Anti-Inflammatory Potential of Monogalactosyl Diacylglycerols and a Monoacylglycerol from the Edible Brown Seaweed Fucus spiralis Linnaeus
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation of Monoacylglycerol and Monogalactosyl Diacylglycerols
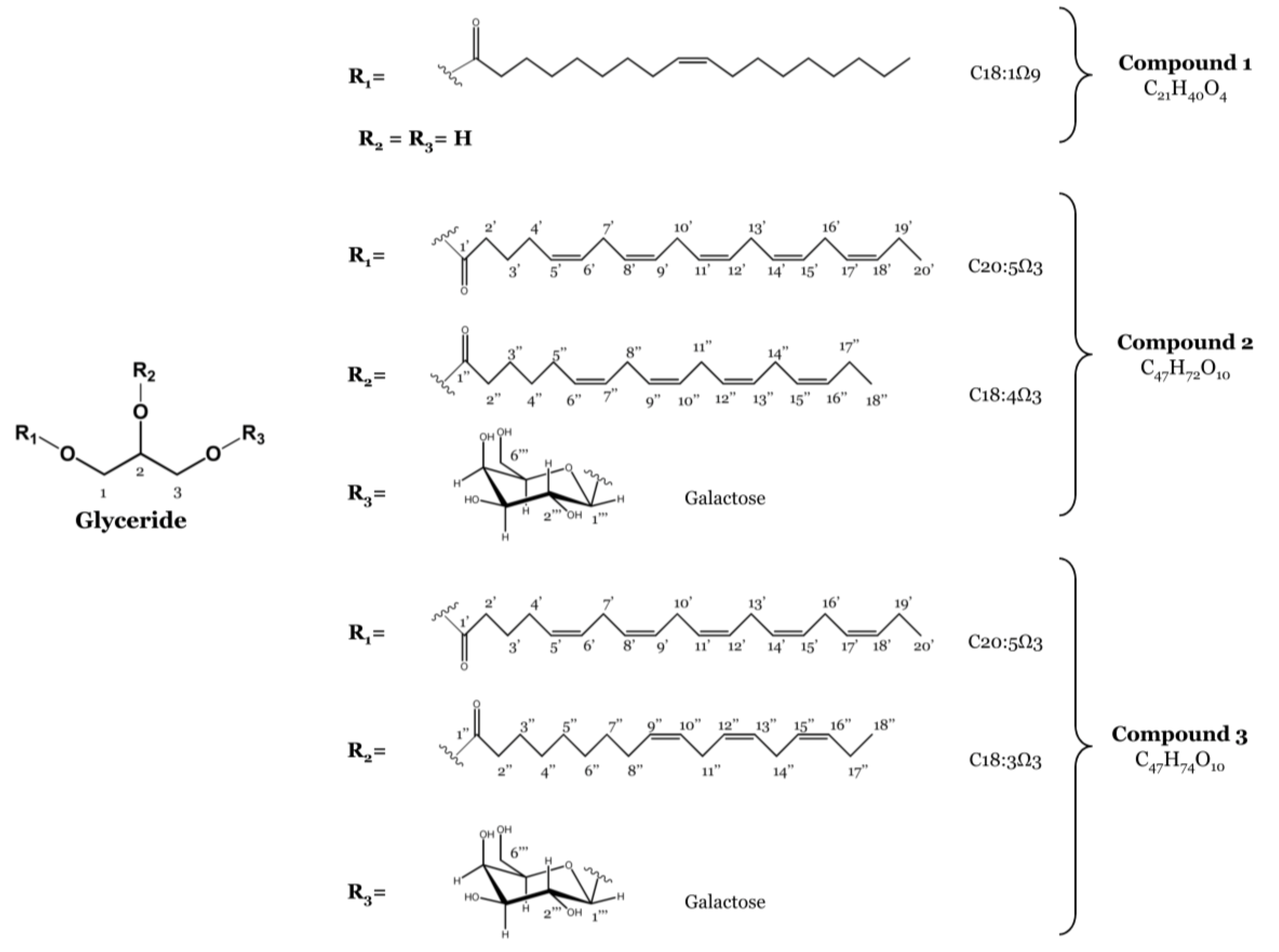
2.2. Anti-Inflammatory Activity
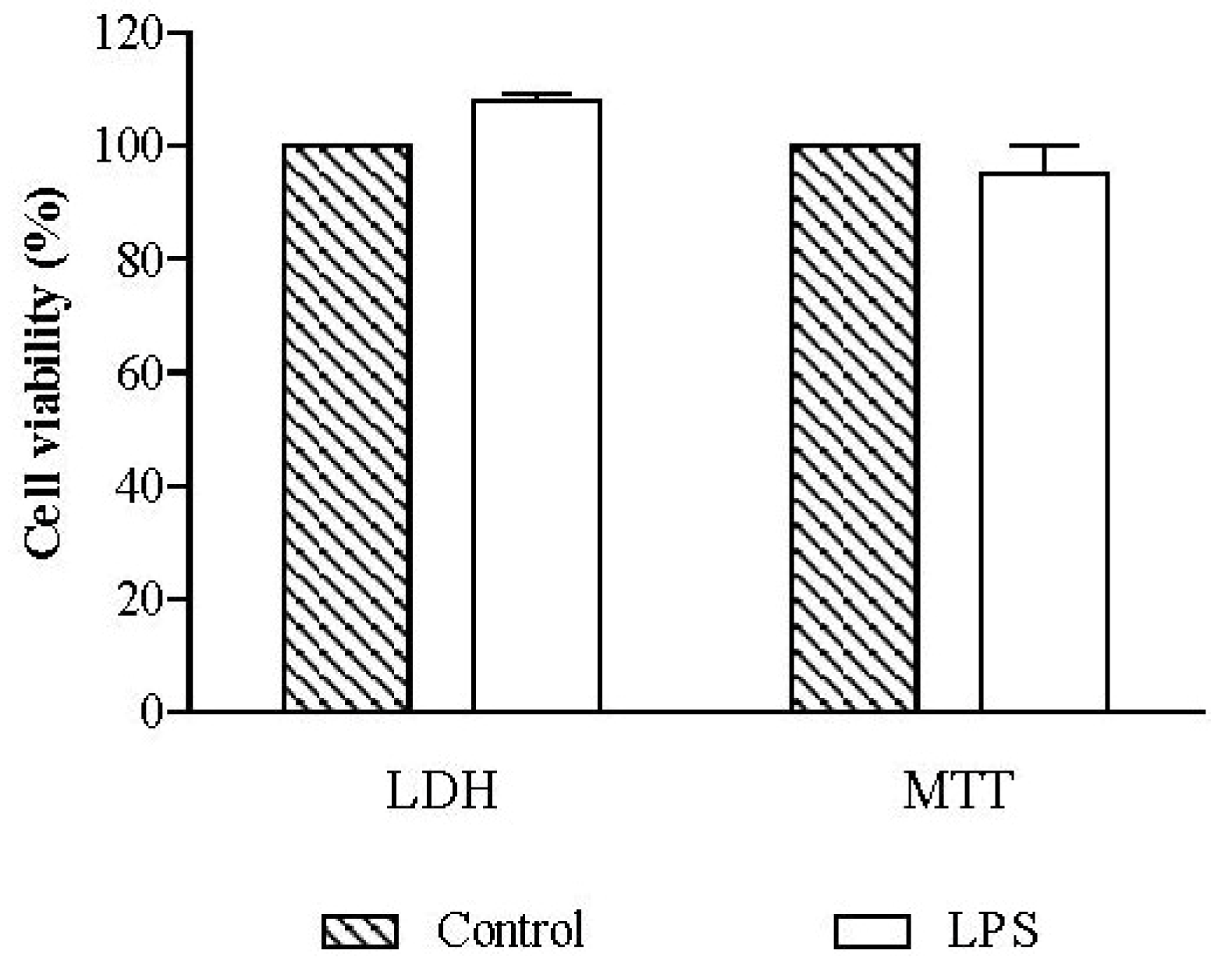
2.2.1. Cell Viability
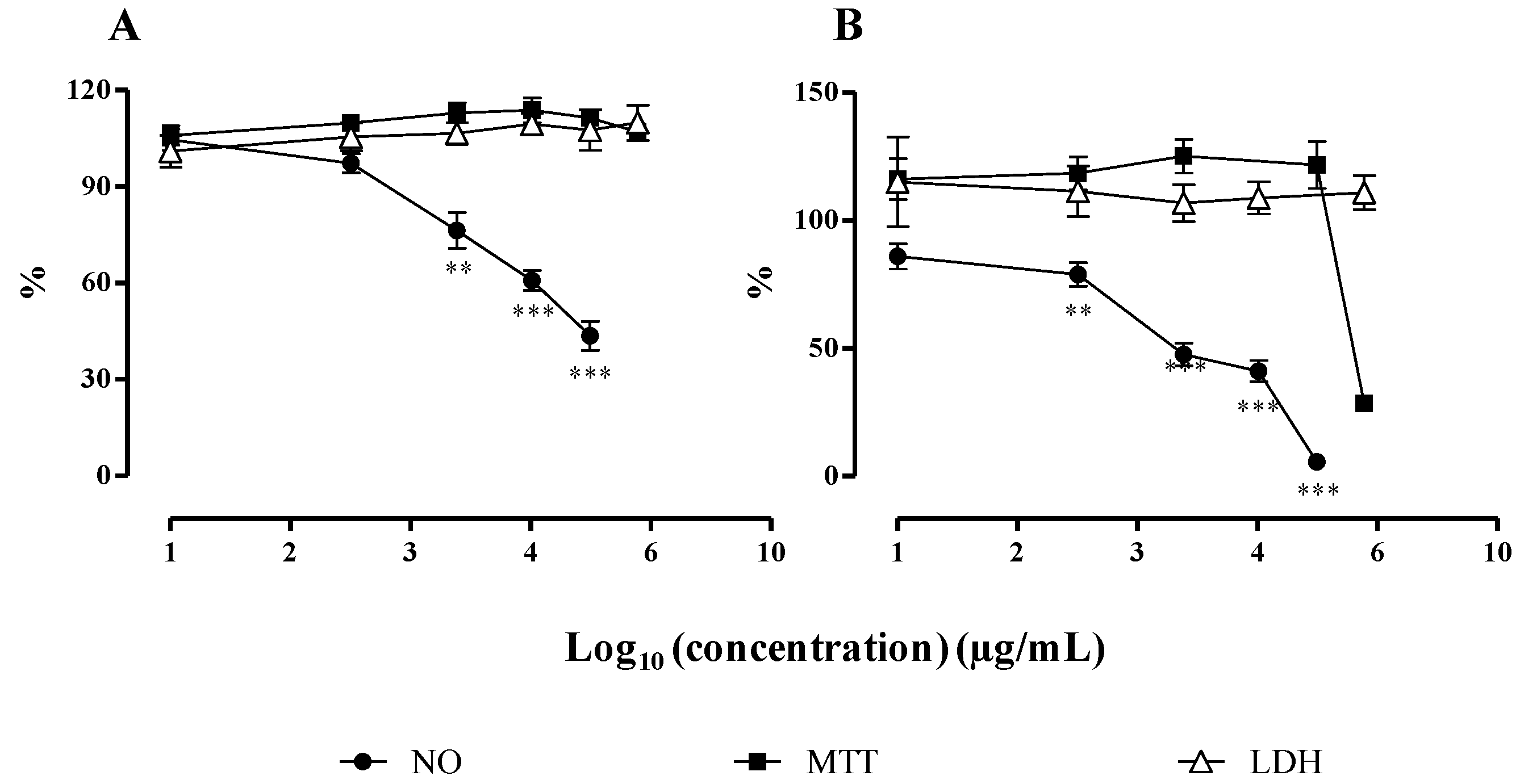
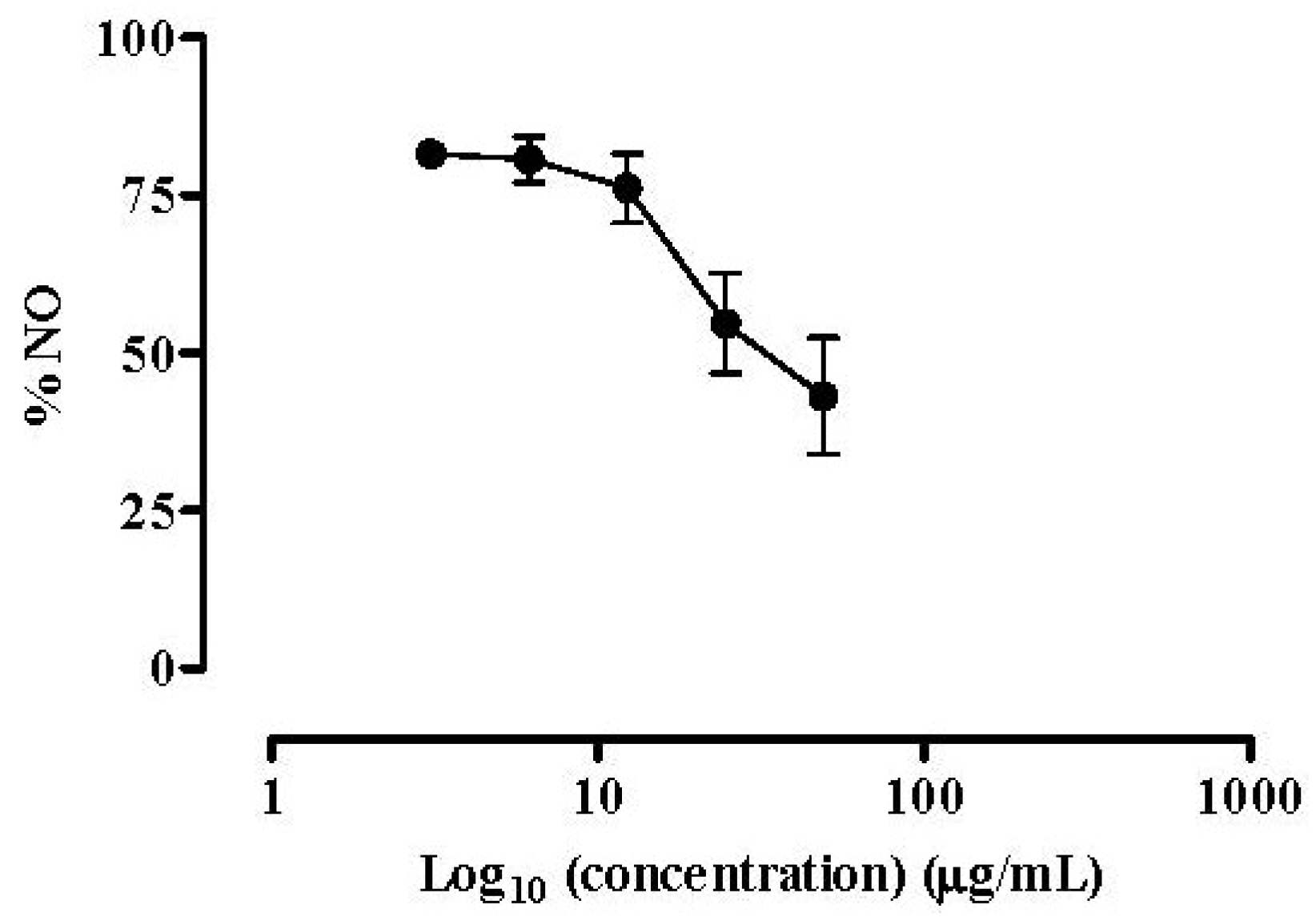
2.2.2. NO Released by RAW 264.7 Macrophages
3. Experimental Section
3.1. Standards and Reagents
3.2. Compounds Characterization
3.2.1. General Experimental Methods
3.2.2. Seaweed Material
3.2.3. Extraction and Isolation
3.2.4. Spectrometric Data of Isolated Compounds
3.3. Anti-Inflammatory Capacity
3.3.1. Cell Culture and Treatments
3.3.2. Cell Viability
3.3.2.1. LDH Assay
3.3.2.2. MTT Assay
3.3.3. NO Release by RAW 264.7 Cells
3.4. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gounaris, K.; Barber, J. Monogalactosyldiacylglycerol: The most abundant polar lipid in nature. Trends Biochem. Sci. 1983, 8, 378–381. [Google Scholar] [CrossRef]
- Khotimchenko, S.V. Distribution of glyceroglycolipids in marine algae and grasses. Chem. Nat. Compd. 2002, 38, 223–229. [Google Scholar] [CrossRef]
- Dormann, P.; Benning, C. Galactolipids role in seed plants. Trends Plant Sci. 2002, 7, 112–118. [Google Scholar] [CrossRef]
- Maeda, N.; Hada, T.; Yoshida, H.; Mizushina, Y. Inhibitory effect on replicative DNA polymerases, human cancer cell proliferation, and in vivo anti-tumor activity by glycolipids from spinach. Curr. Med. Chem. 2007, 14, 955–967. [Google Scholar] [CrossRef]
- Maeda, N.; Kokai, Y.; Hada, T.; Yoshida, H.; Mizushina, Y. Oral administration of monogalactosyl diacylglycerol from spinach inhibits colon tumor growth in mice. Exp. Ther. Med. 2013, 5, 17–22. [Google Scholar]
- Souza, L.M.; Sassaki, G.L.; Romanos, M.T.V.; Barreto-Bergter, E. Structural characterization and anti-HSV-1 and HSV-2 activity of glycolipids from the marine algae Osmundaria obtusiloba isolated from southeastern Brazilian coast. Mar. Drugs 2012, 10, 918–931. [Google Scholar] [CrossRef]
- Hirao, S.; Tara, K.; Kuwano, K.; Tanaka, J.; Ishibashi, F. Algicidal activity of glicerolipids from brown alga Ishige sinicola toward red tide microalgae. Biosci. Biotechnol. Biochem. 2012, 76, 372–374. [Google Scholar] [CrossRef]
- Bruno, A.; Rossi, C.; Marcolongo, G.; di Lena, A.; Venzo, A.; Berrie, C.P.; Corda, D. Selective in vivo anti-inflammatory action of the galactolipid monogalactosyldiacylglycerol. Eur. J. Pharmacol. 2005, 7, 159–168. [Google Scholar]
- Ulivi, V.; Lenti, M.; Gentili, C.; Marcolongo, G.; Cancedda, R.; Cancedda, F.D. Anti-inflammatory activity of monogalactosyldiacylglycerol in human articular cartilage in vitro: Activation of an anti-inflammatory cyclooxygenase-2 (COX-2) pathway. Arthritis Res. Ther. 2011, 13. [Google Scholar] [CrossRef]
- Banskota, A.H.; Gallant, P.; Stefanova, R.; Melanson, R.; O’Leary, S.J.B. Monogalactosyldiacylglycerols, potent nitric oxide inhibitors from the marine macroalga Tetraselmis chui. Nat. Prod. Res. 2012, 27, 1084–1090. [Google Scholar]
- Banskota, A.H.; Stefanova, R.; Sperker, A.; Melanson, R.; Osborne, J.A.; O’Leary, S.J.B. Five new galactolipids from the freshwater microalga Porphyridium aerugineum and their nitric oxide inhibitory activity. J. Appl. Phycol. 2013, 25, 951–960. [Google Scholar] [CrossRef]
- Moncada, S.; Plamer, R.M.J.; Higgs, E.A. Nitric oxide: Physiology, pathophysiology and pharmacology. Pharmacol. Rev. 1996, 43, 109–142. [Google Scholar]
- Moreau, R.A.; Whitaker, B.D.; Hicks, K.B. Phytosterols, phytostanols, and their conjugates in foods: Structural diversity, quantitative analysis, and health promoting uses. Prog. Lipid Res. 2002, 41, 457–500. [Google Scholar] [CrossRef]
- Lopes, G.; Sousa, C.; Bernardo, J.; Andrade, P.B.; Valentão, P.; Ferreres, F.; Mouga, T. Sterol profiles in 18 macroalgae of the Portuguese coast. J. Phycol. 2011, 47, 1210–1218. [Google Scholar] [CrossRef]
- Lopes, G.; Sousa, C.; Silva, L.R.; Pinto, E.; Andrade, P.B.; Valentão, P. Can phlorotannins extracts constitute a novel pharmacological alternative for microbial infections with associated inflammatory conditions? PLoS One 2012, 7, e31145. [Google Scholar]
- Lopes, G.; Pinto, E.; Andrade, P.B.; Valentão, P. Antifungal activity of phlorotannins against dermatophytes and yeasts: Approaches to the mechanism of action and influence on Candida albicans virulence factor. PLoS One 2013, 8, e72203. [Google Scholar]
- Ferreres, F.; Lopes, G.; Izquierdo, A.G.; Andrade, P.B.; Sousa, C.; Mouga, T.; Valentão, P. Phlorotannins extracts from Fucales characterized by HPLC-DAD-ESI-MSn: Approaches to hyaloronidase innibitory capacity and antioxidant properties. Mar. Drugs 2012, 10, 2766–2781. [Google Scholar] [CrossRef]
- Andrade, P.B.; Barbosa, M.; Matos, R.P.; Lopes, G.; Vinholes, J.; Mouga, T.; Valentão, P. Valuable compounds in macroalgae extracts. Food Chem. 2013, 138, 1819–1828. [Google Scholar] [CrossRef]
- Ren, Y.; Houghton, P.; Hider, R.C. Relevant activities of extracts and constituents of animals used in traditional Chinese medicine for central nervous system effects associated with Alzheimer’s disease. J. Pharm. Pharmacol. 2006, 58, 989–996. [Google Scholar] [CrossRef]
- Miyazawa, M.; Yagi, N.; Taguchi, K. Inhibitory compounds of alpha-glucosidase activity from Arctium lappa L. J. Oleo Sci. 2005, 54, 589–594. [Google Scholar] [CrossRef]
- Kim, Y.H.; Kim, E.-H.; Lee, C.; Kim, M.-H.; Rho, J.-R. Two new monogalactosyl diacylglycerols from brown alga Sargassum thunbergii. Lipids 2007, 42, 395–399. [Google Scholar] [CrossRef]
- Kobayashi, M.; Hayashi, K.; Kawazoe, K.; Kitagawa, I. Marine Natural Products XXIX. Heterosigma-glycolipids I, II, III and IV, four diacylglyceroglycolipids prossessing ω3-polyunsaturated fatty acid residues, from the raphidophycean dinoflagellate Heterosigma akashiwo. Chem. Pharm. Bull. 1992, 40, 1404–1410. [Google Scholar] [CrossRef]
- Guella, G.; Frassanito, R.; Mancini, I. A new solution for an old problem: The regiochemical distribution of the acyl chains in galactolipids can be established by electrospray ionization tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2003, 17, 1982–1994. [Google Scholar] [CrossRef]
- Leblond, J.D.; Timofte, H.I.; Roche, S.A.; Porter, N.M. Mono- and digalactosyldiacylglycerol composition of glaucocystophytes (Glaucophyta): A modern interpretation using positive-ion electrospray ionization/mass spectrometry/mass spectrometry. Phycol. Res. 2010, 58, 222–229. [Google Scholar] [CrossRef]
- Lobner, D. Comparison of the LDH and MTT assays for quantifying cell death: Validity for neuronal apoptosis? J. Neurosci. Methods 2000, 96, 147–152. [Google Scholar] [CrossRef]
- Pinho, B.R.; Sousa, C.; Valentão, P.; Andrade, P.B. Is nitric oxide decrease observed with naphtoquinones in LPS stimulated RAW 264.7 macrophagues a beneficial property? PLoS One 2011, 6, e24098. [Google Scholar]
- Kim, H.K.; Cheon, B.S.; Kim, Y.H.; Kim, H.P. Effects of naturally occurring flavonoids on nitric oxide production in the macrophague cell line RAW 264.7 and their structure-activity relationships. Biochem. Pharmacol. 1999, 58, 759–765. [Google Scholar] [CrossRef]
- Banskota, A.H.; Stefanova, R.; Gallant, P.; McGinn, P. Monogalactosyldiacylglycerols: Potent nitric oxide inhibitors from the marine microalga Nannochloropsis granulata. J. Appl. Phycol. 2013, 25, 349–357. [Google Scholar] [CrossRef]
- Viguerie, N.; Picard, F.; Hul, G.; Roussel, B.; Barbe, P.; Iacovoni, J.S.; Valle, C.; Langin, D.; Saris, W.H.M. Multiple effects of a short-term dexamethasone treatment in human skeletal muscle and adipose tissue. Physiol. Genomics 2011, 44, 141–151. [Google Scholar]
- Lenti, M.; Gentili, C.; Pianezzi, A.; Marcolongo, G.; Lalli, A.; Cancedda, R.; Cancedda, F.D. Monogalactosyldiacylglycerol anti-inflammatory activity on adult articular cartilage. Nat. Prod. Res. 2009, 23, 754–762. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lopes, G.; Daletos, G.; Proksch, P.; Andrade, P.B.; Valentão, P. Anti-Inflammatory Potential of Monogalactosyl Diacylglycerols and a Monoacylglycerol from the Edible Brown Seaweed Fucus spiralis Linnaeus. Mar. Drugs 2014, 12, 1406-1418. https://doi.org/10.3390/md12031406
Lopes G, Daletos G, Proksch P, Andrade PB, Valentão P. Anti-Inflammatory Potential of Monogalactosyl Diacylglycerols and a Monoacylglycerol from the Edible Brown Seaweed Fucus spiralis Linnaeus. Marine Drugs. 2014; 12(3):1406-1418. https://doi.org/10.3390/md12031406
Chicago/Turabian StyleLopes, Graciliana, Georgios Daletos, Peter Proksch, Paula B. Andrade, and Patrícia Valentão. 2014. "Anti-Inflammatory Potential of Monogalactosyl Diacylglycerols and a Monoacylglycerol from the Edible Brown Seaweed Fucus spiralis Linnaeus" Marine Drugs 12, no. 3: 1406-1418. https://doi.org/10.3390/md12031406





