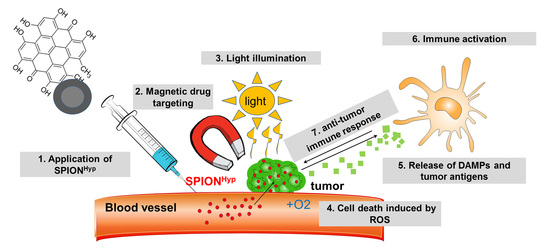
Analysis of Hypericin-Mediated Effects and Implications for Targeted Photodynamic Therapy
Abstract
:1. Introduction
2. Results
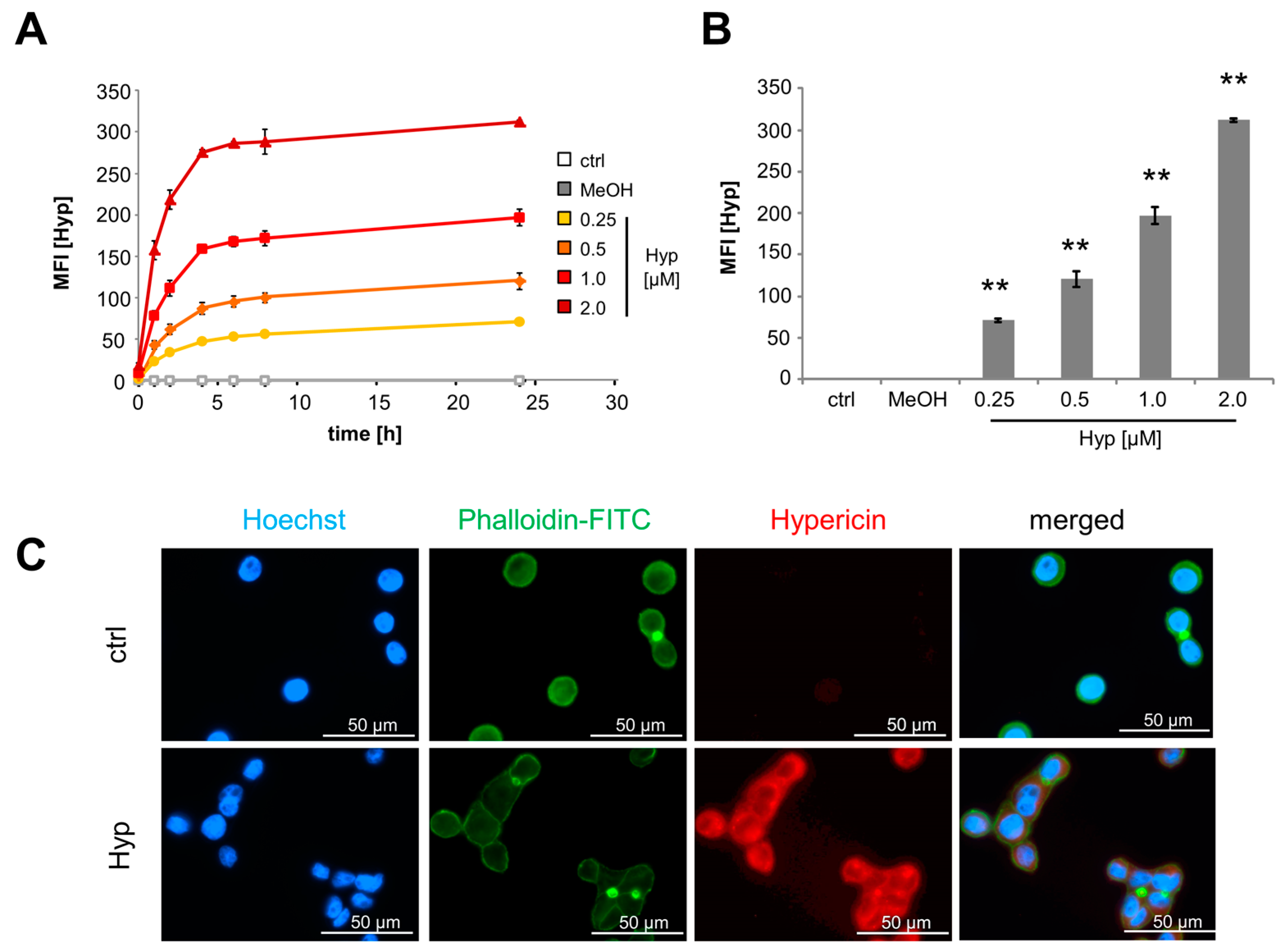
2.1. Intracellular Accumulation of Hypericin
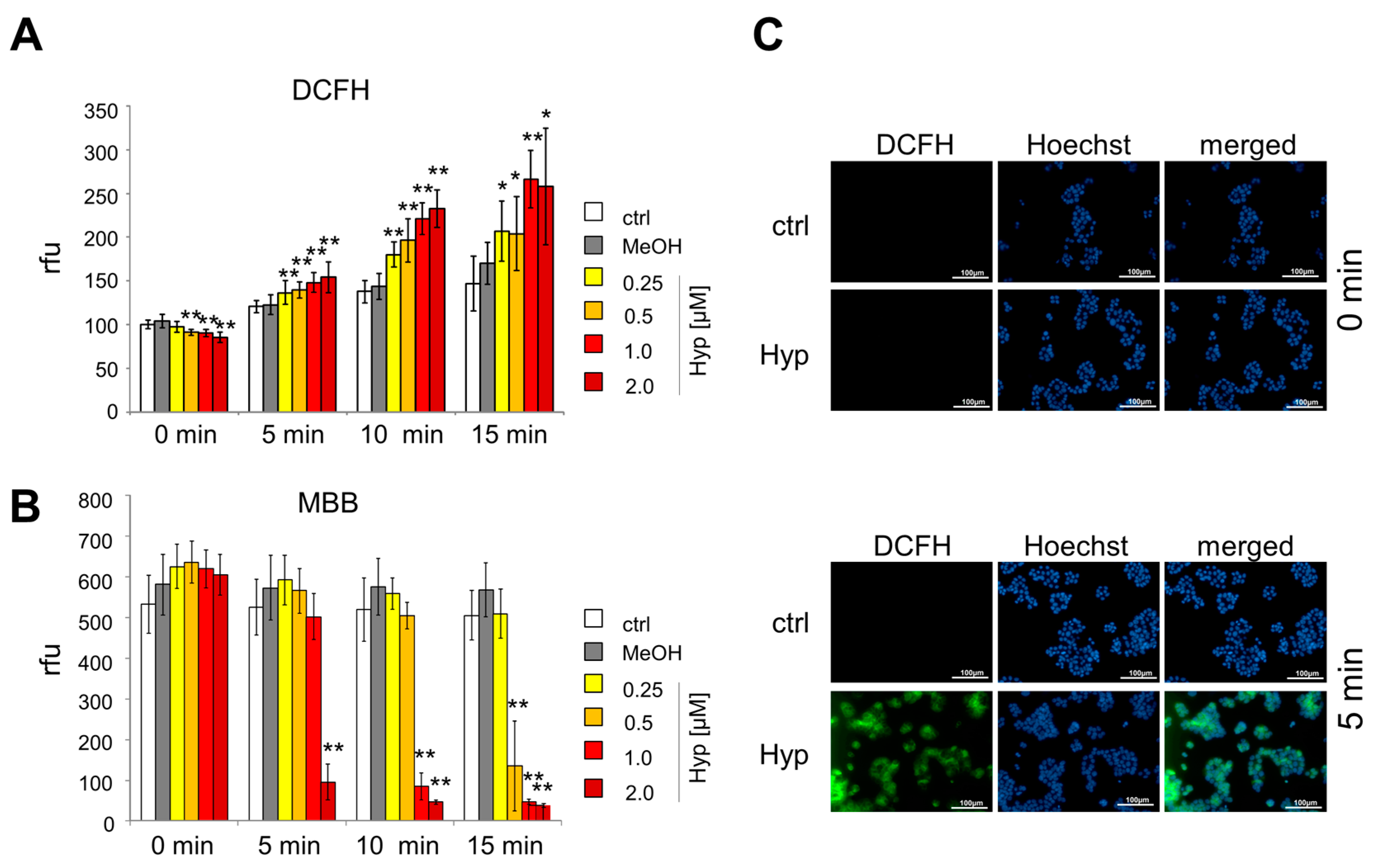
2.2. Oxidative Stress Induced by Hypericin
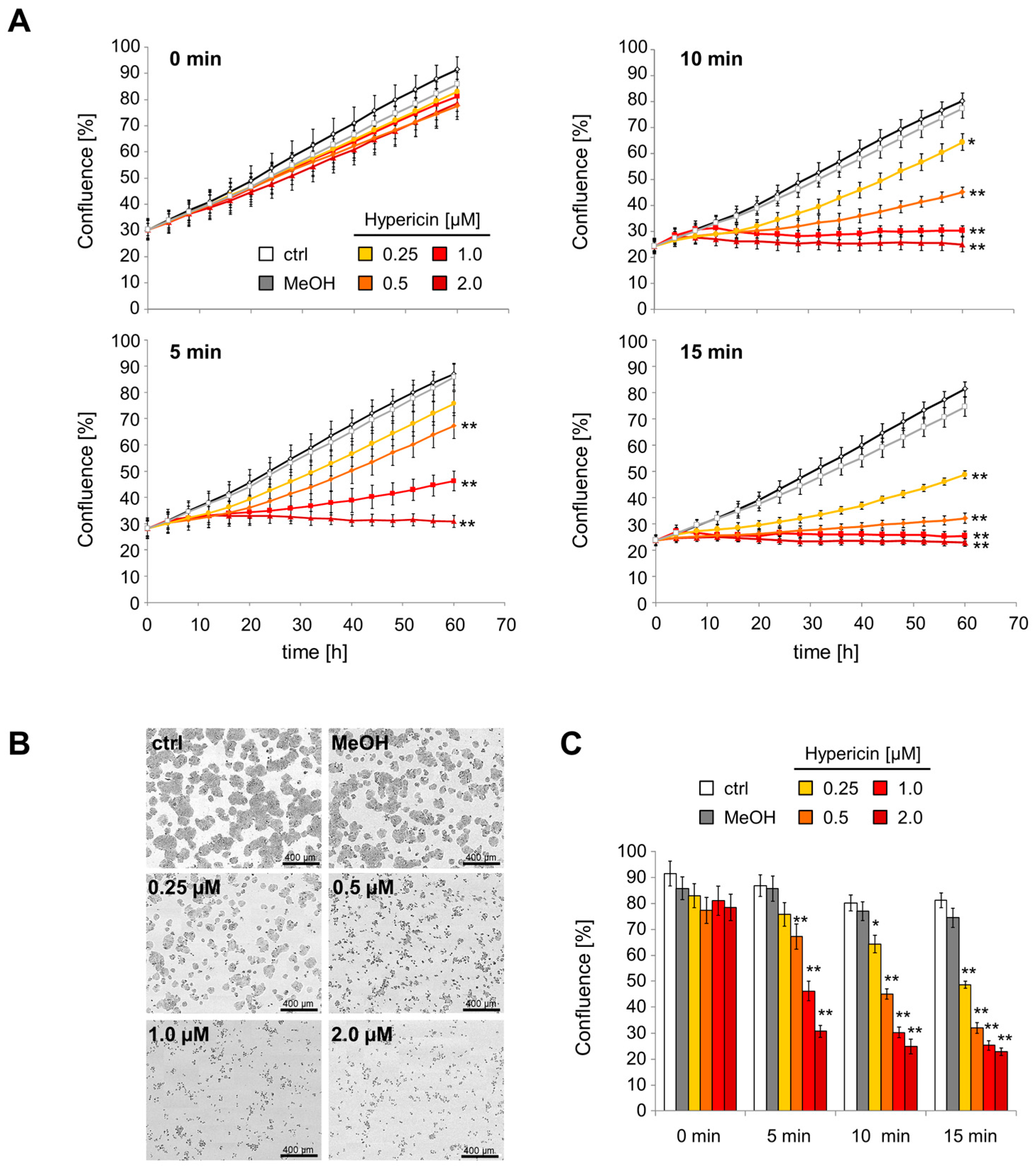
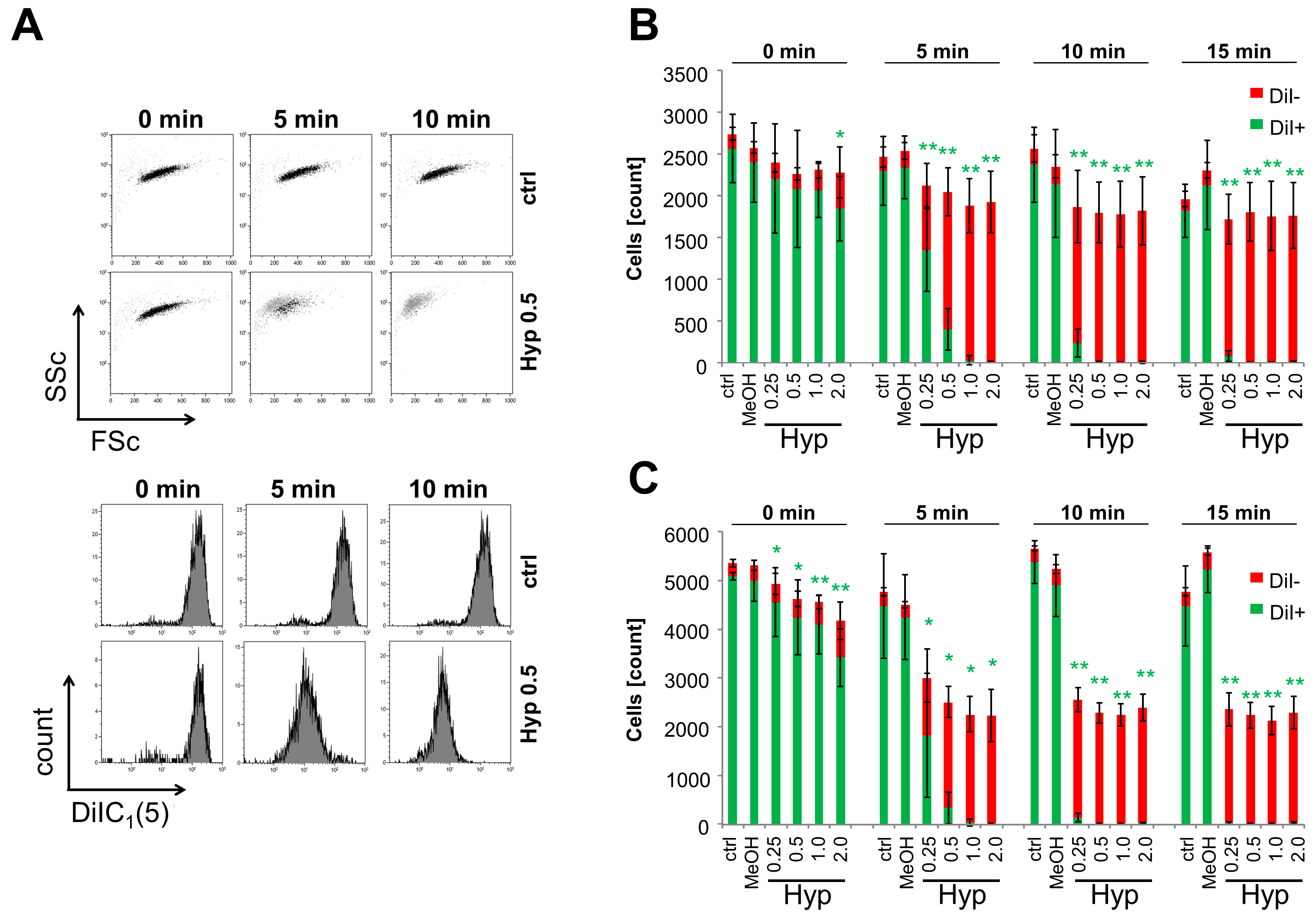
2.3. Inhibition of Cell Proliferation and Induction of Cell Death by Hypericin
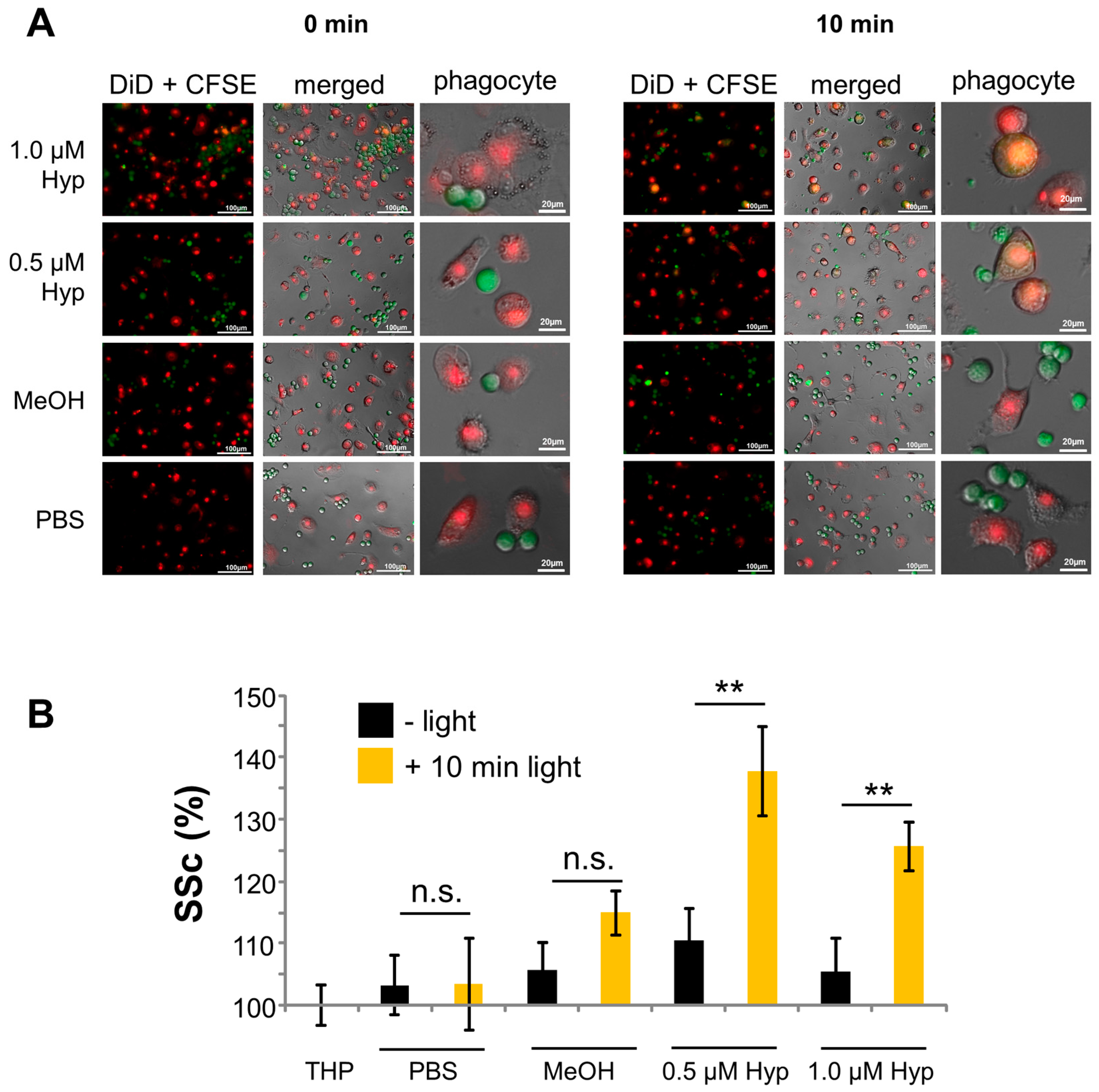
2.4. Phagocytosis of Hypericin-Treated JURKAT Cells by Macrophages
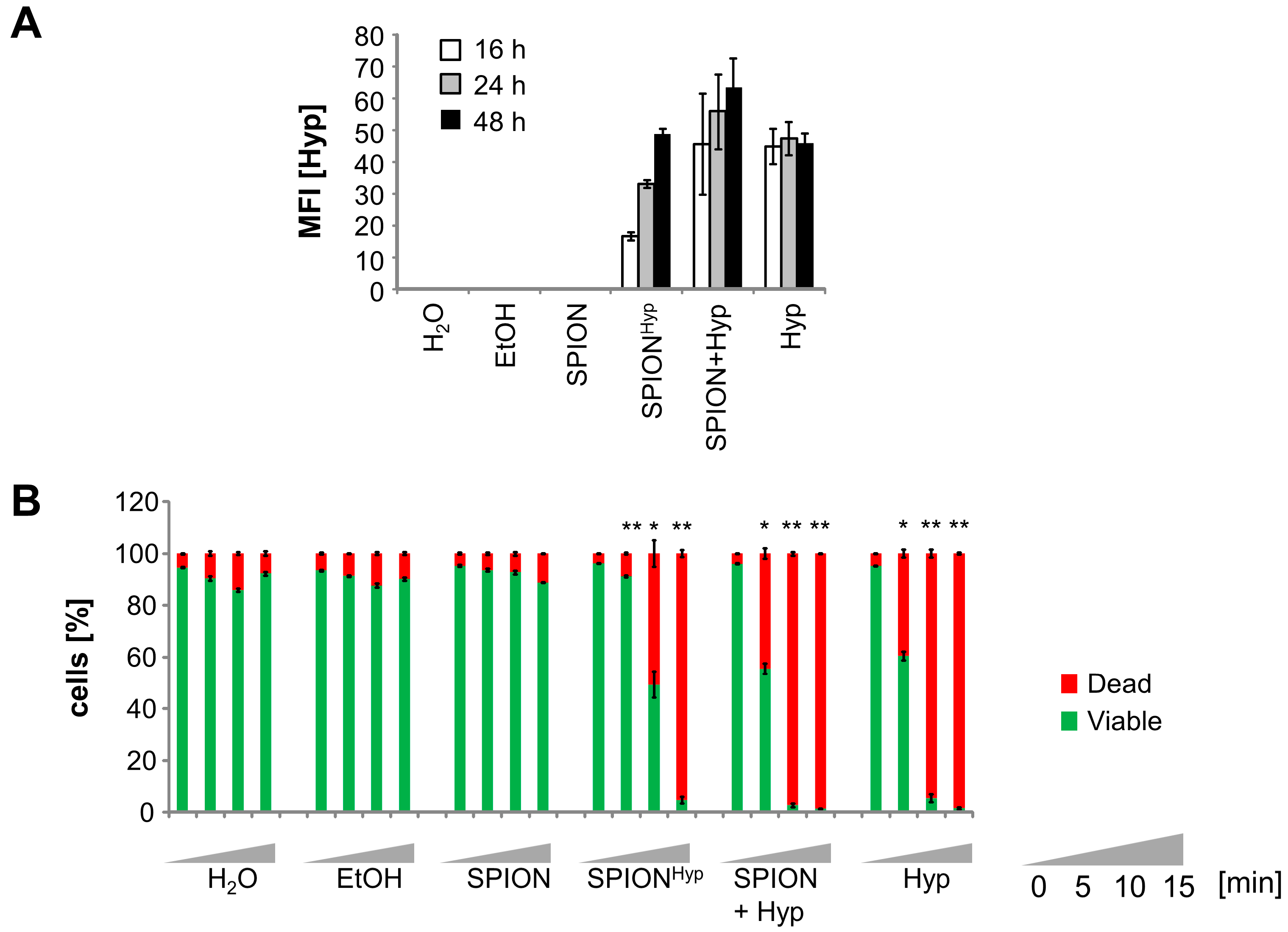
2.5. Comparison of Free Hypericin and SPIONHyp
3. Discussion
4. Materials and Methods
4.1. Hypericin and Hypericin-Lloaded Superparamagnetic Iron Oxide Nanoparticles (SPIONHyp)
4.2. Cells and Culture Conditions
4.3. Hypericin Treatment of Cells
4.4. Fluorescence Microscopy
4.5. Analysis of Viability and Drug Penetration into Cells by Flow Cytometry
4.6. Cell Proliferation after Treatment with Hypericin
4.7. Analysis of ROS Generation Using DCFH-DA
4.8. Phagocytosis of Hypericin-Treated Cells by Macrophages
4.9. Comparison of Jurkat Cells Treated with Free Hypericin and SPIONHyp
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| SEON | Section of Experimental Oncology and Nanomedicine |
| SPIONs | Superparamagnetic iron oxide nanoparticles |
| PDT | Photodynamic therapy |
| MDT | Magnetic drug targeting |
| ROS | Reactive oxygen species |
| ER | Endoplasmic reticulum |
| DCFH-DA | 2′,7′-Dichlorodihydrofluorescein diacetate |
| MBB | Monobromobimane |
| GSH | Glutathione |
| GSSG | Glutathione disulfide |
| MeOH | Methanol |
| DiIC1(5) | hexamethylindodicarbocyanine iodide dye |
| FSc | Forward scatter |
| SSc | Side scatter |
| DiD | C67H103ClN2O3S |
| CFSE | Carboxyfluorescein succinimidyl ester |
| SPIONHyp | Hypericin loaded superparamagnetic iron oxide nanoparticles |
| Hyp | Hypericin |
| DAMPs | Damage-associated molecular patterns |
| FCS | Fetal calf serum |
| PCR | Polymerase chain reaction |
| PBS | Phosphate buffered saline |
| LED | Light-emitting diode |
| FA | Formaldehyde |
| Fitc | Fluorescein isothiocyanate |
| FL | Fluorescence |
| PMA | Phorbol 12-myristate 13-acetate |
| MFI | Mean fluorescence index |
| ctrl | Control |
References
- Madeddu, C.; Deidda, M.; Piras, A.; Cadeddu, C.; Demurtas, L.; Puzzoni, M.; Piscopo, G.; Scartozzi, M.; Mercuro, G. Pathophysiology of cardiotoxicity induced by nonanthracycline chemotherapy. J. Cardiovasc. Med. 2016, 17, S12–S18. [Google Scholar] [CrossRef] [PubMed]
- Ballestrero, A.; Ferrando, F.; Garuti, A.; Basta, P.; Gonella, R.; Esposito, M.; Vannozzi, M.O.; Sorice, G.; Friedman, D.; Puglisi, M.; et al. High-dose mitoxantrone with peripheral blood progenitor cell rescue: Toxicity, pharmacokinetics and implications for dosage and schedule. Br. J. Cancer 1997, 76, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Crawford, J.; Dale, D.C.; Lyman, G.H. Chemotherapy-induced neutropenia: Risks, consequences, and new directions for its management. Cancer 2004, 100, 228–237. [Google Scholar] [CrossRef] [PubMed]
- Vantieghem, A.; Assefa, Z.; Vandenabeele, P.; Declercq, W.; Courtois, S.; Vandenheede, J.R.; Merlevede, W.; de Witte, P.; Agostinis, P. Hypericin-induced photosensitization of HeLa cells leads to apoptosis or necrosis. Involvement of cytochrome c and procaspase-3 activation in the mechanism of apoptosis. FEBS Lett. 1998, 440, 19–24. [Google Scholar] [CrossRef]
- Jendzelovska, Z.; Jendzelovsky, R.; Kucharova, B.; Fedorocko, P. Hypericin in the light and in the dark: Two sides of the same coin. Front. Plant Sci. 2016, 7, 560. [Google Scholar] [CrossRef] [PubMed]
- Ebermann, R.; Alth, G.; Kreitner, M.; Kubin, A. Natural products derived from plants as potential drugs for the photodynamic destruction of tumor cells. J. Photochem. Photobiol. B 1996, 36, 95–97. [Google Scholar] [CrossRef]
- Lavie, G.; Kaplinsky, C.; Toren, A.; Aizman, I.; Meruelo, D.; Mazur, Y.; Mandel, M. A photodynamic pathway to apoptosis and necrosis induced by dimethyl tetrahydroxyhelianthrone and hypericin in leukaemic cells: Possible relevance to photodynamic therapy. Br. J. Cancer 1999, 79, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.; Macgill, R.S.; Miller, G.C.; Pardini, R.S. Photoactivation of hypericin generates singlet oxygen in mitochondria and inhibits succinoxidase. Photochem. Photobiol. 1992, 55, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Agostinis, P.; Vantieghem, A.; Merlevede, W.; de Witte, P.A. Hypericin in cancer treatment: More light on the way. Int. J. Biochem. Cell Biol. 2002, 34, 221–241. [Google Scholar] [CrossRef]
- Agostinis, P.; Berg, K.; Cengel, K.A.; Foster, T.H.; Girotti, A.W.; Gollnick, S.O.; Hahn, S.M.; Hamblin, M.R.; Juzeniene, A.; Kessel, D.; et al. Photodynamic therapy of cancer: An update. CA Cancer J. Clin. 2011, 61, 250–281. [Google Scholar] [CrossRef] [PubMed]
- Dougherty, T.J.; Gomer, C.J.; Henderson, B.W.; Jori, G.; Kessel, D.; Korbelik, M.; Moan, J.; Peng, Q. Photodynamic therapy. J. Natl. Cancer Inst. 1998, 90, 889–905. [Google Scholar] [CrossRef] [PubMed]
- Henderson, B.W.; Dougherty, T.J. How does photodynamic therapy work? Photochem. Photobiol. 1992, 55, 145–157. [Google Scholar] [CrossRef] [PubMed]
- Ahn, P.H.; Quon, H.; O’Malley, B.W.; Weinstein, G.; Chalian, A.; Malloy, K.; Atkins, J.H.; Sollecito, T.; Greenberg, M.; McNulty, S.; et al. Toxicities and early outcomes in a phase 1 trial of photodynamic therapy for premalignant and early stage head and neck tumors. Oral Oncol. 2016, 55, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Castano, A.P.; Demidova, T.N.; Hamblin, M.R. Mechanisms in photodynamic therapy: Part one—Photosensitizers, photochemistry and cellular localization. Photodiagn. Photodyn. Ther. 2004, 1, 279–293. [Google Scholar] [CrossRef]
- Huntosova, V.; Stroffekova, K. Hypericin in the dark: Foe or ally in photodynamic therapy? Cancers 2016, 8, E93. [Google Scholar] [CrossRef] [PubMed]
- Barathan, M.; Mariappan, V.; Shankar, E.M.; Abdullah, B.J.; Goh, K.L.; Vadivelu, J. Hypericin-photodynamic therapy leads to interleukin-6 secretion by HepG2 cells and their apoptosis via recruitment of BH3 interacting-domain death agonist and caspases. Cell Death Dis. 2013, 4, e697. [Google Scholar] [CrossRef] [PubMed]
- Almeida, R.D.; Manadas, B.J.; Carvalho, A.P.; Duarte, C.B. Intracellular signaling mechanisms in photodynamic therapy. Biochim. Biophys. Acta 2004, 1704, 59–86. [Google Scholar] [CrossRef] [PubMed]
- Buytaert, E.; Dewaele, M.; Agostinis, P. Molecular effectors of multiple cell death pathways initiated by photodynamic therapy. Biochim. Biophys. Acta 2007, 1776, 86–107. [Google Scholar] [CrossRef] [PubMed]
- Siboni, G.; Weitman, H.; Freeman, D.; Mazur, Y.; Malik, Z.; Ehrenberg, B. The correlation between hydrophilicity of hypericins and helianthrone: Internalization mechanisms, subcellular distribution and photodynamic action in colon carcinoma cells. Photochem. Photobiol. Sci. 2002, 1, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Uzdensky, A.B.; Ma, L.-W.; Iani, V.; Hjortland, G.O.; Steen, H.B.; Moan, J. Intracellular localisation of hypericin in human glioblastoma and carcinoma cell lines. Lasers Med. Sci. 2001, 16, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Lyer, S.; Tietze, R.; Unterweger, H.; Zaloga, J.; Singh, R.; Matuszak, J.; Poettler, M.; Friedrich, R.P.; Duerr, S.; Cicha, I.; et al. Nanomedical innovation: The SEON-concept for an improved cancer therapy with magnetic nanoparticles. Nanomedicine 2015, 10, 3287–3304. [Google Scholar] [CrossRef] [PubMed]
- Tietze, R.; Lyer, S.; Durr, S.; Struffert, T.; Engelhorn, T.; Schwarz, M.; Eckert, E.; Goen, T.; Vasylyev, S.; Peukert, W.; et al. Efficient drug-delivery using magnetic nanoparticles—Biodistribution and therapeutic effects in tumour bearing rabbits. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 961–971. [Google Scholar] [CrossRef] [PubMed]
- Chertok, B.; David, A.E.; Yang, V.C. Brain tumor targeting of magnetic nanoparticles for potential drug delivery: Effect of administration route and magnetic field topography. J. Controll. Release Off. J. Controll. Release Soc. 2011, 155, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Sabnis, S.; Sabnis, N.A.; Raut, S.; Lacko, A.G. Superparamagnetic reconstituted high-density lipoprotein nanocarriers for magnetically guided drug delivery. Int. J. Nanomed. 2017, 12, 1453–1464. [Google Scholar] [CrossRef] [PubMed]
- Unterweger, H.; Subatzus, D.; Tietze, R.; Janko, C.; Poettler, M.; Stiegelschmitt, A.; Schuster, M.; Maake, C.; Boccaccini, A.R.; Alexiou, C. Hypericin-bearing magnetic iron oxide nanoparticles for selective drug delivery in photodynamic therapy. Int. J. Nanomed. 2015, 10, 6985–6996. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.D.; Agostinis, P. ER stress, autophagy and immunogenic cell death in photodynamic therapy-induced anti-cancer immune responses. Photochem. Photobiol. Sci. 2014, 13, 474–487. [Google Scholar] [CrossRef] [PubMed]
- Panzarini, E.; Inguscio, V.; Dini, L. Immunogenic cell death: Can it be exploited in photodynamic therapy for cancer? BioMed Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Kleemann, B.; Loos, B.; Scriba, T.J.; Lang, D.; Davids, L.M. St John’s Wort (Hypericum perforatum L.) photomedicine: Hypericin-photodynamic therapy induces metastatic melanoma cell death. PLoS ONE 2014, 9, e103762. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Joseph, J.A. Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radic. Biol. Med. 1999, 27, 612–616. [Google Scholar] [CrossRef]
- Yakubu, S.I.; Yakasai, I.A.; Musa, A. Spectrofluorimetric assay method for glutathione and glutathione transferase using monobromobimane. J. Basic Clin. Pharm. 2011, 2, 151–158. [Google Scholar] [PubMed]
- Rastogi, R.P.; Singh, S.P.; Hader, D.P.; Sinha, R.P. Detection of reactive oxygen species (ROS) by the oxidant-sensing probe 2′,7′-dichlorodihydrofluorescein diacetate in the cyanobacterium Anabaena variabilis PCC 7937. Biochem. Biophys. Res. Commun. 2010, 397, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Cossarizza, A.; Ferraresi, R.; Troiano, L.; Roat, E.; Gibellini, L.; Bertoncelli, L.; Nasi, M.; Pinti, M. Simultaneous analysis of reactive oxygen species and reduced glutathione content in living cells by polychromatic flow cytometry. Nat. Protoc. 2009, 4, 1790–1797. [Google Scholar] [CrossRef] [PubMed]
- Zitka, O.; Skalickova, S.; Gumulec, J.; Masarik, M.; Adam, V.; Hubalek, J.; Trnkova, L.; Kruseova, J.; Eckschlager, T.; Kizek, R. Redox status expressed as GSH:GSSG ratio as a marker for oxidative stress in paediatric tumour patients. Oncol. Lett. 2012, 4, 1247–1253. [Google Scholar] [PubMed]
- Mattiasson, G. Flow cytometric analysis of isolated liver mitochondria to detect changes relevant to cell death. Cytom. Part A J. Int. Soc. Anal. Cytol. 2004, 60, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.D.; Krysko, D.V.; Vandenabeele, P.; Agostinis, P. Hypericin-based photodynamic therapy induces surface exposure of damage-associated molecular patterns like HSP70 and calreticulin. Cancer Immunol. Immunother. 2012, 61, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Vandenberk, L.; Belmans, J.; Van Woensel, M.; Riva, M.; Van Gool, S.W. Exploiting the immunogenic potential of cancer cells for improved dendritic cell Vaccines. Front. Immunol. 2015, 6, 663. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, R.P.; Janko, C.; Poettler, M.; Tripal, P.; Zaloga, J.; Cicha, I.; Durr, S.; Nowak, J.; Odenbach, S.; Slabu, I.; et al. Flow cytometry for intracellular SPION quantification: Specificity and sensitivity in comparison with spectroscopic methods. Int. J. Nanomed. 2015, 10, 4185–4201. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Toyooka, T.; Ibuki, Y. Simple and easy method to evaluate uptake potential of nanoparticles in mammalian cells using a flow cytometric light scatter analysis. Environ. Sci. Technol. 2007, 41, 3018–3024. [Google Scholar] [CrossRef] [PubMed]
- Toduka, Y.; Toyooka, T.; Ibuki, Y. Flow cytometric evaluation of nanoparticles using side-scattered light and reactive oxygen species-mediated fluorescence-correlation with genotoxicity. Environ. Sci. Technol. 2012, 46, 7629–7636. [Google Scholar] [CrossRef] [PubMed]
- Zucker, R.M.; Daniel, K.M.; Massaro, E.J.; Karafas, S.J.; Degn, L.L.; Boyes, W.K. Detection of silver nanoparticles in cells by flow cytometry using light scatter and far-red fluorescence. Cytom. Part A J. Int. Soc. Anal. Cytol. 2013, 83, 962–972. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mühleisen, L.; Alev, M.; Unterweger, H.; Subatzus, D.; Pöttler, M.; Friedrich, R.P.; Alexiou, C.; Janko, C. Analysis of Hypericin-Mediated Effects and Implications for Targeted Photodynamic Therapy. Int. J. Mol. Sci. 2017, 18, 1388. https://doi.org/10.3390/ijms18071388
Mühleisen L, Alev M, Unterweger H, Subatzus D, Pöttler M, Friedrich RP, Alexiou C, Janko C. Analysis of Hypericin-Mediated Effects and Implications for Targeted Photodynamic Therapy. International Journal of Molecular Sciences. 2017; 18(7):1388. https://doi.org/10.3390/ijms18071388
Chicago/Turabian StyleMühleisen, Laura, Magdalena Alev, Harald Unterweger, Daniel Subatzus, Marina Pöttler, Ralf P. Friedrich, Christoph Alexiou, and Christina Janko. 2017. "Analysis of Hypericin-Mediated Effects and Implications for Targeted Photodynamic Therapy" International Journal of Molecular Sciences 18, no. 7: 1388. https://doi.org/10.3390/ijms18071388