Inhibitors of Acetylcholinesterase and Butyrylcholinesterase Meet Immunity
Abstract
:1. Introduction
2. The Cholinergic Anti-Inflammatory Pathway
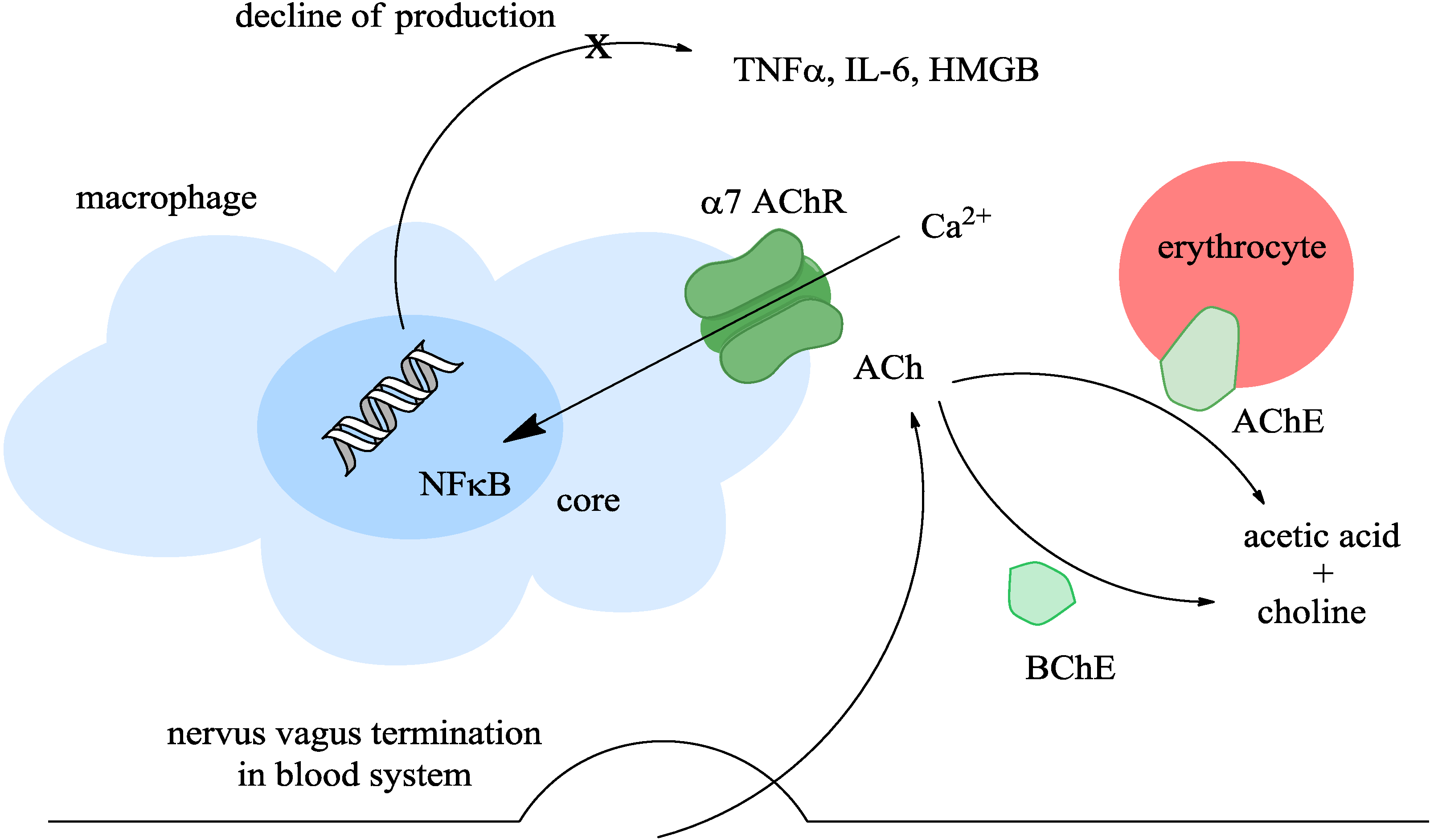
3. Division of Inhibitors
| Group of Compounds | Compounds (Examples) | Mechanism of Inhibition | Inhibition of AChE and BChE | Penetration through Blood Brain Barrier | Importance as Drugs | References |
|---|---|---|---|---|---|---|
| Organophosphates | sarin, soman, tabun, malaoxon | irreversible | equal to AChE and BChE | Good | Low | [6,43,44,45,46,47,48,49,50] |
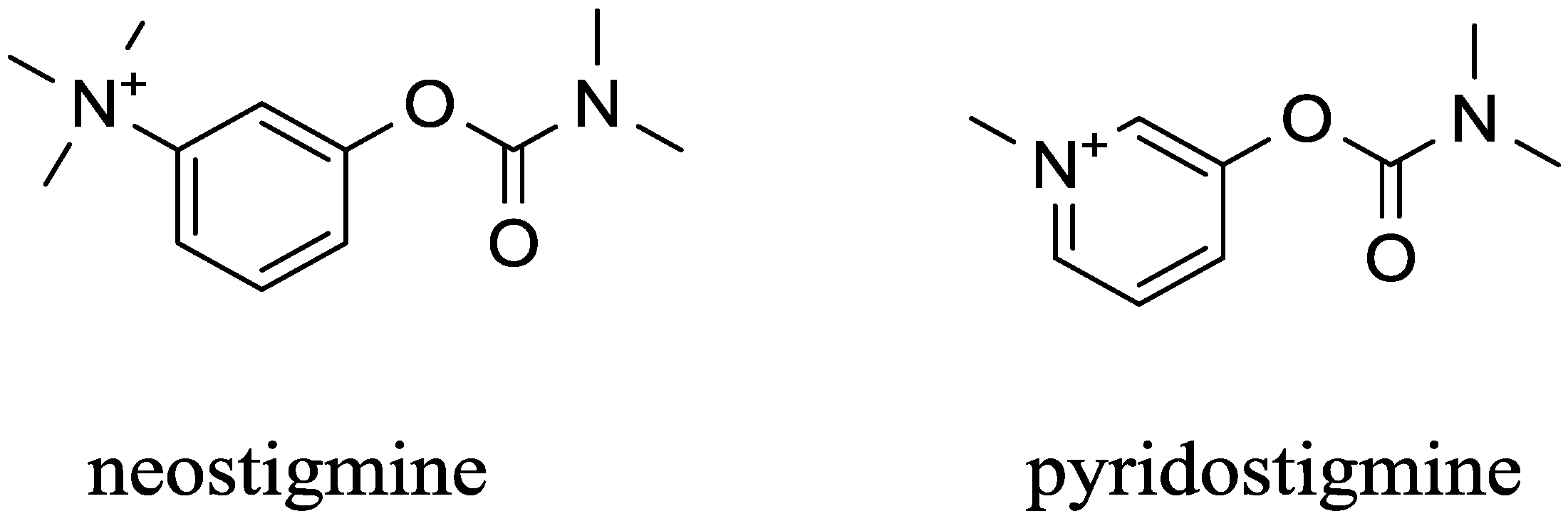
| Carbamates | pyridostigmine, physostigmine neostigmine, rivastigmine | pseudo-irreversible | equal to AChE and BChE | low (pyridostigmine, neostigmine), good (physostigmine, rivastigmine) | High | [29,53,54,55,56,57,58,59,60] |
| - | Tacrine | non-competitive | AChE > BChE | Good | former drug, discontinued now | [53,66,68,69] |
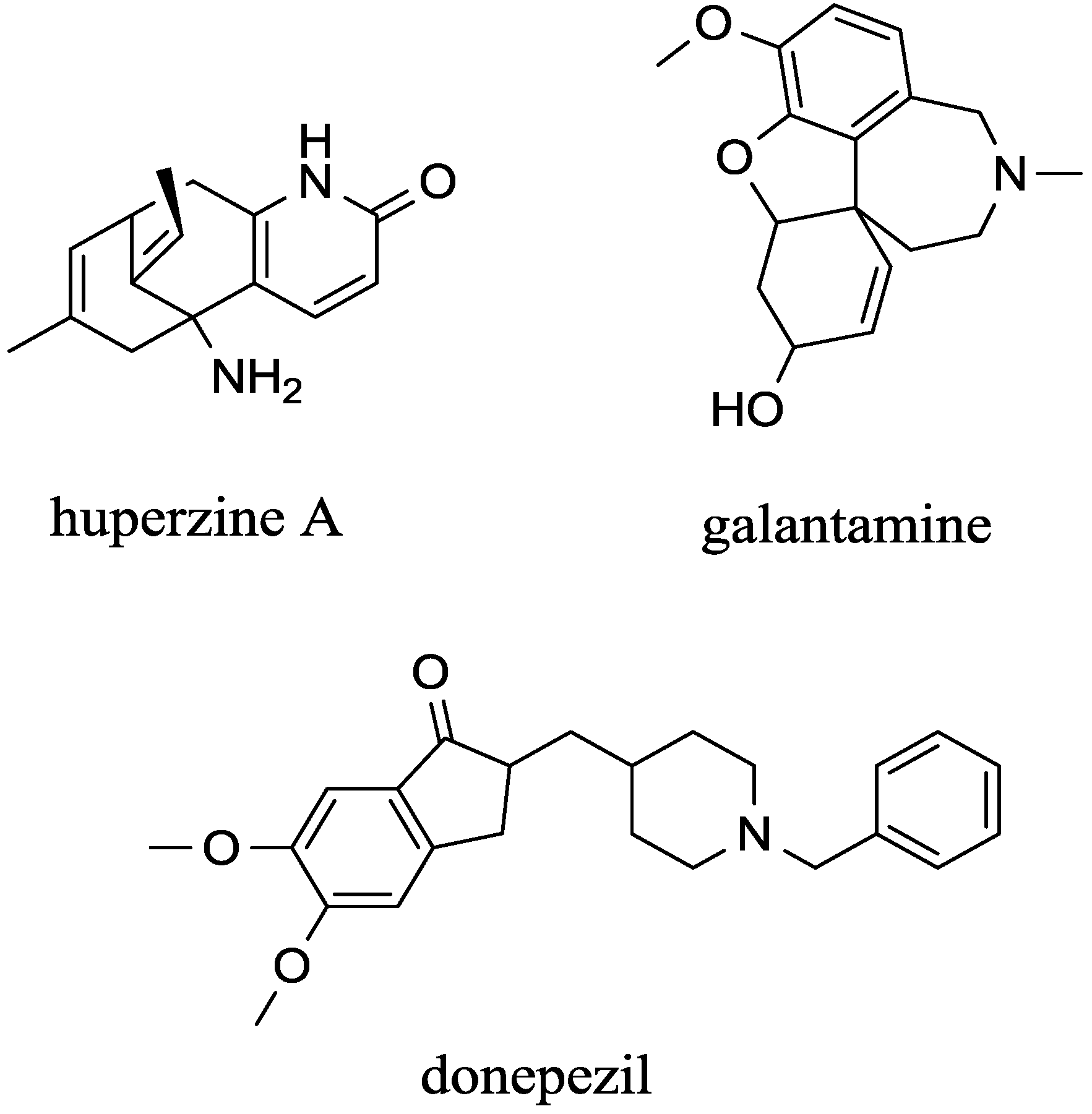
| - | Galantamine | competitive | AChE | Good | High | [73,74] |
| - | Donepezil | non-competitive | AChE | Good | High | [75,76] |
| - | huperzine A | non-competitive | AChE >> BChE | Good | will increase | [29,75,77,78,79] |
4. Inhibitors that Cross the Blood Brain Barrier
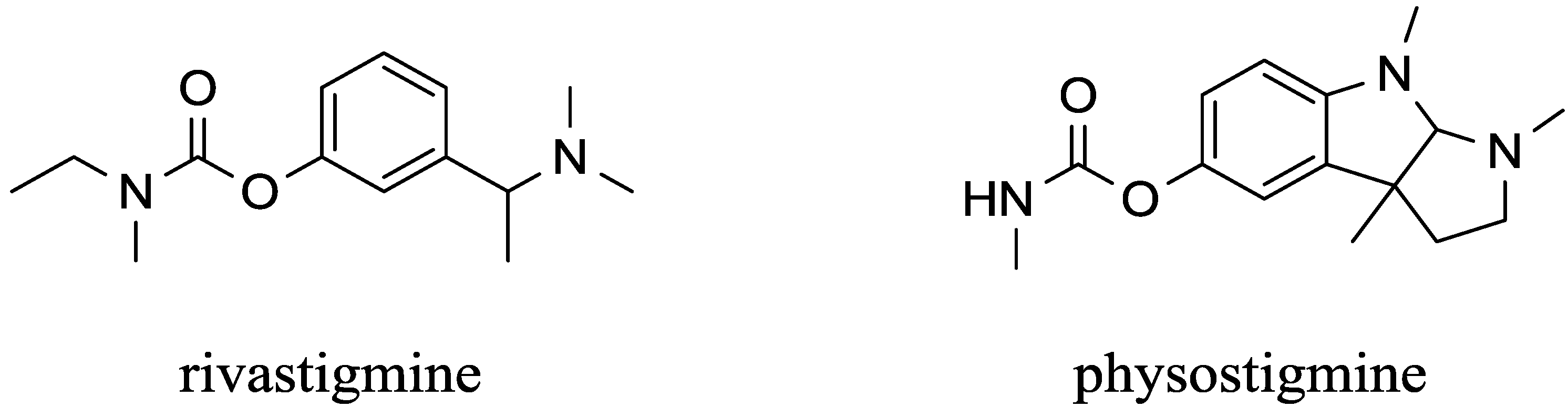
5. Peripherally Acting Carbamates-Parasympathomimetics
6. Conclusions

Acknowledgments
Conflicts of Interest
References
- Ofek, K.; Soreq, H. Cholinergic involvement and manipulation approaches in multiple system disorders. Chem. Biol. Interact. 2013, 203, 113–119. [Google Scholar] [CrossRef]
- De los Rios, C. Cholinesterase inhibitors: A patent review (2007–2011). Expert Opin. Ther. Pat. 2012, 22, 853–869. [Google Scholar] [CrossRef]
- Silman, I.; Sussman, J.L. Acetylcholinesterase: “Classical” and “non-classical” functions and pharmacology. Curr. Opin. Pharmacol. 2005, 5, 293–302. [Google Scholar] [CrossRef]
- Tayeb, H.O.; Yang, H.D.; Price, B.H.; Tarazi, F.I. Pharmacotherapies for Alzheimer’s disease: Beyond cholinesterase inhibitors. Pharmacol. Ther. 2012, 134, 8–25. [Google Scholar]
- Haines, S.R.; Thurtell, M.J. Treatment of ocular myasthenia gravis. Curr. Treat. Option Neurol. 2012, 14, 103–112. [Google Scholar]
- Marrs, T.C.; Maynard, R.L. Neurotranmission systems as targets for toxicants: A review. Cell Biol. Toxicol. 2013, 29, 381–396. [Google Scholar] [CrossRef]
- Colovic, M.B.; Krstic, D.Z.; Lazarevic-Pasti, T.D.; Bondzic, A.M.; Vasic, V.M. Acetylcholinesterase inhibitors: Pharmacology and toxicology. Curr. Neuropharmacol. 2013, 11, 315–335. [Google Scholar] [CrossRef]
- Rakowski, F.; Srinivasan, J.; Sternberg, P.W.; Karbowski, J. Synaptic polarity of the interneuron circuit controlling c. Elegans locomotion. Front. Comput. Neurosci. 2013, 7, 128. [Google Scholar]
- Kosinski, R.A.; Zaremba, M. Dynamics of the model of the caenorhabditis elegans neural network. Acta Phys. Pol. B 2007, 38, 2201–2210. [Google Scholar]
- Rand, J.B. Acetylcholine. WormBook 2007. [Google Scholar] [CrossRef]
- Pohanka, M. Alpha7 nicotinic acetylcholine receptor is a target in pharmacology and toxicology. Int. J. Mol. Sci. 2012, 13, 2219–2238. [Google Scholar]
- Bellier, J.P.; Kimura, H. Peripheral type of choline acetyltransferase: Biological and evolutionary implications for novel mechanisms in cholinergic system. J. Chem. Neuroanat. 2011, 42, 225–235. [Google Scholar] [CrossRef]
- Wessler, I.; Kirkpatrick, C.J. Acetylcholine beyond neurons: The non-neuronal cholinergic system in humans. Br. J. Pharmacol. 2008, 154, 1558–1571. [Google Scholar] [CrossRef]
- Rosas-Ballina, M.; Tracey, K.J. Cholinergic control of inflammation. J. Intern. Med. 2009, 265, 663–679. [Google Scholar]
- Bernik, T.R.; Friedman, S.G.; Ochani, M.; DiRaimo, R.; Ulloa, L.; Yang, H.; Sudan, S.; Czura, C.J.; Ivanova, S.M.; Tracey, K.J. Pharmacological stimulation of the cholinergic antiinflammatory pathway. J. Exp. Med. 2002, 195, 781–788. [Google Scholar] [CrossRef]
- Borovikova, L.V.; Ivanova, S.; Zhang, M.H.; Yang, H.; Botchkina, G.I.; Watkins, L.R.; Wang, H.C.; Abumrad, N.; Eaton, J.W.; Tracey, K.J. Vagus nerve stimulation attenuates the systemic inflammatory response to endotoxin. Nature 2000, 405, 458–462. [Google Scholar] [CrossRef]
- Ofek, K.; Krabbe, K.S.; Evron, T.; Debecco, M.; Nielsen, A.R.; Brunnsgaad, H.; Yirmiya, R.; Soreq, H.; Pedersen, B.K. Cholinergic status modulations in human volunteers under acute inflammation. J. Mol. Med. 2007, 85, 1239–1251. [Google Scholar]
- Andersson, U.; Tracey, K.J. Neural reflexes in inflammation and immunity. J. Exp. Med. 2012, 209, 1057–1068. [Google Scholar] [CrossRef]
- Pavlov, V.A.; Ochani, M.; Gallowitsch-Puerta, M.; Ochani, K.; Huston, J.M.; Czura, C.J.; Al-Abed, Y.; Tracey, K.J. Central muscarinic cholinergic regulation of the systemic inflammatory response during endotoxemia. Proc. Natl. Acad. Sci. USA 2006, 103, 5219–5223. [Google Scholar] [CrossRef]
- Pavlov, V.A.; Parrish, W.R.; Rosas-Ballina, M.; Ochani, M.; Puerta, M.; Qchani, K.; Chavan, S.; Al-Abed, Y.; Tracey, K.J. Brain acetylcholinesterase activity controls systemic cytokine levels through the cholinergic anti-inflammatory pathway. Brain Behav. Immun. 2009, 23, 41–45. [Google Scholar] [CrossRef]
- Tracey, K.J. The inflammatory reflex. Nature 2002, 420, 853–859. [Google Scholar] [CrossRef]
- Tracey, K.J. Reflex control of immunity. Nat. Rev. Immunol. 2009, 9, 418–428. [Google Scholar] [CrossRef]
- Wang, H.; Yu, M.; Ochani, M.; Amella, C.A.; Tanovic, M.; Susarla, S.; Li, J.H.; Wang, H.C.; Yang, H.; Ulloa, L.; et al. Nicotinic acetylcholine receptor alpha 7 subunit is an essential regulator of inflammation. Nature 2003, 421, 384–388. [Google Scholar] [CrossRef]
- Wang, H.; Liao, H.; Ochani, M.; Justiniani, M.; Lin, X.C.; Yang, L.H.; Al-Abed, Y.; Wang, H.C.; Metz, C.; Miller, E.J.; et al. Cholinergic agonists inhibit hmgb1 release and improve survival in experimental sepsis. Nat. Med. 2004, 10, 1216–1221. [Google Scholar] [CrossRef]
- Silva-Herdade, A.S.; Saldanha, C. Effects of acetylcholine on an animal mode of inflammation. Clin. Hemorheol. Microcirc. 2013, 53, 209–216. [Google Scholar]
- Coin, A.; Perissinotto, E.; Catanzaro, S.; Mosele, M.; de Rui, M.; Girardi, A.; Inelmen, E.M.; Toffanello, E.D.; Manzato, E.; Sergi, G. Effects of 21 months of cholinesterase inhibitors on cognitive and functional decline in demented patients. Aging Clin. Exp. Res. 2012, 24, 14–16. [Google Scholar]
- Iwasaki, T.; Yoneda, M.; Nakajima, A.; Terauchi, Y. Serum butyrylcholinesterase is strongly associated with adiposity, the serum lipid profile and insulin resistance. Intern. Med. 2007, 46, 1633–1639. [Google Scholar] [CrossRef]
- Karlsson, D.; Fallarero, A.; Brunhofer, G.; Mayer, C.; Prakash, O.; Mohan, C.G.; Vuorela, P.; Erker, T. The exploration of thienothiazines as selective butyrylcholinesterase inhibitors. Eur. J. Pharm. Sci. 2012, 47, 190–205. [Google Scholar] [CrossRef]
- Pohanka, M. Cholinesterases, a target of pharmacology and toxicology. Biomed. Pap. 2011, 155, 219–229. [Google Scholar] [CrossRef]
- Noelker, C.; Stuckenholz, V.; Reese, J.P.; Alvarez-Fischer, D.; Sankowski, R.; Rausch, T.; Oertel, W.H.; Hartmann, A.; van Patten, S.; Al-Abed, Y.; et al. Cni-1493 attenuates neuroinflammation and dopaminergic neurodegeneration in the acute mptp mouse model of Parkinson’s disease. Neurodegener. Dis. 2013, 12, 103–110. [Google Scholar] [CrossRef]
- Lee, L.; Kosuri, P.; Arancio, O. Picomolar amyloid-beta peptides enhance spontaneous astrocyte calcium transients. J. Alzheimers Dis. 2014, 38, 49–62. [Google Scholar]
- Sun, P.; Zhou, K.W.; Wang, S.; Li, P.; Chen, S.J.; Lin, G.P.; Zhao, Y.; Wang, T.H. Involvement of mapk/NF-κB signaling in the activation of the cholinergic anti-inflammatory pathway in experimental colitis by chronic vagus nerve stimulation. PLoS One 2013, 8, e69424. [Google Scholar]
- De Haan, J.J.; Hadfoune, M.; Lubbers, T.; Hodin, C.; Lenaerts, K.; Ito, A.; Verbaeys, I.; Skynner, M.J.; Cailotto, C.; van der Vliet, J.; et al. Lipid-rich enteral nutrition regulates mucosal mast cell activation via the vagal anti-inflammatory reflex. Am. J. Physiol.-Gastroint. Liver Physiol. 2013, 305, G383–G391. [Google Scholar] [CrossRef]
- Starec, M.; Sinet, M.; Kodym, P.; Rosina, J.; Fiserova, A.; Desforges, B.; Rouveix, B. The effect of drugs on the mortality of mice inoculated with friend leukaemia virus or toxoplasma gondii. Physiol. Res. 1997, 46, 107–111. [Google Scholar]
- Pohanka, M. Role of oxidative stress in infectious diseases. A review. Folia Microbiol. 2013, 58, 503–513. [Google Scholar] [CrossRef]
- Smirnov, I.; Belogurov, A.; Friboulet, A.; Masson, P.; Gabibov, A.; Renard, P.Y. Strategies for the selection of catalytic antibodies against organophosphorus nerve agents. Chem.-Biol. Interact. 2013, 203, 196–201. [Google Scholar] [CrossRef]
- Da Silva, V.B.; de Andrade, P.; Kawano, D.F.; Morais, P.A.B.; de Almeida, J.R.; Carvalho, I.; Taft, C.A.; da Silva, C. In silico design and search for acetylcholinesterase inhibitors in Alzheimer’s disease with a suitable pharmacokinetic profile and low toxicity. Future Med. Chem. 2011, 3, 947–960. [Google Scholar] [CrossRef]
- Krall, W.J.; Sramek, J.J.; Cutler, N.R. Cholinesterase inhibitors: A therapeutic strategy for alzheimer disease. Ann. Pharmacother. 1999, 33, 441–450. [Google Scholar] [CrossRef]
- Arduini, F.; Errico, I.; Amine, A.; Micheli, L.; Palleschi, G.; Moscone, D. Enzymatic spectrophotometric method for aflatoxin b detection based on acetylcholinesterase inhibition. Anal. Chem. 2007, 79, 3409–3415. [Google Scholar] [CrossRef]
- Arduini, F.; Amine, A.; Moscone, D.; Palleschi, G. Biosensors based on cholinesterase inhibition for insecticides, nerve agents and aflatoxin b-1 detection (review). Microchim. Acta 2010, 170, 193–214. [Google Scholar] [CrossRef] [Green Version]
- Brazzolotto, X.; Wandhammer, M.; Ronco, C.; Trovaslet, M.; Jean, L.; Lockridge, O.; Renard, P.Y.; Nachon, F. Human butyrylcholinesterase produced in insect cells: Huprine-based affinity purification and crystal structure. FEBS J. 2012, 279, 2905–2916. [Google Scholar] [CrossRef]
- Massoulie, J. The origin of the molecular diversity and functional floating of cholinesterases. Neurosignals 2002, 11, 130–143. [Google Scholar] [CrossRef]
- Marrs, T.C. Organophosphate poisoning. Pharmacol. Ther. 1993, 58, 51–66. [Google Scholar] [CrossRef]
- Furtado, M.D.; Rossetti, F.; Chanda, S.; Yourick, D. Exposure to nerve agents: From status epilepticus to neuroinflammation, brain damage, neurogenesis and epilepsy. Neurotoxicology 2012, 33, 1476–1490. [Google Scholar] [CrossRef]
- Dorandeu, F.; Foquin, A.; Briot, R.; Delacour, C.; Denis, J.; Alonso, A.; Froment, M.T.; Renault, F.; Lallement, G.; Masson, P. An unexpected plasma cholinesterase activity rebound after challenge with a high dose of the nerve agent vx. Toxicology 2008, 248, 151–157. [Google Scholar] [CrossRef]
- Raghu, P.; Madhusudana Reddy, T.; Reddaiah, K.; Swamy, B.E.; Sreedhar, M. Acetylcholinesterase based biosensor for monitoring of malathion and acephate in food samples: A voltammetric study. Food Chem. 2014, 142, 188–196. [Google Scholar] [CrossRef]
- Ahmed, M.; Rocha, J.B.; Mazzanti, C.M.; Morsch, A.L.; Cargnelutti, D.; Correa, M.; Loro, V.; Morsch, V.M.; Schetinger, M.R. Malathion, carbofuran and paraquat inhibit bungarus sindanus (krait) venom acetylcholinesterase and human serum butyrylcholinesterase in vitro. Ecotoxicology 2007, 16, 363–369. [Google Scholar] [CrossRef]
- Tian, Y.; Zhou, S.F.; Gao, Y.; Zhou, Y.J.; Shi, R.; Heiger-Bernays, W.; Ding, Y.; Wang, P.; Han, S.; Shen, X.M. Effects of repeated maternal oral exposure to low levels of trichlorfon on development and cytogenetic toxicity in 3-day mouse embryos. Food Chem. Toxicol. 2011, 49, 2655–2659. [Google Scholar] [CrossRef]
- Becker, R.E.; Unni, L.K.; Greig, N.H. Resurrecting clinical pharmacology as a context for alzheimer disease drug development. Curr. Alzheimer Res. 2009, 6, 79–81. [Google Scholar] [CrossRef]
- López-Arrieta, J.M.; Schneider, L. Metrifonate for Alzheimer’s disease. Cochrane Database Syst. Rev. 2006, CD003155. [Google Scholar]
- Yang, Y.X.; Niu, L.Z.; Li, S.N. Purification and studies on characteristics of cholinesterases from daphnia magna. J. Zhejiang Univ.-Sci. B 2013, 14, 325–335. [Google Scholar] [CrossRef]
- Oropesa, A.L.; Gravato, C.; Sanchez, S.; Soler, F. Characterization of plasma cholinesterase from the white stork (ciconia ciconia) and its in vitro inhibition by anticholinesterase pesticides. Ecotoxicol. Environ. Saf. 2013, 97, 131–138. [Google Scholar] [CrossRef]
- Zawadzka, A.; Lózinska, I.; Moleda, Z.; Panasiewicz, M.; Czarnocki, Z. Highly selective inhibition of butyrylcholinesterase by a novel melatonin-tacrine heterodimers. J. Pineal Res. 2013, 54, 435–441. [Google Scholar]
- Bartolucci, C.; Stojan, J.; Yu, Q.S.; Greig, N.H.; Lamba, D. Kinetics of torpedo californica acetylcholinesterase inhibition by bisnorcymserine and crystal structure of the complex with its leaving group. Biochem. J. 2012, 444, 269–277. [Google Scholar] [CrossRef]
- Jokanovic, M. Medical treatment of acute poisoning with organophosphorus and carbamate pesticides. Toxicol. Lett. 2009, 190, 107–115. [Google Scholar] [CrossRef]
- Ballatore, C.; Zhang, B.; Trojanowski, J.Q.; Lee, V.M.Y.; Smith, A.B. In situ blood-brain barrier permeability of a c-10 paclitaxel carbamate. Bioorg. Med. Chem. Lett. 2008, 18, 6119–6121. [Google Scholar] [CrossRef]
- Prades, R.; Guerrero, S.; Araya, E.; Molina, C.; Salas, E.; Zurita, E.; Selva, J.; Egea, G.; Lopez-Iglesias, C.; Teixido, M.; et al. Delivery of gold nanoparticles to the brain by conjugation with a peptide that recognizes the transferrin receptor. Biomaterials 2012, 33, 7194–7205. [Google Scholar] [CrossRef]
- Lee, N.Y.; Kang, Y.S. The inhibitory effect of rivastigmine and galantamine on choline transport in brain capillary endothelial cells. Biomol. Ther. 2010, 18, 65–70. [Google Scholar] [CrossRef]
- Di Stefano, A.; Iannitelli, A.; Laserra, S.; Sozio, P. Drug delivery strategies for Alzheimer’s disease treatment. Expert Opin. Drug Deliv. 2011, 8, 581–603. [Google Scholar] [CrossRef]
- Beilin, B.; Bessler, H.; Papismedov, L.; Weinstock, M.; Shavit, Y. Continuous physostigmine combined with morphine-based patient-controlled analgesia in the postoperative period. Acta Anaesthesiol. Scand. 2005, 49, 78–84. [Google Scholar] [CrossRef]
- Arkypova, V.N.; Dzyadevych, S.V.; Soldatkin, A.P.; El’skaya, A.V.; Martelet, C.; Jaffrezic-Renault, N. Development and optimisation of biosensors based on ph-sensitive field effect transistors and cholinesterases for sensitive detection of solanaceous glycoalkaloids. Biosens. Bioelectron. 2003, 18, 1047–1053. [Google Scholar] [CrossRef]
- Dzyadevich, S.V.; Arkhypova, V.N.; Soldatkin, A.P.; El’skaya, A.V.; Martelet, C.; Jaffrezic-Renault, N. Enzyme biosensor for tomatine detection in tomatoes. Anal. Lett. 2004, 37, 1611–1624. [Google Scholar] [CrossRef]
- Benilova, I.V.; Arkhypova, V.N.; Dzyadeviych, S.V.; Jaffrezic-Renault, N.; Martelet, C.; Soldatkin, A.P. Kinetics of human and horse sera cholinesterases inhibition with solanaceous glycoalkaloids: Study by potentiometric biosensor. Pest. Biochem. Physiol. 2006, 86, 203–210. [Google Scholar] [CrossRef]
- Ingkaninan, K.; Phengpa, P.; Yuenyongsawad, S.; Khorana, N. Acetylcholinesterase inhibitors from stephania venosa tuber. J. Pharm. Pharmacol. 2006, 58, 695–700. [Google Scholar] [CrossRef]
- Xiao, H.T.; Peng, J.; Liang, Y.; Yang, J.; Bai, X.; Hao, X.Y.; Yang, F.M.; Sun, Q.Y. Acetylcholinesterase inhibitors from corydalis yanhusuo. Nat. Prod. Res. 2011, 25, 1418–1422. [Google Scholar] [CrossRef]
- Jann, M.W.; Shirley, K.L.; Small, G.W. Clinical pharmacokinetics and pharmacodynamics of cholinesterase inhibitors. Clin. Pharmacokinet. 2002, 41, 719–739. [Google Scholar] [CrossRef]
- Pohanka, M. Acetylcholinesterase inhibitors: A patent review (2008-present). Expert Opin. Ther. Pat. 2012, 22, 871–886. [Google Scholar] [CrossRef]
- Alfirevic, A.; Mills, T.; Carr, D.; Barratt, B.J.; Jawaid, A.; Sherwood, J.; Smith, J.C.; Tugwood, J.; Hartkoorn, R.; Owen, A.; et al. Tacrine-induced liver damage: An analysis of 19 candidate genes. Pharmacogenet. Genomics 2007, 17, 1091–1100. [Google Scholar]
- Carr, D.F.; Alfirevic, A.; Tugwood, J.D.; Barratt, B.J.; Sherwood, J.; Smith, J.; Pirmohamed, M.; Park, B.K. Molecular and genetic association of interleukin-6 in tacrine-induced hepatotoxicity. Pharmacogenet. Genomics 2007, 17, 961–972. [Google Scholar] [CrossRef]
- Pathak, M.K.; Fareed, M.; Srivastava, A.K.; Pangtey, B.S.; Bihari, V.; Kuddus, M.; Kesavachandran, C. Seasonal variations in cholinesterase activity, nerve conduction velocity and lung function among sprayers exposed to mixture of pesticides. Environ. Sci. Pollut. Res. 2013, 20, 7296–7300. [Google Scholar] [CrossRef]
- Berg, R.M.G.; Ofek, K.; Qvist, T.; Tofteng, F.; Soreq, H.; Moller, K. Cholinesterase modulations in patients with acute bacterial meningitis. Scand. J. Clin. Lab. Investig. 2011, 71, 350–352. [Google Scholar] [CrossRef]
- Sonali, N.; Tripathi, M.; Sagar, R.; Velpandian, T.; Subbiah, V. Clinical effectiveness of rivastigmine monotherapy and combination therapy in Alzheimer’s patients. CNS Neurosci. Ther. 2013, 19, 91–97. [Google Scholar] [CrossRef]
- Bond, M.; Rogers, G.; Peters, J.; Anderson, R.; Hoyle, M.; Miners, A.; Moxham, T.; Davis, S.; Thokala, P.; Wailoo, A.; et al. The effectiveness and cost-effectiveness of donepezil, galantamine, rivastigmine and memantine for the treatment of Alzheimer’s disease (review of technology appraisal no. 111): A systematic review and economic model. Health Technol. Asses. 2012, 16, 1–470. [Google Scholar]
- Lilienfeld, S. Galantamine—A novel cholinergic drug with a unique dual mode of action for the treatment of patients with Alzheimer’s disease. CNS Drug Rev. 2002, 8, 159–176. [Google Scholar] [CrossRef]
- Bai, D.L.; Tang, X.C.; He, X.C. Huperzine a, a potential therapeutic agent for treatment of Alzheimer’s disease. Curr. Med. Chem. 2000, 7, 355–374. [Google Scholar] [CrossRef]
- Cheewakriengkrai, L.; Gauthier, S. A 10-year perspective on donepezil. Expert Opin. Pharmacother. 2013, 14, 331–338. [Google Scholar] [CrossRef]
- Zhang, H.C.; Liang, H.; Kuang, P.Q.; Yuan, Q.P.; Wang, Y. Simultaneously preparative purification of huperzine a and huperzine b from huperzia serrata by macroporous resin and preparative high performance liquid chromatography. J. Chromatogr. B 2012, 904, 65–72. [Google Scholar] [CrossRef]
- Zhang, J.M.; Hu, G.Y. Huperzine a, a nootropic alkaloid, inhibits n-methyl-d-aspartate-induced current in rat dissociated hippocampal neurons. Neuroscience 2001, 105, 663–669. [Google Scholar] [CrossRef]
- Zhang, Y.H.; Zhao, X.Y.; Chen, X.Q.; Wang, Y.; Yang, H.H.; Hu, G.Y. Spermidine antagonizes the inhibitory effect of huperzine a on h-3 dizocilpine (mk-801) binding in synaptic membrane of rat cerebral cortex. Neurosci. Lett. 2002, 319, 107–110. [Google Scholar] [CrossRef]
- Heinrich, M.; Teoh, H.L. Galanthamine from snowdrop—The development of a modern drug against Alzheimer’s disease from local caucasian knowledge. J. Ethnopharmacol. 2004, 92, 147–162. [Google Scholar] [CrossRef]
- Rainer, M. Galanthamine in Alzheimer’s disease-A new alternative to tacrine? CNS Drugs 1997, 7, 89–97. [Google Scholar] [CrossRef]
- Pavlov, V.A.; Ochani, M.; Parrish, W.R.; Rosas-Ballina, M.; Ochani, K.; Al-Abed, Y.; Tracey, K.J. The anti-inflammatory efficacy of galantamine is dependent on the integrity of the cholinergic anti-inflammatory pathway. Shock 2007, 27, 23. [Google Scholar]
- Takata, K.; Kitamura, Y.; Saeki, M.; Terada, M.; Kagitani, S.; Kitamura, R.; Fujikawa, Y.; Maelicke, A.; Tomimoto, H.; Taniguchi, T.; et al. Galantamine-induced amyloid-beta clearance mediated via stimulation of microglial nicotinic acetylcholine receptors. J. Biol. Chem. 2010, 285, 40180–40191. [Google Scholar] [CrossRef]
- Liu, Z.H.; Ma, Y.F.; Wu, J.S.; Gan, J.X.; Xu, S.W.; Jiang, G.Y. Effect of cholinesterase inhibitor galanthamine on circulating tumor necrosis factor alpha in rats with lipopolysaccharide-induced peritonitis. Chin. Med. J. 2010, 123, 1727–1730. [Google Scholar]
- Hwang, J.; Hwang, H.; Lee, H.W.; Suk, K. Microglia signaling as a target of donepezil. Neuropharmacology 2010, 58, 1122–1129. [Google Scholar] [CrossRef]
- Sochocka, M.; Zaczynska, E.; Leszek, J.; Siemieniec, W.; Blach-Olszewska, Z. Effect of donepezil on innate antiviral immunity of human leukocytes. J. Neurol. Sci. 2008, 273, 75–80. [Google Scholar]
- Sochocka, M.; Zaczynska, E.; Tabol, A.; Czarny, A.; Leszek, J.; Sobczynski, M. The influence of donepezil and egb 761 on the innate immunity of human leukocytes effect on the NF-κB system. Int. Immunopharmacol. 2010, 10, 1505–1513. [Google Scholar] [CrossRef]
- Tang, X.C.; Kindel, G.H.; Kozikowski, A.P.; Hanin, I. Comparison of the effects of natural and synthetic huperzine-a on rat brain cholinergic function in vitro and in vivo. J. Enthnopharmacol. 1994, 44, 147–155. [Google Scholar] [CrossRef]
- White, J.D.; Li, Y.; Kim, J.; Terinek, M. A novel synthesis of (−)-huperzine a via tandem intramolecular aza-prins cyclization-cyclobutane fragmentation. Org. Lett. 2013, 15, 882–885. [Google Scholar] [CrossRef]
- Ma, X.; Gang, D.R. In vitro production of huperzine a, a promising drug candidate for Alzheimer’s disease. Phytochemistry 2008, 69, 2022–2028. [Google Scholar]
- Leman, L.; Kitson, S.L.; Brown, R.T.; Cairns, J.; Watters, W.; McMordie, A.; Murrell, V.L.; Marfurt, J. Synthesis of isotopically labelled c-14 zt-1 (debio-9902), d(3) zt-1 and (−)-d(3) huperzinea, a new generation of acetylcholinesterase inhibitors. J. Labelledc Compd. Rad. 2011, 54, 720–730. [Google Scholar] [CrossRef]
- Hedberg, M.M.; Clos, M.V.; Ratia, M.; Gonzalez, D.; Lithner, C.U.; Camps, P.; Munoz-Torrero, D.; Badia, A.; Gimenez-Llort, L.; Nordberg, A. Effect of huprine x on beta-amyloid, synaptophysin and alpha 7 neuronal nicotinic acetylcholine receptors in the brain of 3xtg-ad and appswe transgenic mice. Neurodegener. Dis. 2010, 7, 379–388. [Google Scholar] [CrossRef]
- Wang, J.; Chen, F.; Zheng, P.; Deng, W.J.; Yuan, J.; Peng, B.; Wang, R.C.; Liu, W.J.; Zhao, H.; Wang, Y.Q.; et al. Huperzine a ameliorates experimental autoimmune encephalomyelitis via the suppression of t cell-mediated neuronal inflammation in mice. Exp. Neurol. 2012, 236, 79–87. [Google Scholar]
- Wang, Z.F.; Tang, X.C. Huperzine a protects c6 rat glioma cells against oxygen-glucose deprivation-induced injury. FEBS Lett. 2007, 581, 596–602. [Google Scholar] [CrossRef]
- Wang, Z.F.; Wang, J.; Zhang, H.Y.; Tang, X.C. Huperzine a exhibits anti-inflammatory and neuroprotective effects in a rat model of transient focal cerebral ischemia. J. Neurochem. 2008, 106, 1594–1603. [Google Scholar] [CrossRef]
- Darreh-Shori, T.; Soininen, H. Effects of cholinesterase inhibitors on the activities and protein levels of cholinesterases in the cerebrospinal fluid of patients with Alzheimer’s disease: A review of recent clinical studies. Curr. Alzheimer Res. 2010, 7, 67–73. [Google Scholar]
- Darreh-Shori, T.; Hellstrom-Lindahl, E.; Flores-Flores, C.; Guan, Z.Z.; Soreq, H.; Nordberg, A. Long-lasting acetylcholinesterase splice variations in anticholinesterase-treated Alzheimer’s disease patients. J. Neurochem. 2004, 88, 1102–1113. [Google Scholar] [CrossRef]
- Bastiat, G.; Plourde, F.; Motulsky, A.; Furtos, A.; Dumont, Y.; Quirion, R.; Fuhrmann, G.; Leroux, J.C. Tyrosine-based rivastigmine-loaded organogels in the treatment of Alzheimer’s disease. Biomaterials 2010, 31, 6031–6038. [Google Scholar] [CrossRef] [Green Version]
- Field, R.H.; Gossen, A.; Cunningham, C. Prior pathology in the basal forebrain cholinergic system predisposes to inflammation-induced working memory deficits: Reconciling inflammatory and cholinergic hypotheses of delirium. J. Neurosci. 2012, 32, 6288–6294. [Google Scholar] [CrossRef]
- Nizri, E.; Irony-Tur-Sinai, M.; Faranesh, N.; Lavon, I.; Lavi, E.; Weinstock, M.; Brenner, T. Suppression of neuroinflammation and immunomodulation by the acetylcholinesterase inhibitor rivastigmine. J. Neuroimmunol. 2008, 203, 12–22. [Google Scholar] [CrossRef]
- Chiou, W.H.; Kao, C.L.; Tsai, J.C.; Chang, Y.M. Domino rh-catalyzed hydroformylation-double cyclization of o-amino cinnamyl derivatives: Applications to the formal total syntheses of physostigmine and physovenine. Chem. Commun. 2013, 49, 8232–8234. [Google Scholar]
- Realini, T. A history of glaucoma pharmacology. Optom. Vis. Sci. 2011, 88, 36–38. [Google Scholar] [CrossRef]
- Greig, N.H.; Becker, R.E.; Yu, Q.; Holloway, H.W.; Tweedie, D.; Luo, W.; Utsuki, T.; Ingram, D.K.; Maccecchini, M.L.; Rogers, J.T.; et al. From natural products to alzheimer experimental therapeutics, eserine based drugs as symptomatic and disease altering agents. Prog. Nutr. 2010, 12, 58–63. [Google Scholar]
- Russo, P.; Frustaci, A.; del Bufalo, A.; Fini, M.; Cesario, A. Multitarget drugs of plants origin acting on Alzheimer’s disease. Curr. Med. Chem. 2013, 20, 1686–1693. [Google Scholar] [CrossRef]
- Bitzinger, D.I.; Zausig, Y.A.; Paech, C.; Gruber, M.; Busse, H.; Sinner, B.; Graf, B.M.; Trabold, B. Modulation of immune functions in polymorphonuclear neutrophils induced by physostigmine, but not neostigmine, independent of cholinergic neurons. Immunobiology 2013, 218, 1049–1054. [Google Scholar] [CrossRef]
- Peter, C.; Schmidt, K.; Hofer, S.; Stephan, M.; Martin, E.; Weigand, M.A.; Walther, A. Effects of physostigmine on microcirculatory alterations during experimental endotoxemia. Shock 2010, 33, 405–411. [Google Scholar] [CrossRef]
- Kutsuna, S.; Tsuruta, R.; Fujita, M.; Todani, M.; Yagi, T.; Ogino, Y.; Igarashi, M.; Takahashi, K.; Izumi, T.; Kasaoka, S.; et al. Cholinergic agonist physostigmine suppresses excessivesuperoxide anion radical generation in blood, oxidative stress, early inflammation, and endothelial injury in rats with forebrain ischemia/reperfusion. Brain Res. 2010, 1313, 242–249. [Google Scholar] [CrossRef]
- Benatar, M.; Kaminski, H. Medical and surgical treatment for ocular myasthenia. Cochrane Database Syst. Rev. 2012, 12, CD005081. [Google Scholar]
- Chambers, D.; Paulden, M.; Paton, F.; Heirs, M.; Duffy, S.; Craig, D.; Hunter, J.; Wilson, J.; Sculpher, M.; Woolacott, N. Sugammadex for the reversal of muscle relaxation in general anaesthesia: A systematic review and economic assessment. Health Technol. Assess. 2010, 14, 1–211. [Google Scholar]
- Cossins, J.; Belaya, K.; Zoltowska, K.; Koneczny, I.; Maxwell, S.; Jacobson, L.; Leite, M.I.; Waters, P.; Vincent, A.; Beeson, D. The search for new antigenic targets in myasthenia gravis. Ann. N. Y. Acad. Sci. 2012, 1275, 123–128. [Google Scholar] [CrossRef]
- Vyskocil, F.; Malomouzh, A.I.; Nikolsky, E.E. Non-quantal acetylcholine release at the neuromuscular junction. Physiol. Res. 2009, 58, 763–784. [Google Scholar]
- Walker, S.M.; Yaksh, T.L. Neuraxial analgesia in neonates and infants: A review of clinical and preclinical strategies for the development of safety and efficacy data. Anesth. Analg. 2012, 115, 638–662. [Google Scholar]
- Fisher, D.M. Clinical pharmacology of neuromuscular blocking agents. Am. J. Health Syst. Pharm. 1999, 56, S4–S9. [Google Scholar]
- Yang, L.P.; Keam, S.J. Sugammadex: A review of its use in anaesthetic practice. Drugs 2009, 69, 919–942. [Google Scholar] [CrossRef]
- Friedman, A.; Kaufer, D.; Shemer, J.; Hendler, I.; Soreq, H.; TurKaspa, I. Pyridostigmine brain penetration under stress enhances neuronal excitability and induces early immediate transcriptional response. Nat. Med. 1996, 2, 1382–1385. [Google Scholar]
- Kalb, A.; von Haefen, C.; Sifringer, M.; Tegethoff, A.; Paeschke, N.; Kostova, M.; Feldheiser, A.; Spies, C.D. Acetylcholinesterase inhibitors reduce neuroinflammation and -degeneration in the cortex and hippocampus of a surgery stress rat model. PLoS One 2013, 8, e62679. [Google Scholar]
- Kalb, A.; von Haefen, C.; Sifringer, M.; Tegethoff, A.; Paeschke, N.; Kostova, M.; Feldheiser, A.; Spies, C.D. Physostigmine and neostigmine reduce the increased expression of il-1beta in the hippocampus and cortex after surgery combined with lps-treatment. Eur. J. Neurol. 2012, 19, 756. [Google Scholar]
- Pohanka, M.; Pavlis, O. Neostigmine modulates tularemia progression in balb/c mice. Afr. J. Pharm. Pharmacol. 2012, 6, 1317–1322. [Google Scholar]
- Sun, L.; Zhang, G.F.; Zhang, X.; Liu, Q.; Liu, J.G.; Su, D.F.; Liu, C. Combined administration of anisodamine and neostigmine produces anti-shock effects: Involvement of alpha 7 nicotinic acetylcholine receptors. Acta Pharm. Sin. 2012, 33, 761–766. [Google Scholar] [CrossRef]
- Kox, M.; Pompe, J.C.; Peters, E.; Vaneker, M.; van der Laak, J.W.; van der Hoeven, J.G.; Scheffer, G.J.; Hoedemaekers, C.W.; Pickkers, P. Alpha 7 nicotinic acetylcholine receptor agonist gts-21 attenuates ventilator-induced tumour necrosis factor-alpha production and lung injury. Br. J. Anaesth. 2011, 107, 559–566. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Pohanka, M. Inhibitors of Acetylcholinesterase and Butyrylcholinesterase Meet Immunity. Int. J. Mol. Sci. 2014, 15, 9809-9825. https://doi.org/10.3390/ijms15069809
Pohanka M. Inhibitors of Acetylcholinesterase and Butyrylcholinesterase Meet Immunity. International Journal of Molecular Sciences. 2014; 15(6):9809-9825. https://doi.org/10.3390/ijms15069809
Chicago/Turabian StylePohanka, Miroslav. 2014. "Inhibitors of Acetylcholinesterase and Butyrylcholinesterase Meet Immunity" International Journal of Molecular Sciences 15, no. 6: 9809-9825. https://doi.org/10.3390/ijms15069809





