Multi-instrumental Analysis of Tissues of Sunflower Plants Treated with Silver(I) Ions – Plants as Bioindicators of Environmental Pollution
Abstract
:1. Introduction
1.1 Silver ions and their effects on organisms
1.2 Bio-indicators
1.3 Silver ions analysis
2. Material and Methods
2.1 Chemicals and pH measurements
2.2 Plants, cultivation conditions and a sample preparation
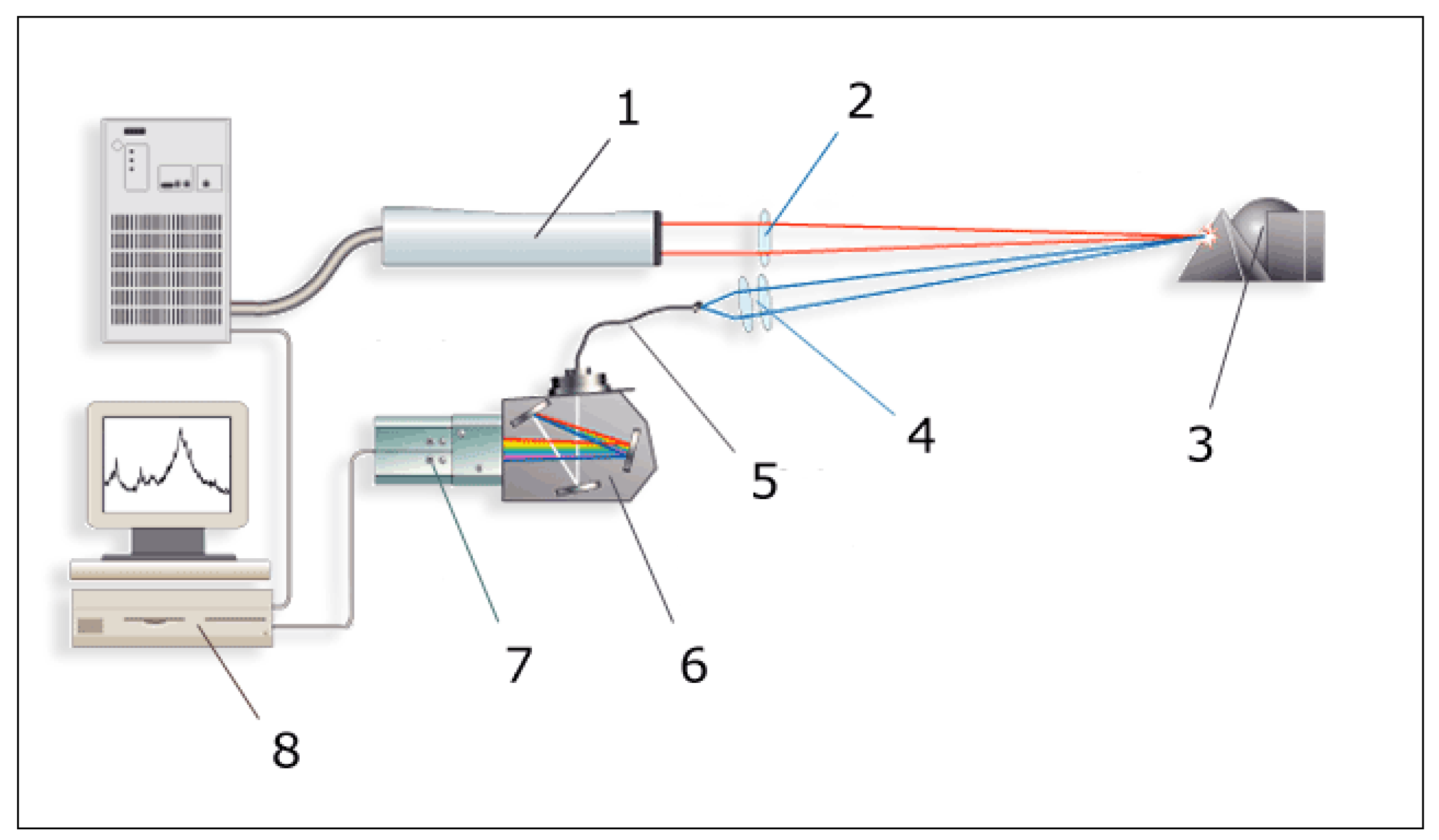
2.3 Laser spectrometry
2.4 Automated spectrometric measurements
2.4.1 Urease activity determination – An indophenol assay (Berthelot method)
2.4.2 Protein determination – Biuret test
2.5 Anatomical analysis of plant samples
2.6 Statistical analysis
3. Results and Discussion
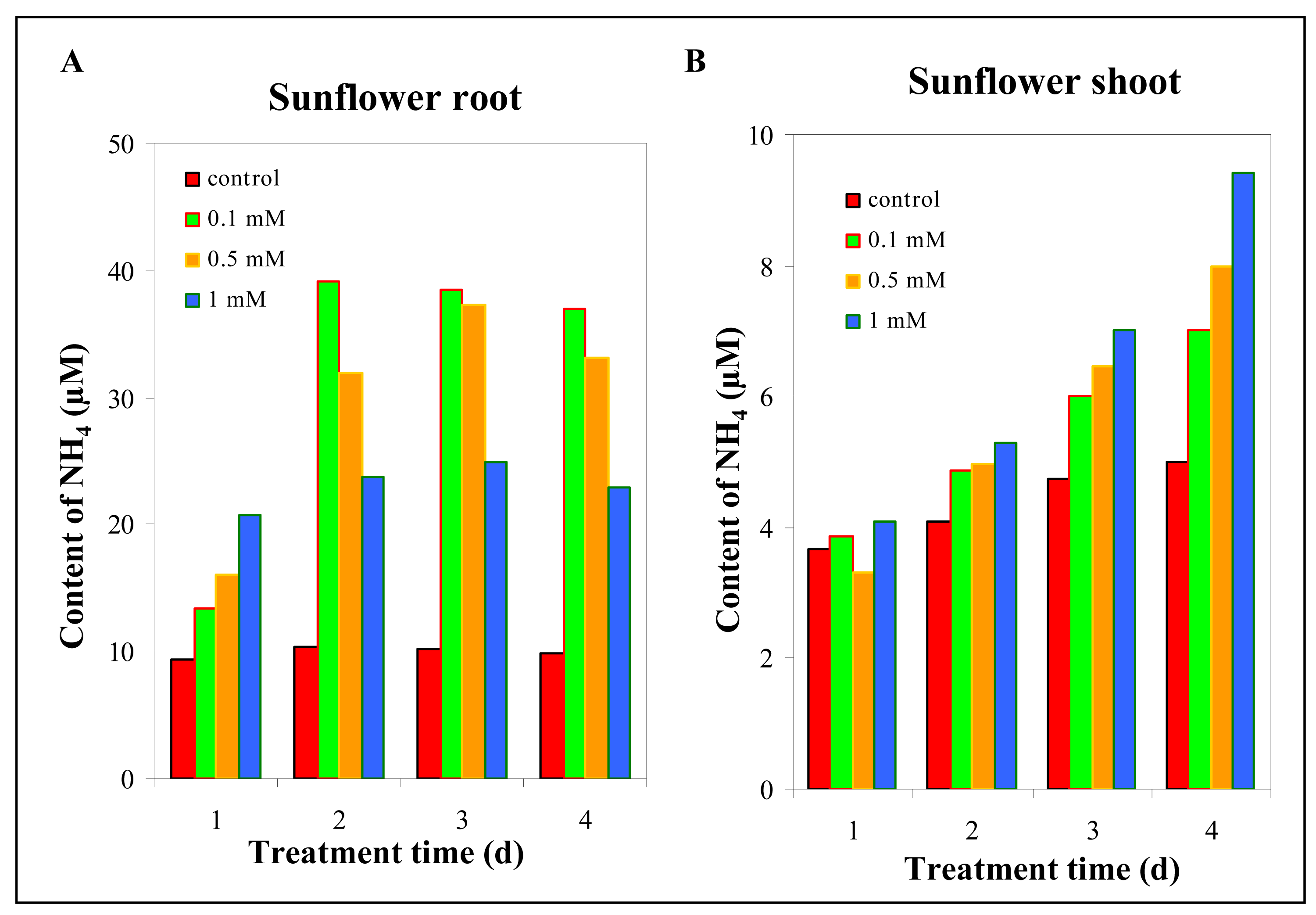
3.1 Physiological changes in sunflower plants exposed to silver(I) ions
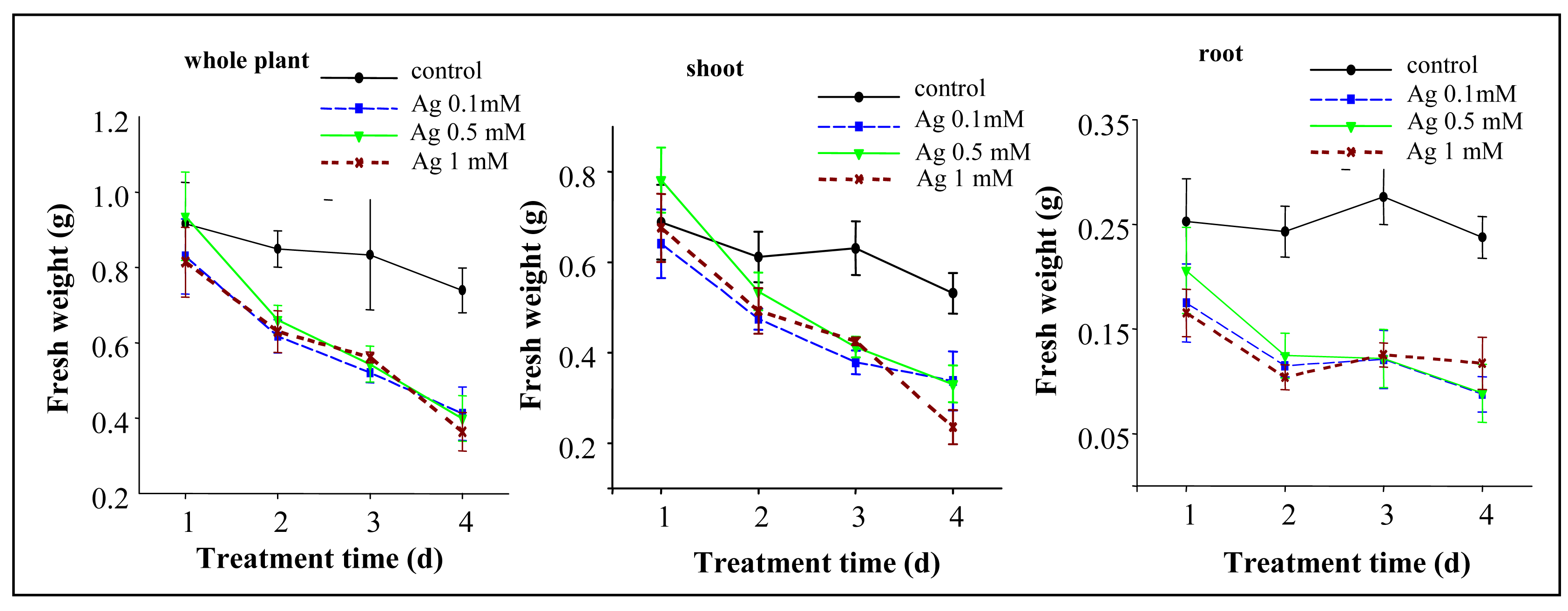
3.2 Changes in the treated plants fresh weight
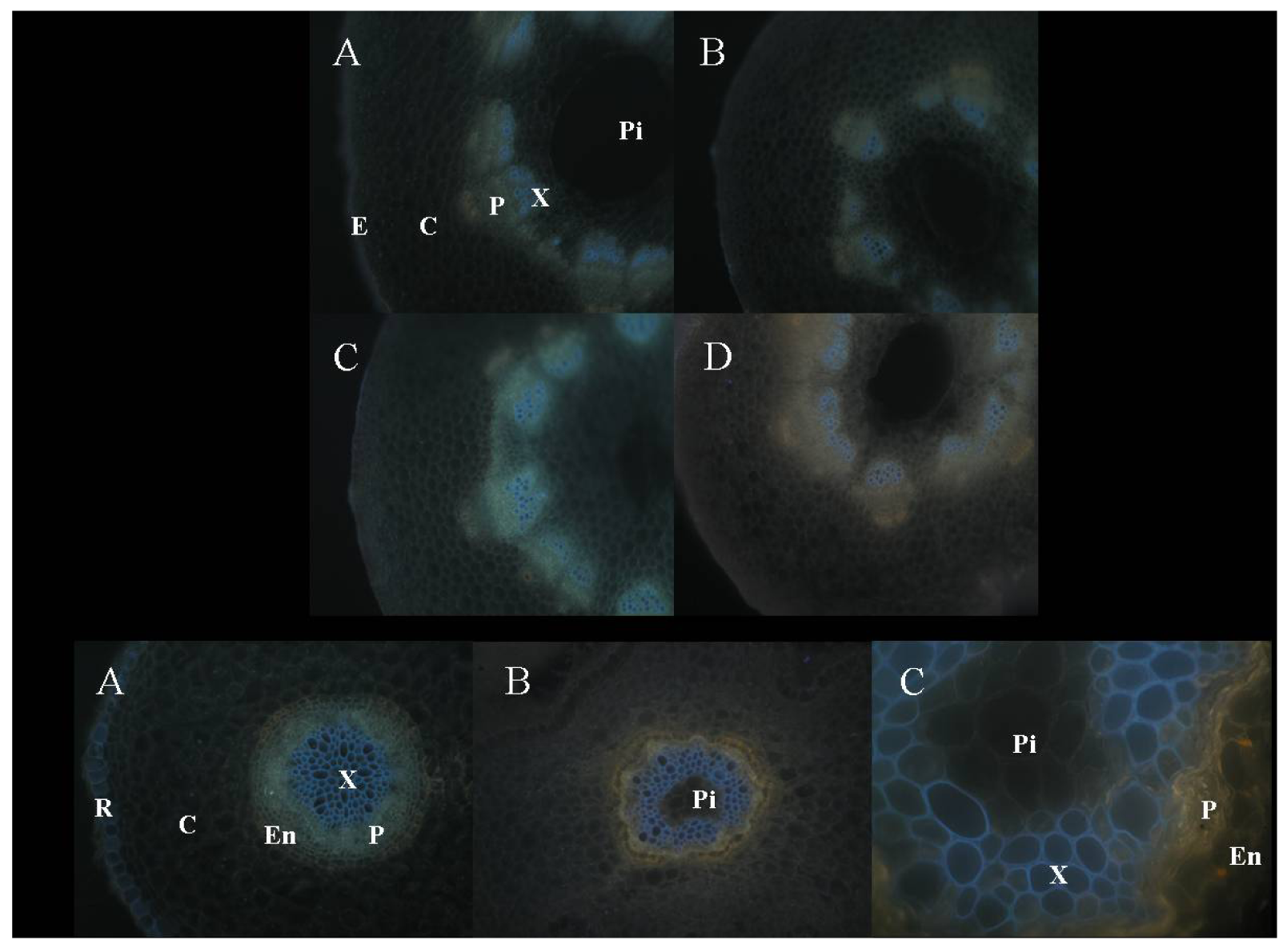
3.3 Anatomical changes of sunflower shoots (hypocotyls, stems) and roots
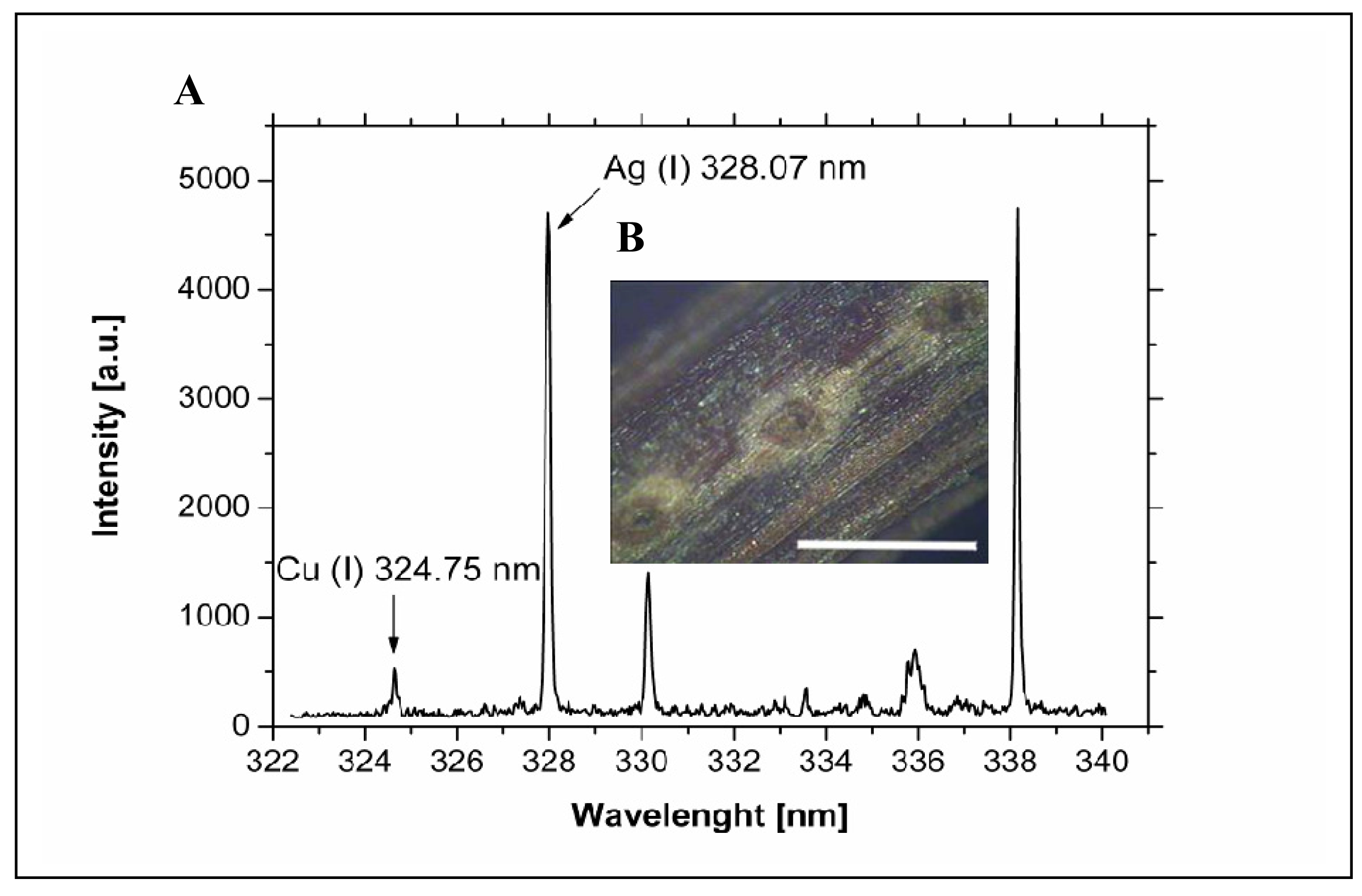
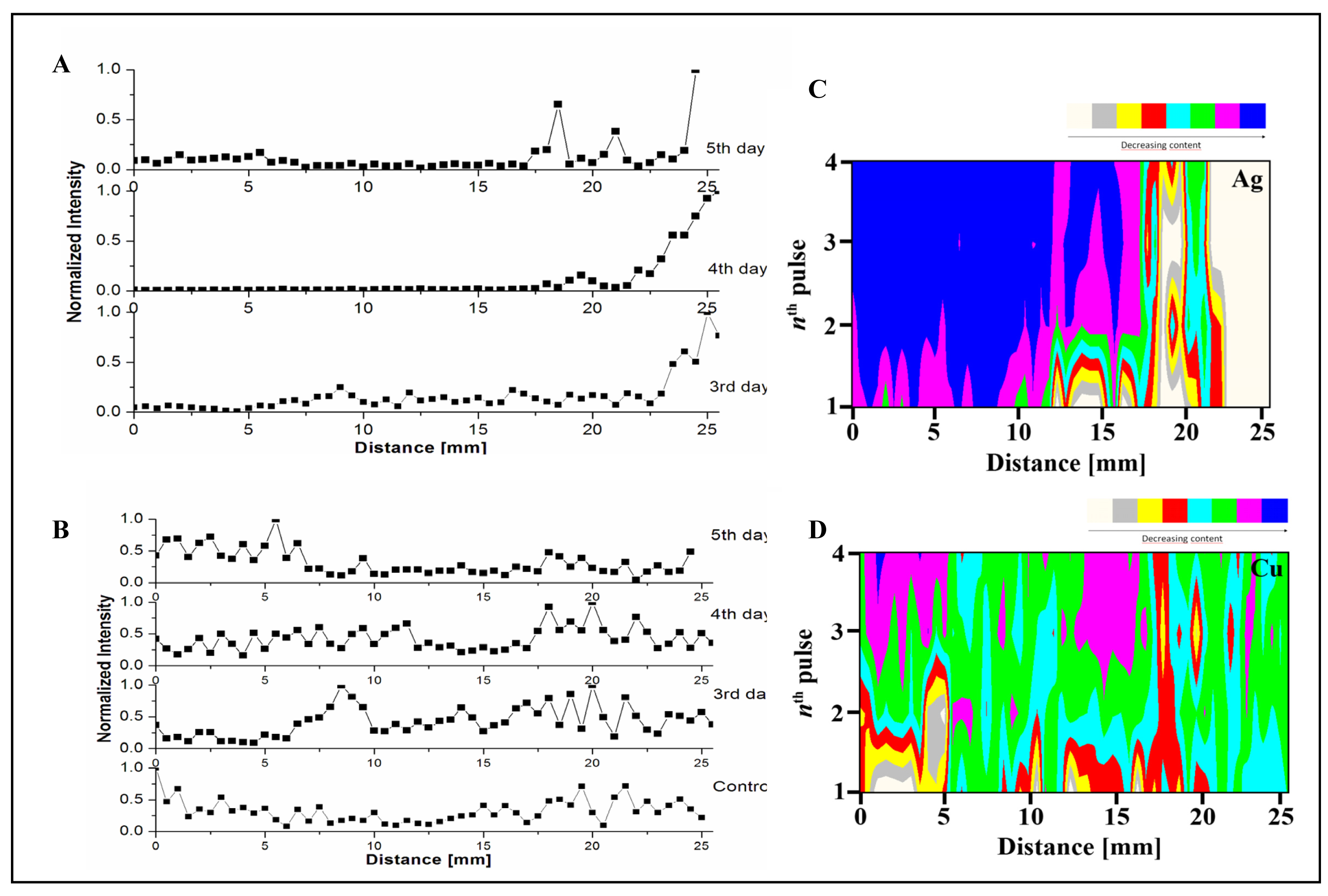
3.4 Spatial distribution of silver(I) ions in the plant tissues
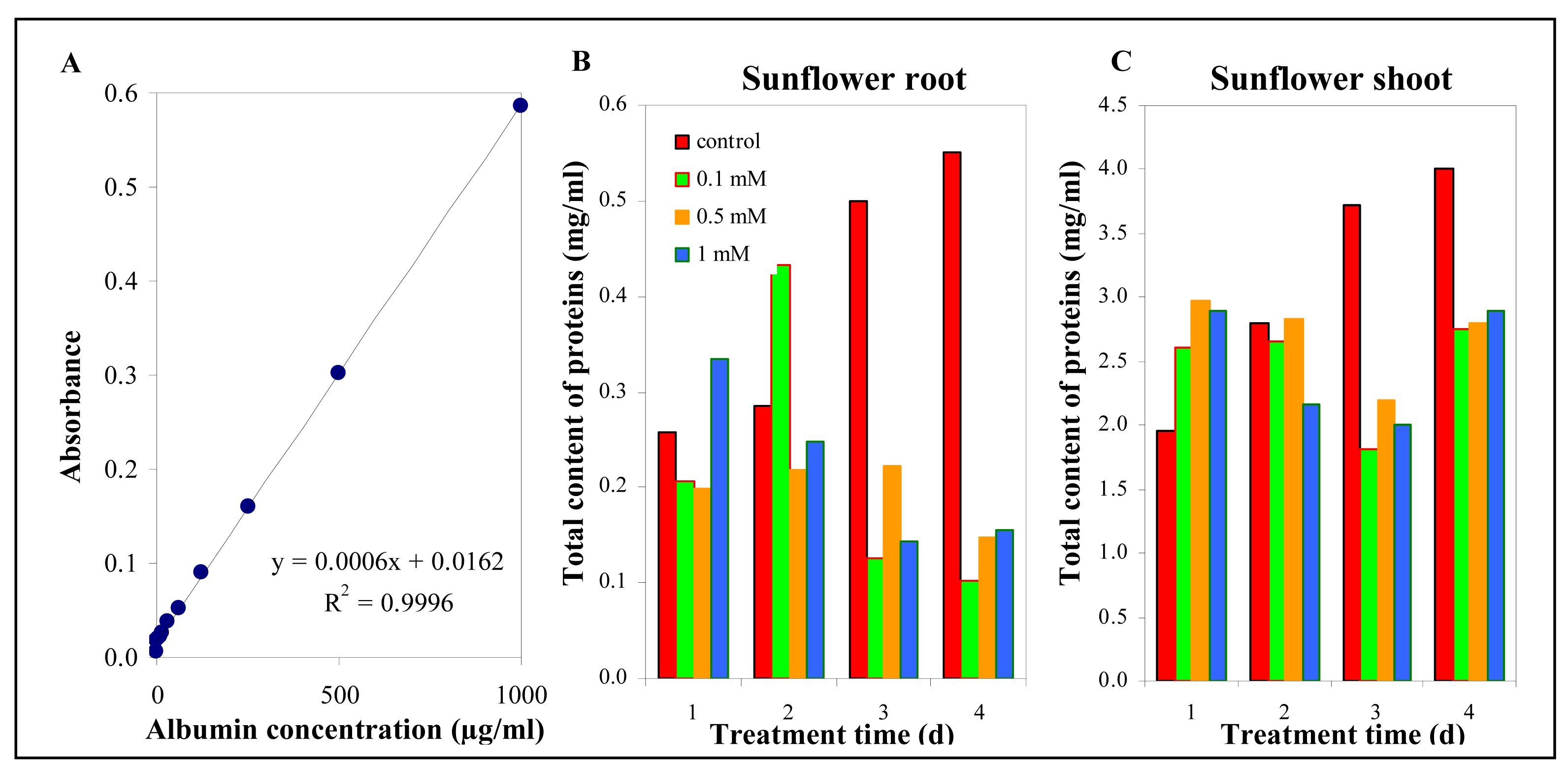
3.5.1 The total protein content
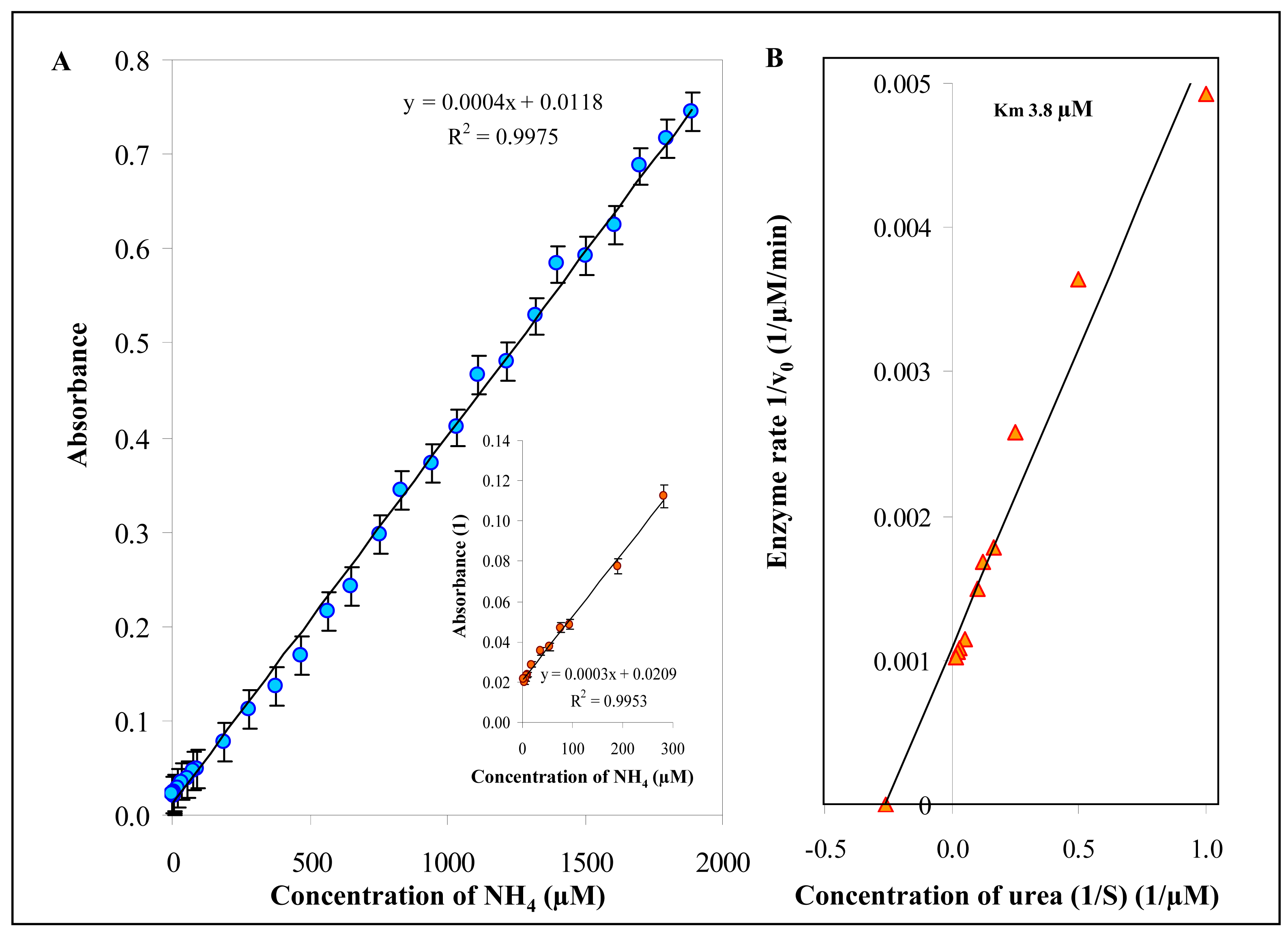
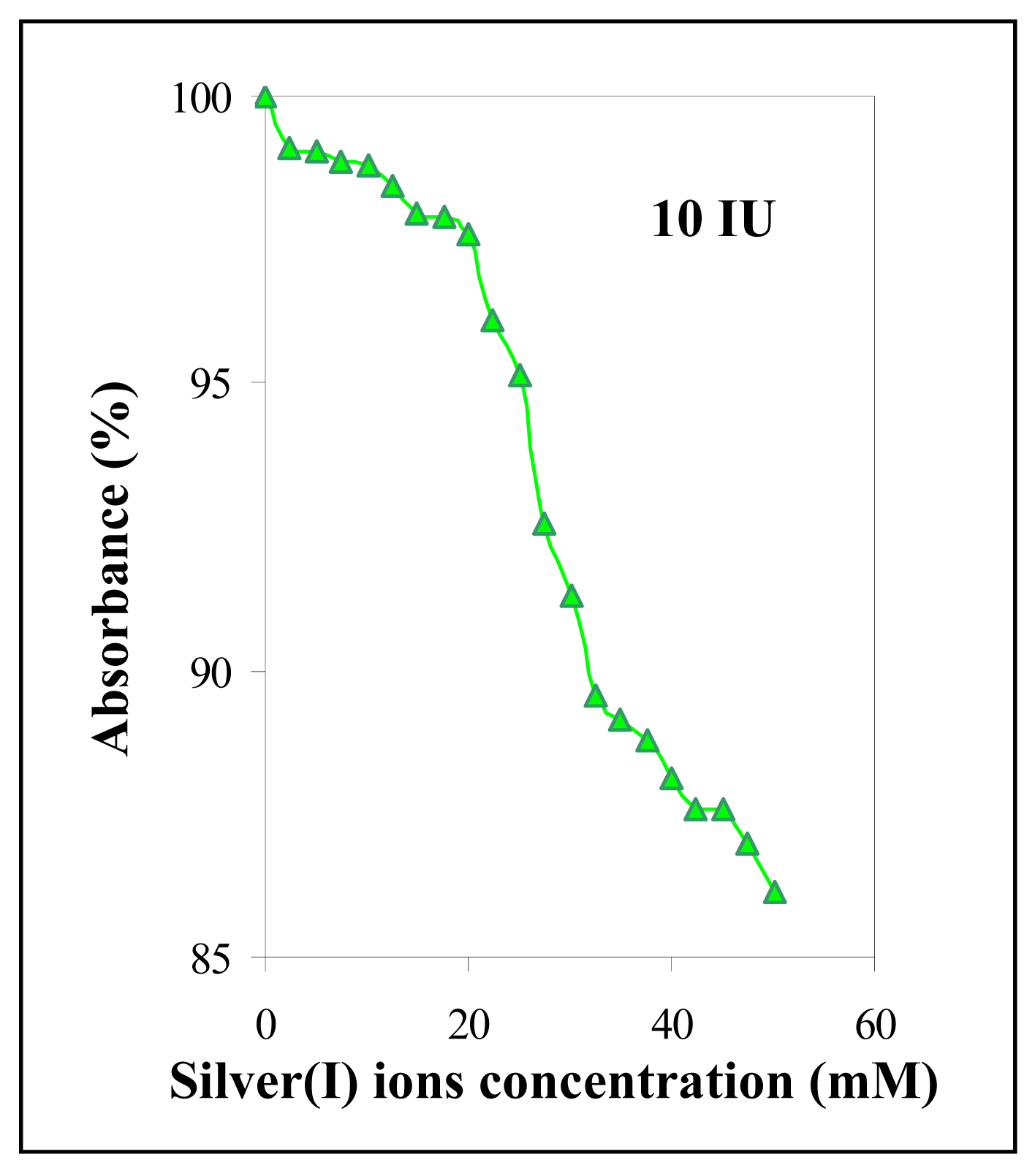
3.5.2 Urease activity
3.6 The changes of urease activity in the presence of silver(I) ions
4. Conclusion
Acknowledgments
References
- Galvez, F.; Hogstrand, C.; McGeer, J.C.; Wood, C.M. The physiological effects of a biologically incorporated silver diet on rainbow trout (Oncorhynchus mykiss). Aquat. Toxicol. 2001, 55, 95–112. [Google Scholar]
- Davis, A.; Drexler, J.W.; Ruby, M.V.; Nicholson, A. Micromineralogy of Mine Wastes in Relation to Lead Bioavailability, Butte, Montana. Environ. Sci. Technol. 1993, 27, 1415–1425. [Google Scholar]
- Hogstrand, C.; Wood, C.M. Toward a better understanding of the bioavailability, physiology and toxicity of silver in fish: Implications for water quality criteria. Environ. Toxicol. Chem. 1998, 17, 547–561. [Google Scholar]
- Mann, R.M.; Ernste, M.J.; Bell, R.A.; Kramer, J.R.; Wood, C.M. Evaluation of the protective effects of reactive sulfide on the acute toxicity of silver to rainbow trout (Oncorhynchus mykiss). Environ. Toxicol. Chem. 2004, 23, 1204–1210. [Google Scholar]
- Hogstrand, C.; Wood, C.M. Toward a better understanding of the bioavailability, physiology and toxicity of silver in fish: Implications for water quality criteria. Environ. Toxicol. Chem. 1998, 17, 547–561. [Google Scholar]
- Gorsuch, J.W.; Klaine, S.J. Toxicity and fate of silver in the environment. Environ. Toxicol. Chem. 1998, 17, 537–538. [Google Scholar]
- Song, Y.M.; Lu, X.L.; Yang, M.L.; Zheng, X.R. Study on the interaction of platinum(IV), gold(III) and silver(I) ions with DNA. Transit. Met. Chem. 2005, 30, 499–502. [Google Scholar]
- Hossain, Z.; Huq, F. Studies on the interaction between Ag+ and DNA. J. Inorg. Biochem. 2002, 91, 398–404. [Google Scholar]
- Clemens, S. Toxic metal accumulation, responses to exposure and mechanisms of tolerance in plants. Biochimie 2006, 88, 1707–1719. [Google Scholar]
- Zenk, M.H. Heavy metal detoxification in higher plants - A review. Gene 1996, 179, 21–30. [Google Scholar]
- Hamer, D.H. Metallothionein. Annu. Rev. Biochem. 1986, 55, 913–951. [Google Scholar]
- Giordani, T.; Natali, L.; Maserti, B.E.; Taddei, S.; Cavallini, A. Characterization and expression of DNA sequences encoding putative type-II metallothioneins in the seagrass Posidonia oceanica. Plant Physiol. 2000, 123, 1571–1581. [Google Scholar]
- Sochova, I.; Hofman, J.; Holoubek, I. Using nematodes in soil ecotoxicology. Environ. Int. 2006, 32, 374–383. [Google Scholar]
- Fichez, R.; Adjeroud, M.; Bozec, Y.M.; Breau, L.; Chancerelle, Y.; Chevillon, C.; Douillet, P.; Fernandez, J.M.; Frouin, P.; Kulbicki, M.; Moreton, B.; Ouillon, S.; Payri, C.; Perez, T.; Sasal, P.; Thebault, J. A review of selected indicators of particle, nutrient and metal inputs in coral reef lagoon systems. Aquat. Living Resour. 2005, 18, 125–147. [Google Scholar]
- Bebianno, M.J.; Geret, F.; Hoarau, P.; Serafim, M.A.; Coelho, M.R.; Gnassia-Barelli, M.; Romeo, M. Biomarkers in Ruditapes decussatus: a potential bioindicator species. Biomarkers 2004, 9, 305–330. [Google Scholar]
- Conti, M.E.; Cecchetti, G. Biological monitoring: lichens as bioindicators of air pollution assessment - a review. Environ. Pollut. 2001, 114, 471–492. [Google Scholar]
- McGeoch, M.A. The selection, testing and application of terrestrial insects as bioindicators. Biol. Rev. Cambridge Philosophic. Soc. 1998, 73, 181–201. [Google Scholar]
- Leyval, C.; Turnau, K.; Haselwandter, K. Effect of heavy metal pollution on mycorrhizal colonization and function: physiological, ecological and applied aspects. Mycorrhiza 1997, 7, 139–153. [Google Scholar]
- Petrlova, J.; Krizkova, S.; Zitka, O.; Hubalek, J.; Prusa, R.; Adam, V.; Wang, J.; Beklova, M.; Sures, B.; Kizek, R. Utilizing a chronopotentiometric sensor technique for metallothionein determination in fish tissues and their host parasites. Sens. Actuator B-Chem. 2007, 127, 112–119. [Google Scholar]
- Lessire, F.; Delaunois, A.; Gustin, P.; Ansay, M. Biomarkers and bioindicators in vertebrates: importance in evaluation of quality of an ecosystem. Ann. Med. Vet. 1997, 141, 281–290. [Google Scholar]
- Kafka, Z.; Puncocharova, J. Bioindicators in the environment monitoring. Chem. Listy 2000, 94, 909–912. [Google Scholar]
- Wyrobek, A.J.; Schmid, T.E.; Marchetti, F. Cross-species sperm-FISH assays for chemical testing and assessing paternal risk for chromosomally abnormal pregnancies. Environ. Mol. Mutagen. 2005, 45, 271–283. [Google Scholar]
- Sures, B. Environmental parasitology: relevancy of parasites in monitoring environmental pollution. Trends Parasitol. 2004, 20, 170–177. [Google Scholar]
- Purcell, T.W.; Peters, J.J. Sources of silver in the environment. Environ. Toxicol. Chem. 1998, 17, 539–546. [Google Scholar]
- Purcell, T.W.; Peters, J.J. Historical impacts of environmental regulation of silver. Environ. Toxicol. Chem. 1999, 18, 3–8. [Google Scholar]
- Saeki, S.; Kubota, M.; Asami, T. Determination of silver in plants by flame atomic absorption spectrometry. Int. J. Environ. Anal. Chem. 1996, 64, 179–183. [Google Scholar]
- Shamspur, T.; Mashhadizadeh, M.H.; Sheikhshoaie, I. Flame atomic absorption spectrometric determination of silver ion after preconcentration on octadecyl silica membrane disk modified with bis[5-((4-nitrophenyl)azosalicylaldehyde)] as a new Schiff base ligand. J. Anal. At. Spectrom. 2003, 18, 1407–1410. [Google Scholar]
- Raoof, J.B.; Ojani, R.; Kiani, A. Kinetic determination of silver ion by its perturbation on Belousov-Zhabotinskii oscillating chemical reaction using potentiometric method. Anal. Sci. 2004, 20, 883–886. [Google Scholar]
- Safavi, A.; Iranpoor, N.; Saghir, N. Directly silica bonded analytical reagents: synthesis of 2-mercaptobenzothiazole-silica gel and its application as a new sorbent for preconcentration and determination of silver ion using solid-phase extraction method. Sep. Purif. Technol. 2004, 40, 303–308. [Google Scholar]
- Schildkraut, D.E.; Dao, P.T.; Twist, J.P.; Davis, A.T.; Robillard, K.A. Determination of silver ions at sub microgram-per-liter levels using anodic square-wave stripping voltammetry. Environ. Toxicol. Chem. 1998, 17, 642–649. [Google Scholar]
- Zhang, S.B.; Zhang, X.J.; Lin, X.Q. An ethylenediaminetetraacetic acid modified carbon paste electrode for the determination of silver ion. Chin. J. Anal. Chem. 2002, 30, 745–747. [Google Scholar]
- Guo, S.X.; Khoo, S.B. Highly selective and sensitive determination of silver(I) at a poly(8-mercaptoquinoline) film modified glassy carbon electrode. Electroanalysis 1999, 11, 891–898. [Google Scholar]
- Ye, X.Z.; Yang, Q.H.; Wang, Y.; Li, N.Q. Electrochemical behaviour of gold, silver, platinum and palladium on the glassy carbon electrode modified by chitosan and its application. Talanta 1998, 47, 1099–1106. [Google Scholar]
- Wang, J.; Lu, J.M.; Farias, P.A.M. Remote electrochemical monitoring of trace silver. Anal. Chim. Acta 1996, 318, 151–157. [Google Scholar]
- Mikelova, R.; Baloun, J.; Petrlova, J.; Adam, V.; Havel, L.; Petrek, J.; Horna, A.; Kizek, R. Electrochemical determination of Ag-ions in environment waters and their action on plant embryos. Bioelectrochemistry 2007, 70, 508–518. [Google Scholar]
- Stejskal, K.; Krizkova, S.; Adam, V.; Sures, B.; Trnkova, L.; Zehnalek, J.; Hubalek, J.; Beklova, M.; Hanustiak, P.; Svobodova, Z.; Horna, A.; Kizek, R. Bio-assessing of environmental pollution via monitoring of metallothionein level using electrochemical detection. IEEE Sens. J. 2007, in press. [Google Scholar]
- Svancara, I.; Ogorevc, B.; Hocevar, S.B.; Vytras, K. Perspectives of carbon paste electrodes in stripping potentiometry. Anal. Sci. 2002, 18, 301–305. [Google Scholar]
- Svancara, I.; Ogorevc, B.; Novic, M.; Vytras, K. Simple and rapid determination of iodide in table salt by stripping potentiometry at a carbon-paste electrode. Anal. Bioanal. Chem. 2002, 372, 795–800. [Google Scholar]
- Barek, J.; Muck, A.; Wang, J.; Zima, J. Study of voltammetric determination of carcinogenic 1-nitropyrene and 1-aminopyrene using a glassy carbon paste electrode. Sensors 2004, 4, 47–57. [Google Scholar]
- Svancara, I.; Kalcher, K.; Diewald, W.; Vytras, K. Voltammetric determination of silver at ultratrace levels using a carbon paste electrode with improved surface characteristics. Electroanalysis 1996, 8, 336–342. [Google Scholar]
- Svancara, I.; Vytras, K.; Barek, J.; Zima, J. Carbon paste electrodes in modern electroanalysis. Crit. Rev. Anal. Chem. 2001, 31, 311–345. [Google Scholar]
- Lawrence, N.S.; Deo, R.P.; Wang, J. Biocatalytic carbon paste sensors based on a mediator pasting liquid. Anal. Chem. 2004, 76, 3735–3739. [Google Scholar]
- Blaedel, W.J.; Wang, J. Mixed Immobilized Enzyme-Porous Electrode Reactor. Anal. Chem. 1980, 52, 1426–1429. [Google Scholar]
- Kizek, R.; Vacek, J.; Trnkova, L.; Klejdus, B.; Kuban, V. Electrochemical biosensors in agricultural and environmental analysis. Chem. Listy 2003, 97, 1003–1006. [Google Scholar]
- Masarik, M.; Kizek, R.; Kramer, K.J.; Billova, S.; Brazdova, M.; Vacek, J.; Bailey, M.; Jelen, F.; Howard, J.A. Application of avidin-biotin technology and adsorptive transfer stripping square-wave voltammetry for detection of DNA hybridization and avidin in transgenic avidin maize. Anal. Chem. 2003, 75, 2663–2669. [Google Scholar]
- Petrlova, J.; Masarik, M.; Potesil, D.; Adam, V.; Trnkova, L.; Kizek, R. Zeptomole detection of streptavidin using carbon paste electrode and square wave voltammetry. Electroanalysis 2007, 19, 1177–1182. [Google Scholar]
- Berry, W.J.; Cantwell, M.G.; Edwards, P.A.; Serbst, J.R.; Hansen, D.J. Predicting toxicity of sediments spiked with silver. Environ. Toxicol. Chem. 1999, 18, 40–48. [Google Scholar]
- Bury, N.R.; McGeer, J.C.; Wood, C.M. Effects of altering freshwater chemistry on physiological responses of rainbow trout to silver exposure. Environ. Toxicol. Chem. 1999, 18, 49–55. [Google Scholar]
- Bury, N.R.; Galvez, F.; Wood, C.M. Effects of chloride, calcium, and dissolved organic carbon on silver toxicity: Comparison between rainbow trout and fathead minnows. Environ. Toxicol. Chem. 1999, 18, 56–62. [Google Scholar]
- Bianchini, A.; Wood, C.M. Mechanism of acute silver toxicity in Daphnia magna. Environ. Toxicol. Chem. 2003, 22, 1361–1367. [Google Scholar]
- Bianchini, A.; Bowles, K.C.; Brauner, C.J.; Gorsuch, J.W.; Kramer, J.R.; Wood, C.M. Evaluation of the effect of reactive sulfide on the acute toxicity of silver (I) to Daphnia magna. part 2: Toxicity results. Environ. Toxicol. Chem. 2002, 21, 1294–1300. [Google Scholar]
- Bianchini, A.; Grosell, M.; Gregory, S.M.; Wood, C.M. Acute silver toxicity in aquatic animals is a function of sodium uptake rate. Environ. Sci. Technol. 2002, 36, 1763–1766. [Google Scholar]
- Call, D.J.; Polkinghorne, C.N.; Markee, T.P.; Brooke, L.T.; Geiger, D.L.; Gorsuch, J.W.; Robillard, K.A. Silver toxicity to Chironomus tentans in two freshwater sediments. Environ. Toxicol. Chem. 1999, 18, 30–39. [Google Scholar]
- Karen, D.J.; Ownby, D.R.; Forsythe, B.L.; Bills, T.P.; La Point, T.W.; Cobb, G.B.; Klaine, S.J. Influence of water quality on silver toxicity to rainbow trout (Oncorhynchus mykiss), fathead minnows (Pimephales promelas), and water fleas (Daphnia magna). Environ. Toxicol. Chem. 1999, 18, 63–70. [Google Scholar]
- Morgan, T.P.; Wood, C.M. A relationship between gill silver accumulation and acute silver toxicity in the freshwater rainbow trout: Support for the acute silver biotic ligand model. Environ. Toxicol. Chem. 2004, 23, 1261–1267. [Google Scholar]
- Ratte, H.T. Bioaccumulation and toxicity of silver compounds: A review. Environ. Toxicol. Chem. 1999, 18, 89–108. [Google Scholar]
- Miziolek, A.W.; Palleschi, V.; Schecher, I. Laser-Induced Breakdown Spectroscopy (LIBS); Cambridge University Press, 2006. [Google Scholar]
- Buckley, S.G. LIBS comes on strong. Laser Focus World 2006, 42, 95–98. [Google Scholar]
- Radziemski, L.J.; Loree, T.R.; Cremers, D.A.; Hoffman, N.M. Time-Resolved Laser-Induced Breakdown Spectrometry of Aerosols. Anal. Chem. 1983, 55, 1246–1252. [Google Scholar]
- Cremers, D.A.; Radziemski, L.J. Detection of Chlorine and Fluorine in Air by Laser-Induced Breakdown Spectrometry. Anal. Chem. 1983, 55, 1252–1256. [Google Scholar]
- Martin, M.Z.; Wullschleger, S.D.; Garten, C.T.; Palumbo, A.V.; Smith, J.G. Elemental analysis of environmental and biological samples using laser-induced breakdown spectroscopy and pulsed Raman spectroscopy. J. Dispersion Sci. Technol. 2004, 25, 687–694. [Google Scholar]
- Samek, O.; Lambert, J.; Hergenroder, R.; Liska, M.; Kaiser, J.; Novotny, K.; Kukhlevsky, S. Femtosecond laser spectrochemical analysis of plant samples. Laser Phys. Lett. 2006, 3, 21–25. [Google Scholar]
- Kaiser, J.; Samek, O.; Reale, L.; Liska, M.; Malina, R.; Ritucci, A.; Poma, A.; Tucci, A.; Flora, F.; Lai, A.; Mancini, L.; Tromba, G.; Zanini, F.; Faenov, A.; Pikuz, T.; Cinque, G. Monitoring of the heavy-metal hyperaccumulation in vegetal tissues by X-ray radiography and by femto-second laser induced breakdown spectroscopy. Microsc. Res. Tech. 2007, 70, 147–153. [Google Scholar]
- Limpert, E.; Stahel, W.A.; Abbt, M. Log-normal distributions across the sciences: Keys and clues. Bioscience 2001, 51, 341–352. [Google Scholar]
- Hubalek, J.; Hradecky, J.; Adam, V.; Krystofova, O.; Huska, D.; Masarik, M.; Trnkova, L.; Horna, A.; Klosova, K.; Adamek, M.; Zehnalek, J.; Kizek, R. Spectrometric and voltammetric analysis of urease – Nickel nanoelectrode as an electrochemical sensor. Sensors 2007, 7, 1238–1255. [Google Scholar]
- Xu, Z.; Chen, X.; Qu, X.H.; Jia, J.B.; Dong, S.J. Single-wall carbon nanotube-based voltammetric sensor and biosensor. Biosens. Bioelectron. 2004, 20, 579–584. [Google Scholar]
- Witte, C.P.; Medina-Escobar, N. In-gel detection of urease with nitroblue tetrazolium and quantification of the enzyme from different crop plants using the indophenol reaction. Anal. Biochem. 2001, 290, 102–107. [Google Scholar]
- Petrek, J.; Vitecek, J.; Vlasinova, H.; Kizek, R.; Kramer, K.J.; Adam, V.; Klejdus, B.; Havel, L. Application of computer imaging, stripping voltammetry and mass spectrometry for study of the effect of lead (Pb-EDTA) on growth and viability of early somatic embryos of Norway spruce (Picea abies /L./ Karst.). Anal. Bioanal. Chem. 2005, 383, 576–586. [Google Scholar]
- Supalkova, V.; Beklova, M.; Baloun, J.; Singer, C.; Sures, B.; Adam, V.; Huska, D.; Pikula, J.; Rauscherova, L.; Havel, L.; Zehnalek, J.; Kizek, R. Affecting of aquatic vascular plant Lemna minor by cisplatin revealed by voltammetry. Bioelectrochemistry 2007, in press. [Google Scholar]
- Supalkova, V.; Huska, D.; Diopan, V.; Hanustiak, P.; Zitka, O.; Stejskal, K.; Baloun, J.; Pikula, J.; Havel, L.; Zehnalek, J.; Adam, V.; Trnkova, L.; Beklova, M.; Kizek, R. Electroanalysis of plant thiols. Sensors 2007, 7, 932–959. [Google Scholar]
- Supalkova, V.; Petrek, J.; Baloun, J.; Adam, V.; Bartusek, K.; Trnkova, L.; Beklova, M.; Diopan, V.; Havel, L.; Kizek, R. Multi-instrumental investigation of affecting of early somatic embryos of Spruce by cadmium(II) and lead(II) ions. Sensors 2007, 7, 743–759. [Google Scholar]
- Vitecek, J.; Adam, V.; Petrek, J.; Vacek, J.; Kizek, R.; Havel, L. Esterases as a marker for the growth of BY-2 tobacco cells and early somatic embryos of the norway spruce. Plant. Cell. Tiss. Org. 2004, 79, 195–201. [Google Scholar]
- Zitka, O.; Stejskal, K.; Kleckerova, A.; Adam, V.; Beklova, M.; Horna, A.; Havel, L.; Kizek, R. Utilizing of electrochemical techniques for detection of biological samples. Chem. Listy 2007, 101, 225–231. [Google Scholar]


© 2008 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Krizkova, S.; Ryant, P.; Krystofova, O.; Adam, V.; Galiova, M.; Beklova, M.; Babula, P.; Kaiser, J.; Novotny, K.; Novotny, J.; et al. Multi-instrumental Analysis of Tissues of Sunflower Plants Treated with Silver(I) Ions – Plants as Bioindicators of Environmental Pollution. Sensors 2008, 8, 445-463. https://doi.org/10.3390/s8010445
Krizkova S, Ryant P, Krystofova O, Adam V, Galiova M, Beklova M, Babula P, Kaiser J, Novotny K, Novotny J, et al. Multi-instrumental Analysis of Tissues of Sunflower Plants Treated with Silver(I) Ions – Plants as Bioindicators of Environmental Pollution. Sensors. 2008; 8(1):445-463. https://doi.org/10.3390/s8010445
Chicago/Turabian StyleKrizkova, Sona, Pavel Ryant, Olga Krystofova, Vojtech Adam, Michaela Galiova, Miroslava Beklova, Petr Babula, Jozef Kaiser, Karel Novotny, Jan Novotny, and et al. 2008. "Multi-instrumental Analysis of Tissues of Sunflower Plants Treated with Silver(I) Ions – Plants as Bioindicators of Environmental Pollution" Sensors 8, no. 1: 445-463. https://doi.org/10.3390/s8010445



