Fluorescent Polymer Incorporating Triazolyl Coumarin Units for Cu2+ Detection via Planarization of Ict-Based Fluorophore
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Measurement
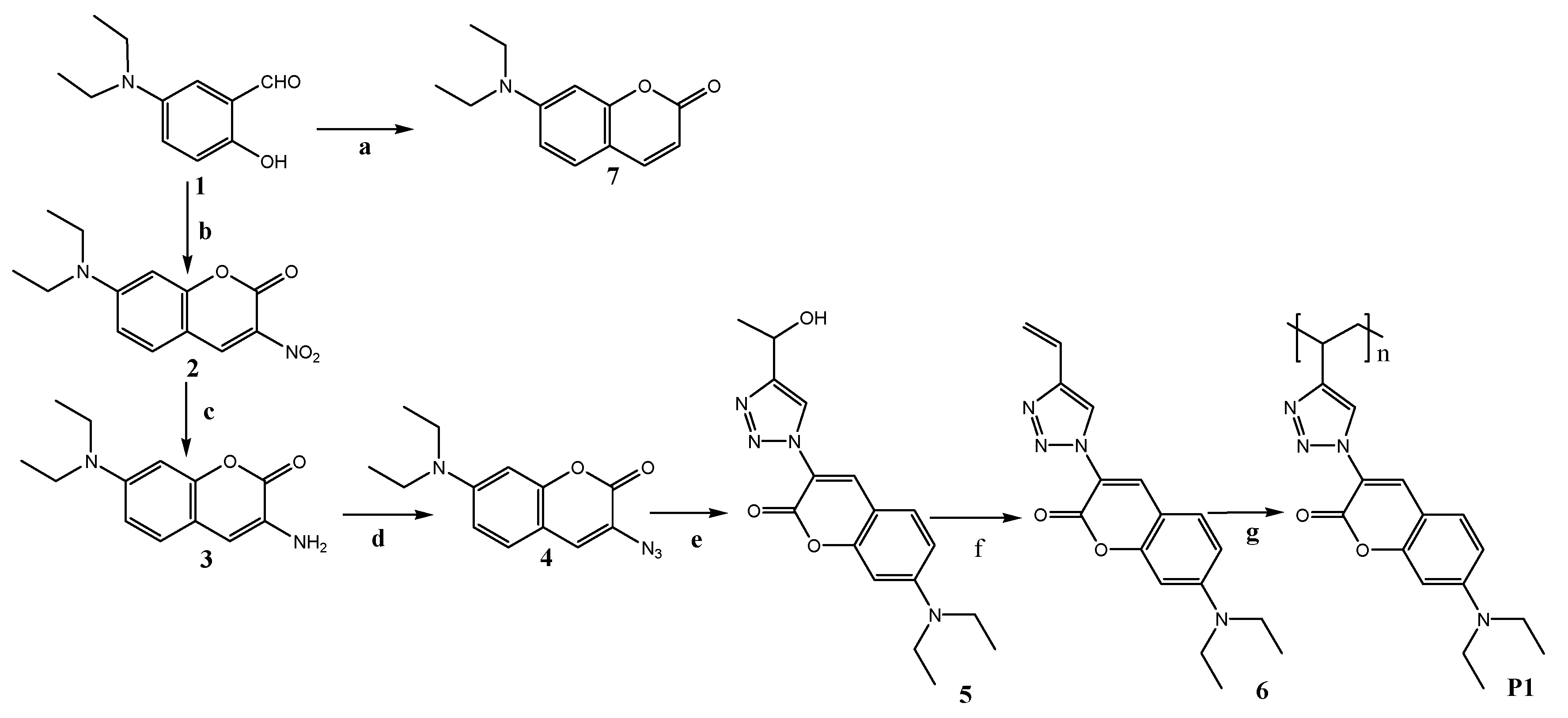
2.3. Polymer Synthesis
2.3.1. Synthesis of Compound 5
2.3.2. Synthesis of Monomer 6
2.3.3. Synthesis of P1
3. Results and Discussion
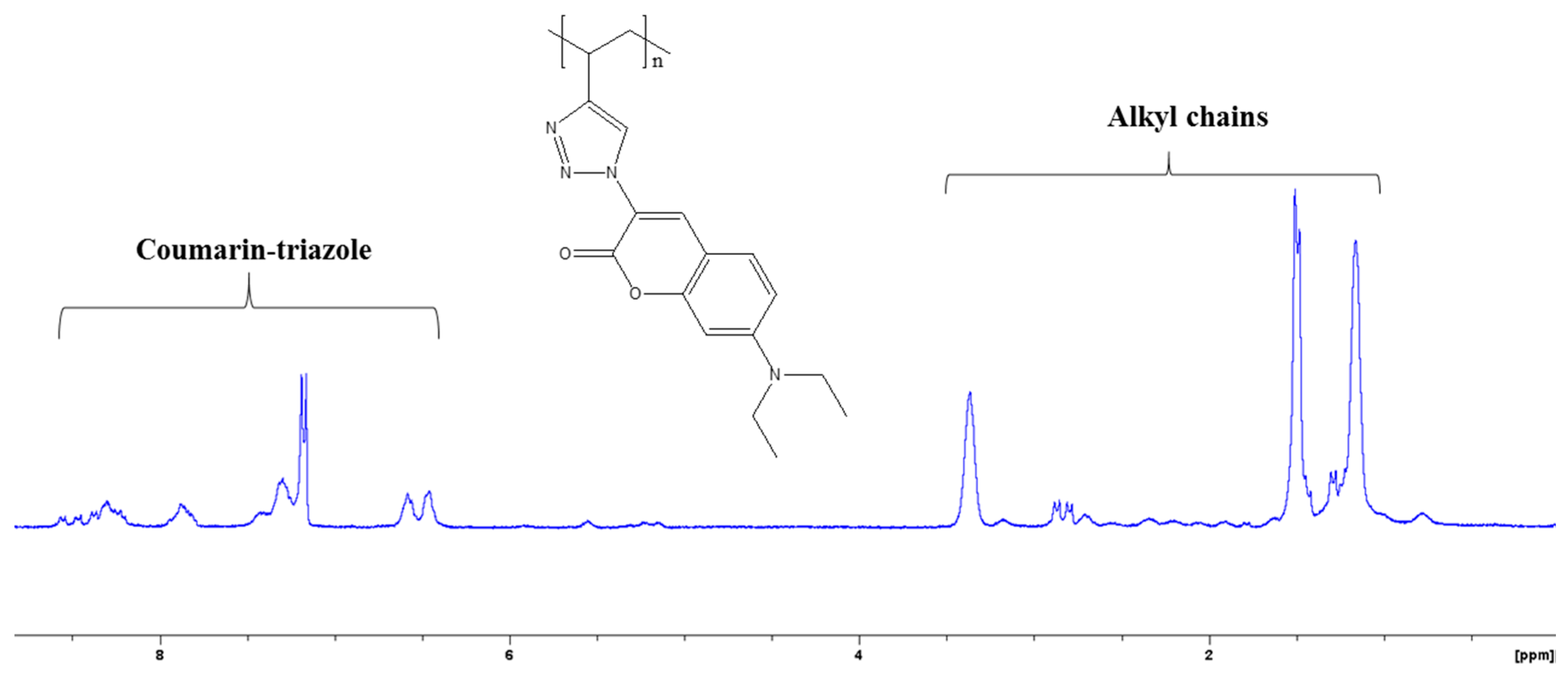
3.1. Synthesis and Characterization P1
3.2. Absorption Spectra Analysis
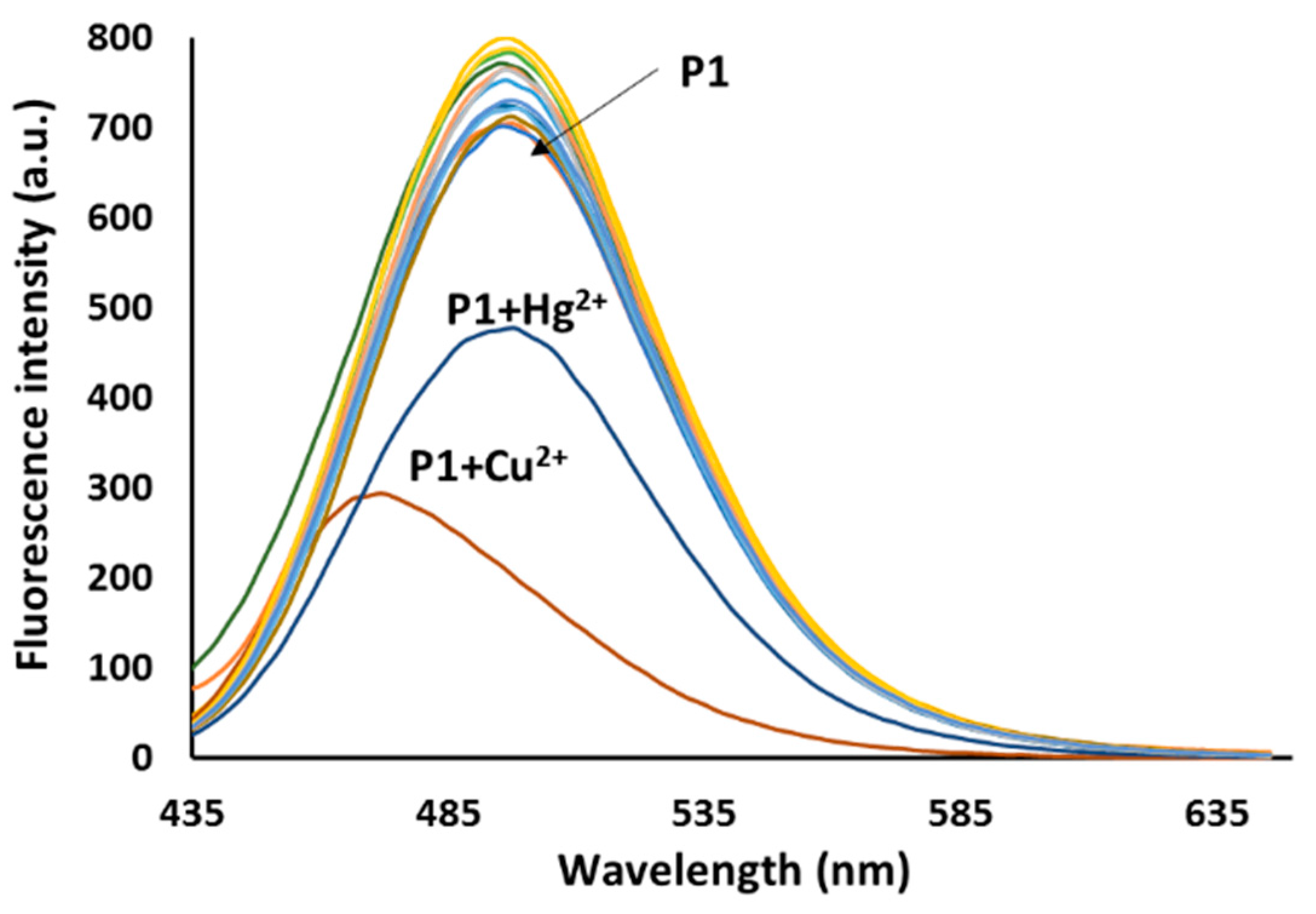
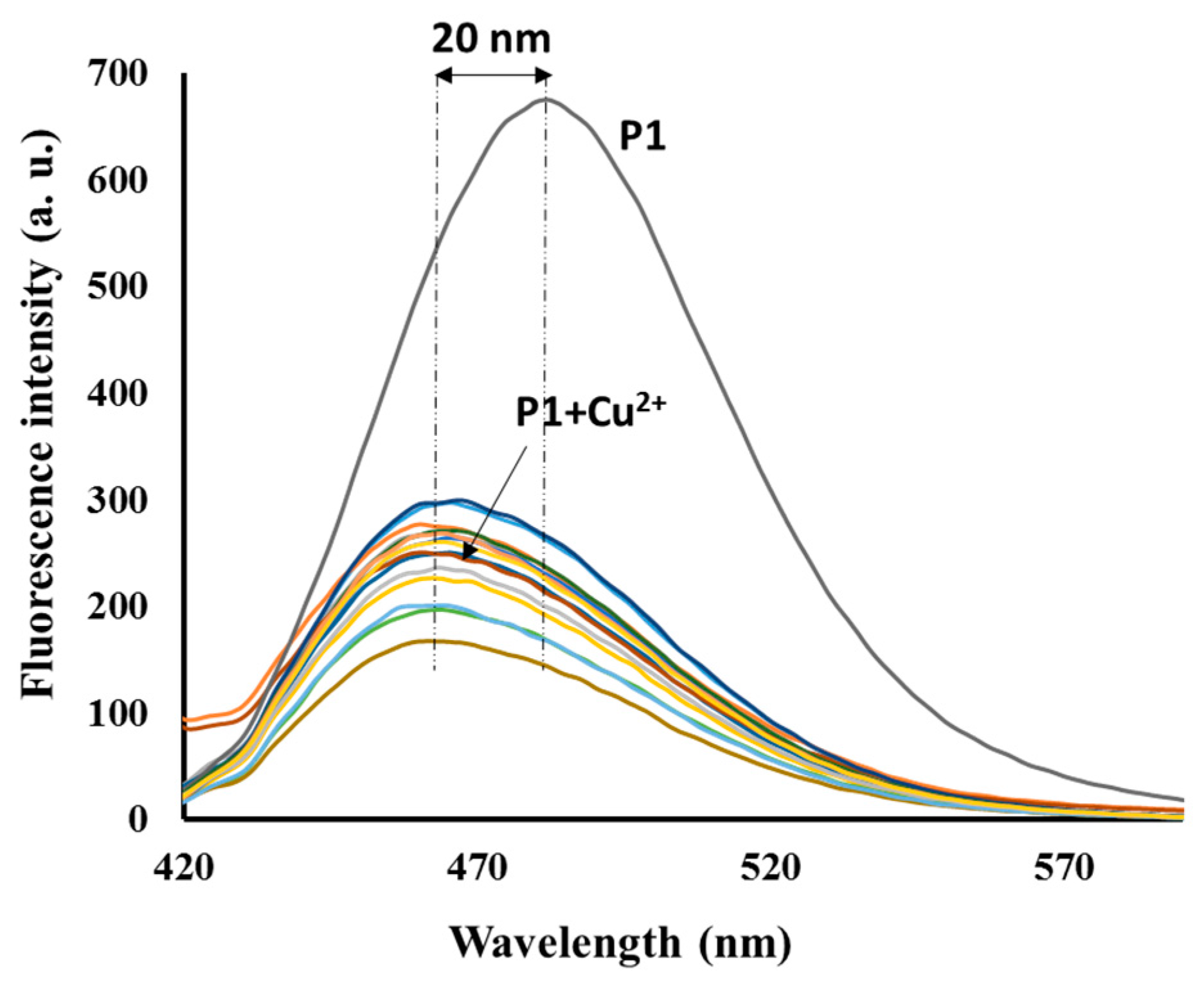
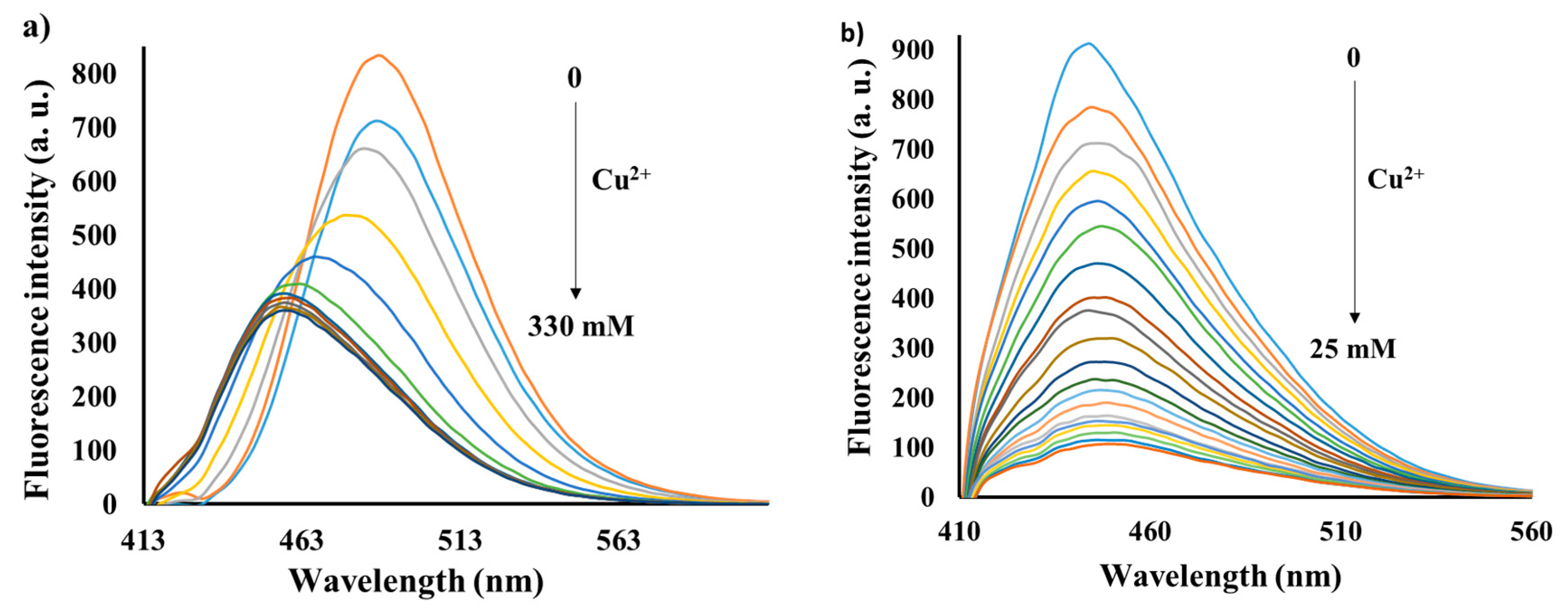
3.3. Emission Spectra Analysis
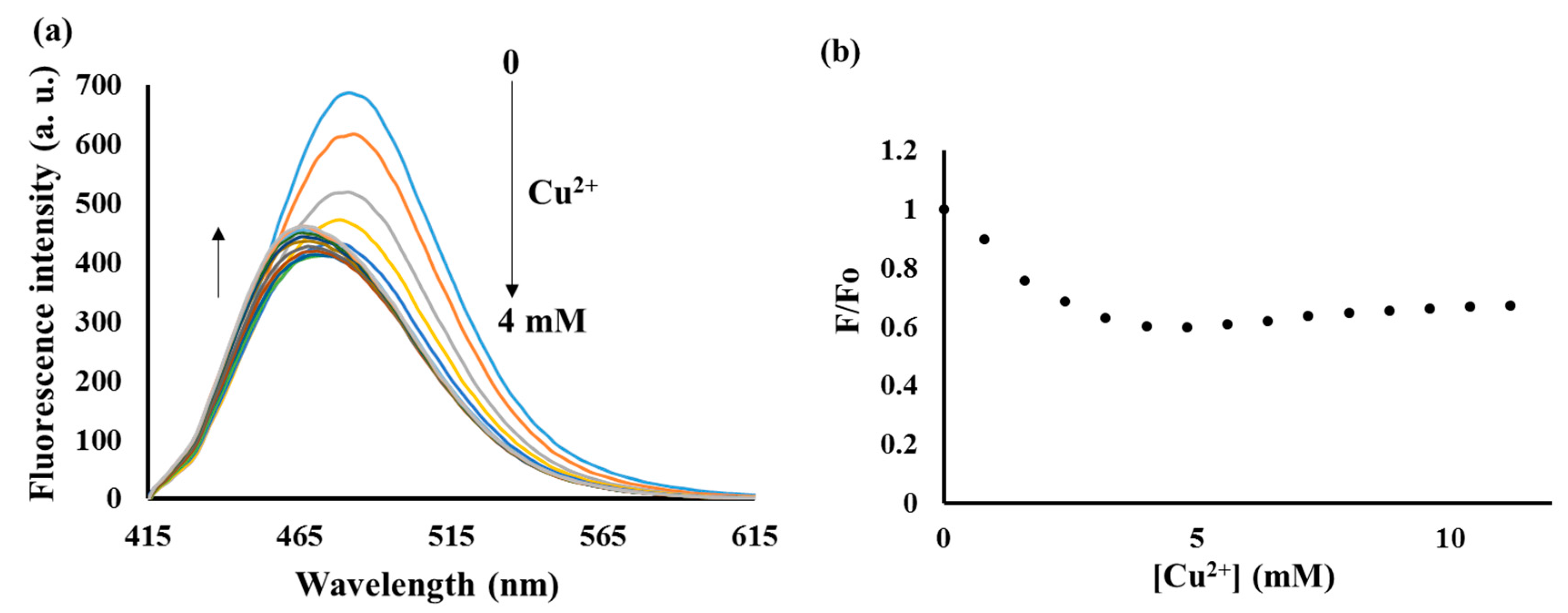
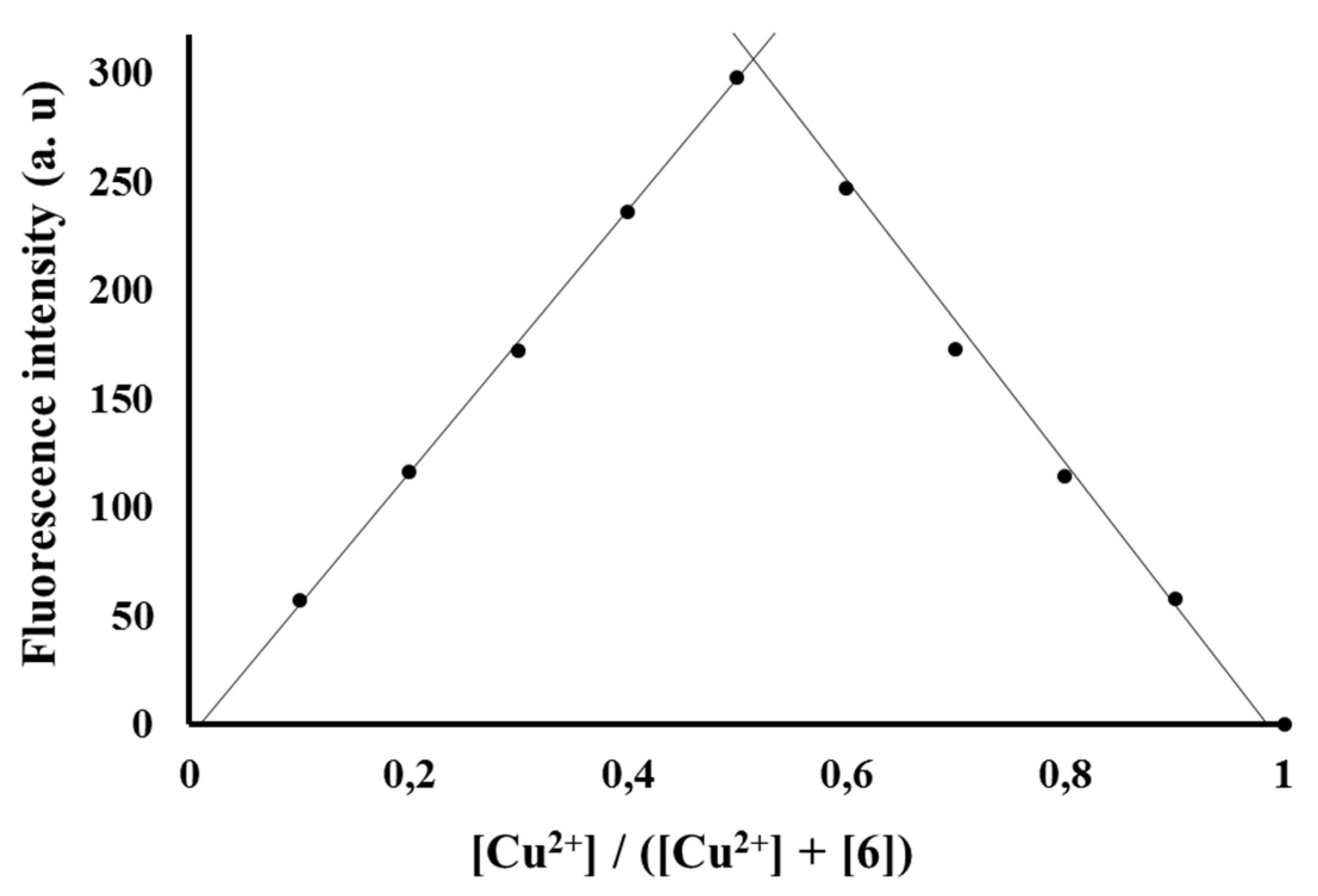
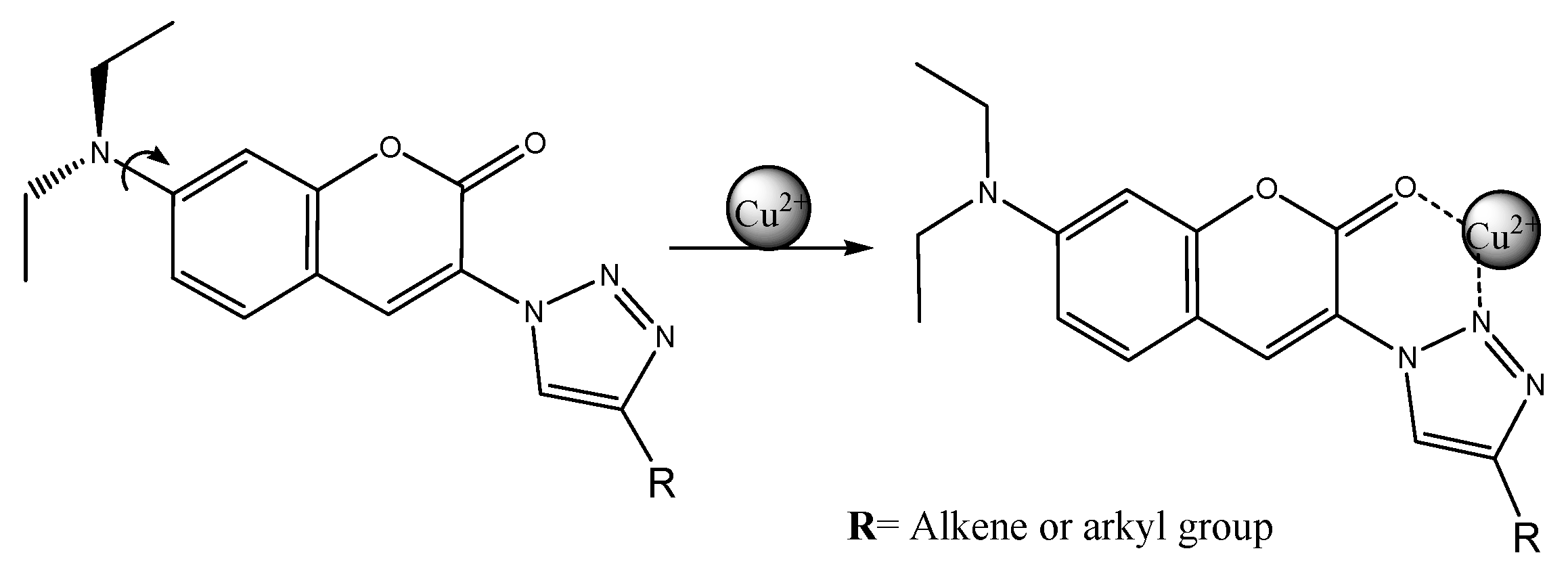
3.4. Sensing Mechanism of P1 with Cu2+
3.5. Reversibility and Response Time
3.6. Preliminary Analytical Application
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cowan, J.A. Inorganic Biochemistry: An Introduction, 2nd ed.; Wiley-VCH: New York, NY, USA, 1997; pp. 133–134. [Google Scholar]
- Linder, M.C.; Azam, M.H. Copper biochemistry and molecular biology. Am. J. Clin. Nutr. 1996, 63, 797S–811S. [Google Scholar] [PubMed]
- Multhaup, G.; Schlicksupp, A.; Hesse, L.; Beher, D.; Ruppert, T.; Masters, C.L.; Beyreuther, K. The amyloid precursor protein of Alzheimer’s disease in the reduction of copper(II) to copper(I). Science 1996, 271, 1406–1409. [Google Scholar] [CrossRef] [PubMed]
- Deraeve, C.; Boldron, C.; Maraval, A.; Mazarguil, H.; Gornitzka, H.; Vendier, L.; Pitie, M.; Meunier, B. Preparation and study of new poly-8-hydroxyquinoline chelators for an anti-Alzheimer strategy. Chem. Eur. J. 2008, 14, 682–696. [Google Scholar] [CrossRef] [PubMed]
- Bruijn, L.I.; Miller, T.M.; Cleveland, D.W. Unraveling the mechanisms involved in motor neuron degeneration in ALS. Annu. Rev. Neurosci. 2004, 27, 723–749. [Google Scholar] [CrossRef] [PubMed]
- Waldermar, M.; Ryszard, R.; Teresa, U. Effect of excess Cu on the photosynthetic apparatus of runner bean leaves treated at two different growth stages. Physiol. Plant. 1994, 91, 715–721. [Google Scholar]
- Georgopoulos, P.G.; Roy, A.; Yonone-Lioy, M.J.; Opiekun, R.E.; Lioy, P.J. Environmental copper: Its dynamics and human exposure issues. J. Toxicol. Environ. Health Part B 2001, 4, 341–394. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, A.P.S.; Firmino, M.A.; Nomura, C.S.; Rocha, F.R.P.; Oliveira, P.V.; Gaubeur, I. Peat as a natural solid-phase for copper preconcentration and determination in a multicommuted flow system coupled to flame atomic absorption spectrometry. Anal. Chim. Acta 2009, 636, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liang, P.; Guo, L. Nanometer titanium dioxide immobilized on silica gel as sorbent for preconcentration of metal ions prior to their determination by inductively coupled plasma atomic emission spectrometry. Talanta 2005, 68, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Pathirathna, P.; Yang, Y.; Forzley, K.; McElmurry, S.P.; Hashemi, P. Fast-scan deposition-stripping voltammetry at carbon-fiber microelectrodes: Real-time, subsecond, mercury free measurements of copper. Anal. Chem. 2012, 84, 6298–6302. [Google Scholar] [CrossRef] [PubMed]
- Mülazımoğlu, İ.E. Electrochemical determination of copper(II) ions at naringenin-modified glassy carbon electrode: Application in lake water sample. Desalin. Water Treat. 2012, 44, 161–167. [Google Scholar] [CrossRef]
- Wheelock, C.E. The Fluorescence of Some Coumarins1. Am. Chem. Soc. 1959, 81, 1348–1352. [Google Scholar] [CrossRef]
- Yoshimura, I.; Miyahara, Y.; Kasagi, N.; Yamane, H.; Ojida, A.; Hamachi, I. Molecular recognition in a supramolecular hydrogel to afford a semi-wet sensor chip. J. Am. Chem. Soc. 2004, 126, 12204–12205. [Google Scholar] [CrossRef] [PubMed]
- Lv, F.; Feng, X.; Tang, H.; Liu, L.; Yang, Q.; Wang, S. Development of film sensors based on conjugated polymers for copper(II) ion detection. Adv. Funct. Mat. 2011, 21, 845–850. [Google Scholar] [CrossRef]
- García, J.M.; García, F.C.; Serna, F.; de la Peña, J.L. High-performance aromatic polyamides. Prog. Polym. Sci. 2010, 35, 623–686. [Google Scholar]
- Anzenbacher, P.; Liuand, Y.; Kozelkova, M.E. Hydrophilic polymer matrices in optical array sensing. Curr. Opin. Chem. Biol. 2010, 14, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Rotello, V.; Thayumanavan, S. Molecular Recognition and Polymers: Control of Polymer Structure and Self-Assembly; Rotello, V., Thayumanavan, S., Eds.; Wiley: Hoboken, NJ, USA, 2008. [Google Scholar]
- Gu, C.; Huang, N.; Wu, Y.; Xu, H.; Jiang, D. Design of Highly Photofunctional Porous Polymer Films with Controlled Thickness and Prominent Microporosity. Angew. Chem. Int. Ed. 2015, 54, 11540–11544. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Wang, L.; Xiang, G.; Zhou, L.; An, X.; Cao, D. A colorimetric and fluorescence “turn-off” chemosensor for the detection of silver ion based on a conjugated polymer containing 2, 3-di(pyridin-2-yl) quinoxaline. Sens. Actuators B Chem. 2015, 207, 281–290. [Google Scholar] [CrossRef]
- Luo, J.; Jiang, S.; Qin, S.; Wu, H.; Wang, Y.; Jiang, J.; Liu, X. Highly sensitive and selective turn-on fluorescent chemosensor for Hg2+ in pure water based on a rhodamine containing water-soluble copolymer. Sens. Actuators B Chem. 2011, 160, 1191–1197. [Google Scholar] [CrossRef]
- Wang, B.; Liu, X.; Hu, Y.; Su, Z. Synthesis and photophysical behavior of a water-soluble coumarin-bearing polymer for proton and Ni2+ ion sensing. Polym. Int. 2009, 58, 703–709. [Google Scholar] [CrossRef]
- Guo, Z.Q.; Zhu, W.H.; Tian, H. Hydrophilic copolymer bearing dicyanomethylene-4 H-pyran moiety as fluorescent film sensor for Cu2+ and pyrophosphate anion. Macromolecules 2010, 43, 739–744. [Google Scholar] [CrossRef]
- Dong, Y.; Koken, B.; Ma, X.; Wang, L.; Cheng, Y.; Zhu, C. Polymer-based fluorescent sensor incorporating 2, 2′-bipyridyl and benzo[2,1,3]thiadiazole moieties for Cu2+ detection. Inorg. Chem. Commun. 2011, 14, 1719–1722. [Google Scholar] [CrossRef]
- Rostovtsev, V.V.; Green, L.G.; Fokin, V.V.; Sharpless, K.B. A stepwise huisgen cycloaddition process: Copper(I)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew. Chem. Int. Ed. 2002, 41, 2596–2599. [Google Scholar] [CrossRef]
- Meldal, M.; Tornøe, C.W. Cu-catalyzed azide-alkyne cycloaddition. Chem. Rev. 2008, 108, 2952–3015. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, K.; Xie, F.; Cash, B.M.; Long, S.; Barnhill, H.N.; Wang, Q. A fluorogenic 1, 3-dipolar cycloaddition reaction of 3-azidocoumarins and acetylenes. Org. Lett. 2004, 6, 4603–4606. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Liu, W.; Zhuang, X.; Wang, F.; Wang, P.; Tao, S.; Zhang, X.; Wu, S.; Lee, S. Fluorescence turn on of coumarin derivatives by metal cations: A new signaling mechanism based on C=N isomerization. Org. Lett. 2007, 9, 33–36. [Google Scholar] [CrossRef] [PubMed]
- Hasirci, V.; Yilgor, P.; Endogan, T.; Eke, G.; Hasirci, N. Polymer fundamentals: Polymer synthesis. Compr. Biomater. 2011, 1, 349–371. [Google Scholar]
- Goswami, S.; Das, A.K.; Maity, S. ‘PET’ vs. ‘push–pull’ induced ICT: A remarkable coumarinyl-appended pyrimidine based naked eye colorimetric and fluorimetric sensor for the detection of Hg2+ ions in aqueous media with test trip. Dalton Trans. 2013, 42, 16259–16263. [Google Scholar] [CrossRef] [PubMed]
- McClure, D.S. Spin-orbit interaction in aromatic molecules. J. Chem. Phys. 1952, 20, 682–686. [Google Scholar] [CrossRef]
- Masuhara, H.; Shioyama, H.; Saito, T.; Hamada, K.; Yasoshima, S.; Mataga, N. Fluorescence quenching mechanism of aromatic hydrocarbons by closed-shell heavy metal ions in aqueous and organic solutions. J. Phys. Chem. 1984, 88, 5868–5873. [Google Scholar] [CrossRef]
- Wang, R.; Wan, Q.; Feng, F.; Bai, Y. A novel coumarin-based fluorescence chemosensor for Fe3+. Chem. Res. Chin. Univ. 2014, 30, 560–565. [Google Scholar] [CrossRef]
- Hong, Y.; Lam, J.W.; Tang, B.Z. Aggregation-induced emission: Phenomenon, mechanism and applications. Chem. Commun. 2009, 29, 4332–4353. [Google Scholar] [CrossRef] [PubMed]
- Satpati, A.K.; Kumbhakar, M.; Nath, S.; Pal, H. Photophysical Properties of Coumarin-7 Dye: Role of twisted intramolecular charge transfer state in high polarity protic solvents. Photochem. Photobiol. 2009, 85, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Nag, A.; Bhattacharyya, K. Role of twisted intramolecular charge transfer in the fluorescence sensitivity of biological probes: Diethylaminocoumarin laser dyes. Chem. Phys. Lett. 1990, 169, 12–16. [Google Scholar] [CrossRef]
- Ramakrishna, G.; Ghosh, H.N. Efficient electron injection from twisted intramolecular charge transfer (TICT) state of 7-diethyl amino coumarin 3-carboxylic acid (D-1421) dye to TiO2 nanoparticle. J. Phys. Chem. A 2002, 106, 2545–2553. [Google Scholar] [CrossRef]
- Sasaki, S.; Drummen, G.P.; Konishi, G.I. Recent advances in twisted intramolecular charge transfer (TICT) fluorescence and related phenomena in materials chemistry. J. Mater. Chem. C 2016, 4, 2731–2743. [Google Scholar] [CrossRef]
- Nad, S.; Kumbhakar, M.; Pal, H. Photophysical properties of coumarin-152 and coumarin-481 dyes: Unusual behavior in nonpolar and in higher polarity solvents. J. Phys. Chem. A 2003, 107, 4808–4816. [Google Scholar] [CrossRef]
- Kikuchi, K.P.K. Design, synthesis and biological application of chemical probes for bio-imaging. Chem. Soc. Rev. 2010, 39, 2048–2053. [Google Scholar] [CrossRef] [PubMed]
- Carter, K.P.; Young, A.M.; Palmer, A.E. Fluorescent sensors for measuring metal ions in living systems. Chem. Rev. 2014, 114, 4564–4601. [Google Scholar] [CrossRef] [PubMed]
- Renny, J.S.; Tomasevich, L.L.; Tallmadge, E.H.; Collum, D.B. Method of continuous variations: Applications of job plots to the study of molecular associations in organometallic chemistry. Angew. Chem. Int. Ed. 2013, 52, 11998–12013. [Google Scholar] [CrossRef] [PubMed]
- Benesi, H.A.; Hildebrand, J.H. A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. J. Am. Chem. Soc. 1949, 71, 2703–2707. [Google Scholar] [CrossRef]
- Barra, M.; Bohne, C.; Scaiano, J.C. Effect of cyclodextrin complexation on the photochemistry of xanthone. Absolute measurement of the kinetics for triplet-state exit. J. Am. Chem. Soc. 1990, 112, 8075–8579. [Google Scholar] [CrossRef]
- Wang, L.; Ye, D.; Cao, D. A novel coumarin Schiff-base as a Ni(II) ion colorimetric sensor. Spectrochim. Acta Part A 2012, 90, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Würthner, F.; Kaiser, T.E.; Saha-Möller, C.R. J-Aggregates: From Serendipitous Discovery to Supramolecular Engineering of Functional Dye Materials. Angew. Chem. Int. Ed. 2011, 50, 3376–3410. [Google Scholar] [CrossRef] [PubMed]


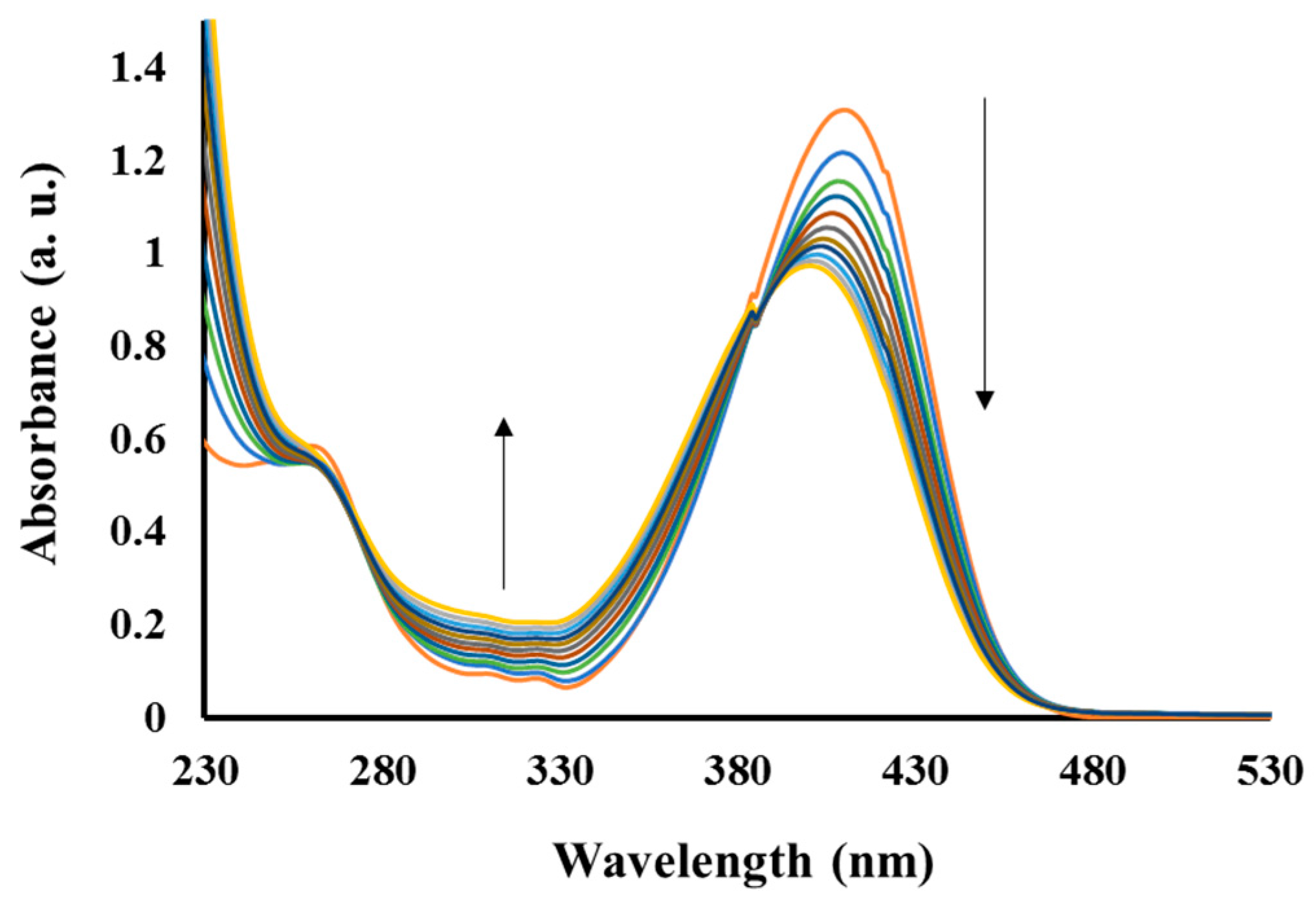
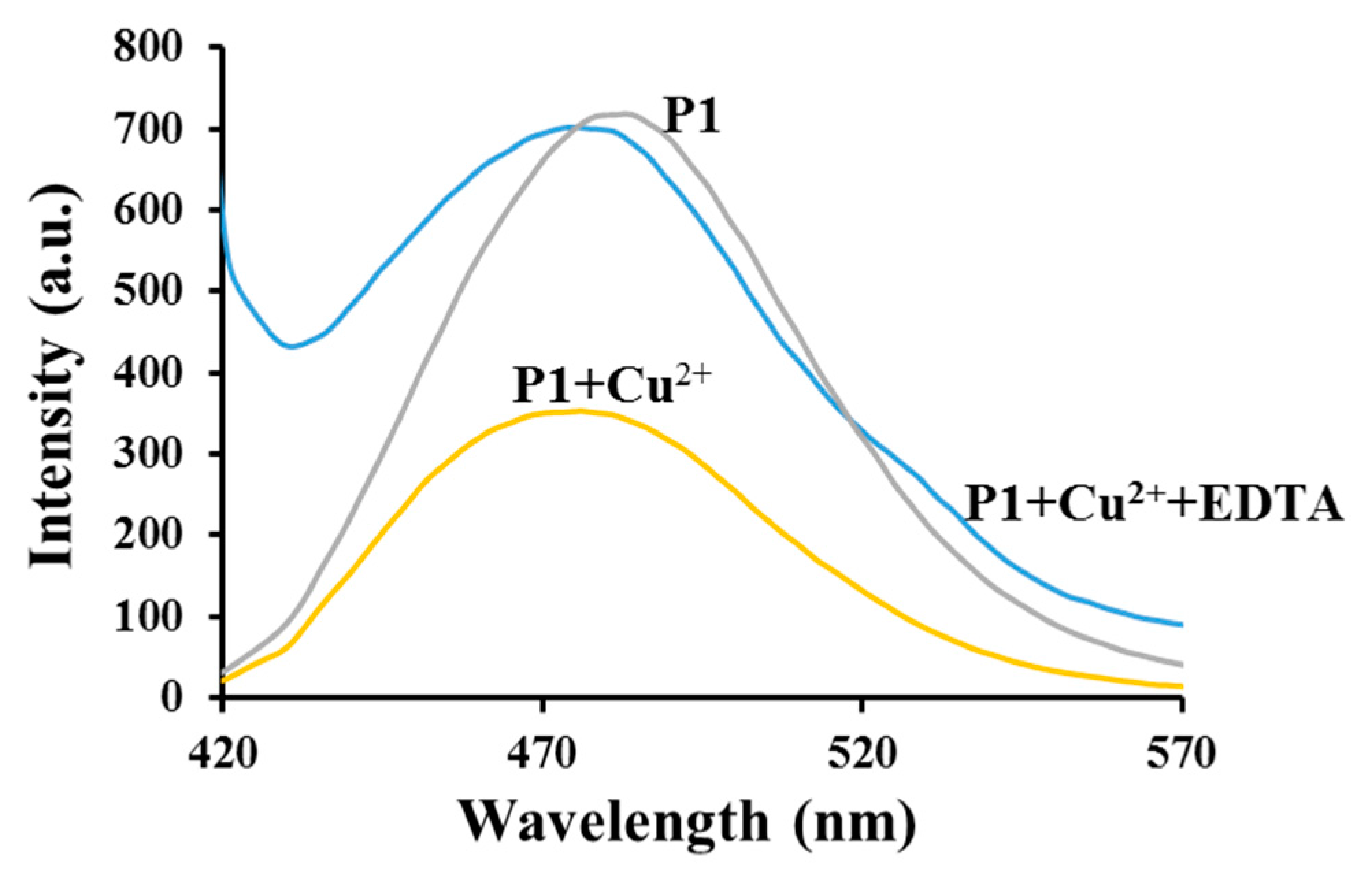
| Polymer | Mw * | Mn * | PDI * | λabs (nm) | λex (nm) | λemit (nm) | Stock Shift (nm) |
|---|---|---|---|---|---|---|---|
| P1 | 2.17 × 103 | 1.19 × 103 | 1.92 | 265 and 396 | 395 | 484 | 90 |
| Sample | 1 | 2 | 3 |
|---|---|---|---|
| Concentration (ppm) | 0.255 | 0.317 | 0.328 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ngororabanga, J.M.V.; Du Plessis, J.; Mama, N. Fluorescent Polymer Incorporating Triazolyl Coumarin Units for Cu2+ Detection via Planarization of Ict-Based Fluorophore. Sensors 2017, 17, 1980. https://doi.org/10.3390/s17091980
Ngororabanga JMV, Du Plessis J, Mama N. Fluorescent Polymer Incorporating Triazolyl Coumarin Units for Cu2+ Detection via Planarization of Ict-Based Fluorophore. Sensors. 2017; 17(9):1980. https://doi.org/10.3390/s17091980
Chicago/Turabian StyleNgororabanga, Jean Marie Vianney, Jacolien Du Plessis, and Neliswa Mama. 2017. "Fluorescent Polymer Incorporating Triazolyl Coumarin Units for Cu2+ Detection via Planarization of Ict-Based Fluorophore" Sensors 17, no. 9: 1980. https://doi.org/10.3390/s17091980




