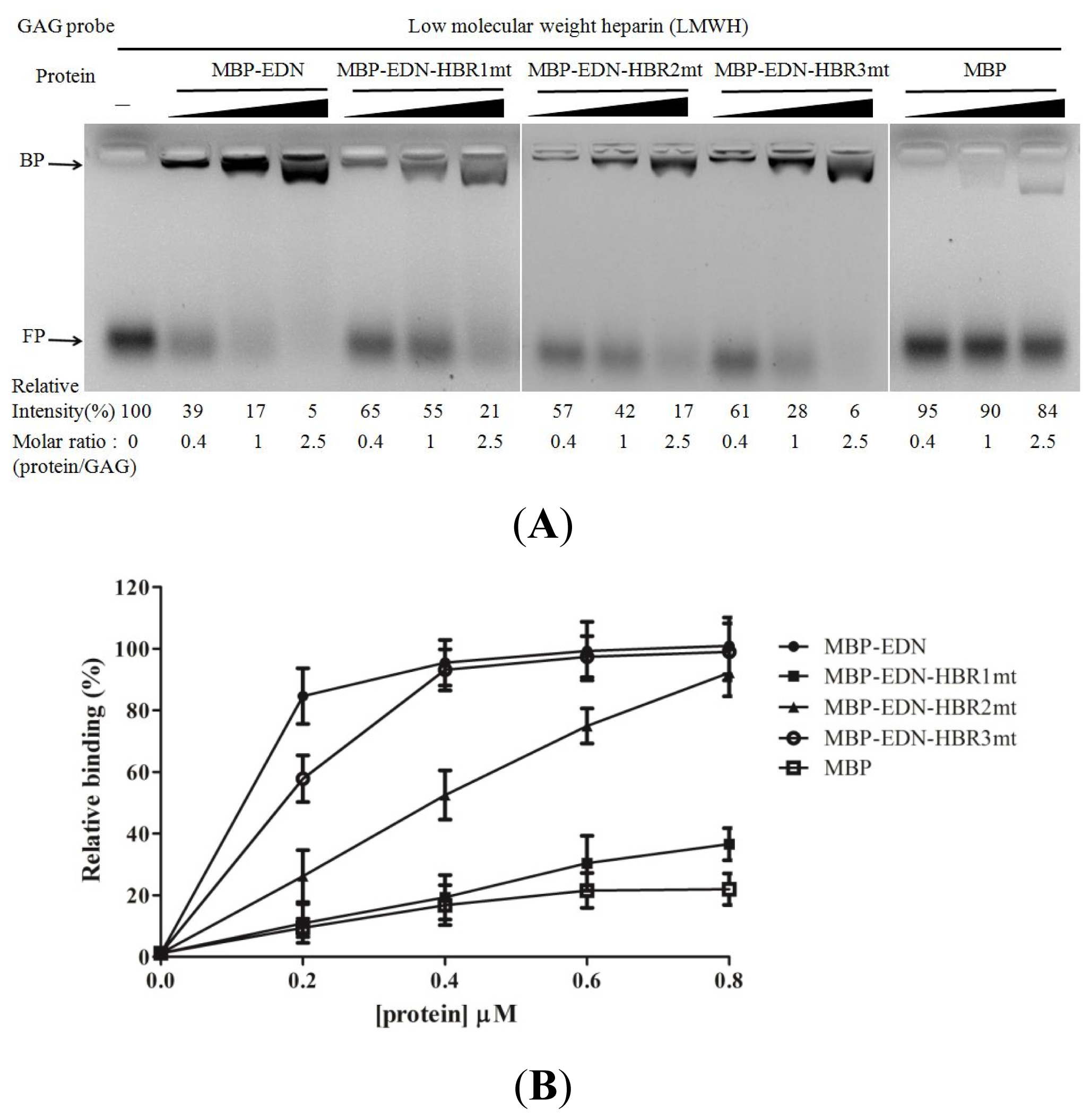
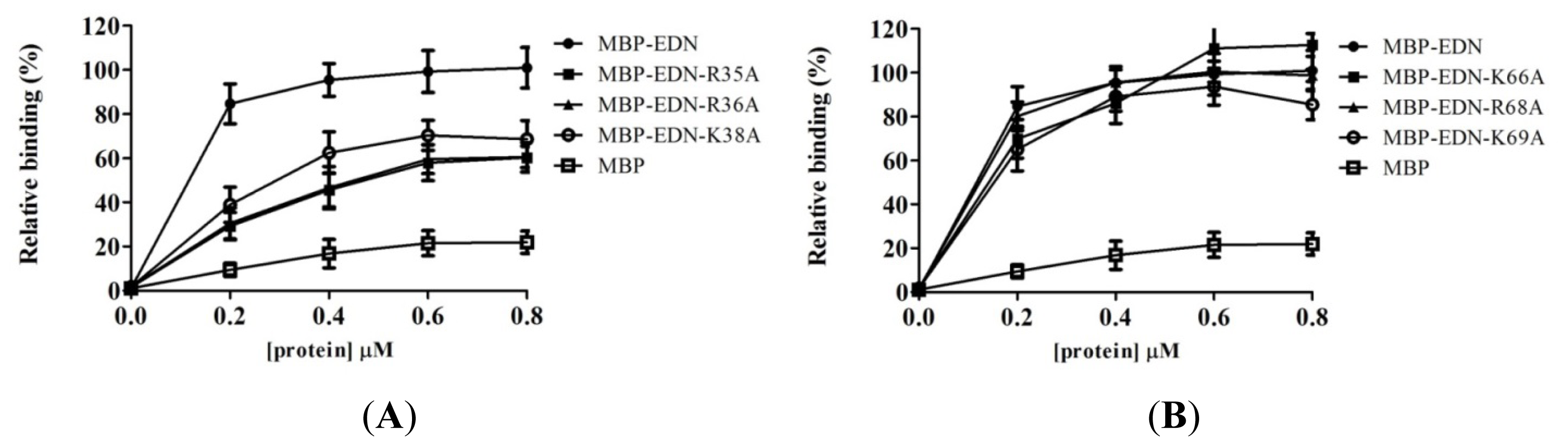
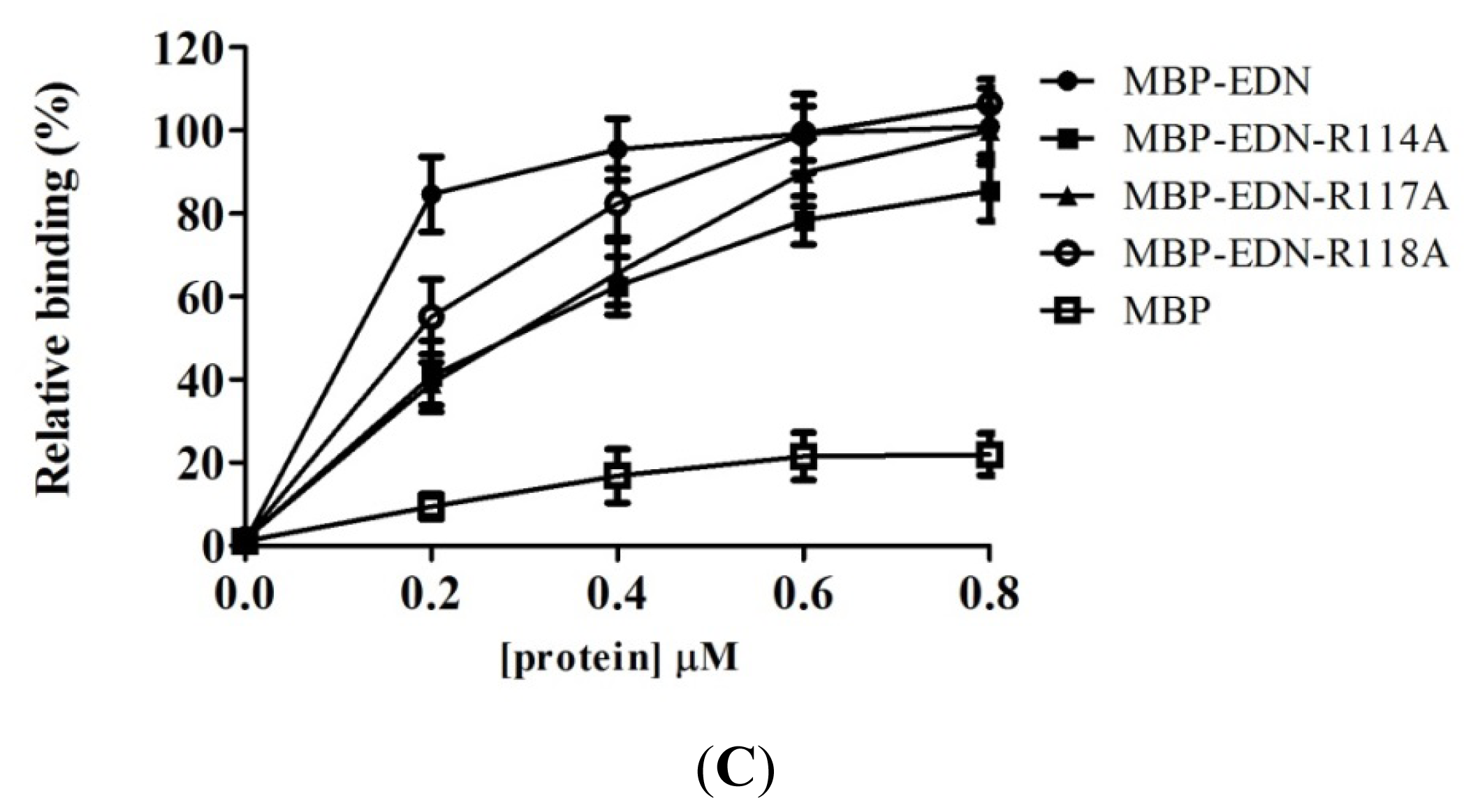
| Protein (0.2 μM) | Mean ± SD (%) |
|---|---|
| MBP-EDN | 84.6 ± 9.0 |
| R35A | 28.5 ± 5.8 *** |
| R36A | 31.0 ± 7.0 *** |
| K38A | 38.9 ± 8.0 *** |
| K66A | 69.9 ± 8.8 * |
| R68A | 80.1 ± 6.5 |
| K69A | 65.1 ± 9.8 *** |
| R114A | 40.8 ± 8.5 *** |
| R117A | 39.0 ± 5.1 *** |
| R118A | 55.1 ± 9.0 *** |
| MBP-EDN-HBR1mt | 10.9 ± 6.3 *** |
| MBP-EDN-HBR2mt | 26.2 ± 8.4 *** |
| MBP-EDN-HBR3mt | 57.8 ± 7.6 *** |
| MBP | 9.4 ± 3.0 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hung, T.-J.; Chang, W.-T.; Tomiya, N.; Lee, Y.-C.; Chang, H.-T.; Chen, C.-J.; Kuo, P.-H.; Fan, T.-c.; Chang, M.D.-T. Basic Amino Acid Residues of Human Eosinophil Derived Neurotoxin Essential for Glycosaminoglycan Binding. Int. J. Mol. Sci. 2013, 14, 19067-19085. https://doi.org/10.3390/ijms140919067
Hung T-J, Chang W-T, Tomiya N, Lee Y-C, Chang H-T, Chen C-J, Kuo P-H, Fan T-c, Chang MD-T. Basic Amino Acid Residues of Human Eosinophil Derived Neurotoxin Essential for Glycosaminoglycan Binding. International Journal of Molecular Sciences. 2013; 14(9):19067-19085. https://doi.org/10.3390/ijms140919067
Chicago/Turabian StyleHung, Ta-Jen, Wei-Tang Chang, Noboru Tomiya, Yuan-Chuan Lee, Hao-Teng Chang, Chien-Jung Chen, Ping-Hsueh Kuo, Tan-chi Fan, and Margaret Dah-Tsyr Chang. 2013. "Basic Amino Acid Residues of Human Eosinophil Derived Neurotoxin Essential for Glycosaminoglycan Binding" International Journal of Molecular Sciences 14, no. 9: 19067-19085. https://doi.org/10.3390/ijms140919067





