A Non-invasive and Real-time Monitoring of the Regulation of Photosynthetic Metabolism Biosensor Based on Measurement of Delayed Fluorescence in Vivo
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and NaHSO3 Treatments
2.2. Measurement of Photosynthesis Rate (Pn)
2.3. In Vivo DF Biosensor System and in Vivo DF Measurement
2.3.1. Process of Operation
2.3.2. LED Driver
2.3.3. CO2 and Humidity Controller
2.3.4. Temperature Controller
3. Results
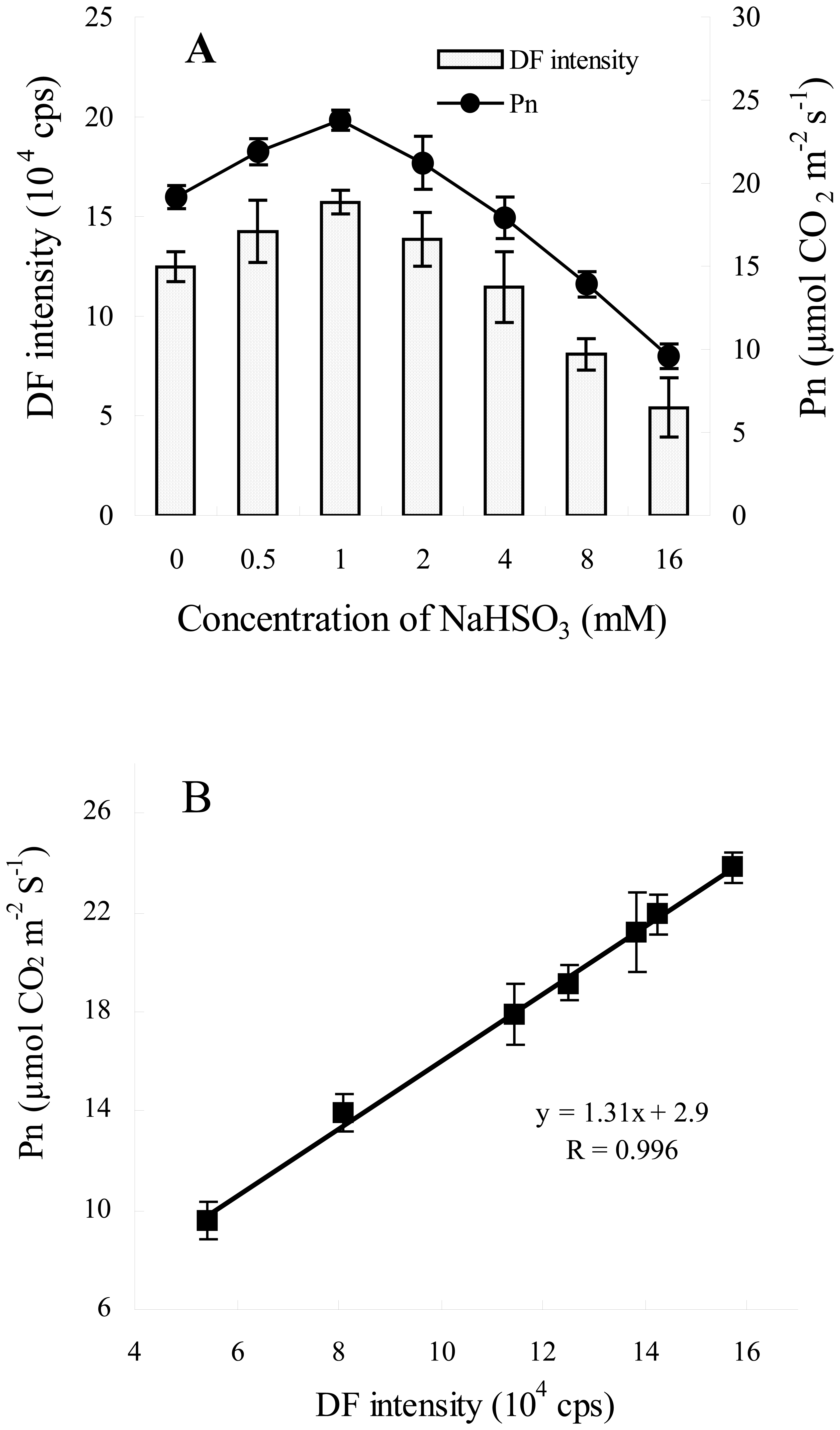
3.1. Correlation between DF Intensity and Pn after Different NaHSO3 Concentrations Treatment
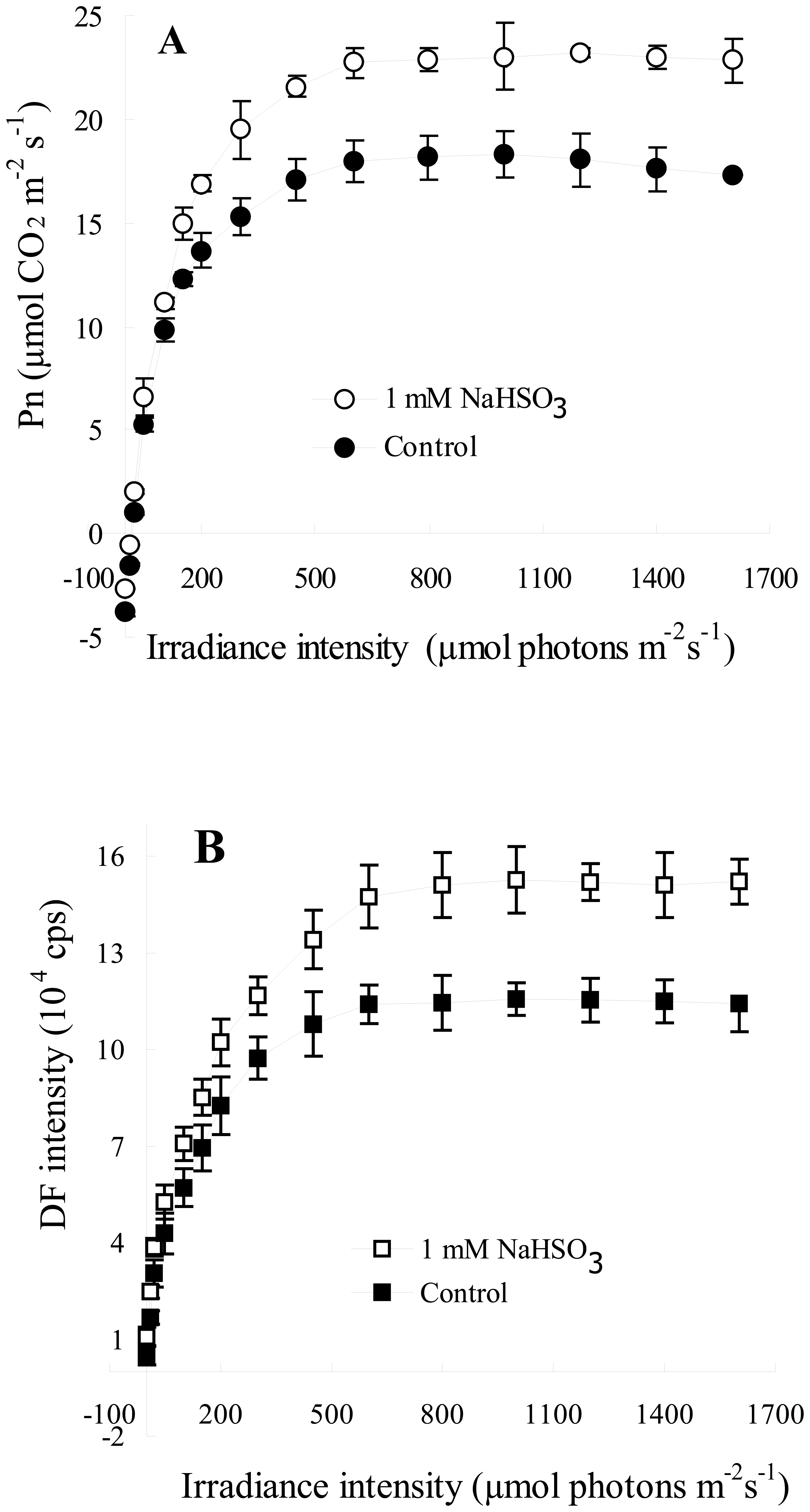
3.2. The Responses of DF Intensity and Pn to Irradiance Intensity after 1mM NaHSO3 Treatment
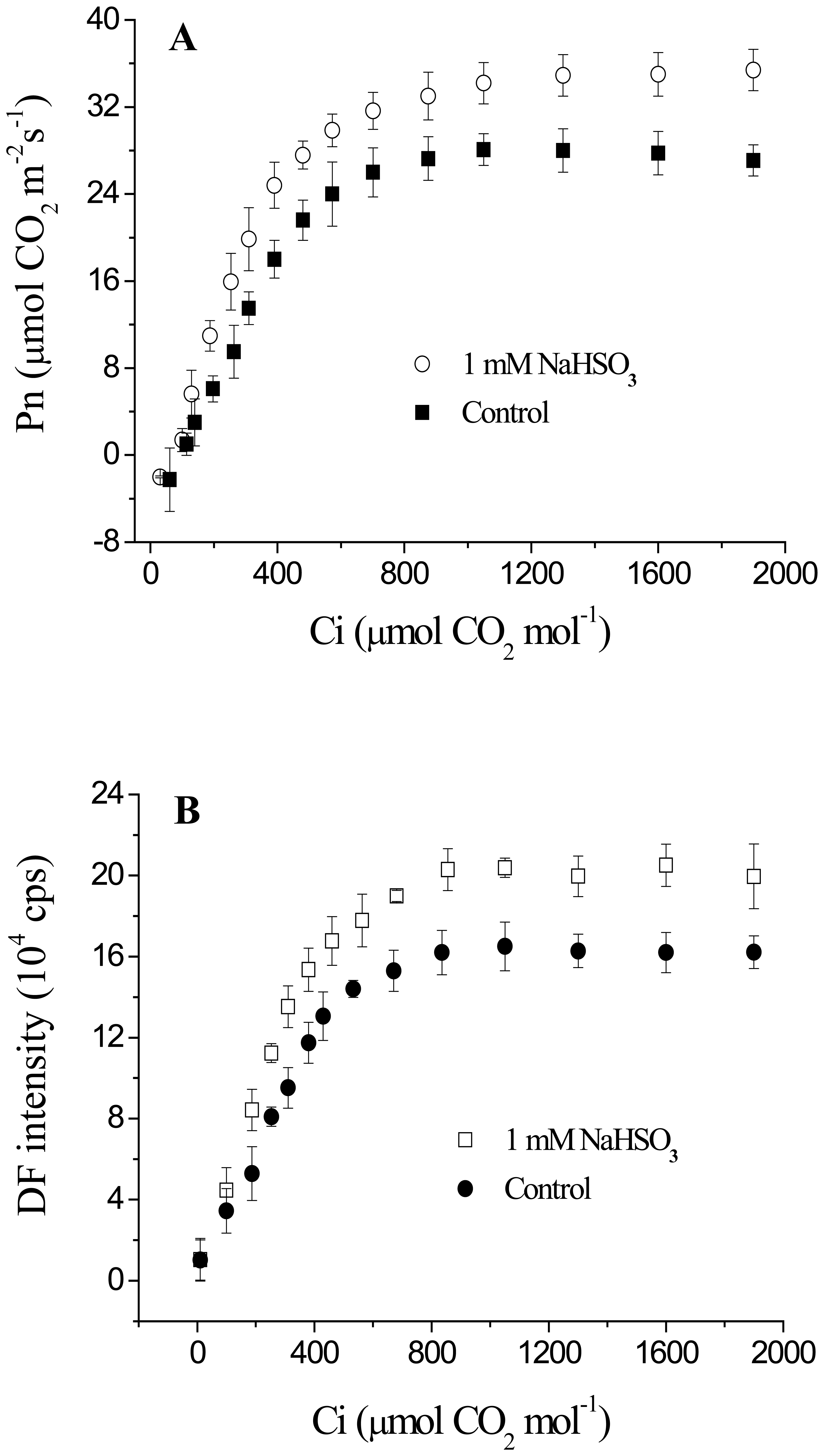
3.3. The Responses of DF Intensity and Pn to Intercellular CO2 Concentration (Ci) after 1mM NaHSO3 Treatment
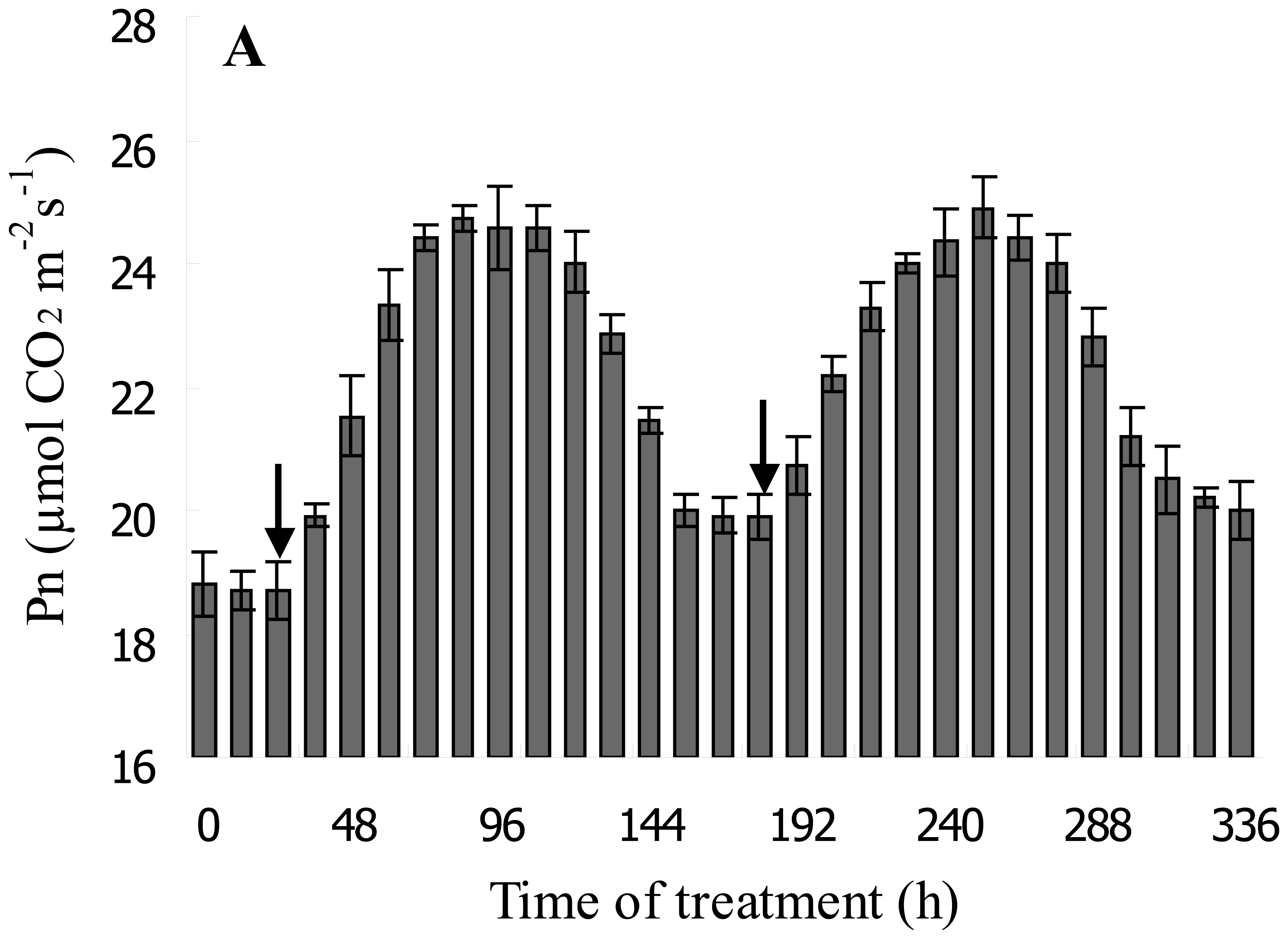
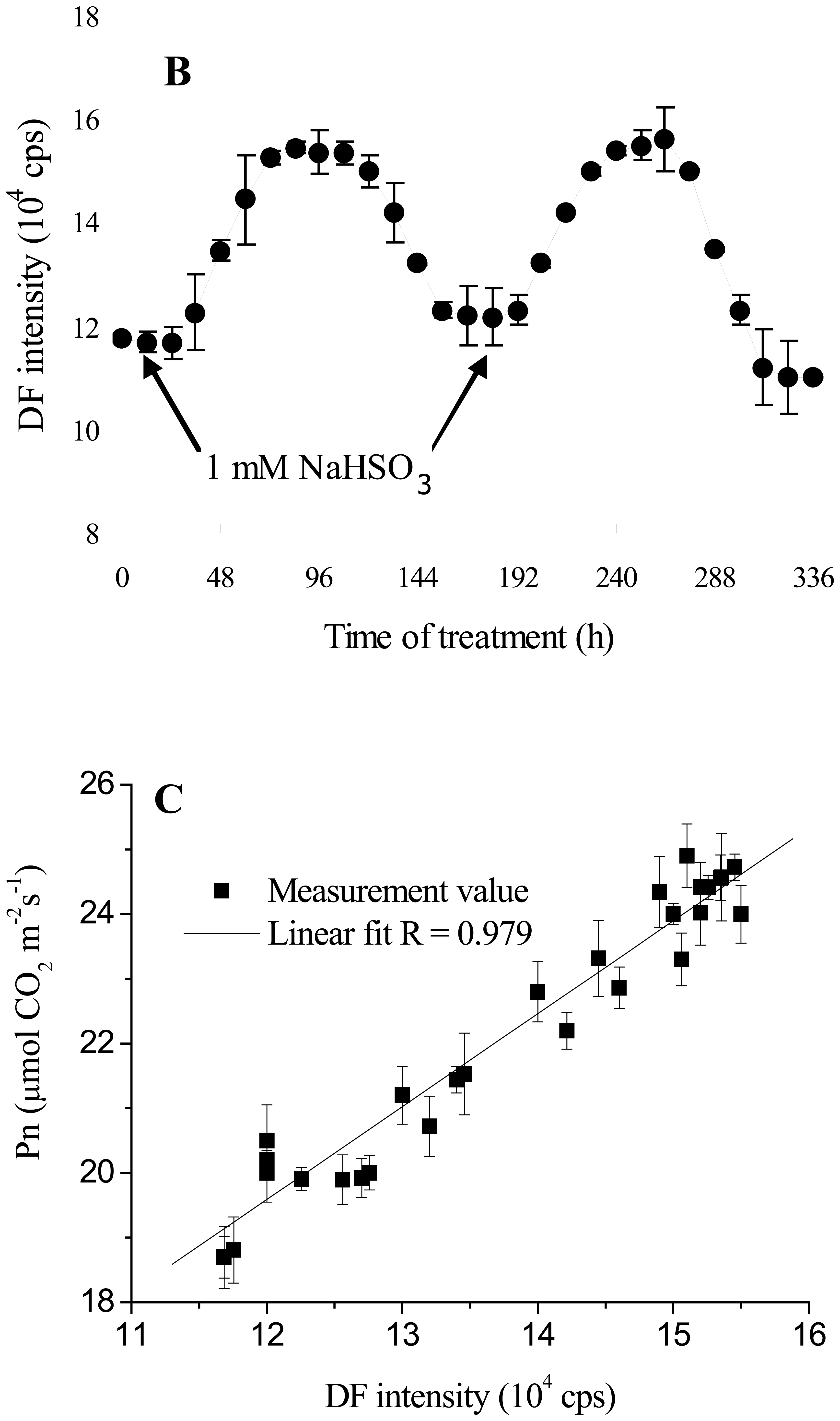
3.4. DF Monitoring the Dynamic Process of NaHSO3 on Soybean Seedling Photosynthesis
4. Discussion
5. Conclusions
Acknowledgments
References
- Zhang, X.; Zhang, L.; Dong, F. C.; Gao, J. F.; Galbraith, D. W.; Song, C. P. Hydrogen Peroxide Is Involved in Abscisic Acid-Induced Stomatal Closure in Vicia faba. Plant Physiology 2001, 126, 1438–1448. [Google Scholar]
- Long, S. P.; Zhu, X. G.; Naidu, S. L.; Ort, D. R. Can Improvement in Photosynthesis Increase Crop Yields? Plant Cell and Environment 2006, 293, 315–330. [Google Scholar]
- Wang, H. W.; Wei, J. M.; Shen, Y. G. Enhancement in Wheat Leaf Photophosphorylation and Photosynthesis by Spraying Low Concentration of NaHSO3. Chinese Science Bulletin 2000, 45, 1308–1311. [Google Scholar]
- Wang, H. W.; Wei, J. M.; Shen, Y. G.; Zhang, R. X.; Yang, T. N. Enhancement of Photophosphorylation and Photosynthesis in Rice by Low Concentrations of NaHSO3 under Field Conditions. Acta Botanica Sinica 2000, 42, 1295–1299. [Google Scholar]
- Asada, K.; Deura, R.; Kasai, Z. Effects of Sulfate Ions on Photophosphorylation by Spinach Chloroplasts. Plant Cell Physiology 1968, 9, 143–146. [Google Scholar]
- Wang, C. L.; Xing, D.; Zeng, L. Z.; Ding, C. F.; Chen, Q. Effect of Artificial Acid Rain and SO2 on Characteristics of Delayed Light Emission. Luminescence 2005, 20, 51–56. [Google Scholar]
- Barbagallo, R. P.; Oxborough, K.; Pallett, K. E.; Baker, N. R. Rapid, Noninvasive Screening for Perturbations of Metabolism and Plant Growth Using Chlorophyll Fluorescence Imaging. Plant Physiology 2003, 132, 485–493. [Google Scholar]
- Wang, C. L.; Xing, D.; Chen, Q. A Novel Method for Photosynthesis Measuring Using Chloroplasts Delayed Fluorescence. Biosensors and Bioelectronics 2004, 20, 454–459. [Google Scholar]
- Christen, G.; Steffen, R.; Renger, G. Delayed Fluorescence Emitted from Light Harvesting Complex II and Photosystem II of Higher Plants in the 100 ns–5 micros Time Domain. FEBS Letters 2000, 475, 103–106. [Google Scholar]
- Wang, C. L.; Ma, G. X.; Fan, D. W.; Xia, R. B. Spectroscopy Research on the Origin Mechanism for Light Induced Delayed Fluorescence of Chloroplast. Spectroscopy and Spectral Analysis 2005, 25, 1262–1265. [Google Scholar]
- Wang, C. L.; Xing, D.; Zeng, L. Z. Spectroscopy Research on the Mechanism for 730 nm Component Delayed Fluorescence of Chloroplast. Spectroscopy and Spectral Analysis 2006, 26, 6–10. [Google Scholar]
- Chaerle, L.; Van der straeten, D. Seeing Is Believing: Imaging to Monitor Plant Health. Biochimica et Biophysica Acta-Gene Structure and Expression 2001, 1519, 153–166. [Google Scholar]
- Badretdinov, D. Z.; Kuznetsova, S. A.; Poltev, S. V.; Kukushkin, A. K. Backward Electron Transport in Photosystem 2 Reaction Center and Temperature Dependence of Delayed Luminescence Characteristics. Bioelectrochemistry 2002, 56, 13–16. [Google Scholar]
- Anderson, J. M.; Boardman, N. K. Fractionation of the Photochemical Systems of Photosynthesis. I. Chlorophyll Contents and Photochemical Activities of Particles Isolated from Spinach Chloroplasts. Biochimica et Biophysica Acta-Gene Structure and Expression 1996, 112, 403–421. [Google Scholar]
- Biswal, A. K.; Dilnawaz, F.; David, K. A.; Ramaswamy, N. K.; Misra, A. N. Increase in the Intensity of Thermoluminescence Q-Band During Leaf Ageing Is Due to a Block in the Electron Transfer from QA to QB. Luminescence 2001, 16, 309–313. [Google Scholar]
- Badretdinov, D. Z.; Baranova, E. A.; Kukushkin, A. K. Study of Temperature Influence on Electron Transport in Higher Plants via Delayed Luminescence Method: Experiment, Theory. Bioelectrochemistry 2004, 63, 67–71. [Google Scholar]
- Harbinson, J.; Genty, B.; Baker, N. R. Relationship between the Quantum Efficiencies of Photosystems I and II in Pea Leaves. Plant Physiology 1989, 90, 1029–1034. [Google Scholar]
- Fracheboud, Y.; Ribaut, J. M.; Vargas, M.; Messmer, R.; Stamp, P. Identification of Quantitative Trait Loci for Cold-Tolerance of Photosynthesis in Maize (Zea mays L.). The Journal of Experimental Botany 2002, 53, 1967–1977. [Google Scholar]
- Zhang, L. R.; Xing, D.; Wang, J. S.; Zeng, L. Z.; Li, Q. Light-Induced Delayed Fluorescence as an Indicator for the Effects of Salt Stress on Plant Physiology. Acta Photonica Sinica 2007, 10. in press. [Google Scholar]
- Law, R. D.; Crafts-Brandner, S. J. Inhibition and Acclimation of Photosynthesis to Heat Stress Is Closely Correlated with Activation of Ribulose-1,5-Bisphosphate Carboxylase/Oxygenase. Plant Physiol 1999, 120, 173–181. [Google Scholar]
- Goltsev, V.; Zaharieva, I.; Lambrev, P.; Yordanov, I.; Strasser, R. Simultaneous Analysis of Prompt and Delayed Chlorophyll a Fluorescence in Leaves During the Iinduction Period of Dark to Light Adaptation. Journal of Theoretical Biology 2003, 225, 171–183. [Google Scholar]
- Wang, H. W.; Mi, H.; Ye, J. Y.; Deng, Y.; Shen, Y. G. Low Concentrations of NaHSO3 Increase Cyclic Photophosphorylation and Photosynthesis in Cyanobacterium Synechocystis PCC6803. Photosynthesis Research 2003, 75, 151–159. [Google Scholar]

© 2007 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Zhang, L.; Xing, D.; Wang, J. A Non-invasive and Real-time Monitoring of the Regulation of Photosynthetic Metabolism Biosensor Based on Measurement of Delayed Fluorescence in Vivo. Sensors 2007, 7, 52-66. https://doi.org/10.3390/s7010052
Zhang L, Xing D, Wang J. A Non-invasive and Real-time Monitoring of the Regulation of Photosynthetic Metabolism Biosensor Based on Measurement of Delayed Fluorescence in Vivo. Sensors. 2007; 7(1):52-66. https://doi.org/10.3390/s7010052
Chicago/Turabian StyleZhang, Lingrui, Da Xing, and Junsheng Wang. 2007. "A Non-invasive and Real-time Monitoring of the Regulation of Photosynthetic Metabolism Biosensor Based on Measurement of Delayed Fluorescence in Vivo" Sensors 7, no. 1: 52-66. https://doi.org/10.3390/s7010052



