Food-Safety Hazards in the Pork Chain in Nagaland, North East India: Implications for Human Health
Abstract
:1. Introduction
2. Experimental Section
2.1. Selection of Hazards for Investigation
2.2. Study Design, Sampling Locations
- (a)
- Slaughterhouses: We visited the four largest slaughter places in different areas of Kohima, and also five village slaughters performed directly on-farm. We took faecal and blood samples and lingual palpation was performed on the fresh slaughtered pigs. As well, meat samples were taken from direct sale from the fresh slaughtered pigs on farms.
- (b)
- Butchers: We first conducted a census of all pork selling stalls in Kohima. From this list, 25 stalls were chosen by simple random sampling. Each of these stalls was visited three times. At the first visit, the aim of the study was explained to the retailers and their approval requested. The three visits of each stall were coordinated so that they took place once early (7–9 am, E), once mid-morning (11 am–1 pm, H) and once after noon (after 1 pm–2:30 pm, L) and at each time, a meat sample (50 g) was purchased from the meat pieces displayed for sale. The samples were taken by two trained local project assistants from pieces of muscle meat displayed for sale. The pieces which were for sale varied in amount and anatomic origin between sampling times and between retailers; to minimize this bias, meat was requested from the largest piece of muscle meat present.
2.3. Sampling and Diagnostic Testing
- (a)
- Blood samples were taken from the vena cava during slaughtering process in a 5 mL or 10 mL sterile syringe out of the abdominal cavity and stored in 7 mL sterile plastic tubes without additives. It was stored on ice and allowed to clot for approximately 12 h at 4 °C followed by centrifugation at ~1200 rpm for 10 min. The serum was pipetted into sterile 2 mL tubes and frozen at −18 °C until utilization in the Brucella suis serologic flow assay as described in the manufacturer’s instructions.
- (b)
- Faecal samples were taken during slaughter out of the rectal part of the intestine when it had been taken out of the abdominal cavity and before it was processed further. Approx. 10 g of faeces were put in a plastic bag which was closed and stored in a cool box and later in the refrigerator. Within 48 h following the sampling, the faeces underwent a sedimentation-flotation technique and were observed under the microscope for eggs/oocysts of protozoa and helminth species (Only results for Fasciola spp. eggs are reported here).
- (c)
- Lingual palpation for cysticercosis was carried out by project veterinarians in situ in fresh slaughtered animals before rigor mortis set in.
| Hazard /indicator | Diagnostic testing method | n samples positive/ n samples tested (%) | Quality categories 1 | % samples per categories |
|---|---|---|---|---|
| Total aerobic bacteria | Himedia HiTouchTM Flexi Plate Agar plate culture | 89/91 (97.8%) | Satisfactory: <104 cfu/g | 24.7% |
| Acceptable: 104–105 cfu/g | 52.8% | |||
| Unsatisfactory: >105 cfu/g | 20.2% | |||
| Not classifiable | 2.25% | |||
| Enterobacteriaceae | Himedia HiTouchTM Flexi Plate Selective agar plate culture | 88/91 (94.5%) | Satisfactory: <102 cfu/g | 5.7% |
| Acceptable: 102–103 cfu/g | 5.7% | |||
| Unsatisfactory: >103 cfu/g | 88.6% | |||
| Listeria spp. | Petrifilm® selective culture system | 36/91 (39.6%) | Acceptable: <102 cfu/g | 97.7% |
| Unacceptable/potentially hazardous: >102 cfu/g | 2.3% | |||
| Staphylococcus aureus | Petrifilm® selective culture system | 9/19 (47.4%) | Acceptable: <102 cfu/g | 10.5% |
| Unsatisfactory: <102 cfu/g | 36.8% | |||
| Antibiotic residues | Premi® Test (DSM) 2 | 4/88 (4.5%) | Any positive unacceptable | 4.5% |
| Cysticercus cellulosae | Lingual palpation post mortem; occasional findings in meat samples | 7/80 (8.8%) (palpation]) | Any positive unacceptable | 8.8% |
| 2/91 (2.2%) (found when cutting meat) | 2.2% | |||
| Brucella suis | Brucella IgG flow assay (KIT Biomedical Research) | 3/53 (5.6%) | Any positive unacceptable | 5.6% |
| Fasciola spp. | Sedimentation | 18/88 (20.6%) | No threshold set |
- (d)
- The meat samples were placed in sterile plastic bags, placed on ice and transported to the laboratory within one hour of time where they were processed immediately to give an account of bacterial colonization at sales point. Diagnostic testing using routine tests such as (selective) colony plate count methods followed the respective manufacturer’s instructions. Classification thresholds were based on internationally recognized standards (food safety criteria/process hygiene criteria) for destructive sampling techniques for pig carcasses [11] or dissected, portioned retail meat [12] (see Table 1). Meat juice needed for the microbial screening test for antibiotic residues was gained through freezing and subsequent thawing of a small piece of each meat sample.
2.4. Questionnaire and Direct Observation Check List
2.5. Statistical Analysis
2.6. Risk Assessment
| Hazard identification & characterization | Exposure assessment | Risk characterization | |
|---|---|---|---|
| Enterobacteriaceae | Bacteria of this group representing hazards for human health include Salmonella spp., Klebsiella spp. and toxigenic E. coli. These cause a range of symptoms from diarrhoea to urinary tract and soft tissue infection and septicaemia. General morbidity to infection with clinical symptoms is moderate, mortality is low. | In the questionnaire survey, pork consumers from Nagaland indicated that boiling of meat was the common method of preparing meat (98%). Ninety-eight % of questionnaire respondents cook pork between 30 and 190 min. This means that pork is generally well cooked which will kill Enterobacteriaceae. However there are chances for cross contamination, which is most problematic for hazards where the infectious dose is very low: notably pathogenic E. coli strains. | Because of thorough cooking, we consider the risk of ingestion of harmful Enterobacteriaceae from cooked pork was low with potentially severe health consequences. The risk of ingesting Enterobacteriaceae on other aliments which have been cross-contaminated through pork can be estimated higher. However we did not directly investigate these processes in this study. The uncertainty of these estimates is high. |
| Taenia solium (Cysticercus cellulosae) | Infection with the larval stage (cysticerci) of the tapeworm T. solium can lead to establishment of a tapeworm in the gut. Through the ingestion of tapeworm eggs, human cysticercosis, a serious disease, develops: Inside the body, cysticerci can develop in a number of tissues; muscle tissue can be affected as well as the eyes and the central nervous system. Neurocysticercosis [16] including epilepsy [17] is the most severe form of the disease. | Proof of highly frequent presence of the parasite was found manifold within this study: through lingual palpation and detection of viable cysticerci in meat. Also, 57% of consumers reported white, rice grain like cysts in the meat they purchased. Based on this, prevalence of T. solium stages in pork designated for human alimentation can be estimated as high. Thorough cooking will kill cysticerci and only 1% pork consumers in the study indicated they would eat raw meat. It is common practice in Nagaland to consume smoked pork products (indicated by 43% of consumers). | The risk of ingestion of viable cysticerci from cooked meat has to be considered low, because meat tends to be thoroughly cooked. The direct health impacts can be estimated as low and the indirect health impacts as severe with moderate associated uncertainties. Regarding the possibility of infection through cysticerci in raw/smoked meat products, the uncertainties are higher. |
| Antibiotic residues | Antibiotic residues favour the development of antibiotic resistance in bacteria and can cause allergies in sensitive persons who consume meat containing residues. Prevalence of such incidents is low when searching the literature for case descriptions; but this can only be stated with a high level of uncertainty. | Of the pork samples tested in this study, 4.5% were positive and thus contained traces of antibiotic residues. Good hygiene practices and heat treatment do not eliminate the residues and thus, the consumer is not able to influence or mitigate this risk. | We considered the risk of ingestion of antibiotic residues in pork with a direct impact on consumer’s health (anaphylactic reaction) as very low but severe. We considered indirect health impacts as moderate. Uncertainty of these estimates was moderate. |
3. Results
3.1. Pork Consumption in Nagaland
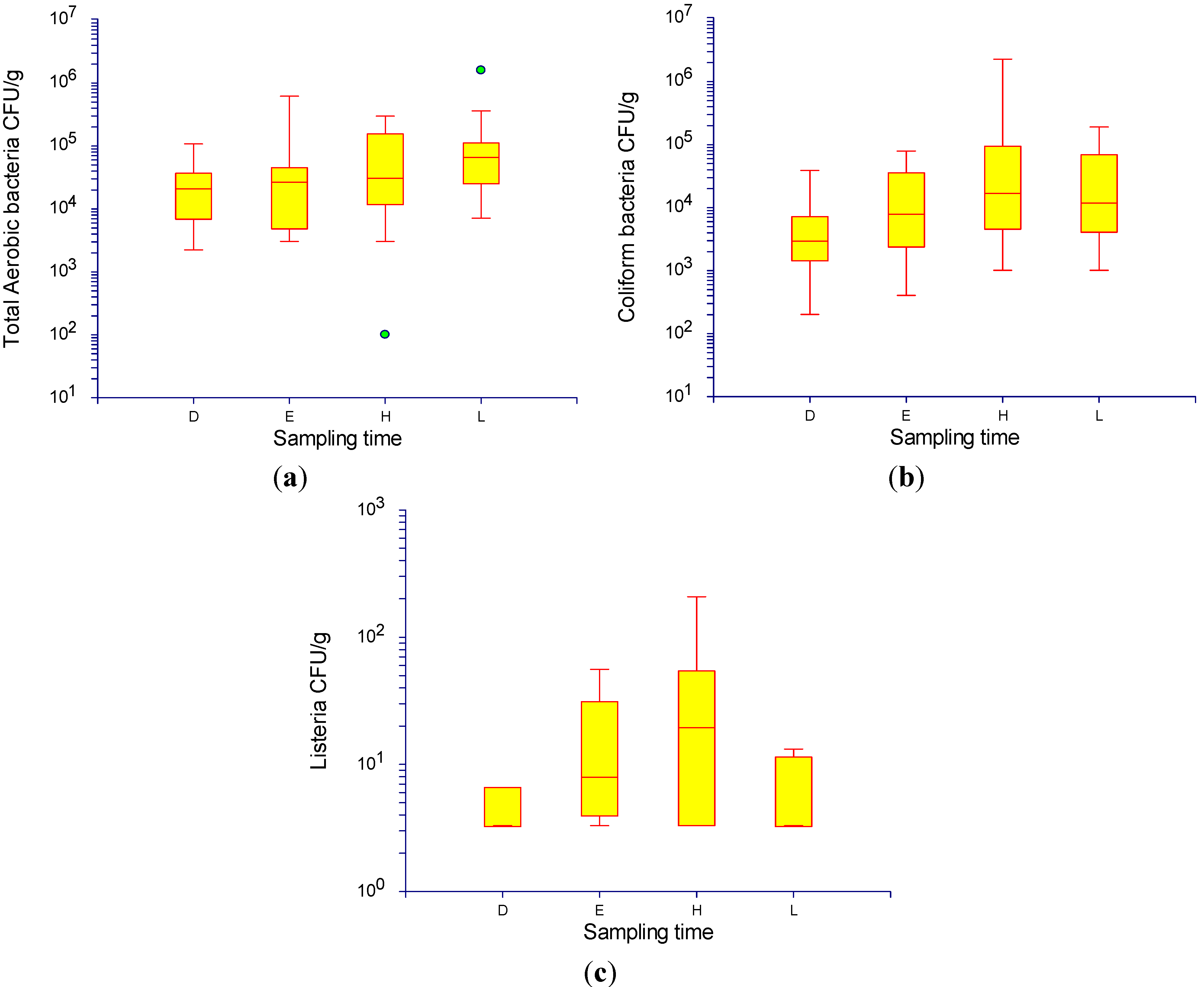
3.2. Numbers of Samples Taken and Results of Laboratory Testing
3.3. Risk Assessment (See Table 2)
- The most common health effects of Enterobacteriaceae are gastro-intestinal illness. This is often most serious in young, old, pregnant and immune-compromised people. A minority of cases can have severe and even fatal illness (haemolytic uremia syndrome from STEC or septicaemia from salmonellosis). Only a few cells of some Salmonella and STEC strains are sufficient to initiate disease in susceptible humans and levels of Enterobacteriaceae were very high.
- Ingestion of viable T. solium cysts can result in establishment of a tapeworm in the gut. Tapeworms can cause gastrointestinal symptoms but these are usually not severe. A more significant health impact is neurocysticercosis as the result of auto-infection. An indirect impact is when the eggs of the tapeworm are ingested by a pig which then develops cysts, allowing the cycle of the parasite to continue.
- Antibiotic residues can directly cause allergies in sensitive persons, however, cases are quite rare. There is a potential for antibiotic residues in food to promote antibiotic resistance in bacteria pathogenic to humans. However, this has not been quantified.
- We found that levels of contamination with Enterobacteriaceae were unacceptably high at all steps along the value chain. The likelihood of consumption of pathogenic organisms was reduced by the long period of cooking, but the likelihood of cross contamination in the kitchen was high as most people did not separate pork and vegetables during preparation. In addition, we found a local practice of storing pork for several weeks in the chimneys and this smoked pork was not always cooked before eating. Less than 10% of households have refrigeration and owners of refrigerators stored meat three times longer than people without. According to secondary data, mothers report 1.6 cases of diarrhoea in infants each year [19].
- Taenia solium prevalence in meat and by lingual palpation were high, as has been reported for pigs in the rural North East of India [7,20]. Prolonged cooking is effective at inactivating cysts and cross-contamination is not likely to be a problem. However, the effect of traditional smoking of pork in chimneys on cyst viability is not known. Moreover, secondary data shows a high prevalence of asymptomatic and clinical neurocysticercosis in Northern India, especially in pig farming communities [21,22]. This suggests that people are exposed to tape-worm eggs which are shed by people who have consumed pork with cysts. The implication is that viable cysts are being ingested in Nagaland.
- Antibiotic residues are not eliminated through good hygiene practices and heat treatment and thus, the consumer is not able to influence this risk. Around one in 20 pork samples had unacceptable levels which in conjunction with the high consumption of pork, suggests that exposure is common.
4. Discussion
4.1. Importance of Systematic Selection of Hazards
4.2. Importance of Pork-Borne Disease in Nagaland
4.3. Risk Mitigation
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Report of a WHO Consultation on Public Health Implications of Consumption of Raw Milk and Meat and Their Products.; World Health Organization, Emerging and other Communicable Diseases, Surveillance and Control: Geneva, Switzerland, 1995.
- Stein, C.; Kuchenmuller, T.; Hendrickx, S.; Pruss-Ustun, A.; Wolfson, L.; Engels, D.; Schlundt, J. The global burden of disease assessments—who is responsible? PLoS Negl Trop Dis 2007, 1. [Google Scholar] [CrossRef]
- Chugh, T.D. Emerging and re-emerging bacterial diseases in India. J Biosci. 2008, 33, 549–555. [Google Scholar] [CrossRef]
- Nayak, P. Growth and Human Development in North-East India; Oxford University Press: New Delhi, 2010; p. 349. [Google Scholar]
- Kumaresan, A.; Bujarbaruah, K.M.; Pathak, K.A.; Das, A.; Bardoloi, R.K. Integrated resource-driven pig production systems in a mountainous area of northeast India: Production practices and pig performance. Trop. Anim. Health Prod. 2009, 41, 1187–1196. [Google Scholar] [CrossRef]
- Deka, R.; Thorpe, W. Nagaland’s Pig Sub-Sector: Current Status, Constraints and Opportunities; International Livestock Research Institute: New Delhi, India, 2008. [Google Scholar]
- Roy, B.; Tendon, V. Metacestodiasis in north east India: A study on the prevalence of hydatidosis and cysticercosis in Mizoram, Nagaland and Assam. Indian J. Anim. Health 1989, 28, 4–10. [Google Scholar]
- Singh, B.B.; Sharma, R.; Sharma, J.K.; Juyal, P.D. Parasitic zoonoses in India: An overview. Rev. Sci. Tech. 2010, 29, 629–637. [Google Scholar]
- Cameron, R. A Review of the Industrialisation of Pig Production Worldwide with Particular Reference to the Asian Region. In Animal Health and Area-wide Integration; FAO: Brisbane, Australia, 2000; p. 29. [Google Scholar]
- Fosse, J.; Seegers, H.; Magras, C. Prevalence and risk factors for bacterial food-borne zoonotic hazards in slaughter pigs: A review. Zoonoses Public Health 2009, 56, 429–454. [Google Scholar] [CrossRef]
- Zweifel, C.; Fischer, R.; Stephan, R. Microbiological contamination of pig and cattle carcasses in different small-scale swiss abattoirs. Meat Sci 2008, 78, 225–231. [Google Scholar] [CrossRef] [Green Version]
- Anonymous. Commission regulation (EC) No. 2073/2005 of 15 november 2005 on microbiological criteria for foodstuffs. Off. J. Eur. Union 2005, L338, 1–26. [Google Scholar]
- Hintze, J. Ncss 2007; NCSS, LCC.: Kaysville, UT, USA, 2007. [Google Scholar]
- Anonymous. Principles and Guidelines for the Conduct of Microbial Risk Assessment; Codex Alimentarius Commission: Rome, Italy, 1999. [Google Scholar]
- Grace, D.; Makita, K.; Kang’ethe, E.; Bonfoh, B. Safe food, fair food: Participatory risk analysis for improving the safety of informally produced and marketed food in Sub-saharan Africa. Rev. Afr. Santé Prod. Anim. 2010, 8, 3–11. [Google Scholar]
- Eckert, J.; Friedhoff, K.T.; Zahner, H.; Deplazes, P. Lehrbuch der Parasitologie für die Tiermedizin; (in German). Enke: Stuttgart, Germany, 2005; p. 575. [Google Scholar]
- Garcia, H.H.; Gonzalez, A.E.; Evans, C.A.; Gilman, R.H. Taenia solium cysticercosis. Lancet 2003, 362, 547–556. [Google Scholar] [CrossRef]
- Mataragas, M.; Skandamis, P.N.; Drosinos, E.H. Risk profiles of pork and poultry meat and risk ratings of various pathogen/product combinations. Int. J. Food Microbiol. 2008, 126, 1–12. [Google Scholar] [CrossRef]
- Anonymous. Reproductive and Child Health District Level Household Survey (dhls-2) Nagaland 2002–2004; Ministry of Health and Family Welfare: New Delhi, India, 2006.
- Prasad, K.N.; Chawla, S.; Jain, D.; Pandey, C.M.; Pal, L.; Pradhan, S.; Gupta, R.K. Human and porcine taenia solium infection in rural north India. Trans. Roy. Soc. Trop. Med. Hyg. 2002, 96, 515–516. [Google Scholar] [CrossRef]
- Prasad, K.N.; Verma, A.; Srivastava, S.; Gupta, R.K.; Pandey, C.M.; Paliwal, V.K. An epidemiological study of asymptomatic neurocysticercosis in a pig farming community in northern India. Trans. Roy. Soc. Trop. Med. Hyg. 2011, 105, 531–536. [Google Scholar] [CrossRef]
- Kumar, A.; Khan, S.A.; Khan, S.; Das, S.; Bhargava, A.; Negi, K.S. A study of neurocysticercosis in the foothills of the Himalayas. Int. J. Infect. Dis. 2006, 10, 79–82. [Google Scholar] [CrossRef]
- Knight-Jones, T.J.; Mylrea, G.E.; Kahn, S. Animal production food safety: Priority pathogens for standard setting by the World Organisation for Animal Health. Rev. Sci. Tech. 2010, 29, 523–535. [Google Scholar]
- Grace, D.; Omore, A.; Randolph, T.; Kang’ethe, E.; Nasinyama, G.W.; Mohammed, H.O. Risk assessment for Escherichia coli O157:H7 in marketed unpasteurized milk in selected east African countries. J. Food Protect. 2008, 71, 257–263. [Google Scholar]
- Fahrion, A.S.; Lapar, M.L.; Ngoc, T.N.; Ngoc, T.D.; Grace, D. Food-borne hazards in a transforming pork value chain in Hanoi: Basis for future risk assessments in Vietnam. J. Prev. Med. 2013, 23, 18–25. [Google Scholar]
- Chau, T.T.; Campbell, J.I.; Schultsz, C.; Chau, N.V.; Diep, T.S.; Baker, S.; Chinh, N.T.; Farrar, J.J.; van Doorn, H.R. Three adult cases of listeria monocytogenes meningitis in Vietnam. PLoS Med. 2010, 7. [Google Scholar] [CrossRef]
- Report of the WHO Consultation on Foodborne Trematode Infections and Taeniasis/Cysticercosis; World Health Organization: Geneva, Switzerland, 2011.
- Furst, T.; Sayasone, S.; Odermatt, P.; Keiser, J.; Utzinger, J. Manifestation, diagnosis, and management of foodborne trematodiasis. BMJ 344. [CrossRef]
- Poester, F.P.; Samartino, L.E.; Santos, R.L. Pathogenesis and pathobiology of brucellosis in livestock. Rev. Sci. Tech. 2013, 32, 105–115. [Google Scholar]
- Mantur, B.G.; Amarnath, S.K. Brucellosis in India—A review. J. Biosci. 2008, 33, 539–547. [Google Scholar] [CrossRef]
- Thakur, D.; Ravikumar, R.K.; Kumar, P.; Gupta, A.; Sharma, A.; Katoch, S.; Bodh, V.K. Meat inspection and animal welfare practices: Evidences from the north-western Himalayan region, India. Vet. World 2012, 5, 718–722. [Google Scholar] [CrossRef]
- Grace, D.; Dipeolu, M.; Olawoye, J.; Ojo, E.; Odebode, S.; Agbaje, M.; Akindana, G.; Randolph, T. Evaluating a group-based intervention to improve the safety of meat in Bodija market, Ibadan, Nigeria. Trop. Anim. Health Prod. 2012, 44, S61–S66. [Google Scholar] [CrossRef]
- Von Holy, A.; Makhoane, F.M. Improving street food vending in South Africa: Achievements and lessons learned. Int. J. Food Microbiol. 2006, 111, 89–92. [Google Scholar] [CrossRef]
- Jabbar, M.A.; Baker, D.; Fadiaga, M.L. Demand for Livestock Products in Developing Countries with a Focus on Quality and Safety Attributes: Evidence from Case Studies; ILRI: Nairobi, Kenya, 2010. [Google Scholar]
- Islam, M.S.; Mahmud, Z.H.; Gope, P.S.; Zaman, R.U.; Hossain, Z.; Mondal, D.; Sharker, M.A.; Islam, K.; Jahan, H.; Bhuiya, A.; et al. Hygiene intervention reduces contamination of weaning food in Bangladesh. Trop. Med. Int. Health 2013, 18, 250–258. [Google Scholar]
- Chelleng, P.K.; Narain, K.; Das, H.K.; Chetia, M.; Mahanta, J. Risk factors for cancer nasopharynx: A case-control study from Nagaland, India. Natl. Med. J. India 2000, 13, 6–8. [Google Scholar]
- Makita, K.; Desissa, F.; Teklu, A.; Zewde, G.; Grace, D. Risk assessment of staphylococcal poisoning due to consumption of informally-marketed milk and home-made yoghurt in Debre Zeit, Ethiopia. Int. J. Food Microbiol. 2012, 153, 135–141. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fahrion, A.S.; Jamir, L.; Richa, K.; Begum, S.; Rutsa, V.; Ao, S.; Padmakumar, V.P.; Deka, R.P.; Grace, D. Food-Safety Hazards in the Pork Chain in Nagaland, North East India: Implications for Human Health. Int. J. Environ. Res. Public Health 2014, 11, 403-417. https://doi.org/10.3390/ijerph110100403
Fahrion AS, Jamir L, Richa K, Begum S, Rutsa V, Ao S, Padmakumar VP, Deka RP, Grace D. Food-Safety Hazards in the Pork Chain in Nagaland, North East India: Implications for Human Health. International Journal of Environmental Research and Public Health. 2014; 11(1):403-417. https://doi.org/10.3390/ijerph110100403
Chicago/Turabian StyleFahrion, Anna Sophie, Lanu Jamir, Kenivole Richa, Sonuwara Begum, Vilatuo Rutsa, Simon Ao, Varijaksha P. Padmakumar, Ram Pratim Deka, and Delia Grace. 2014. "Food-Safety Hazards in the Pork Chain in Nagaland, North East India: Implications for Human Health" International Journal of Environmental Research and Public Health 11, no. 1: 403-417. https://doi.org/10.3390/ijerph110100403





