Immobilization of HRP in Mesoporous Silica and Its Application for the Construction of Polyaniline Modified Hydrogen Peroxide Biosensor
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterizations of SBA-15 Mesoporous Silica
2.2. Immobilization of HRP in SBA-15 Mesopores
2.3. The Surface Morphology of Electrodes
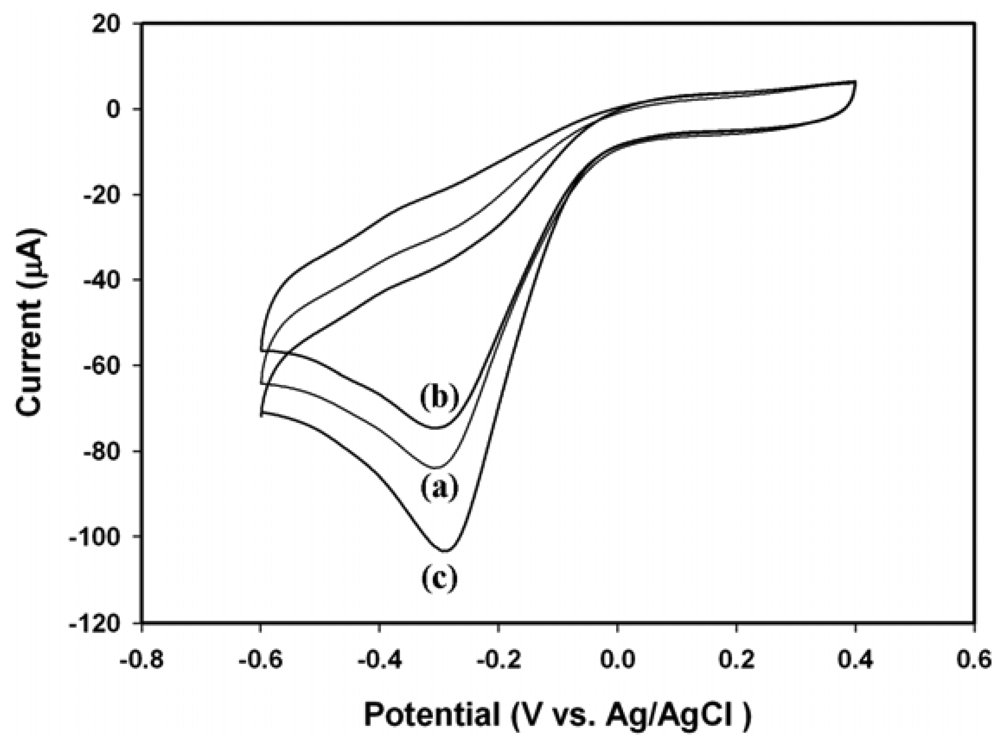
2.4. Electrochemical Response of Electrodes towards H2O2
2.5. The Stability of Constructed Electrode
3. Experimental Section
3.1. Chemicals
3.2. Preparation of SBA-15 Mesoporous Silica
3.3. Characterizations of SBA-15 Mesoporous Silica
3.4. Assay of Protein Activity
3.5. Immobilization of HRP
3.6. Fabrication of the Biosensor
3.7. Electrochemical Measurement
4. Conclusions
Acknowledgments
References
- Chen, C.; Gu, Y. Enhancing the sensitivity and stability of HRP/PANI/Pt electrode by implanted bovine serum albumin. Biosens. Bioelectron. 2008, 23, 765–770. [Google Scholar]
- Gerard, M.; Chaubey, A.; Malhotra, B.D. Application of conducting polymers to biosensors. Biosens. Bioelectron. 2002, 17, 345–359. [Google Scholar]
- Karyakin, A.A.; Bobrova, O.A.; Luckachova, L.V.; Karyakina, E.E. Potentiometric biosensors based on polyaniline semiconductor films. Sens. Actuat. B: Chem. 1996, 33, 34–38. [Google Scholar]
- Kang, X.F.; Cheng, G.J.; Dong, S.J. A novel electrochemical SPR biosensor. Electrochem. Commun. 2001, 3, 489–493. [Google Scholar]
- Kan, J.Q.; Pan, X.H.; Chen, C. Polyaniline-uricase biosensor prepared with template process. Biosens. Bioelectron. 2004, 19, 1635–1640. [Google Scholar]
- Xian, Y.Z.; Hu, Y.; Liu, F.; Xian, Y.; Wang, H.T.; Jin, L.T. Glucose biosensor based on Au nanoparticles-conductive polyaniline nanocomposite. Biosens. Bioelectron. 2006, 21, 1996–2000. [Google Scholar]
- Arora, K.; Sumana, G.; Saxena, V.; Gupta, R.K.; Gupta, S.K.; Yakhmi, J.V.; Pandey, M.K.; Chand, S.; Malhotra, B.D. Improved performance of polyaniline-uricase biosensor. Anal. Chim. Acta. 2007, 594, 17–23. [Google Scholar]
- Luo, Y.C.; Do, J.S. Urea biosensor based on PANi(urease)-Nation (R)/Au composite electrode. Biosens. Bioelectron. 2004, 20, 15–23. [Google Scholar]
- Parente, A.H.; Marques, E.T.A.; Azevedo, W.M.; Diniz, F.B.; Melo, E.H.M.; Filho, J.L.L. Glucose biosensor using glucose-oxidase immobilized in polyaniline. Appl. Biochem. Biotechnol. 1992, 37, 267–273. [Google Scholar]
- Migneault, I.; Dartiguenave, C.; Bertrand, M.J.; Waldron, K.C. Glutaraldehyde: behavior in aqueous solution, reaction with proteins, and application to enzyme crosslinking. Biotechniques 2004, 37, 790–802. [Google Scholar]
- Fernandes, K.F.; Lima, C.S.; Pinho, H.; Collins, C.H. Immobilization of horseradish peroxidase onto polyaniline polymers. Process Biochem. 2003, 38, 1379–1384. [Google Scholar]
- Takahashi, H.; Li, B.; Sasaki, T.; Miyazaki, C.; Kajino, T.; Inagaki, S. Catalytic activity in organic solvents and stability of immobilized enzymes depend on the pore size and surface characteristics of mesoporous silica. Chem. Mater. 2000, 12, 3301–3305. [Google Scholar]
- Pandya, P.H.; Jasra, R.V.; Newalkar, B.L.; Bhatt, P.N. Studies on the activity and stability of immobilized alpha-amylase in ordered mesoporous silicas. Microporous Mesoporous Mat. 2005, 77, 67–77. [Google Scholar]
- Hartmann, M. Ordered mesoporous materials for bioadsorption and biocatalysis. Chem. Mater. 2005, 17, 4577–4593. [Google Scholar]
- Diaz, J.F.; Balkus, K.J. Enzyme immobilization in MCM-41 molecular sieve. J. Mol. Catal. B: Enzym. 1996, 2, 115–126. [Google Scholar]
- Takahashi, H.; Li, B.; Sasaki, T.; Miyazaki, C.; Kajino, T.; Inagaki, S. Immobilized enzymes in ordered mesoporous silica materials and improvement of their stability and catalytic activity in an organic solvent. Microporous Mesoporous Mat. 2001, 44, 755–762. [Google Scholar]
- Lettow, J.S.; Han, Y.J.; Schmidt-Winkel, P.; Yang, P.D.; Zhao, D.Y.; Stucky, G.D.; Ying, J.Y. Hexagonal to mesocellular foam phase transition in polymer-templated mesoporous silicas. Langmuir 2000, 16, 8291–8295. [Google Scholar]
- Zhao, D.Y.; Huo, Q.S.; Feng, J.L.; Chmelka, B.F.; Stucky, G.D. Nonionic triblock and star diblock copolymer and oligomeric surfactant syntheses of highly ordered, hydrothermally stable, mesoporous silica structures. J. Am. Chem. Soc. 1998, 120, 6024–6036. [Google Scholar]
- Vinu, A.; Murugesan, V.; Hartmann, M. Adsorption of lysozyme over mesoporous molecular sieves MCM-41 and SBA-15: Influence of pH and aluminum incorporation. J. Phys. Chem. B 2004, 108, 7323–7330. [Google Scholar]
- Yiu, H.H.P.; Wright, P.A.; Botting, N.P. Enzyme immobilisation using SBA-15 mesoporous molecular sieves with functionalised surfaces. J. Mol. Catal. B: Enzym. 2001, 15, 81–92. [Google Scholar]
- Lei, J.; Fan, J.; Yu, C.Z.; Zhang, L.Y.; Jiang, S.Y.; Tu, B.; Zhao, D.Y. Immobilization of enzymes in mesoporous materials: controlling the entrance to nanospace. Microporous Mesoporous Mat. 2004, 73, 121–128. [Google Scholar]
- Salis, A.; Meloni, D.; Ligas, S.; Casula, M.F.; Monduzzi, M.; Solinas, V.; Dumitriu, E. Physical and chemical adsorption of Mucor javanicus lipase on SBA-15 mesoporous silica. Synthesis, structural characterization, and activity performance. Langmuir 2005, 21, 5511–5516. [Google Scholar]
- Chong, A.S.M.; Zhao, X.S. Functionalized nanoporous silicas for the immobilization of penicillin acylase. Appl. Surf. Sci. 2004, 237, 398–404. [Google Scholar]
- Bai, Y.; Yang, H.; Yang, W.W.; Li, Y.C.; Sun, C.Q. Gold nanoparticles-mesoporous silica composite used as an enzyme immobilization matrix for amperometric glucose biosensor construction. Sens. Actuat. B 2007, 124, 179–186. [Google Scholar]
- Dai, Z.H.; Bao, H.C.; Yang, X.D.; Ju, H.X. A bienzyme channeling glucose sensor with a wide concentration range based on co-entrapment of enzymes in SBA-15 mesopores. Biosens. Bioelectron. 2008, 23, 1070–1076. [Google Scholar]
- Guo, H.S.; He, N.Y.; Ge, S.X.; Yang, D.; Zhang, J.N. Determination of cardiac troponin I by anodic stripping voltammetry at SBA-15 modified carbon paste electrode. Nanopor. Mater. IV 2005, 156, 695–702. [Google Scholar]
- Liu, Y.G.; Xu, Q.; Feng, X.M.; Zhu, J.J.; Hou, W.H. Immobilization of hemoglobin on SBA-15 applied to the electrocatalytic reduction of H2O2. Anal. Bioanal.Chem. 2007, 387, 1553–1559. [Google Scholar]
- Zhang, J.D.; Chi, Q.J.; Dong, S.J.; Wang, E.K. In situ electrochemical scanning tunnelling microscopy investigation of structure for horseradish peroxidase and its electrocatalytic property. Bioelectrochem. Bioenerg. 1996, 39, 267–274. [Google Scholar]
- Dai, Z.H.; Bao, H.C.; Yang, X.D.; Ju, H.X. A bienzyme channeling glucose sensor with a wide concentration range based on co-entrapment of enzymes in SBA-15 mesopores. Biosens. Bioelectron. 2008, 23, 1070–1076. [Google Scholar]
- Dai, Z.H.; Ju, H.X.; Chen, H.Y. Mesoporous materials promoting direct electrochemistry and electrocatalysis of horseradish peroxidase. Electroanalysis 2005, 17, 862–868. [Google Scholar]
- Dai, Z.H.; Ni, J.; Huang, X.H.; Lu, G.F.; Bao, J.C. Direct electrochemistry of glucose oxidase immobilized on a hexagonal mesoporous silica-MCM-41 matrix. Bioelectrochemistry 2007, 70, 250–256. [Google Scholar]
- Liu, H.Y.; Hu, N.F. Study on direct Electrochemistry of glucose oxidase stabilized by cross-linking and immobilized in silica nanoparticle films. Electroanalysis 2007, 19, 884–892. [Google Scholar]
- Walcarius, A. Impact of mesoporous silica-based materials on electrochemistry and feedback from electrochemical science to the characterization of these ordered materials. C. R. Chim. 2005, 8, 693–712. [Google Scholar]
- Budny, A.; Novak, F.; Plumere, N.; Schetter, B.; Speiser, B.; Straub, D.; Mayer, H.A.; Reginek, M. Redox-active silica nanoparticles-part 1: electrochemistry and catalytic activity of spherical, nonporous silica particles with nanometric diameters and covalently bound redox-active modifications. Langmuir 2006, 22, 10605–10611. [Google Scholar]
- Fattakhova-Rohlfing, D.; Rathousky, J.; Rohlfing, Y.; Bartels, O.; Wark, M. Functionalized mesoporous silica films as a matrix for anchoring electrochemically active guests. Langmuir 2005, 21, 11320–11329. [Google Scholar]
- Essa, H.; Magner, E.; Cooney, J.; Hodnett, B.K. Influence of pH and ionic strength on the adsorption, leaching and activity of myoglobin immobilized onto ordered mesoporous silicates. J. Mol. Catal. B: Enzym. 2007, 49, 61–68. [Google Scholar]
- Derylo-Marczewska, A.; Marczewski, A.W.; Skrzypek, I.; Pikus, S.; Kozak, M. Effect of addition of pore expanding agent on changes of structure characteristics of ordered mesoporous silicas. Appl. Surf. Sci. 2008, 255, 2851–2858. [Google Scholar]
- Nguyen, T.P.B.; Lee, J.W.; Shim, W.G.; Moon, H. Synthesis of functionalized SBA-15 with ordered large pore size and its adsorption properties of BSA. Microporous Mesoporous Mat. 2008, 110, 560–569. [Google Scholar]
- Katiyar, A.; Yadav, S.; Smirniotis, P.G.; Pinto, N.G. Synthesis of ordered large pore SBA-15 spherical particles for adsorption of biomolecules. J. Chromatogr A. 2006, 1122, 13–20. [Google Scholar]






| Sample | ABET (m2/g) | Vtotal (cm3/g) | a0 (Å) | D (Å) |
|---|---|---|---|---|
| SBA-15 | 708.5 | 0.92 | 12.56 | 76 |
| SBA-15(HRP) | 632.3 | 0.85 | 12.16 | 76 |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chen, C.-C.; Do, J.-S.; Gu, Y. Immobilization of HRP in Mesoporous Silica and Its Application for the Construction of Polyaniline Modified Hydrogen Peroxide Biosensor. Sensors 2009, 9, 4635-4648. https://doi.org/10.3390/s90604635
Chen C-C, Do J-S, Gu Y. Immobilization of HRP in Mesoporous Silica and Its Application for the Construction of Polyaniline Modified Hydrogen Peroxide Biosensor. Sensors. 2009; 9(6):4635-4648. https://doi.org/10.3390/s90604635
Chicago/Turabian StyleChen, Chien-Chung, Jing-Shan Do, and Yesong Gu. 2009. "Immobilization of HRP in Mesoporous Silica and Its Application for the Construction of Polyaniline Modified Hydrogen Peroxide Biosensor" Sensors 9, no. 6: 4635-4648. https://doi.org/10.3390/s90604635




