Poly(Lactic Acid) Blends with Poly(Trimethylene Carbonate) as Biodegradable Medical Adhesive Material
Abstract
:1. Introduction
2. Results and Discussion
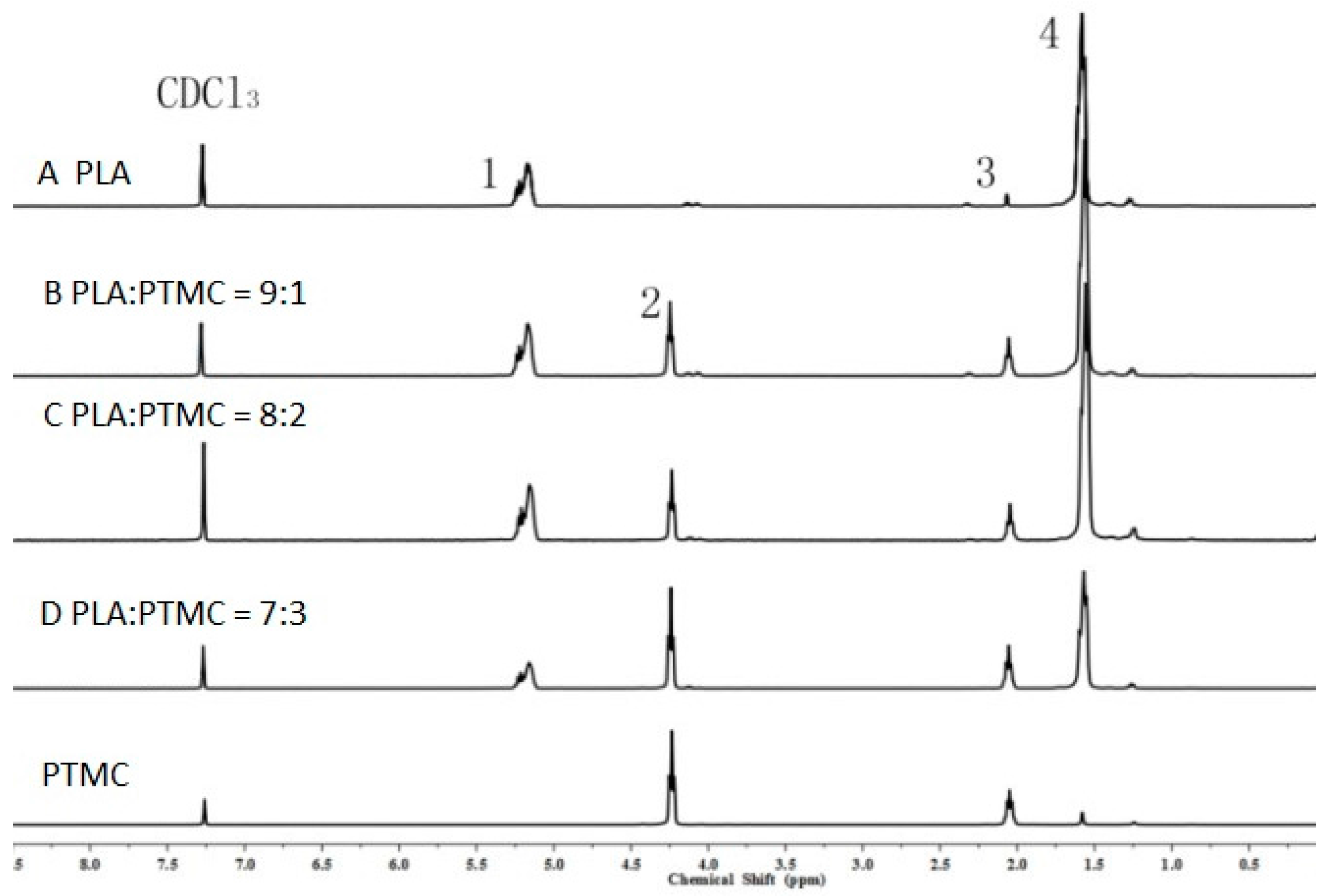
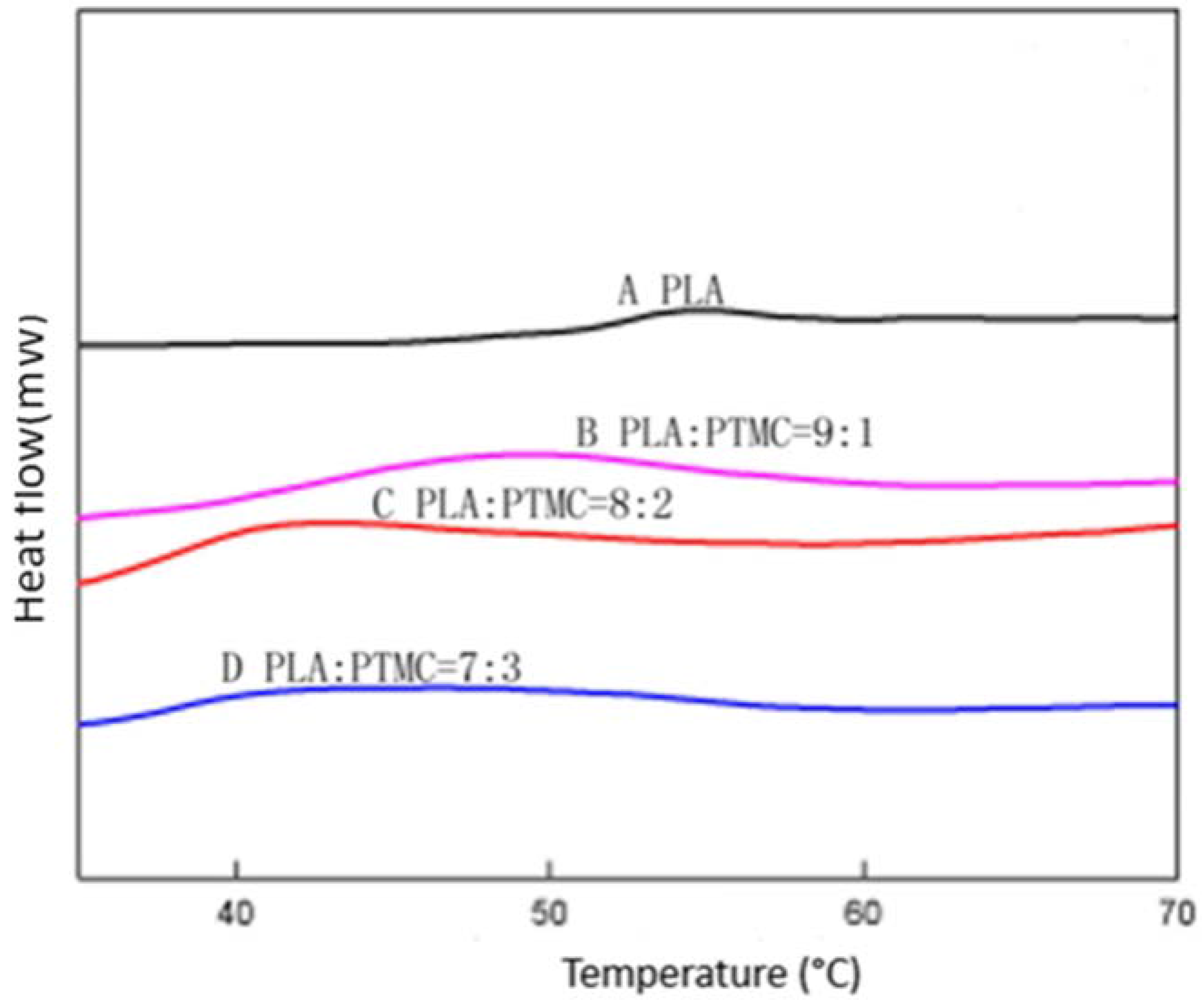
2.1. Characterization of Medical Adhesive Films
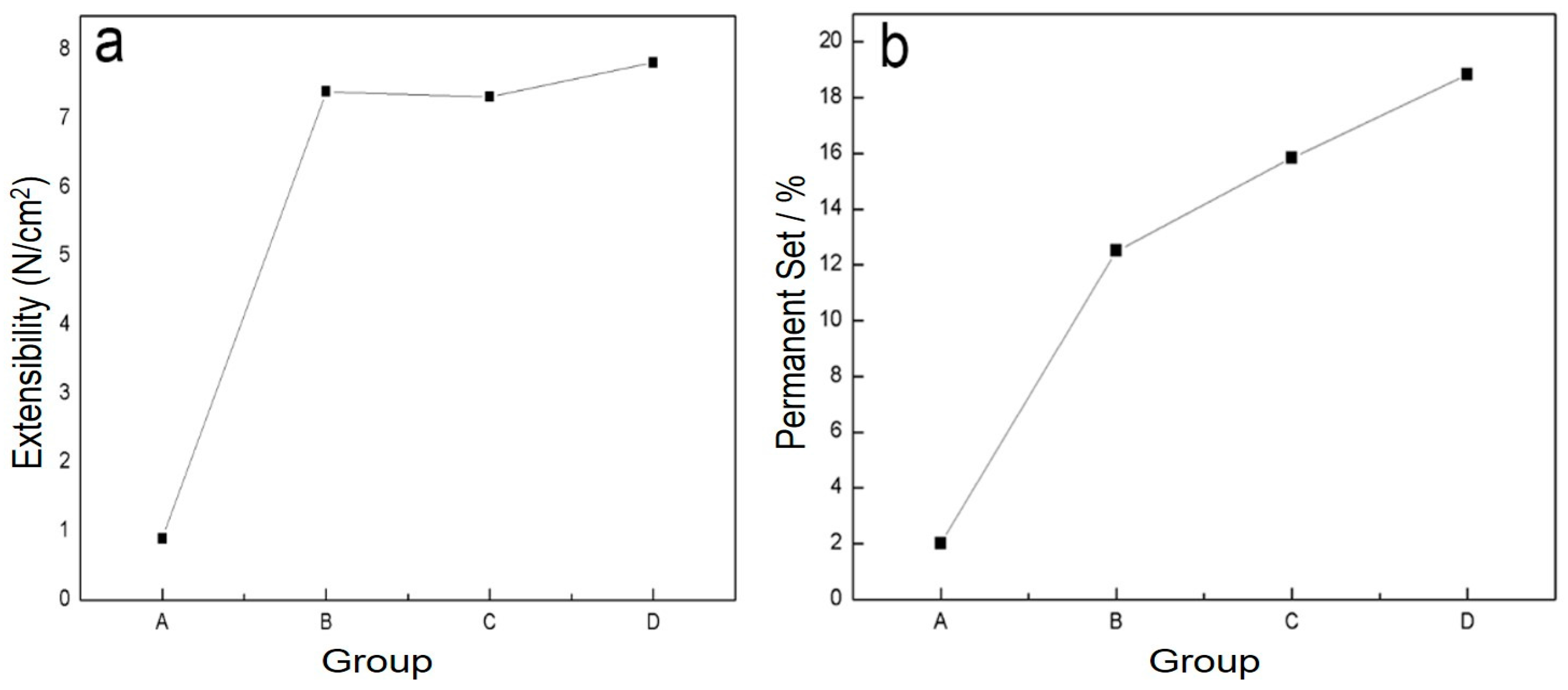
2.2. Evaluation of Physical Properties
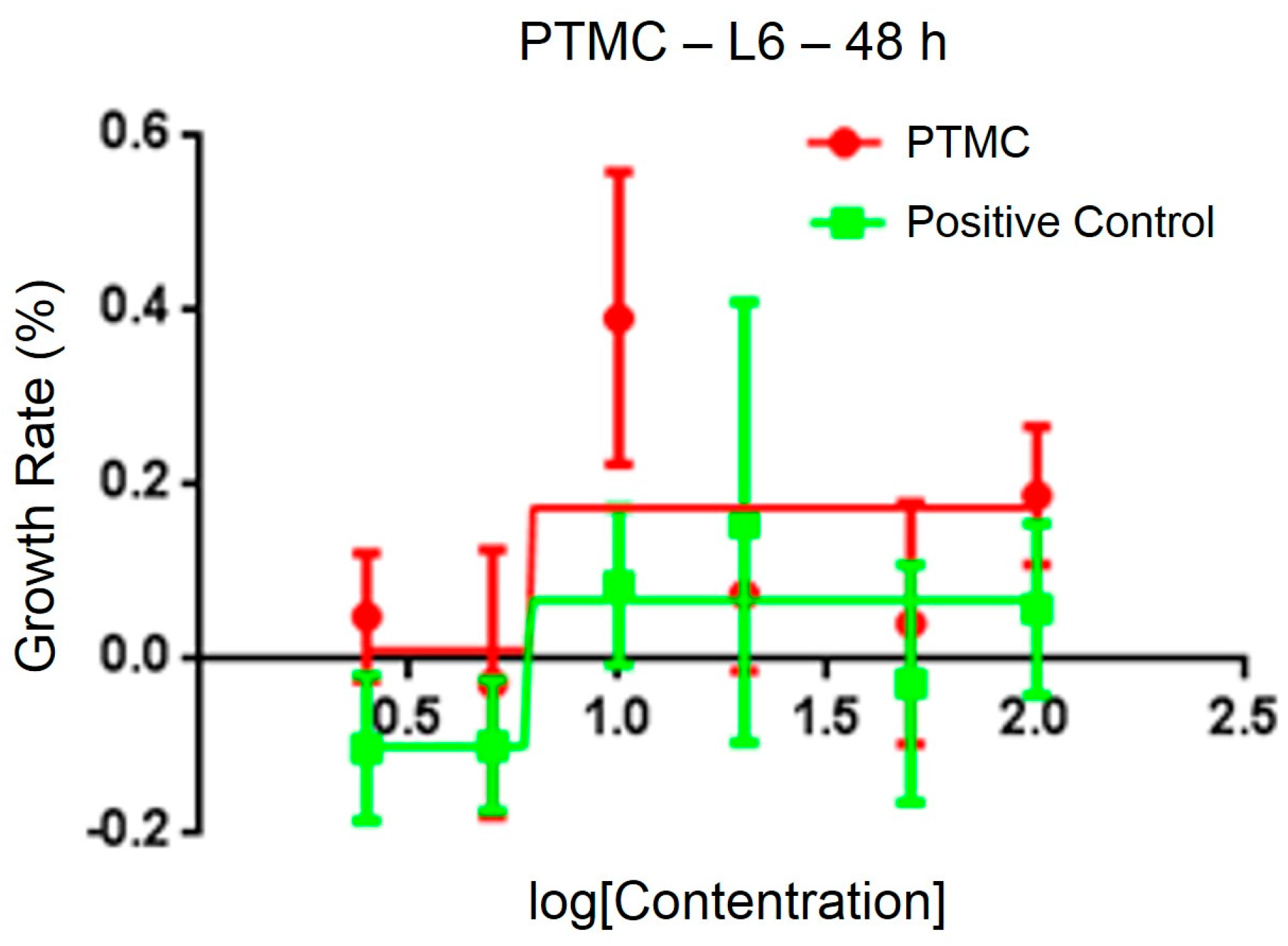
2.3. Biological Performance
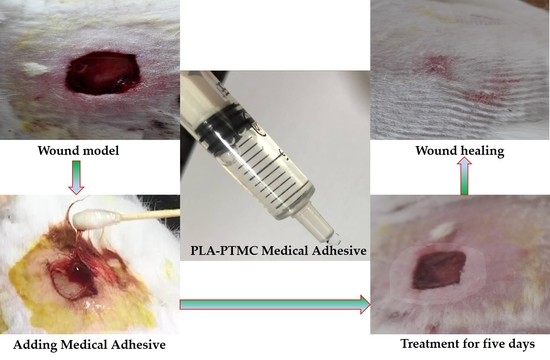
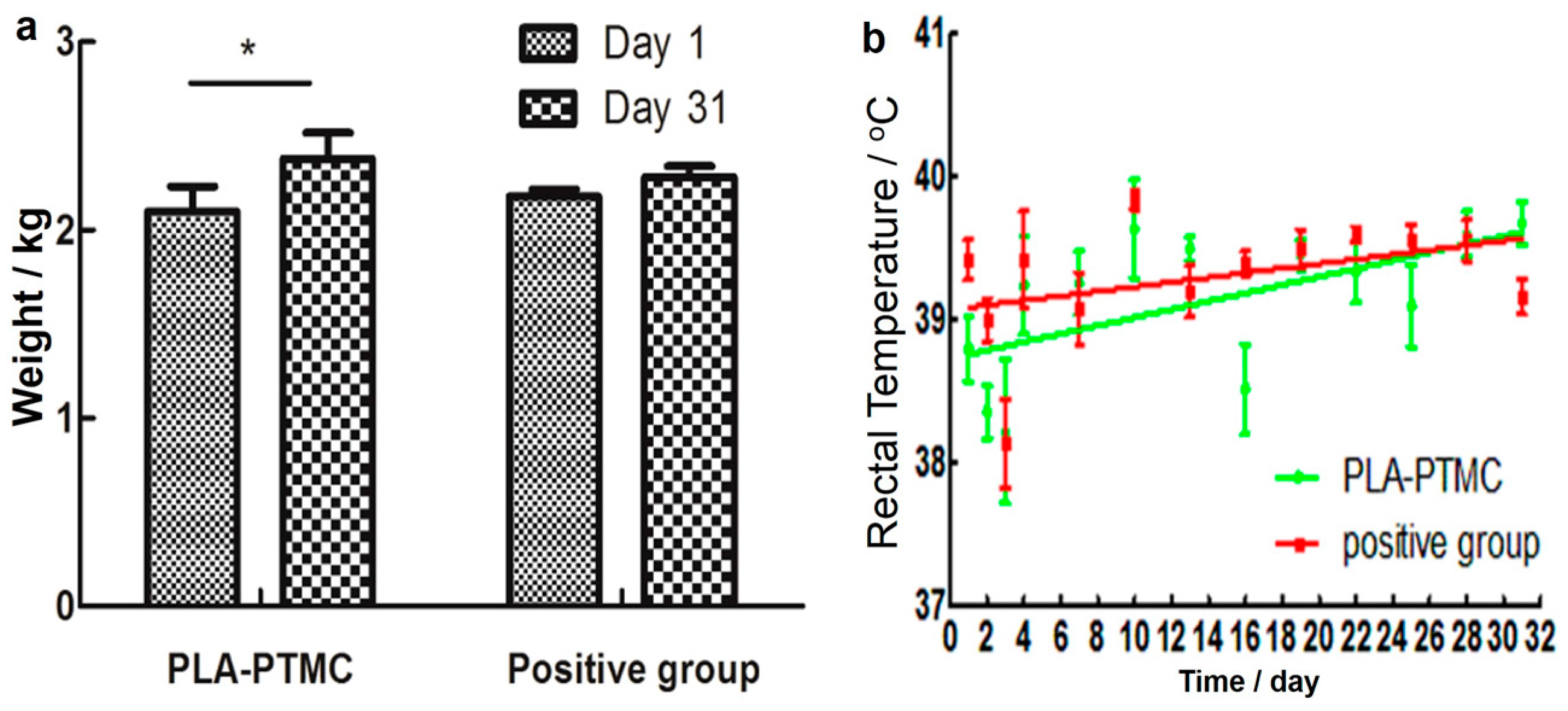
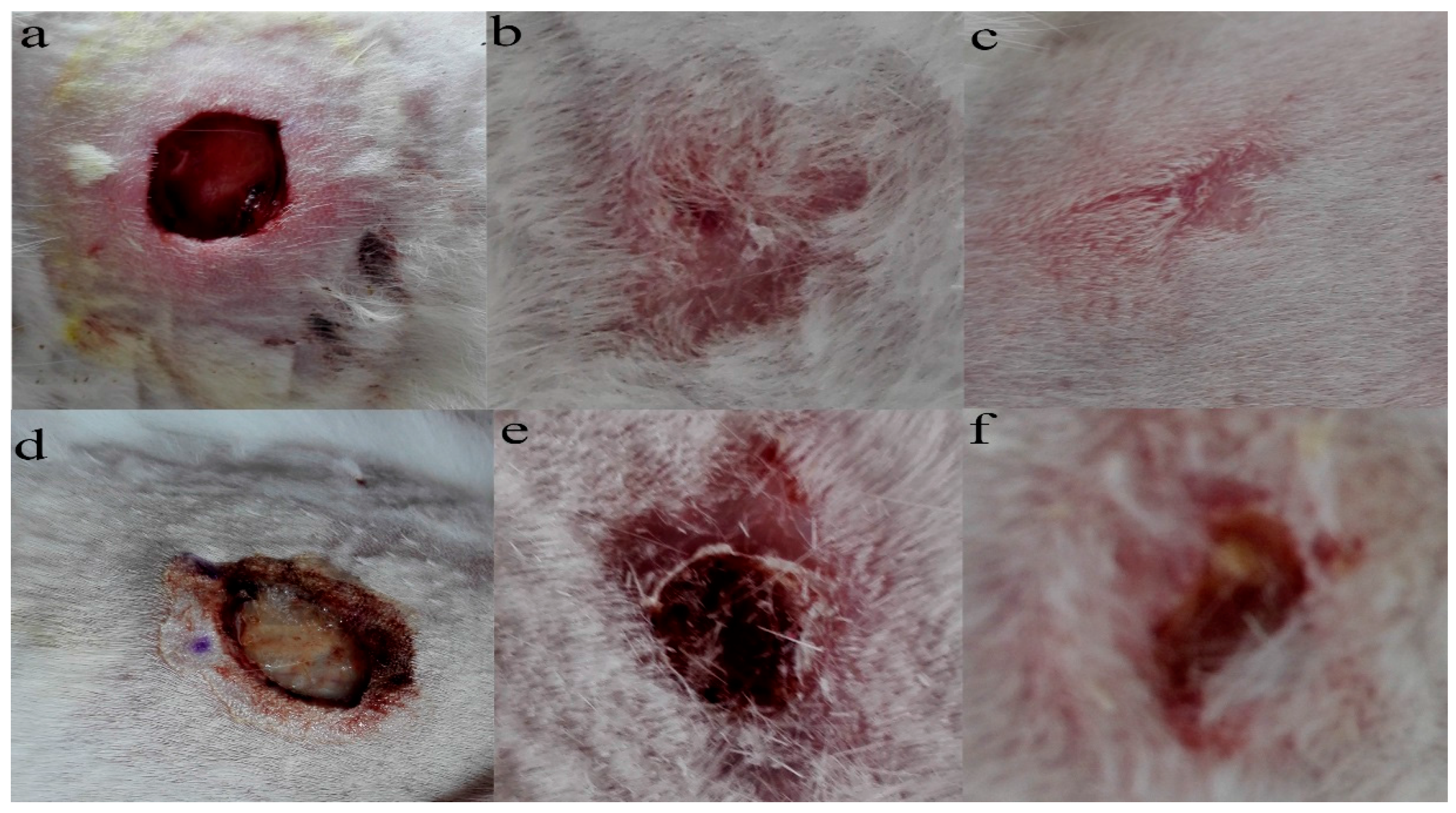
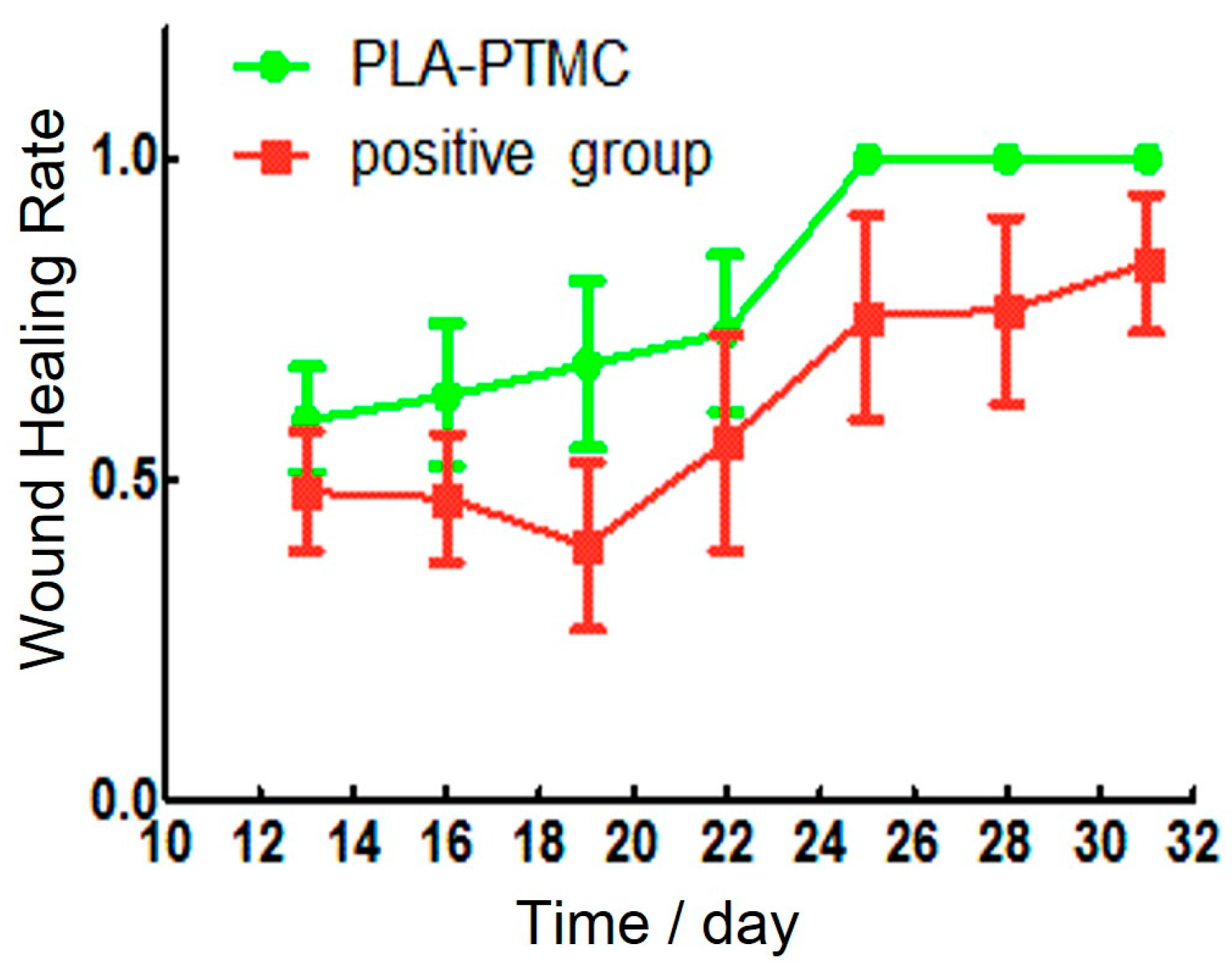
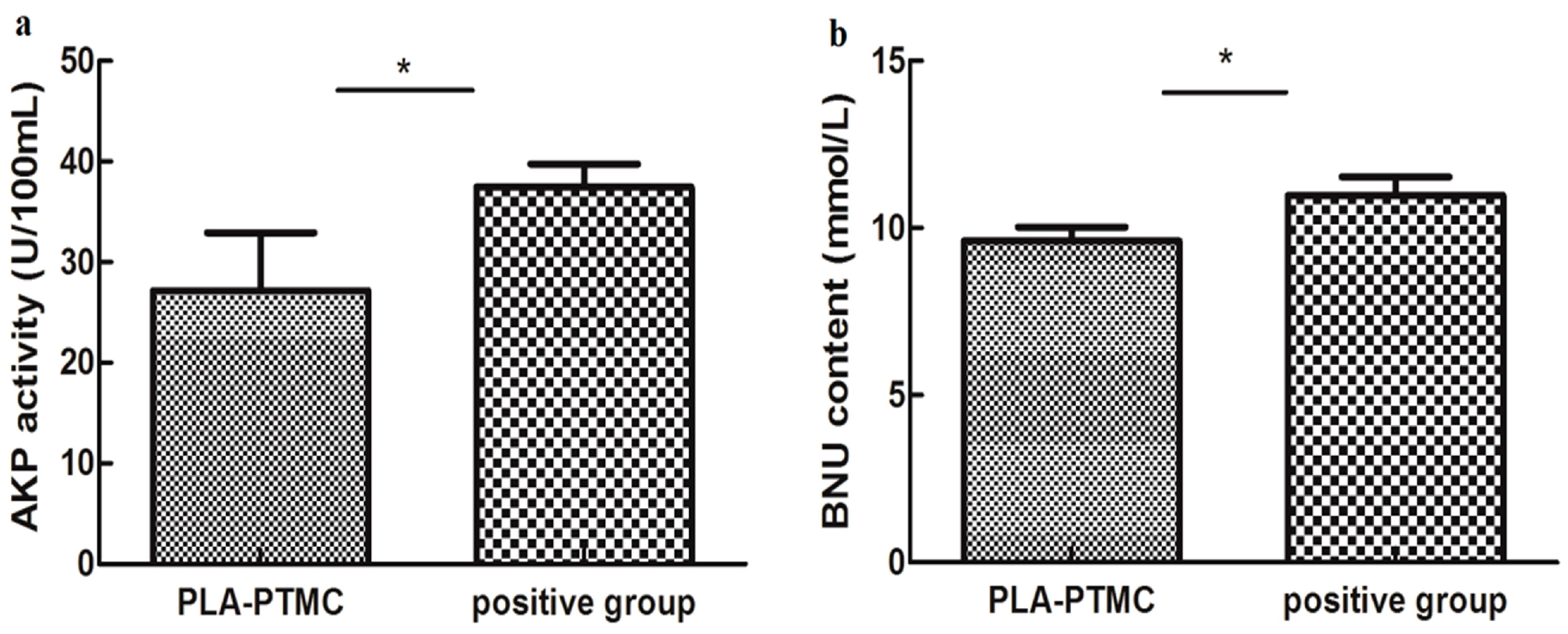
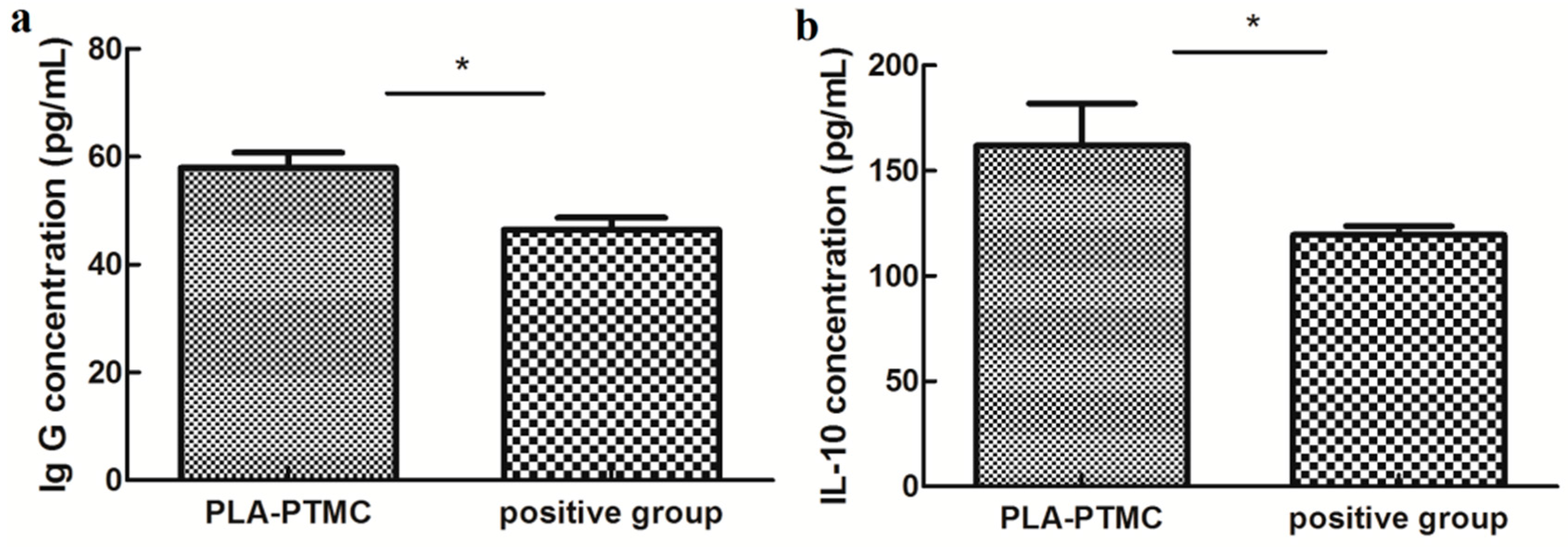
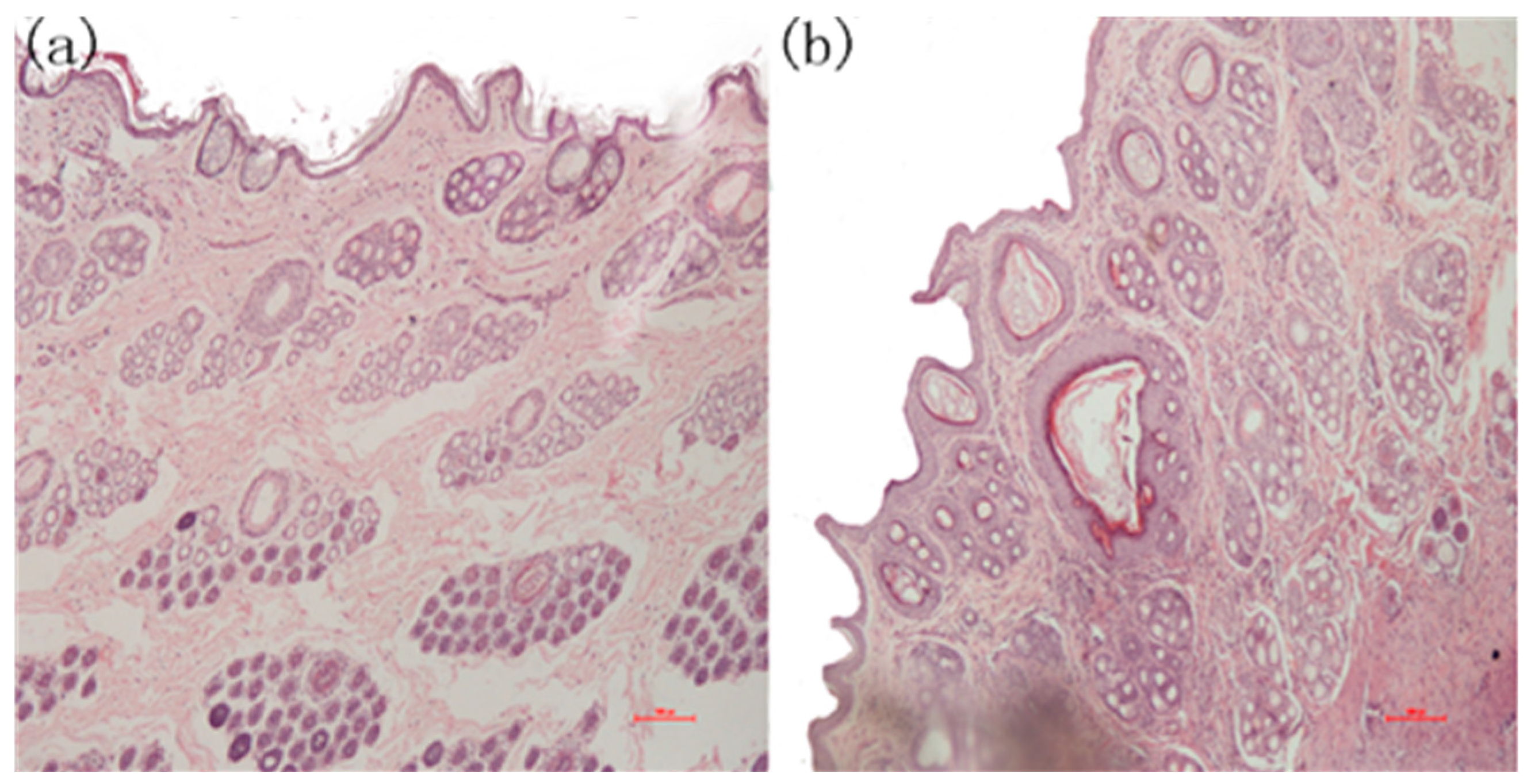
2.4. The Result of Animal Experiments
3. Materials and Methods
3.1. Synthesis of PLA-PTMC Medical Adhesive
3.2. Preparation of Films
3.3. Water Vapor Transmission Rate
3.4. Comfortability Degree of PLA-PTMC Medical Adhesives
3.5. Contact Angle
3.6. Gel Permeation Chromatography
3.7. 1H Nuclear Magnetic Resonance (1HNMR)
3.8. Differential Scanning Calorimetry
3.9. Scanning Electron Microscopy
3.10. Animal Experiments
3.10.1. Cytotoxicity
3.10.2. Test of Sterility
3.10.3. Skin Irritability
3.10.4. Creation of the Wound Model
Animals and Tissue Preparation
Establishment of the Wound Model
Weight and Temperature Transform
Hepatic and Renal Function
ELISA Assay
Skin Pathology Analyses
3.11. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Langer, R.; Tirrell, D.A. Designing materials for biology and medicine. Nature 2004, 428, 487. [Google Scholar] [CrossRef] [PubMed]
- Vacanti, J.P.; Langer, R. Tissue engineering: The design and fabrication of living replacement devices for surgical reconstruction and transplantation. Lancet 1999, 354, S32–S34. [Google Scholar] [CrossRef]
- Somorjai, G.A.; Frei, H.; Park, J.Y. Advancing the frontiers in nanocatalysis, biointerfaces, and renewable energy conversion by innovations of surface techniques. J. Am. Chem. Soc. 2009, 131, 16589–16605. [Google Scholar] [CrossRef] [PubMed]
- Place, E.S.; Evans, N.D.; Stevens, M.M. Complexity in biomaterials for tissue engineering. Nat. Mater. 2009, 8, 457. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gan, H.; Zhang, M.; Sun, T. Modulating cell behaviors on chiral polymer brush films with different hydrophobic side groups. Langmuir 2012, 28, 2791–2798. [Google Scholar] [CrossRef] [PubMed]
- Nel, A.E.; Mädler, L.; Velegol, D.; Xia, T.; Hoek, E.M.; Somasundaran, P.; Klaessig, F.; Castranova, V.; Thompson, M. Understanding biophysicochemical interactions at the nano-bio interface. Nat. Mater. 2009, 8, 543. [Google Scholar] [CrossRef] [PubMed]
- Peppas, N.A.; Langer, R. New challenges in biomaterials. Science 1994, 263, 1715–1719. [Google Scholar] [CrossRef] [PubMed]
- Bouten, P.J.; Zonjee, M.; Bender, J.; Yauw, S.T.; van Goor, H.; van Hest, J.C.; Hoogenboom, R. The chemistry of tissue adhesive materials. Prog. Polym. Sci. 2014, 39, 1375–1405. [Google Scholar] [CrossRef]
- Lloyd, J.D.; Marque, M.J.; Kacprowicz, R.F. Closure techniques. Emerg. Med. Clin. N. Am. 2007, 25, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.; Coelho, J.; Bordado, J.; Cidade, M.; Gil, M. Surgical adhesives: Systematic review of the main types and development forecast. Prog. Polym. Sci. 2012, 37, 1031–1050. [Google Scholar] [CrossRef]
- Tajirian, A.L.; Goldberg, D.J. A review of sutures and other skin closure materials. J. Cosmet. Laser Ther. 2010, 12, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Holm, V.K.; Ndoni, S.; Risbo, J. The stability of poly(lactic acid) packaging films as influenced by humidity and temperature. J. Food Sci. 2006, 71, E40–E44. [Google Scholar] [CrossRef]
- Cava, D.; Gavara, R.; Lagaron, J.; Voelkel, A. Surface characterization of poly(lactic acid) and polycaprolactone by inverse gas chromatography. J. Chromatogr. A 2007, 1148, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, D.J. Bioprocessing–No Longer a Field of Dreams. In Macromolecular Symposia; Wiley Online Library: New York, NY, USA, 2003; pp. 271–282. [Google Scholar]
- Drumright, R.E.; Gruber, P.R.; Henton, D.E. Polylactic acid technology. Adv. Mater. 2000, 12, 1841–1846. [Google Scholar] [CrossRef]
- Dorgan, J.R.; Lehermeier, H.J.; Palade, L.I.; Cicero, J. Polylactides: Properties and prospects of an environmentally benign plastic from renewable resources. In Macromolecular Symposia; Wiley Online Library: New York, NY, USA, 2001; pp. 55–66. [Google Scholar]
- Barrows, T. Degradable implant materials: A review of synthetic absorbable polymers and their applications. Clin. Mater. 1986, 1, 233–257. [Google Scholar] [CrossRef]
- Seyednejad, H.; Gawlitta, D.; Kuiper, R.V.; de Bruin, A.; van Nostrum, C.F.; Vermonden, T.; Dhert, W.J.; Hennink, W.E. In vivo biocompatibility and biodegradation of 3D-printed porous scaffolds based on a hydroxyl-functionalized poly(ε-caprolactone). Biomaterials 2012, 33, 4309–4318. [Google Scholar] [CrossRef] [PubMed]
- Mainil-Varlet, P.; Curtis, R.; Gogolewski, S. Effect of in vivo and in vitro degradation on molecular and mechanical properties of various low-molecular-weight polylactides. J. Biomed. Mater. Res. A 1997, 36, 360–380. [Google Scholar] [CrossRef]
- Tsuji, H.; Sumida, K. Poly(l-lactide): V. Effects of storage in swelling solvents on physical properties and structure of poly(l-lactide). J. Appl. Polym. Sci. 2001, 79, 1582–1589. [Google Scholar] [CrossRef]
- Bleach, N.; Tanner, K.; Kellomäki, M.; Törmälä, P. Effect of filler type on the mechanical properties of self-reinforced polylactide–calcium phosphate composites. J. Mater. Sci. 2001, 12, 911–915. [Google Scholar]
- Eling, B.; Gogolewski, S.; Pennings, A. Biodegradable materials of poly(l-lactic acid): 1. Melt-spun and solution-spun fibres. Polymer 1982, 23, 1587–1593. [Google Scholar] [CrossRef]
- Athanasiou, K.A.; Niederauer, G.G.; Agrawal, C.M. Sterilization, toxicity, biocompatibility and clinical applications of polylactic acid/polyglycolic acid copolymers. Biomaterials 1996, 17, 93–102. [Google Scholar] [CrossRef]
- Janorkar, A.V.; Metters, A.T.; Hirt, D.E. Modification of poly(lactic acid) films: Enhanced wettability from surface-confined photografting and increased degradation rate due to an artifact of the photografting process. Macromolecules 2004, 37, 9151–9159. [Google Scholar] [CrossRef]
- Vink, E.T.; Rabago, K.R.; Glassner, D.A.; Gruber, P.R. Applications of life cycle assessment to natureworks™ polylactide (PLA) production. Polym. Degrad. Stab. 2003, 80, 403–419. [Google Scholar] [CrossRef]
- Pezzin, A.; Ekenstein, V.; Alberda, G.; Zavaglia, C.; Ten Brinke, G.; Duek, E. Poly(para-dioxanone) and poly(l-lactic acid) blends: Thermal, mechanical, and morphological properties. J. Appl. Polym. Sci. 2003, 88, 2744–2755. [Google Scholar] [CrossRef]
- Ma, X.; Yu, J.; Wang, N. Compatibility characterization of poly(lactic acid)/poly(propylene carbonate) blends. J. Polym. Sci. B 2006, 44, 94–101. [Google Scholar] [CrossRef]
- Shibata, M.; Inoue, Y.; Miyoshi, M. Mechanical properties, morphology, and crystallization behavior of blends of poly(l-lactide) with poly(butylene succinate-co-l-lactate) and poly(butylene succinate). Polymer 2006, 47, 3557–3564. [Google Scholar] [CrossRef]
- Chen, G.-X.; Kim, H.-S.; Kim, E.-S.; Yoon, J.-S. Compatibilization-like effect of reactive organoclay on the poly(l-lactide)/poly(butylene succinate) blends. Polymer 2005, 46, 11829–11836. [Google Scholar] [CrossRef]
- Pego, A.; Van Luyn, M.; Brouwer, L.; Van Wachem, P.; Poot, A.A.; Grijpma, D.W.; Feijen, J. In vivo behavior of poly(1,3-trimethylene carbonate) and copolymers of 1,3-trimethylene carbonate with d,l-lactide or ϵ-caprolactone: Degradation and tissue response. J. Biomed. Mater. Res. A 2003, 67, 1044–1054. [Google Scholar] [CrossRef] [PubMed]
- Albertsson, A.C.; Eklund, M. Influence of molecular structure on the degradation mechanism of degradable polymers: In vitro degradation of poly(trimethylene carbonate), poly(trimethylene carbonate-co-caprolactone), and poly(adipic anhydride). J. Appl. Polym. Sci. 1995, 57, 87–103. [Google Scholar] [CrossRef]
- Nederberg, F.; Bowden, T.; Hilborn, J. Induced surface migration of biodegradable phosphoryl choline functional poly(trimethylene carbonate). Polym. Adv. Technol. 2005, 16, 108–112. [Google Scholar] [CrossRef]
- Márquez, Y.; Franco, L.; Puiggalí, J. Thermal degradation studies of poly(trimethylene carbonate) blends with either polylactide or polycaprolactone. Thermochim. Acta 2012, 550, 65–75. [Google Scholar] [CrossRef]
- Rocha, D.N.; Brites, P.; Fonseca, C.; Pêgo, A.P. Poly(trimethylene carbonate-co-ε-caprolactone) promotes axonal growth. PLoS ONE 2014, 9, e88593. [Google Scholar] [CrossRef] [PubMed]
- Rhim, J.-W. Effect of pla lamination on performance characteristics of agar/κ-carrageenan/clay bio-nanocomposite film. Food Res. Int. 2013, 51, 714–722. [Google Scholar] [CrossRef]
- Li, H.; Chang, J.; Qin, Y.; Wu, Y.; Yuan, M.; Zhang, Y. Poly(lactide-co-trimethylene carbonate) and polylactide/polytrimethylene carbonate blown films. Int. J. Mol. Sci. 2014, 15, 2608–2621. [Google Scholar] [CrossRef] [PubMed]
- Martin, O.; Averous, L. Poly(lactic acid): Plasticization and properties of biodegradable multiphase systems. Polymer 2001, 42, 6209–6219. [Google Scholar] [CrossRef]
- Nampoothiri, K.M.; Nair, N.R.; John, R.P. An overview of the recent developments in polylactide (PLA) research. Bioresour. Technol. 2010, 101, 8493–8501. [Google Scholar] [CrossRef] [PubMed]
- Södergård, A.; Stolt, M. Properties of lactic acid based polymers and their correlation with composition. Prog. Polym. Sci. 2002, 27, 1123–1163. [Google Scholar] [CrossRef]
- Taylor, M.; Daniels, A.; Andriano, K.; Heller, J. Six bioabsorbable polymers: In vitro acute toxicity of accumulated degradation products. J. Appl. Biomater. 1994, 5, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Yuan, M.; Li, L.; Guo, S.; Yuan, M.; Li, W.; Xue, J. Use of polylactic acid/polytrimethylene carbonate blends membrane to prevent postoperative adhesions. J. Biomed. Mater. Res. B 2006, 79, 312–319. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Dong, J.H.; Qiu, K.Y. Synthesis and characterization of ABA-type block copolymer of poly(trimethylene carbonate) with poly(ethylene glycol): Bioerodible copolymer. J. Polym. Sci. A 1998, 36, 695–702. [Google Scholar] [CrossRef]
- Żenkiewicz, M.; Richert, J.; Różański, A. Effect of blow moulding ratio on barrier properties of polylactide nanocomposite films. Polym. Test. 2010, 29, 251–257. [Google Scholar] [CrossRef]
- Bockenstedt, P.; Greenberg, J.; Handin, R. Structural basis of von willebrand factor binding to platelet glycoprotein Ib and collagen. Effects of disulfide reduction and limited proteolysis of polymeric von willebrand factor. J. Clin. Investig. 1986, 77, 743. [Google Scholar] [CrossRef] [PubMed]
- Duangjai, A.; Luo, K.; Zhou, Y.; Yang, J.; Kopeček, J. Combination cytotoxicity of backbone degradable hpma copolymer gemcitabine and platinum conjugates toward human ovarian carcinoma cells. Eur. J. Pharm. Biopharm. 2014, 87, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Blake, M.S.; Johnston, K.H.; Russell-Jones, G.J.; Gotschlich, E.C. A rapid, sensitive method for detection of alkaline phosphatase-conjugated anti-antibody on western blots. Anal. Biochem. 1984, 136, 175–179. [Google Scholar] [CrossRef]
- Xiong, L.-H.; He, X.; Xia, J.; Ma, H.; Yang, F.; Zhang, Q.; Huang, D.; Chen, L.; Wu, C.; Zhang, X. Highly sensitive naked-eye assay for enterovirus 71 detection based on catalytic nanoparticle aggregation and immunomagnetic amplification. ACS Appl. Mater. Interfaces 2017, 9, 14691–14699. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Wang, X.; Tang, J. Evaluation of acute kidney injury using contrast ultrasonography in a rabbit model of crush syndrome. Ultrasound Med. Biol. 2017, 43, 494–499. [Google Scholar] [CrossRef] [PubMed]
- Goding, J.W. Use of staphylococcal protein a as an immunological reagent. J. Immunol. Methods 1978, 20, 241–253. [Google Scholar] [CrossRef]
- Habib, S.; Ali, R. Acquired antigenicity of DNA after modification with peroxynitrite. Int. J. Biol. Macromol. 2005, 35, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.G.; Homer, R.J.; Cohn, L.; Link, H.; Jung, S.; Craft, J.E.; Graham, B.S.; Johnson, T.R.; Elias, J.A. Transgenic overexpression of interleukin (IL)-10 in the lung causes mucus metaplasia, tissue inflammation, and airway remodeling via IL-13-dependent and-independent pathways. J. Biol. Chem. 2002, 277, 35466–35474. [Google Scholar] [CrossRef] [PubMed]
- Klompmaker, J.; Jansen, H.; Veth, R.; de Groot, J.; Nijenhuis, A.; Pennings, A. Porous polymer implant for repair of meniscal lesions: A preliminary study in dogs. Biomaterials 1991, 12, 810–816. [Google Scholar] [CrossRef]
- Majola, A.; Vainionpää, S.; Vihtonen, K.; Mero, M.; Vasenius, J.; Törmälä, P.; Rokkanen, P. Absorption, biocompatibility, and fixation properties of polylactic acid in bone tissue: An experimental study in rats. Clin. Orthop. Relat. Res. 1991, 268, 260–269. [Google Scholar]
- Von Schroeder, H.P.; Kwan, M.; Amiel, D.; Coutts, R.D. The use of polylactic acid matrix and periosteal grafts for the reconstruction of rabbit knee articular defects. J. Biomed. Mater. Res. A 1991, 25, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Vásconez, M.B.; Flores, S.K.; Campos, C.A.; Alvarado, J.; Gerschenson, L.N. Antimicrobial activity and physical properties of chitosan–tapioca starch based edible films and coatings. Food Res. Int. 2009, 42, 762–769. [Google Scholar] [CrossRef]
- Nagelschmidt, M.; Becker, D.; Bonninghoff, N.; Engelhardt, G.H. Effect of fibronectin therapy and fibronectin deficiency on wound healing: A study in rats. J. Trauma Acute Care Surg. 1987, 27, 1267–1271. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Verpoorte, R.; Suresh, B. Evaluation of in-vivo wound healing activity of hypericum patulum (family: Hypericaceae) leaf extract on different wound model in rats. J. Ethnopharmacol. 2000, 70, 315–321. [Google Scholar] [CrossRef]


| Groups | PLA/PTMC a | Solvent | Temperature (°C)/Time (h) | Mn b | PDI b |
|---|---|---|---|---|---|
| A | 10:0 | ethyl acetate | 25/6 | 98,972 | 2.052 |
| B | 9:1 | ethyl acetate | 25/6 | 93,193 | 1.957 |
| C | 8:2 | ethyl acetate | 25/6 | 89,888 | 1.871 |
| D | 7:3 | ethyl acetate | 25/6 | 77,253 | 1.846 |
| Group | Contact Angle (Degree) |
|---|---|
| PLA | 72.45 |
| PLA-PTMC 9:1 | 74.96 |
| PLA-PTMC 8:2 | 80.75 |
| PLA-PTMC 7:3 | 87.38 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, S.; Li, H.; Yuan, M.; Yuan, M.; Chen, H. Poly(Lactic Acid) Blends with Poly(Trimethylene Carbonate) as Biodegradable Medical Adhesive Material. Int. J. Mol. Sci. 2017, 18, 2041. https://doi.org/10.3390/ijms18102041
Zhang S, Li H, Yuan M, Yuan M, Chen H. Poly(Lactic Acid) Blends with Poly(Trimethylene Carbonate) as Biodegradable Medical Adhesive Material. International Journal of Molecular Sciences. 2017; 18(10):2041. https://doi.org/10.3390/ijms18102041
Chicago/Turabian StyleZhang, Shuang, Hongli Li, Mingwei Yuan, Minglong Yuan, and Haiyun Chen. 2017. "Poly(Lactic Acid) Blends with Poly(Trimethylene Carbonate) as Biodegradable Medical Adhesive Material" International Journal of Molecular Sciences 18, no. 10: 2041. https://doi.org/10.3390/ijms18102041