Anti-Inflammatory Effect of Titrated Extract of Centella asiatica in Phthalic Anhydride-Induced Allergic Dermatitis Animal Model
Abstract
:1. Introduction
2. Results
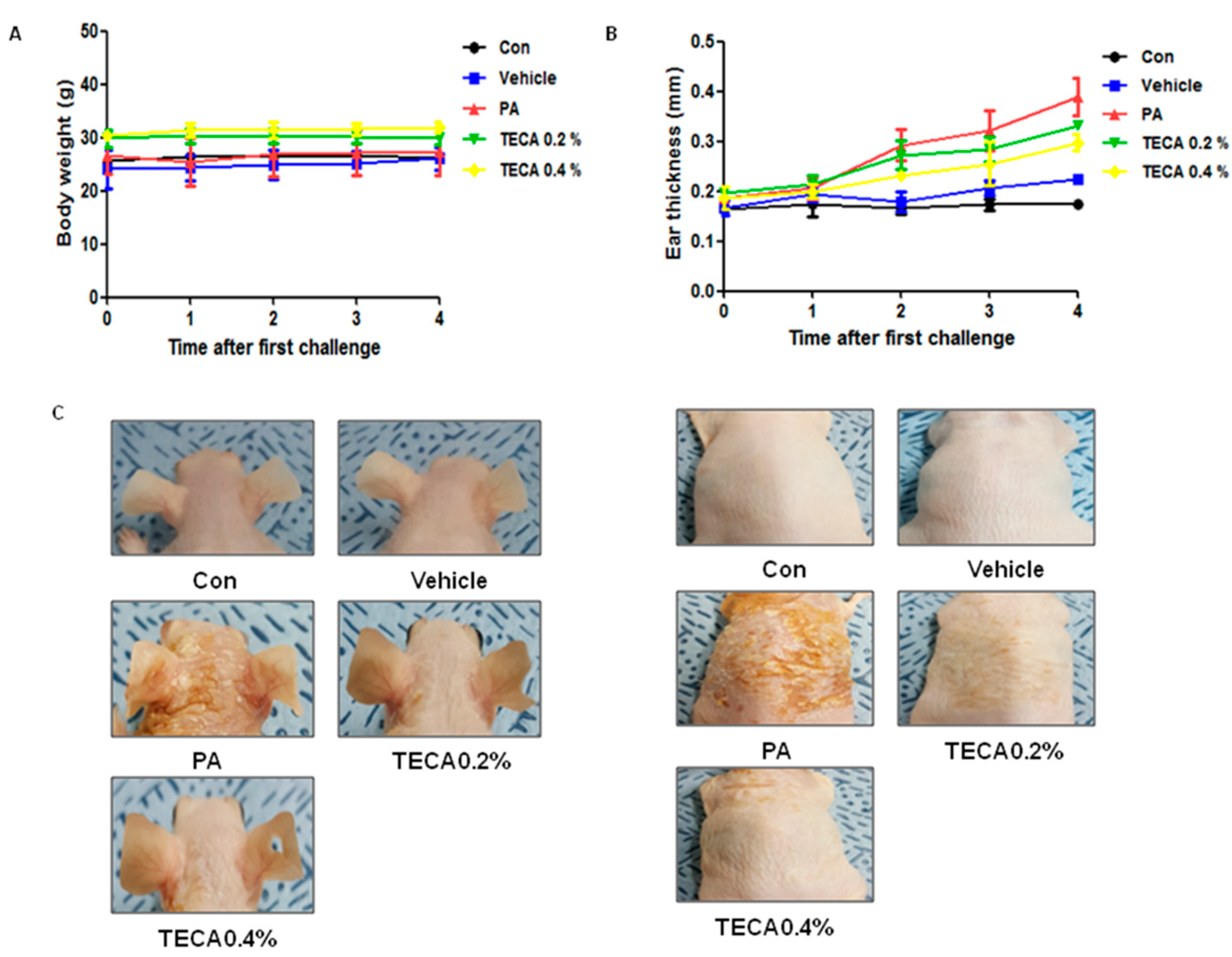
2.1. Effects of TECA Treatment on Ear Thickness and Morphology
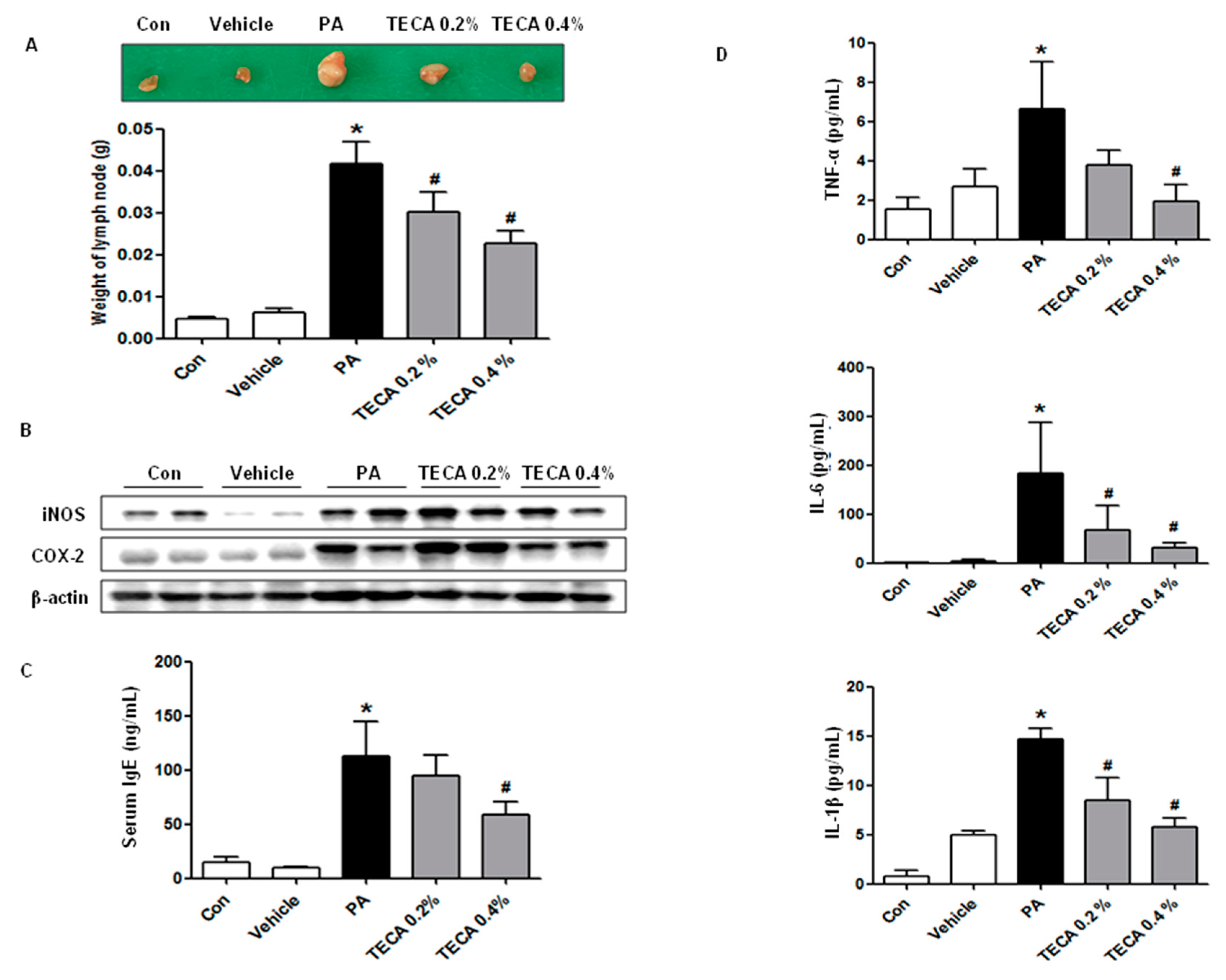
2.2. Effect of TECA Treatment on Lymph Node Weight and IgE Concentration as Well as on Expression of iNOS and COX-2
2.3. Effect of TECA Treatment on the Release of Inflammatory Cytokines
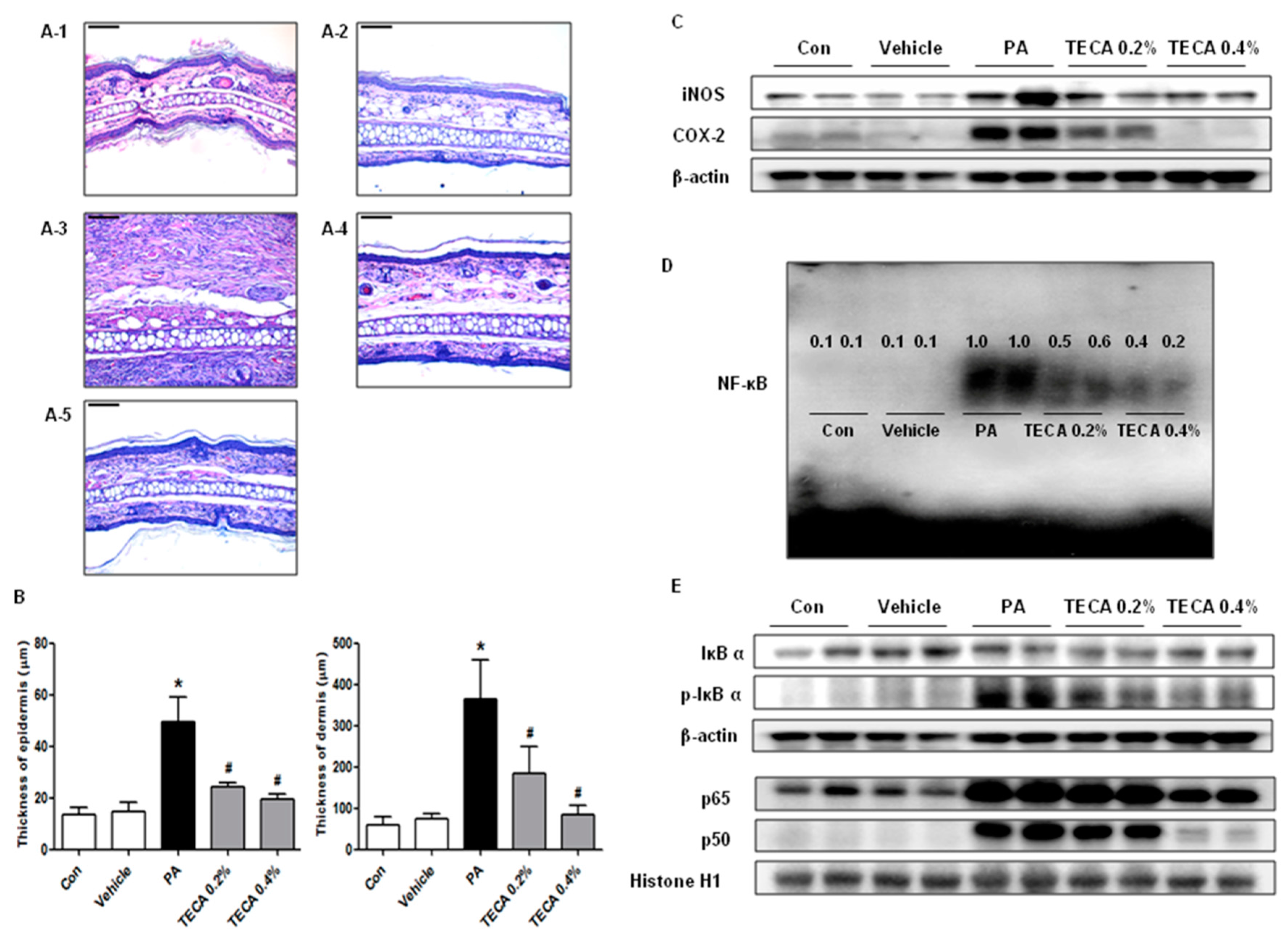
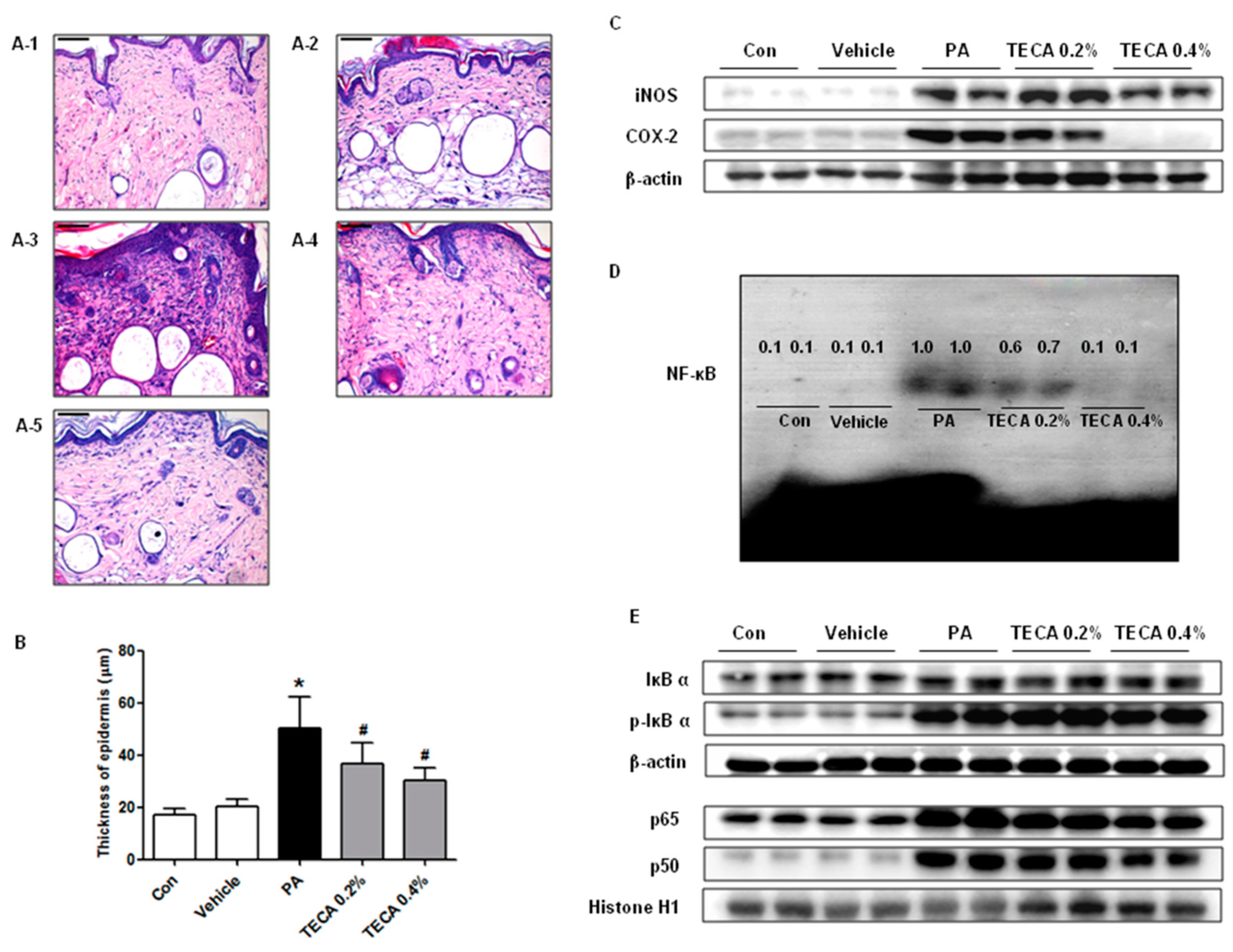
2.4. Effect of TECA Treatment on Inflammatory Responses in Ear and Back
2.5. Effect of TECA on NF-κB DNA Binding Activity in PA-Induced AD Mice
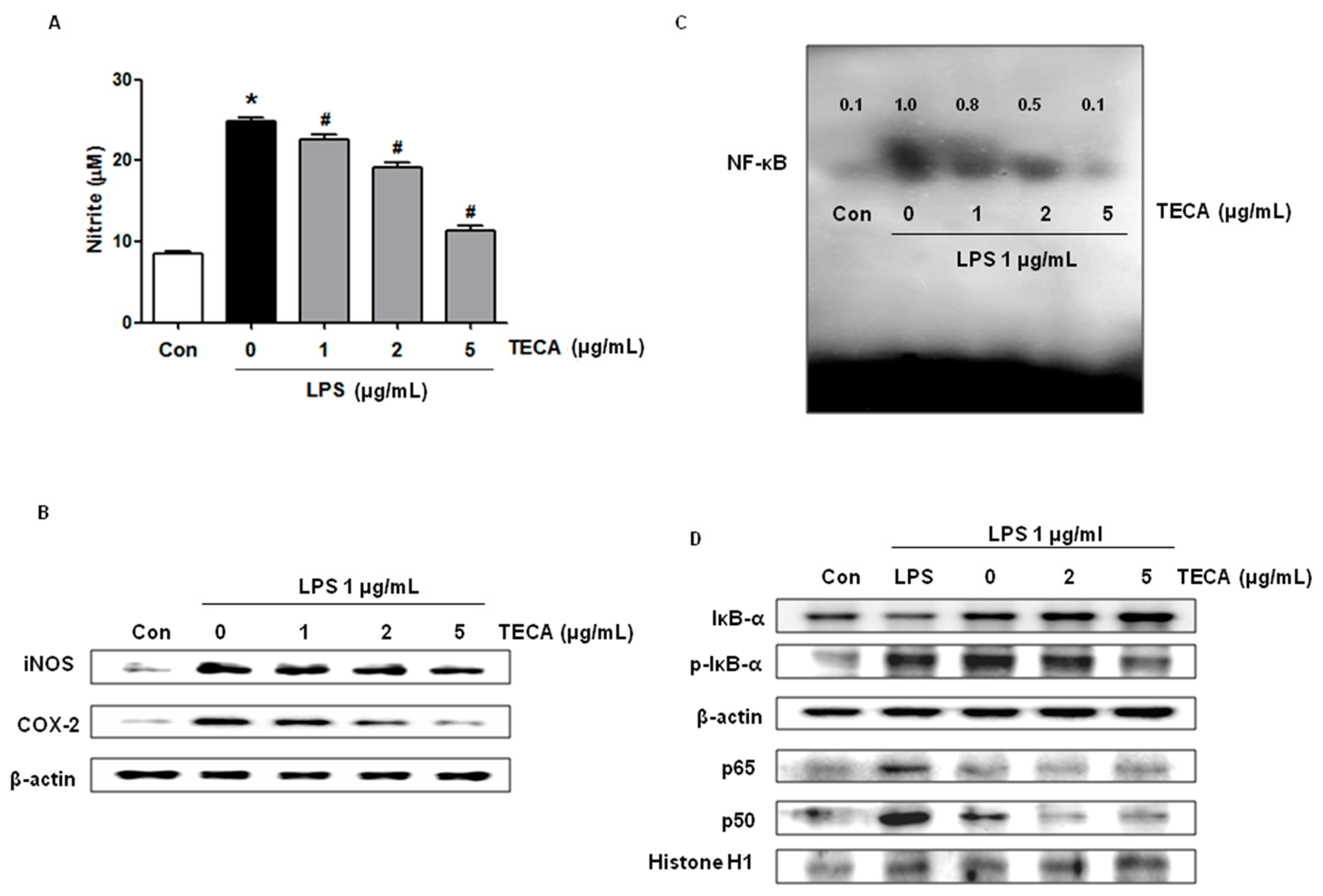
2.6. Effect of TECA on LPS-Induced NO Production, and iNOS and COX-2 Expression in RAW264.7 Cells
2.7. Effect of TECA on NF-κB DNA Binding Activity in RAW 264.7 Cells
3. Discussion
4. Materials and Methods
4.1. Ethical Approval
4.2. Preparation and Extraction of Centella asiatica
4.3. Animal Treatment
4.4. Measurement of Ear Thickness, and Body and Lymph Node Weight
4.5. Histological Techniques
4.6. Mesurement of Serum IgE Concentration
4.7. Cytokine Assay
4.8. Western Blot Analysis
4.9. Gel Electromobility Shift Assay (EMSA)
4.10. Cell Culture
4.11. Nitrite Quantification Assay
4.12. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sehra, S.; Krishnamurthy, P.; Koh, B.; Zhou, H.M.; Seymour, L.; Akhtar, N.; Travers, J.B.; Turner, M.J.; Kaplan, M.H. Increased Th2 activity and diminished skin barrier function cooperate in allergic skin inflammation. Eur. J. Immunol. 2016, 46, 2609–2613. [Google Scholar] [CrossRef] [PubMed]
- Eichenfield, L.F.M.; Friedlander, S.F.M.; Simpson, E.L.M.M.; Irvine, A.D.M. Assessing the New and Emerging Treatments for Atopic Dermatitis. Semin. Cutan. Med. Surg. 2016, 35 (Suppl. S5), S92–S96. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.K.; Kim, S.H. Inhibitory effect of galangin on atopic dermatitis-like skin lesions. Food Chem. Toxicol. 2014, 68, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Kuramoto, T.; Yokoe, M.; Tanaka, D.; Yuri, A.; Nishitani, A.; Higuchi, Y.; Yoshimi, K.; Tanaka, M.; Kuwamura, M.; Hiai, H.; et al. Atopic dermatitis-like skin lesions with IgE hyperproduction and pruritus in KFRS4/Kyo rats. J. Dermatol. Sci. 2015, 80, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Kwak, M.H.; Kim, J.E.; Hwang, I.S.; Lee, Y.J.; An, B.S.; Hong, J.T.; Lee, S.H.; Hwang, D.Y. Quantitative evaluation of therapeutic effect of Liriope platyphylla on phthalic anhydride-induced atopic dermatitis in IL-4/Luc/CNS-1 Tg mice. J. Ethnopharmacol. 2013, 148, 880–889. [Google Scholar] [CrossRef] [PubMed]
- Sung, J.E.; Lee, H.A.; Kim, J.E.; Go, J.; Seo, E.J.; Yun, W.B.; Kim, D.S.; Son, H.J.; Lee, C.Y.; Lee, H.S.; et al. Therapeutic effect of ethyl acetate extract from Asparagus cochinchinensis on phthalic anhydride-induced skin inflammation. Lab. Anim. Res. 2016, 32, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Jang, Y.J.; Yim, J.H.; Lee, H.K.; Pyo, S. Ramalin Isolated from Ramalina Terebrata Attenuates Atopic Dermatitis-like Skin Lesions in Balb/c Mice and Cutaneous Immune Responses in Keratinocytes and Mast Cells. Phytother. Res. 2016, 30, 1978–1987. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.Y.; Wang, A.X.; Xu, L.; Shen, N.; Zhu, J.; Tu, C.X. Characteristics of peripheral blood CD4+CD25+ regulatory T cells and related cytokines in severe atopic dermatitis. Eur. J. Dermatol. 2016, 26, 240–246. [Google Scholar] [PubMed]
- Klewicka, E.; Cukrowska, B.; Libudzisz, Z.; Slizewska, K.; Motyl, I. Changes in gut microbiota in children with atopic dermatitis administered the bacteria Lactobacillus casei DN-114001. Pol. J. Microbiol. 2011, 60, 329–333. [Google Scholar] [PubMed]
- Harada, D.; Takada, C.; Tsukumo, Y.; Takaba, K.; Manabe, H. Analyses of a mouse model of the dermatitis caused by 2,4,6-trinitro-1-chlorobenzene (TNCB)-repeated application. J. Dermatol. Sci. 2005, 37, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Homey, B.; Steinhoff, M.; Ruzicka, T.; Leung, D.Y. Cytokines and chemokines orchestrate atopic skin inflammation. J. Allergy Clin. Immunol. 2006, 118, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Wullaert, A.; Bonnet, M.C.; Pasparakis, M. NF-kappaB in the regulation of epithelial homeostasis and inflammation. Cell Res. 2011, 21, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J.; Karin, M. Nuclear factor-κB: A pivotal transcription factor in chronic inflammatory diseases. N. Engl. J. Med. 1997, 336, 1066–1071. [Google Scholar] [CrossRef]
- Nakamura, H.; Aoki, M.; Tamai, K.; Oishi, M.; Ogihara, T.; Kaneda, Y.; Morishita, R. Prevention and regression of atopic dermatitis by ointment containing NF-κB decoy oligodeoxynucleotides in NC/Nga atopic mouse model. Gene Ther. 2002, 9, 1221–1229. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Konno, M.; Muto, S.; Kambe, N.; Morii, E.; Nakahata, T.; Itai, A.; Matsuda, H. A novel NF-κB inhibitor, IMD-0354, suppresses neoplastic proliferation of human mast cells with constitutively activated C-kit receptors. Blood 2005, 105, 2324–2331. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.Y. Atopic dermatitis: New insights and opportunities for therapeutic intervention. J. Allergy Clin. Immunol. 2000, 105, 860–876. [Google Scholar] [CrossRef] [PubMed]
- Simpson, E.L. Atopic dermatitis: A review of topical treatment options. Curr. Med. Res. Opin. 2010, 26, 633–640. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, F.; Ramdane, N.; Delebarre-Sauvage, C.; Modiano, P.; Duhamel, A.; Lasek, A. Monitoring of topical corticosteroid phobia in a population of parents with children with atopic dermatitis using the TOPICOP(R) scale: Prevalence, risk factors and the impact of therapeutic patient education. J. Eur. Acad. Dermatol. Venereol. 2016, 31, e172–e174. [Google Scholar] [CrossRef] [PubMed]
- Bylka, W.; Znajdek-Awizen, P.; Studzinska-Sroka, E.; Danczak-Pazdrowska, A.; Brzezinska, M. Centella asiatica in dermatology: An overview. Phytother. Res. 2014, 28, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Incandela, L.; Cesarone, M.R.; DeSanctis, M.T.; Belcaro, G.; Dugall, M.; Acerbi, G. Treatment of diabetic microangiopathy and edema with HR (Paroven, Venoruton; O-(β-hydroxyethyl)-rutosides): A prospective, placebo-controlled, randomized study. J. Cardiovasc. Pharmacol. Ther. 2002, 7 (Suppl. S1), S11–S15. [Google Scholar] [CrossRef] [PubMed]
- Subathra, M.; Shila, S.; Devi, M.A.; Panneerselvam, C. Emerging role of Centella asiatica in improving age-related neurological antioxidant status. Exp. Gerontol. 2005, 40, 707–715. [Google Scholar] [CrossRef] [PubMed]
- Chong, N.J.; Aziz, Z. A Systematic Review of the Efficacy of Centella asiatica for Improvement of the Signs and Symptoms of Chronic Venous Insufficiency. Evid. Based Complement. Altern. Med. 2013, 2013, 627182. [Google Scholar] [CrossRef] [PubMed]
- Bylka, W.; Znajdek-Awizen, P.; Studzinska-Sroka, E.; Brzezinska, M. Centella asiatica in cosmetology. Postepy Dermatol. Alergol. 2013, 30, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Makino, Y.; Nagata, M.; Furuta, J.; Enomoto, H.; Hirota, T.; Tamari, M.; Noguchi, E. A rare variant in CYP27A1 and its association with atopic dermatitis with high serum total IgE. Allergy 2016, 71, 1486–1489. [Google Scholar] [CrossRef] [PubMed]
- Takahashi-Ando, N.; Jones, M.A.; Fujisawa, S.; Hama, R. Patient-reported outcomes after discontinuation of long-term topical corticosteroid treatment for atopic dermatitis: A targeted cross-sectional survey. Drug Healthc. Patient Saf. 2015, 7, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Bebawy, J.F. Perioperative steroids for peritumoral intracranial edema: A review of mechanisms, efficacy, and side effects. J. Neurosurg. Anesthesiol. 2012, 24, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Lee, Y.H.; Lee, K.H.; Kim, T.J. Effect of eriodictyol on the development of atopic dermatitis-like lesions in ICR mice. Biol. Pharm. Bull. 2013, 36, 1375–1379. [Google Scholar] [CrossRef] [PubMed]
- Hashim, P.; Sidek, H.; Helan, M.H.; Sabery, A.; Palanisamy, U.D.; Ilham, M. Triterpene composition and bioactivities of Centella asiatica. Molecules 2011, 16, 1310–1322. [Google Scholar] [CrossRef] [PubMed]
- Tang, B.; Zhu, B.; Liang, Y.; Bi, L.; Hu, Z.; Chen, B.; Zhang, K.; Zhu, J. Asiaticoside suppresses collagen expression and TGF-β/Smad signaling through inducing Smad7 and inhibiting TGF-βRI and TGF-βRII in keloid fibroblasts. Arch. Dermatol. Res. 2011, 303, 563–572. [Google Scholar] [CrossRef] [PubMed]
- Nowwarote, N.; Osathanon, T.; Jitjaturunt, P.; Manopattanasoontorn, S.; Pavasant, P. Asiaticoside induces type I collagen synthesis and osteogenic differentiation in human periodontal ligament cells. Phytother. Res. 2013, 27, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Maquart, F.X.; Chastang, F.; Simeon, A.; Birembaut, P.; Gillery, P.; Wegrowski, Y. Triterpenes from Centella asiatica stimulate extracellular matrix accumulation in rat experimental wounds. Eur. J. Dermatol. 1999, 9, 289–296. [Google Scholar] [PubMed]
- Nhiem, N.X.; Tai, B.H.; Quang, T.H.; Kiem, P.V.; Minh, C.V.; Nam, N.H.; Kim, J.H.; Im, L.R.; Lee, Y.M.; Kim, Y.H. A new ursane-type triterpenoid glycoside from Centella asiatica leaves modulates the production of nitric oxide and secretion of TNF-α in activated RAW264.7 cells. Bioorg. Med. Chem. Lett. 2011, 21, 1777–1781. [Google Scholar] [CrossRef] [PubMed]
- Bonte, F.; Dumas, M.; Chaudagne, C.; Meybeck, A. Influence of asiatic acid, madecassic acid, and asiaticoside on human collagen I synthesis. Planta Med. 1994, 60, 133–135. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.; Li, M.; Lu, Y.H.; Liu, D.H.; Li, C.C. Burn wound healing properties of asiaticoside and madecassoside. Exp. Ther. Med. 2016, 12, 1269–1274. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Sumiyoshi, M.; Samukawa, K.; Satake, N.; Sakanaka, M. Facilitating action of asiaticoside at low doses on burn wound repair and its mechanism. Eur. J. Pharmacol. 2008, 584, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Won, J.H.; Shin, J.S.; Park, H.J.; Jung, H.J.; Koh, D.J.; Jo, B.G.; Lee, J.Y.; Yun, K.; Lee, K.T. Anti-inflammatory effects of madecassic acid via the suppression of NF-κB pathway in LPS-induced RAW264.7 macrophage cells. Planta Med. 2010, 76, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Murakami, Y.; Shoji, M.; Hirata, A.; Tanaka, S.; Yokoe, I.; Fujisawa, S. Dehydrodiisoeugenol, an isoeugenol dimer, inhibits lipopolysaccharide-stimulated nuclear factor kappa B activation and cyclooxygenase-2 expression in macrophages. Arch. Biochem. Biophys. 2005, 434, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.T.; Goodarzi, H.; Chen, H.Y. IgE, mast cells, and eosinophils in atopic dermatitis. Clin. Rev. Allergy Immunol. 2011, 41, 298–310. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, S.; Kilgus, O.; Payer, E.; Kutil, R.; Elbe, A.; Mueller, C.; Stingl, G. Cytokine pattern of Langerhans cells isolated from murine epidermal cell cultures. J. Immunol. 1992, 149, 3524–3534. [Google Scholar] [PubMed]
- Friedmann, P.S. Contact sensitisation and allergic contact dermatitis: Immunobiological mechanisms. Toxicol. Lett. 2006, 162, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.D.; Lee, S.E.; Park, Y.S.; Shin, D.H.; Park, G.G.; Park, C.S. Immunosuppressive effects of fisetin against dinitrofluorobenzene-induced atopic dermatitis-like symptoms in NC/Nga mice. Food Chem. Toxicol. 2014, 66, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Choi, C.H.; Lee, K.; Lee, M.; Ham, I.; Choi, H.Y. Effects of Catalpa ovata stem bark on atopic dermatitis-like skin lesions in NC/Nga mice. J. Ethnopharmacol. 2013, 145, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Kim, M.H.; Choi, Y.Y.; Kim, E.H.; Hong, J.; Kim, K.; Yang, W.M. Improvement of atopic dermatitis with topical application of Spirodela polyrhiza. J. Ethnopharmacol. 2016, 180, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Muto, S.; Jung, K.; Itai, A.; Matsuda, H. Topical application with a new NF-κB inhibitor improves atopic dermatitis in NC/NgaTnd mice. J. Investig. Dermatol. 2007, 127, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Kim, M.S.; Jeong, G.S.; Yoon, J. Xanthii fructus extract inhibits TNF-α/IFN-γ-induced Th2-chemokines production via blockade of NF-κB, STAT1 and p38-MAPK activation in human epidermal keratinocytes. J. Ethnopharmacol. 2015, 171, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Lee, E.; Lim, T.; Jung, J.; Lyu, Y. Inhibitory effect of Asparagus cochinchinensis on tumor necrosis factor-α secretion from astrocytes. Int. J. Immunopharmacol. 1998, 20, 153–162. [Google Scholar] [CrossRef]
- Qiu, J.; Yu, L.; Zhang, X.; Wu, Q.; Wang, D.; Wang, X.; Xia, C.; Feng, H. Asiaticoside attenuates lipopolysaccharide-induced acute lung injury via down-regulation of NF-κB signaling pathway. Int. Immunopharmacol. 2015, 26, 181–187. [Google Scholar] [CrossRef] [PubMed]
| Extract | Composition of Extract |
|---|---|
| Titrated extract of Centella asiatica (TECA) | Asiaticoside (40%), Asiatic acid (30%), Madecassic acid (29–30%) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.H.; Choi, J.Y.; Son, D.J.; Park, E.K.; Song, M.J.; Hellström, M.; Hong, J.T. Anti-Inflammatory Effect of Titrated Extract of Centella asiatica in Phthalic Anhydride-Induced Allergic Dermatitis Animal Model. Int. J. Mol. Sci. 2017, 18, 738. https://doi.org/10.3390/ijms18040738
Park JH, Choi JY, Son DJ, Park EK, Song MJ, Hellström M, Hong JT. Anti-Inflammatory Effect of Titrated Extract of Centella asiatica in Phthalic Anhydride-Induced Allergic Dermatitis Animal Model. International Journal of Molecular Sciences. 2017; 18(4):738. https://doi.org/10.3390/ijms18040738
Chicago/Turabian StylePark, Ju Ho, Ji Yeon Choi, Dong Ju Son, Eun Kyung Park, Min Jong Song, Mats Hellström, and Jin Tae Hong. 2017. "Anti-Inflammatory Effect of Titrated Extract of Centella asiatica in Phthalic Anhydride-Induced Allergic Dermatitis Animal Model" International Journal of Molecular Sciences 18, no. 4: 738. https://doi.org/10.3390/ijms18040738







