The New Structure of Core Oligosaccharide Presented by Proteus penneri 40A and 41 Lipopolysaccharides
Abstract
:1. Introduction
2. Results
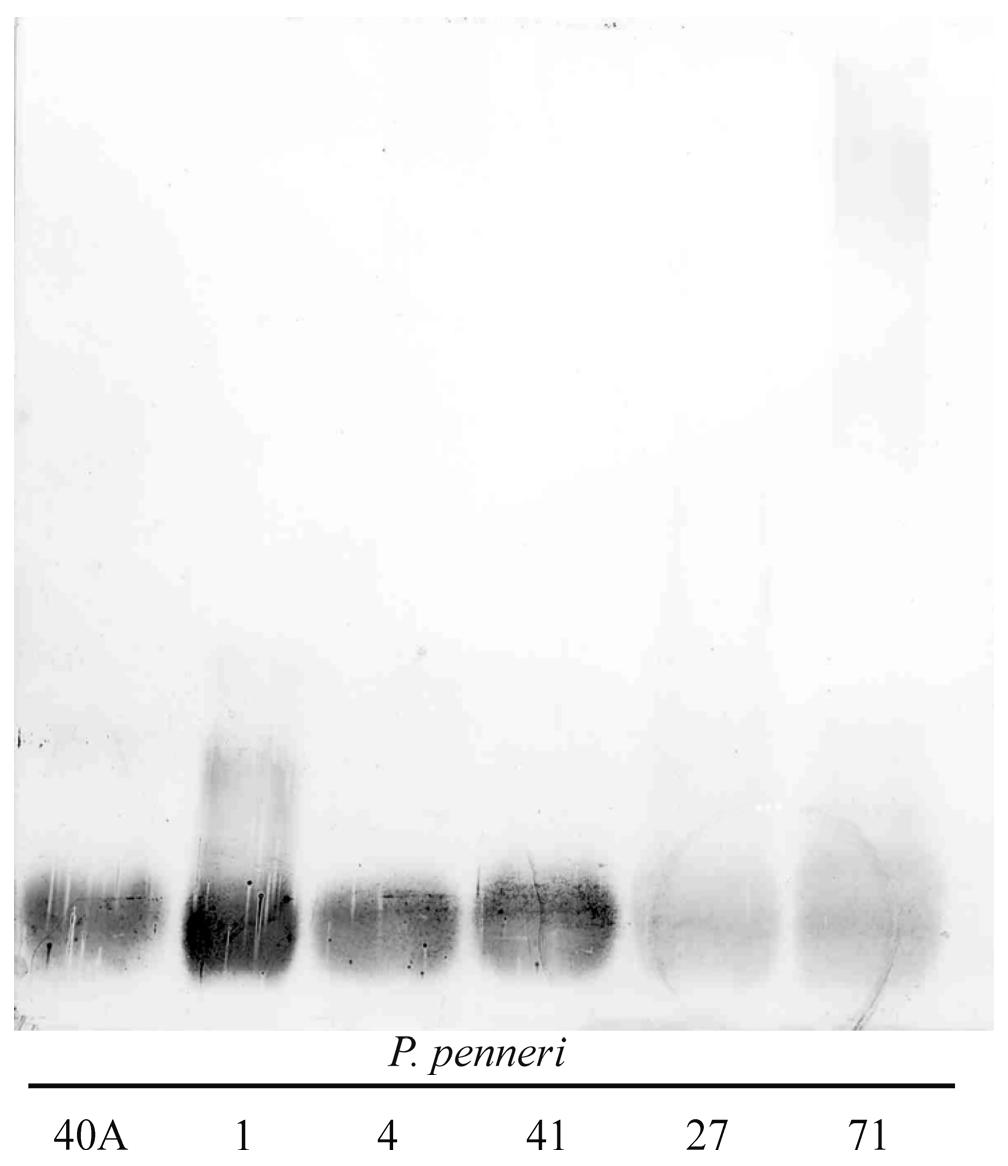
2.1. Serological Studies
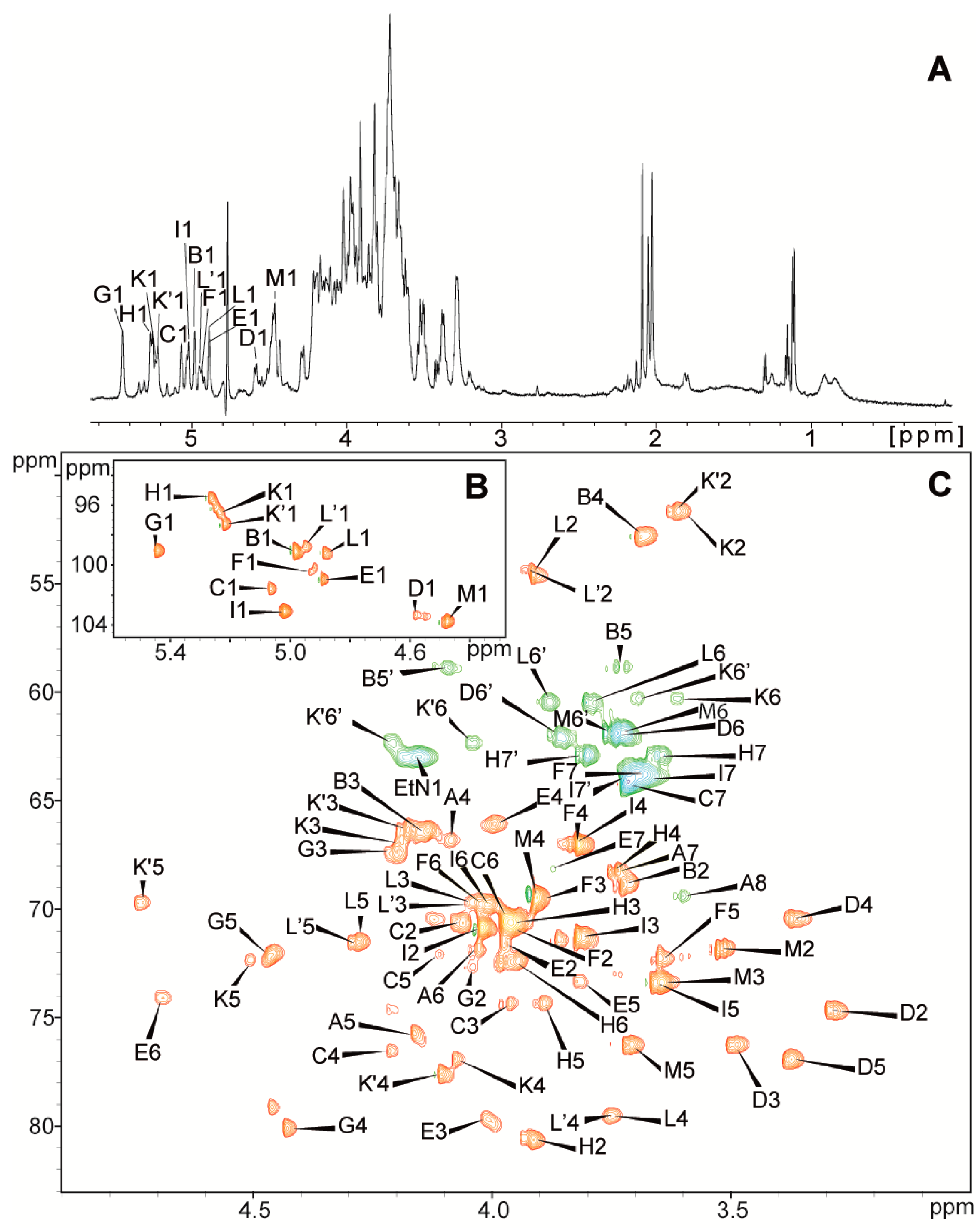
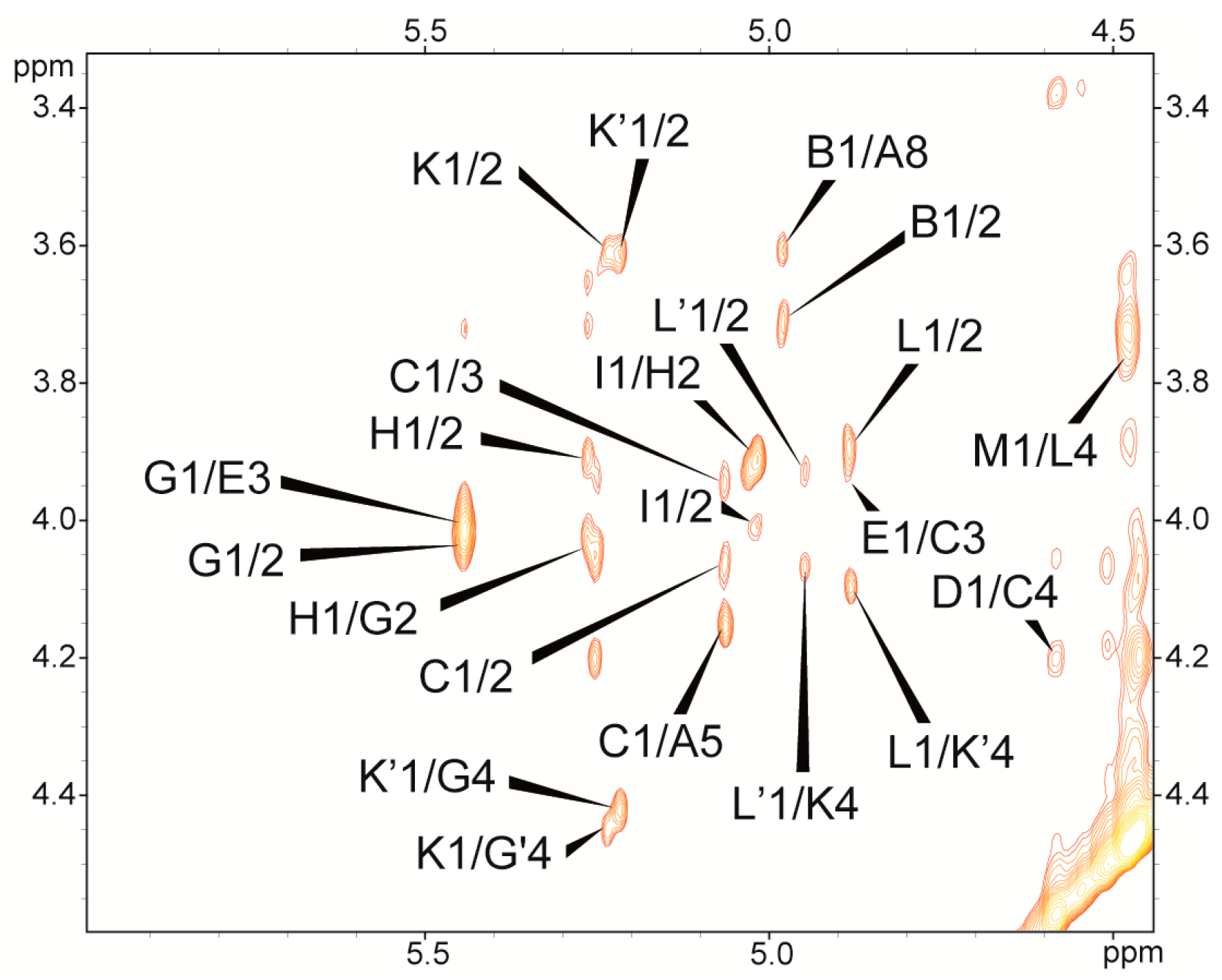
2.2. Structural Studies
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains
4.2. Lipopolysaccharide
4.3. De-O-Acetylation of the Core Oligosaccharide
4.4. P. penneri 40A Core Oligosaccharide Conjugate
4.5. Serological Assays
4.6. Chemical Method
4.7. Instrumental Methods
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Drzewiecka, D.; Sidorczyk, Z. Characterization of Proteus penneri species–Human opportunistic pathogens. Post. Mikrobiol. 2005, 44, 113–126. [Google Scholar]
- Palusiak, A. Immunochemical properties of Proteus penneri lipopolysaccharides—One of the major Proteus sp. virulence factors. Carbohydr. Res. 2013, 380, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Knirel, Y.A.; Perepelov, A.V.; Kondakova, A.N.; Senchenkova, S.N.; Sidorczyk, Z.; Rozalski, A.; Kaca, W. Structure and serology of O-antigens as the basis for classification of Proteus strains. Innate Immun. 2011, 17, 70–96. [Google Scholar] [CrossRef] [PubMed]
- Vinogradov, E.; Sidorczyk, Z.; Knirel, Y.A. Structure of the lipopolysaccharide core region of the bacteria of the genus Proteus. Aust. J. Chem. 2002, 55, 61–67. [Google Scholar] [CrossRef]
- Holst, O. The structures of core regions from enterobacterial lipopolysaccharides—An update. FEMS Microbiol. Lett. 2007, 271, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Siwińska, M.; Levina, E.A.; Ovchinnikova, O.G.; Drzewiecka, D.; Shashkov, A.S.; Różalski, A.; Knirel, Y.A. Classification of a Proteus penneri clinical isolate with a unique O-antigen structure to a new Proteus serogroup, O80. Carbohydr. Res. 2015, 407, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Palusiak, A.; Dzieciątkowska, M.; Sidorczyk, Z. Application of two different kinds of sera against the Proteus penneri lipopolysaccharide core region in search of epitopes determining cross-reactions with antibodies. Arch. Immunol. Ther. Exp. 2008, 56, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Palusiak, A.; Sidorczyk, Z. Characterization of epitope specificity of Proteus penneri 7 lipopolysaccharide core region. Acta Biochim. Pol. 2010, 57, 529–532. [Google Scholar] [PubMed]
- Palusiak, A.; Sidorczyk, Z. Serological characterization of the core region of lipopolysaccharides of rough Proteus sp. strains. Arch. Immunol. Ther. Exp. 2009, 57, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Palusiak, A. Classification of Proteus penneri lipopolysaccharides into core region serotypes. Med. Microbiol. Immunol. 2016, 205, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Vinogradov, E.; Bock, K. The structure of the core part of Proteus vulgaris OX2 lipopolysaccharide. Carbohydr. Res. 1999, 320, 239–243. [Google Scholar] [CrossRef]
- Jansson, P.E.; Kenne, L.; Widmalm, G. Structure of the O-antigen polysaccharide from Escherichia coli O18ac: A revision using computer-assisted structural analysis with the program CASPER. Carbohydr. Res. 1989, 193, 322–325. [Google Scholar] [CrossRef]
- Vinogradov, E. Structure of the core part of the lipopolysaccharide from Proteus mirabilis genomic strain HI4320. Biochemistry 2011, 76, 803–807. [Google Scholar] [CrossRef] [PubMed]
- Vinogradov, E.; Sidorczyk, Z.; Knirel, Y.A. Structure of the core part of the lipopolysaccharides from Proteus penneri strains 7, 8, 14, 15, and 21. Carbohydr. Res. 2002, 337, 643–649. [Google Scholar] [CrossRef]
- Westphal, O.; Jann, K. Bacterial lipopolysaccharides extraction with phenol-water and further applications of the procedure. Methods Carbohydr. Chem. 1965, 5, 83–91. [Google Scholar]
- Sidorczyk, Z.; Zych, K.; Toukach, F.V.; Arbatsky, N.P.; Zabłotni, A.; Shashkov, A.S.; Knirel, Y.A. Structure of the O-polysaccharide and classification of Proteus mirabilis strain G1 in Proteus serogroup O3. Eur. J. Biochem. 2002, 269, 1406–1412. [Google Scholar] [CrossRef] [PubMed]
- Jennings, H.J.; Lugowski, C. Immunochemistry of groups A, B, and C meningococcal polysaccharide-tetanus toxoid conjugates. J. Immunol. 1981, 127, 1011–1018. [Google Scholar] [PubMed]
- Ciucanu, I.; Kerek, F. A simple and rapid method for the permethylation of carbohydrates. Carbohydr. Res. 1984, 131, 209–217. [Google Scholar] [CrossRef]
- Goddard, T.D.; Kneller, D.G. SPARKY 3; University of California: San Francisco, CA, USA, 2001. [Google Scholar]
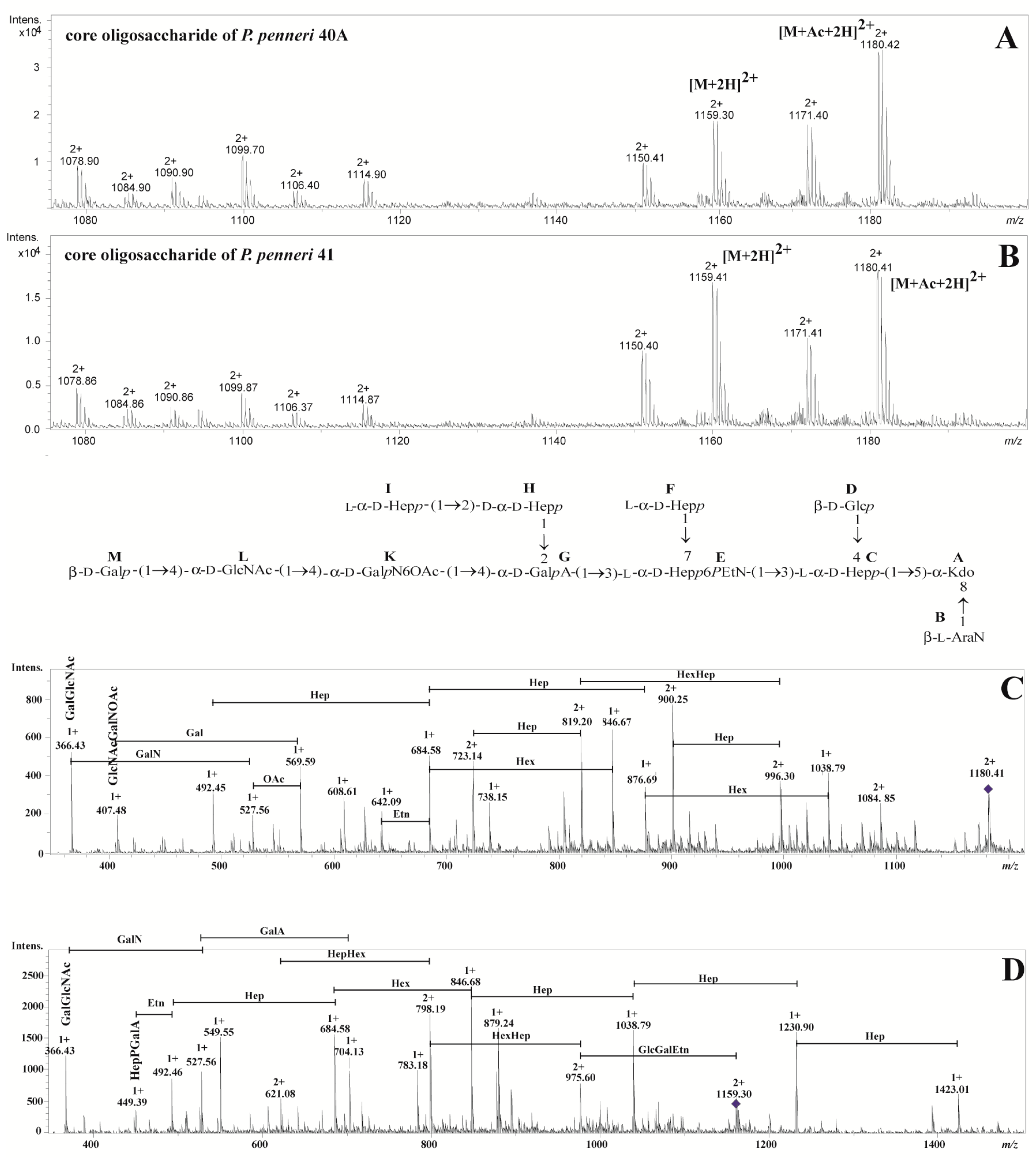
| The Observed Ion (m/z)/The Calculated Mass | The Ion Interpretation | |
|---|---|---|
| P. penneri 40A | P. penneri 41 | |
| 1180.422+/2358.84 | 1180.412+/2358.82 | [M+Ac+2H]2+ |
| 1171.402+/2340.80 | 1171.412+/2340.82 | [M+Ac-H2O+2H]2+ |
| 1159.302+/2316.60 | 1159.412+/2316.82 | [M+2H]2+ |
| 1150.412+/2298.82 | 1150.402+/2298.80 | [M-H2O+2H]2+ |
| 1114.902+/2227.80 | 1114.872+/2227.74 | [M-Ara4N+Ac+2H]2+ |
| 1106.402+/2210.80 | 1106.372+/2210.74 | [M-Ara4N+Ac-H2O+2H]2+ |
| 1099.702+/2197.40 | 1099.872+/2197.74 | [M-Hex+Ac+2H]2+ |
| 1090.902+/2179.80 | 1090.862+/2179.72 | [M-Hex+Ac-H2O+2H]2+ |
| 1084.902+/2167.80 | 1084.862+/2167.72 | [M-Hep+Ac+2H]2+ |
| 1078.902+/2155.80 | 1078.862+/2155.72 | [M-Hex+2H]2+ |
| Residues | Chemical Shifts (ppm) | |||||||
|---|---|---|---|---|---|---|---|---|
| H1/C1 | H2/C2 | H3(H3ax,eq)/C3 | H4/C4 | H5,5′/C5 | H6,6′/C6 | H7,7′/C7 | H8,8′/C8 (NAc, OAc) | |
| A→5,8)-Kdo | (1.80, 2.19) | 4.09 | 4.16 | 4.03 | 3.74 | 3.60, 3.92 | ||
| 35.4 | 66.8 | 75.7 | 71.7 | 68.2 | 69.4 | |||
| B β-l-Ara4N-(1→ | 4.98 | 3.72 | 4.14 | 3.68 | 3.73, 4.10 | |||
| 99.1 | 68.8 | 66.5 | 52.8 | 58.8 | ||||
| C→3,4)-l-glycero-α-d-manno-Hepp-(1→ | 5.07 | 4.06 | 3.96 | 4.21 | 4.11 | 3.97 | 3.72 | |
| 101.6 | 70.6 | 74.4 | 76.5 | 72.1 | 70.5 | 64.3 | ||
| D β-d-Glcp-(1→ | 4.58 | 3.30 | 3.49 | 3.37 | 3.37 | 3.74, 3.86 | ||
| 103.3 | 74.7 | 76.3 | 70.4 | 76.9 | 62.0 | |||
| E→3,7)-l-glycero-α-d-manno-Hepp-(1→ | 4.89 | 3.98 | 4.01 | 3.99 | 3.81 | 4.69 | 3.88 | |
| 101.0 | 71.6 | 79.7 | 66.1 | 73.3 | 74.1 | 68.2 | ||
| F l-glycero-α-d-manno-Hepp-(1→ | 4.93 | 3.96 | 3.89 | 3.82 | 3.65 | 4.02 | 3.69 | |
| 100.4 | 70.9 | 69.5 | 67.1 | 72.4 | 69.8 | 63.8 | ||
| G→2,4)-α-d-GalpA-(1→ | 5.45 | 4.04 | 4.21 | 4.43 | 4.46 | |||
| 99.0 | 72.7 | 67.3 | 80.1 | 72.2 | 176.0 | |||
| H→2)-d-glycero-α-d-manno-Hepp-(1→ | 5.27 | 3.91 | 4.00 | 3.75 | 3.89 | 3.95 | 3.65, 3.81 | |
| 95.5 | 80.6 | 70.6 | 68.3 | 74.3 | 72.4 | 62.9 | ||
| I l-glycero-α-d-manno-Hepp-(1→ | 5.02 | 4.02 | 3.82 | 3.83 | 3.66 | 4.01 | 3.67, 3.72 | |
| 103.1 | 70.8 | 71.3 | 67.0 | 73.4 | 69.8 | 64.0 | ||
| K→4)-α-d-GalpN-(1→ | 5.23 | 3.60 | 4.19 | 4.08 | 4.50 | 3.61, 3.70 | ||
| 96.5 | 51.7 | 66.9 | 76.9 | 72.4 | 60.3 | |||
| K’→4)-α-d-GalpN6OAc-(1→ | 5.22 | 3.62 | 4.18 | 4.11 | 4.73 | 4.04, 4.21 | (2.10) | |
| 97.3 | 51.7 | 66.3 | 77.6 | 69.7 | 62.3 | (21.0, 174.5) | ||
| L→4)-α-d-GlcpNAc-(1→ | 4.88 | 3.92 | 4.04 | 3.75 | 4.28 | 3.79,3.88 | (2.04) | |
| 99.2 | 54.6 | 69.7 | 79.5 | 71.5 | 60.5 | (22.7, 175.3) | ||
| L’→4)-α-d-GlcpNAc-(1→ | 4.95 | 3.93 | 4.05 | 3.78 | 71.5 | (2.06) | ||
| 98.8 | 54.4 | 69.8 | 79.6 | 4.29 | (22.7, 175.4) | |||
| M β-d-Galp-(1→ | 4.48 | 3.53 | 3.64 | 3.91 | 3.71 | 3.72, 3.75 | ||
| 103.7 | 71.8 | 73.4 | 69.5 | 76.3 | 61.9 | |||
| PEtN | 4.16 | 3.28 | ||||||
| 63.0 | 41.0 | |||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palusiak, A.; Maciejewska, A.; Lugowski, C.; Rozalski, A.; Kaszowska, M. The New Structure of Core Oligosaccharide Presented by Proteus penneri 40A and 41 Lipopolysaccharides. Int. J. Mol. Sci. 2018, 19, 676. https://doi.org/10.3390/ijms19030676
Palusiak A, Maciejewska A, Lugowski C, Rozalski A, Kaszowska M. The New Structure of Core Oligosaccharide Presented by Proteus penneri 40A and 41 Lipopolysaccharides. International Journal of Molecular Sciences. 2018; 19(3):676. https://doi.org/10.3390/ijms19030676
Chicago/Turabian StylePalusiak, Agata, Anna Maciejewska, Czeslaw Lugowski, Antoni Rozalski, and Marta Kaszowska. 2018. "The New Structure of Core Oligosaccharide Presented by Proteus penneri 40A and 41 Lipopolysaccharides" International Journal of Molecular Sciences 19, no. 3: 676. https://doi.org/10.3390/ijms19030676





