1950MHz Radio Frequency Electromagnetic Radiation Inhibits Testosterone Secretion of Mouse Leydig Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Cultureand Ethics Statement
2.2. RF-EMR Exposure Equipment and Exposure Parameter
2.3. Cell Proliferation Assay
2.4. Cell Cycle Analysis
2.5. Cell Apoptosis Analysis
2.6. Testosterone Content Assay
2.7. StAR and P450scc mRNA Expression
2.8. Intracellular Cell ROS Analysis
2.9. Statistical Analysis
3. Results
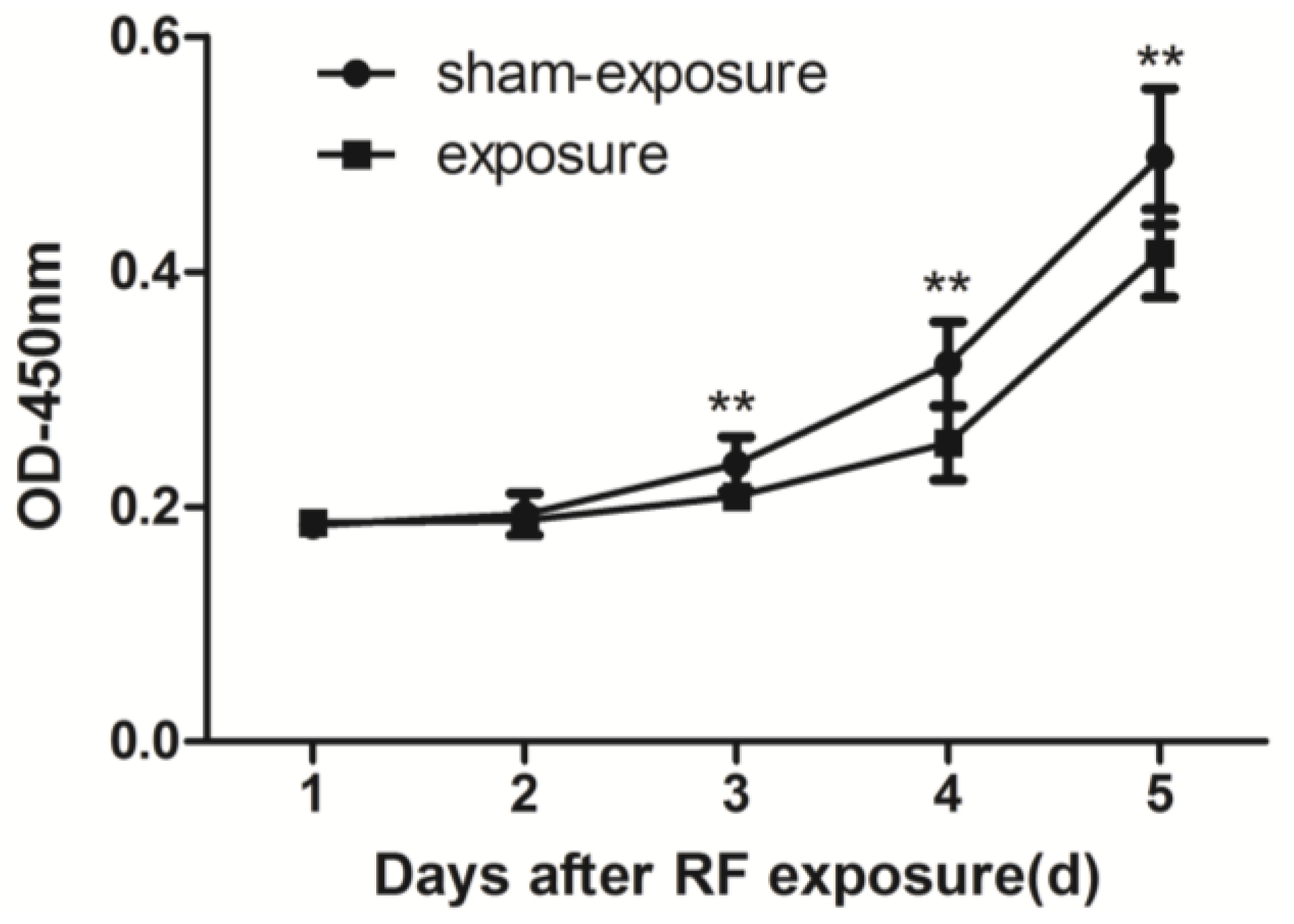
3.1. Proliferation of TM3 Cell was Inhibited by RF Radiation
3.2. RF Radiation Resulted in S Phase Arrest
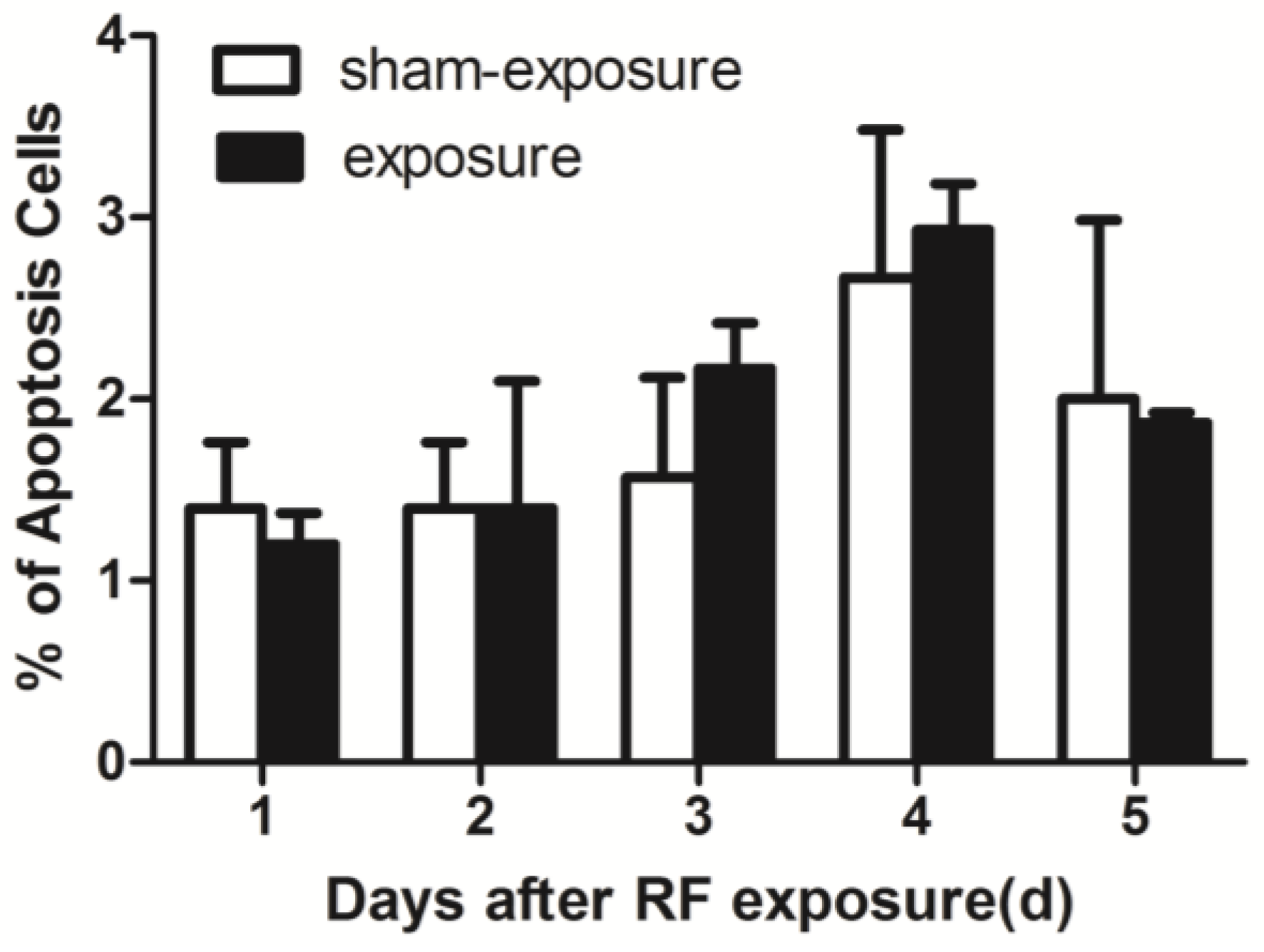
3.3. RF Radiation Produced No Effects on TM3 Cell Apoptosis
3.4. Testosterone Secretion Inhibited by RF Radiation
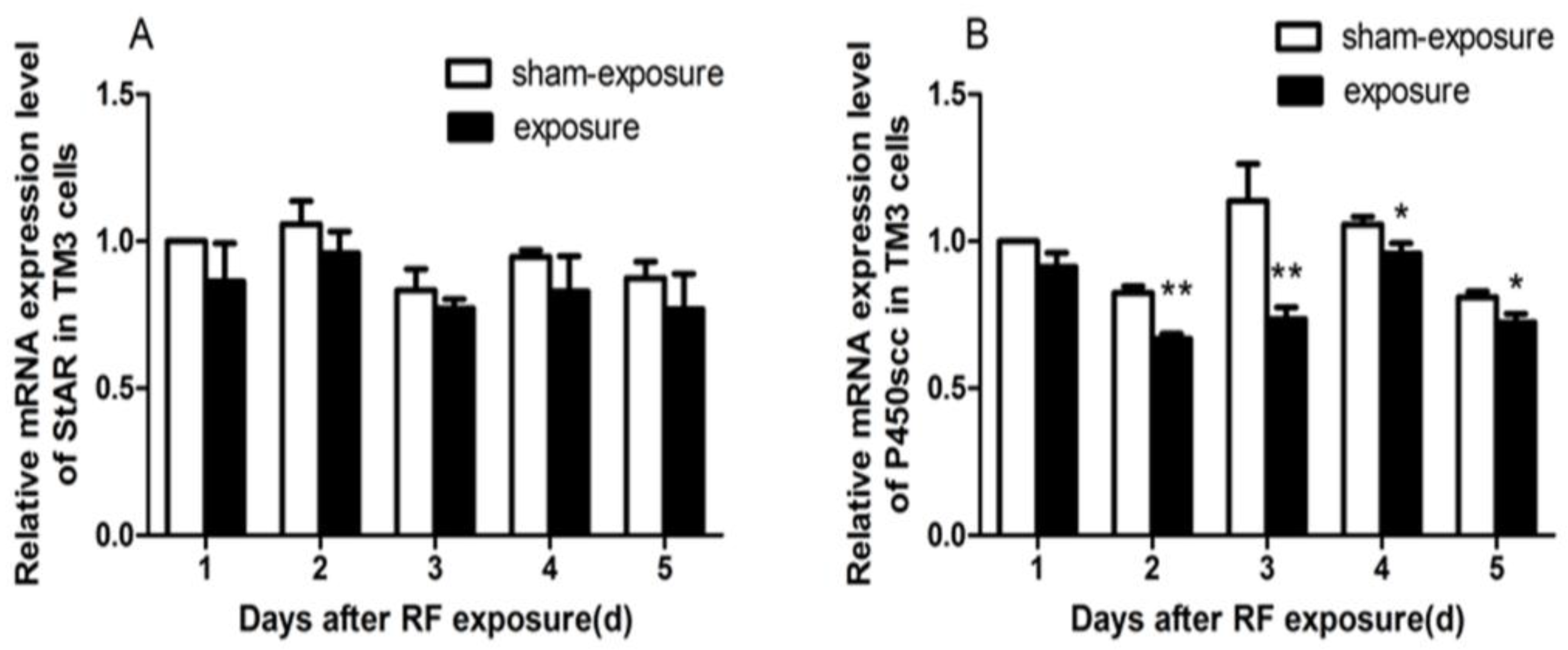
3.5. StAR and P450scc mRNA Expression Downregulated by RF Radiation
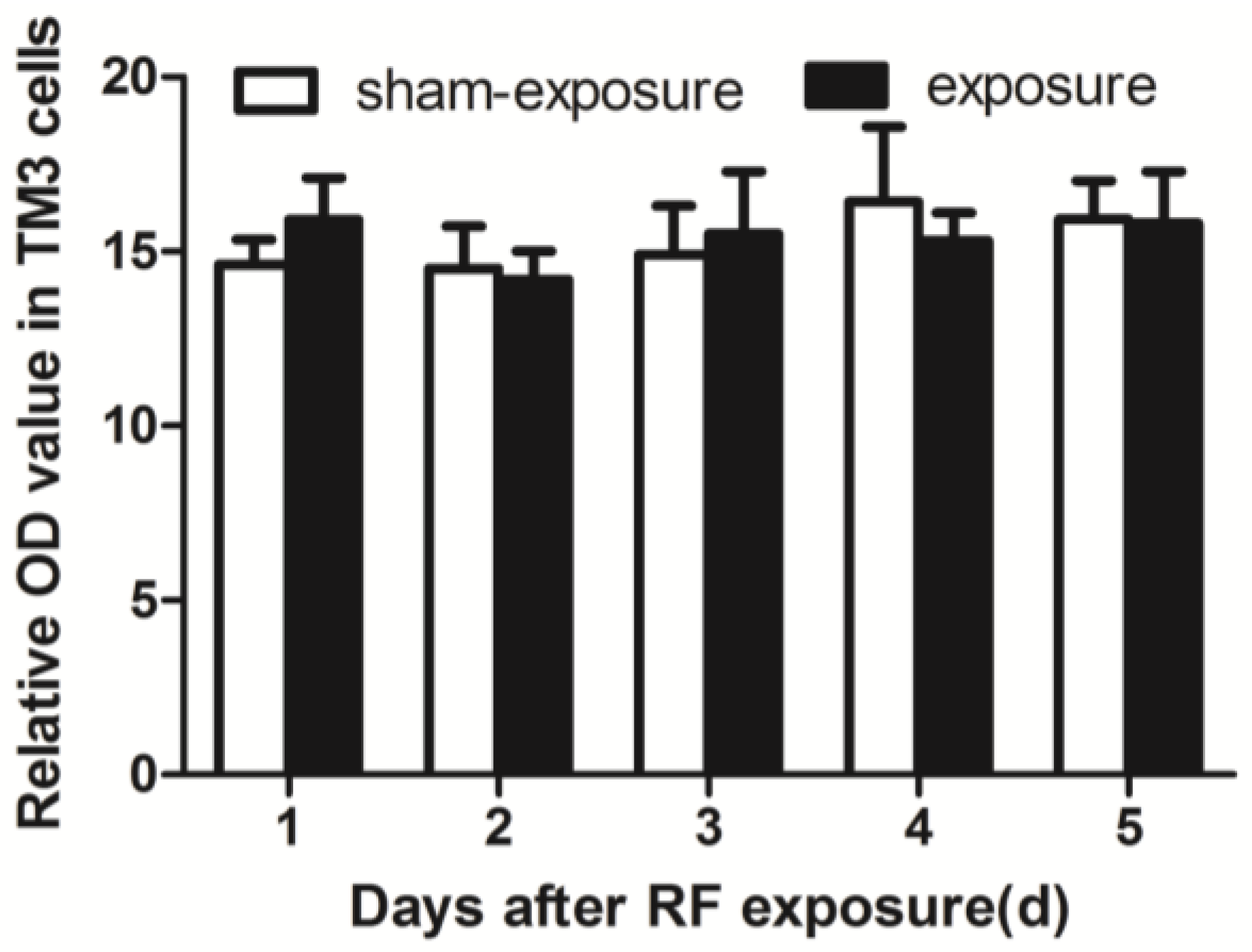
3.6. RF Radiation Produced No Effect on Intracellular ROS Generation
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kesari, K.K.; Siddiqui, M.H.; Meena, R.; Verma, H.N.; Kumar, S. Cell phone radiation exposure on brain and associated biological systems. Indian J. Exp. Biol. 2013, 51, 187–200. [Google Scholar] [PubMed]
- Agarwal, A.; Deepinder, F.; Sharma, R.K.; Ranga, G.; Li, J. Effect of cell phone usage on semen analysis in men attending infertility clinic: An observational study. Fertil. Steril. 2008, 89, 124–128. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, J.A.; Emmerson, R.Y.; Bailey, C.M.; Olsen, R.G.; Gandhi, O.P. Microwave radiation absorption in the rat: Frequency-dependent SAR distribution in body and tail. Bioelectromagnetics 1985, 6, 199–206. [Google Scholar] [CrossRef] [PubMed]
- De la Hoz, A.; Diaz-Ortiz, A.; Moreno, A. Microwaves in organic synthesis. Thermal and non-thermal microwave effects. Chem. Soc. Rev. 2005, 34, 164–178. [Google Scholar] [CrossRef] [PubMed]
- La Vignera, S.; Condorelli, R.A.; Vicari, E.; D’Agata, R.; Calogero, A.E. Effects of the exposure to mobile phones on male reproduction: A review of the literature. J. Androl. 2012, 33, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Hardell, L.; Sage, C. Biological effects from electromagnetic field exposure and public exposure standards. Biomed. Pharmacother. 2008, 62, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Sepehrimanesh, M.; Saeb, M.; Nazifi, S.; Kazemipour, N.; Jelodar, G.; Saeb, S. Impact of 900 MHz electromagnetic field exposure on main male reproductive hormone levels: A Rattusnorvegicus model. Int. J. Biometeorol. 2014, 58, 1657–1663. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, M.E.; Kaynar, M.; Badem, H.; Cavis, M.; Karatas, O.F.; Cimentepe, E. What is harmful for male fertility: Cell phone or the wireless Internet? Kaohsiung J. Med. Sci. 2015, 31, 480–484. [Google Scholar] [CrossRef] [PubMed]
- Bin-Meferij, M.M.; El-Kott, A.F. The radioprotective effects of Moringa oleifera against mobile phone electromagnetic radiation-induced infertility in rats. Int. J. Clin. Exp. Med. 2015, 8, 12487–12497. [Google Scholar] [PubMed]
- Kesari, K.K.; Kumar, S.; Behari, J. Effects of radiofrequency electromagnetic wave exposure from cellular phones on the reproductive pattern in male Wistar rats. Appl. Biochem. Biotechnol. 2011, 164, 546–559. [Google Scholar] [CrossRef] [PubMed]
- Aitken, R.J.; Bennetts, L.E.; Sawyer, D.; Wiklendt, A.M.; King, B.V. Impact of radio frequency electromagnetic radiation on DNA integrity in the male germline. Int. J. Androl. 2005, 28, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Kesari, K.K.; Behari, J. Microwave exposure affecting reproductive system in male rats. Appl. Biochem. Biotechnol. 2010, 162, 416–428. [Google Scholar] [CrossRef] [PubMed]
- Mailankot, M.; Kunnath, A.P.; Jayalekshmi, H.; Koduru, B.; Valsalan, R. Radio frequency electromagnetic radiation (RF-EMR) from GSM (0.9/1.8GHz) mobile phones induces oxidative stress and reduces sperm motility in rats. Clinics 2009, 64, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Naziroglu, M.; Yuksel, M.; Kose, S.A.; Ozkaya, M.O. Recent reports of Wi-Fi and mobile phone-induced radiation on oxidative stress and reproductive signaling pathways in females and males. J. Membr. Biol. 2013, 246, 869–875. [Google Scholar] [CrossRef] [PubMed]
- Qin, F.; Zhang, J.; Cao, H.; Yi, C.; Li, J.X.; Nie, J. Effects of 1800-MHz radiofrequency fields on circadian rhythm of plasma melatonin and testosterone in male rats. J. Toxicol. Environ. Health Part A. 2012, 75, 1120–1128. [Google Scholar] [CrossRef] [PubMed]
- De Iuliis, G.N.; Newey, R.J.; King, B.V.; Aitken, R.J. Mobile phone radiation induces reactive oxygen species production and DNA damage in human spermatozoa in vitro. PLoS ONE 2009, 4. [Google Scholar] [CrossRef] [PubMed]
- O’Shaughnessy, P.J.; Monteiro, A.; Fowler, P.A.; Morris, I.D. Identification of Leydig cell-specific mRNA transcripts in the adult rat testis. Reproduction 2014, 147, 671–682. [Google Scholar] [CrossRef] [PubMed]
- Romano-Spica, V.; Mucci, N.; Ursini, C.L.; Ianni, A.; Bhat, N.K. Ets1 oncogene induction by ELF-modulated 50 MHz radiofrequency electromagnetic field. Bioelectromagnetics 2000, 21, 8–18. [Google Scholar] [CrossRef]
- Ozguner, M.; Koyu, A.; Cesur, G.; Ural, M.; Ozguner, F.; Gokcimen, A. Biological and morphological effects on the reproductive organ of rats after exposure to electromagnetic field. Saudi Med. J. 2005, 26, 405–410. [Google Scholar] [PubMed]
- Dasdag, S.; Tas, M.; Akdag, M.Z.; Yegin, K. Effect of long-term exposure of 2.4 GHz radiofrequency radiation emitted from Wi-Fi equipment on testes functions. Electromagn. Biol. Med. 2015, 34, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Saygin, M.; Caliskan, S.; Karahan, N.; Koyu, A.; Gumral, N.; Uguz, A. Testicular apoptosis and histopathological changes induced by a 2.45 GHz electromagnetic field. Toxicol. Ind. Health 2011, 27, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Wilson, G.D. Radiation and the cell cycle, revisited. Cancer Metastasis Rev. 2004, 23, 209–225. [Google Scholar] [CrossRef] [PubMed]
- Tas, M.; Dasdag, S.; Akdag, M.Z.; Cirit, U.; Yegin, K.; Seker, U.; Ozmen, M.F.; Eren, L.B. Long-term effects of 900 MHz radiofrequency radiation emitted from mobile phone on testicular tissue and epididymalsemen quality. Electromagn. Biol. Med. 2014, 33, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Y.; Wang, S.M.; Peng, R.Y.; Gao, Y.B.; Wang, L.F.; Zhao, L. Long-term microwave radiation affects male reproduction in rats. Zhonghua Nan Ke Xue 2011, 17, 214–218. [Google Scholar] [PubMed]
- Celik, S.; Aridogan, I.A.; Izol, V.; Erdogan, S.; Polat, S.; Doran, S. An evaluation of the effects of long-term cell phone use on the testes via light and electron microscope analysis. Urology 2012, 79, 346–350. [Google Scholar] [CrossRef] [PubMed]
- Werck-Reichhart, D.; Feyereisen, R. Cytochromes P450: A success story. Genome Biol. 2000, 1. [Google Scholar] [CrossRef] [PubMed]
- Mueller, J.W.; Gilligan, L.C.; Idkowiak, J.; Arlt, W.; Foster, P.A. The regulation of steroid action by sulfation and desulfation. Endocr. Rev. 2015, 36, 526–563. [Google Scholar] [CrossRef] [PubMed]
- Clark, B.J.; Cochrum, R.K. The steroidogenic acute regulatory protein as a target of endocrine disruption in male reproduction. Drug Metab. Rev. 2007, 39, 353–370. [Google Scholar] [CrossRef] [PubMed]
- Desai, N.R.; Kesari, K.K.; Agarwal, A. Pathophysiology of cell phone radiation: Oxidative stress and carcinogenesis with focus on male reproductive system. Reprod. Biol. Endocrinol. 2009, 7, 114. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Duan, W.; Xu, S.; Chen, C.; He, M.; Zhang, L. Exposure to 1800 MHz radiofrequency electromagnetic radiation induces oxidative DNA base damage in a mouse spermatocyte-derived cell line. Toxicol. Lett. 2013, 218, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Esmekaya, M.A.; Ozer, C.; Seyhan, N. 900 MHz pulse-modulated radiofrequency radiation induces oxidative stress on heart, lung, testis and liver tissues. Gen. Physiol. Biophys. 2011, 30, 84–89. [Google Scholar] [CrossRef] [PubMed]
- Miao, X.; Wang, Y.F.; Lang, H.Y.; Lin, Y.Y.; Guo, Q.Y.; Yang, M.J.; Guo, J.; Zhang, Y.J.; Zhang, J.; Liu, J.Y.; et al. Preventing electromagnetic pulse irradiation damage on testis using selenium-rich cordyceps fungi. A preclinical study in young male mice. OMICS 2017, 21, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.; Ji, X.; Zhang, Y.; Miao, X.; Zou, C.X.; Lang, H.Y.; Zhang, J.; Li, Y.R.; Wang, X.W.; Qi, H.Y.; et al. MnSOD expression inhibited by electromagnetic pulse radiation in the rat testis. Electromagn. Biol. Med. 2011, 30, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Jiang, D.P.; Li, J.; Zhang, J.; Xu, S.L.; Kuang, F.; Lang, H.Y.; Wang, Y.F.; An, G.Z.; Li, J.H.; Guo, G.Z. Electromagnetic pulse exposure induces overexpression of beta amyloid protein in rats. Arch. Med. Res. 2013, 44, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Lin, Y.Y.; Guo, Q.Y.; Lang, H.Y.; Wang, R.; Liu, J.Y.; Zeng, L.H. Effects of 1.84 GHz radiation-frequency electromagnetic radiation on energy metabolism and anti-oxidative damage of male SD rat testis. High Volt. Eng. 2016, 42, 2571–2576. [Google Scholar]
- Zeni, O.; Sannino, A.; Sarti, M.; Romeo, S.; Massa, R.; Scarfi, M.R. Radiofrequency radiation at 1950 MHz (UMTS) does not affect key cellular endpoints in neuron-like PC12 cells. Bioelectromagnetics 2012, 33, 497–507. [Google Scholar] [CrossRef] [PubMed]
- Kesari, K.K.; Kumar, S.; Nirala, J.; Siddiqui, M.H.; Behari, J. Biophysical evaluation of radiofrequency electromagnetic field effects on male reproductive pattern. Cell Biochem. Biophys. 2013, 65, 85–96. [Google Scholar] [CrossRef] [PubMed]


| Days | G1 | S | G2 | |||
|---|---|---|---|---|---|---|
| Sham | Exposure | Sham | Exposure | Sham | Exposure | |
| 1 | 45.56 ± 1.22 | 43.60 ± 1.54 | 26.75 ± 1.26 | 31.66 ± 1.00 ** | 27.69 ± 2.41 | 24.74 ± 2.19 |
| 2 | 51.16 ± 0.15 | 50.73 ± 2.41 | 20.18 ± 1.10 | 23.76 ± 0.47 ** | 28.66 ± 2.02 | 25.51 ± 2.46 |
| 3 | 51.44 ± 0.81 | 42.36 ± 3.77 * | 27.49 ± 2.40 | 35.20 ± 2.28 * | 21.07 ± 1.56 | 22.44 ± 1.1 |
| 4 | 49.46 ± 3.44 | 45.38 ± 1.44 | 25.62 ± 0.45 | 27.63 ± 1.06 * | 24.92 ± 0.96 | 26.99 ± 1.04 |
| 5 | 55.38 ± 3.15 | 47.80 ± 0.84 * | 20.14 ± 0.49 | 29.88 ± 2.53 ** | 24.21 ± 1.45 | 22.32 ± 0.83 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, Y.-Y.; Wu, T.; Liu, J.-Y.; Gao, P.; Li, K.-C.; Guo, Q.-Y.; Yuan, M.; Lang, H.-Y.; Zeng, L.-H.; Guo, G.-Z. 1950MHz Radio Frequency Electromagnetic Radiation Inhibits Testosterone Secretion of Mouse Leydig Cells. Int. J. Environ. Res. Public Health 2018, 15, 17. https://doi.org/10.3390/ijerph15010017
Lin Y-Y, Wu T, Liu J-Y, Gao P, Li K-C, Guo Q-Y, Yuan M, Lang H-Y, Zeng L-H, Guo G-Z. 1950MHz Radio Frequency Electromagnetic Radiation Inhibits Testosterone Secretion of Mouse Leydig Cells. International Journal of Environmental Research and Public Health. 2018; 15(1):17. https://doi.org/10.3390/ijerph15010017
Chicago/Turabian StyleLin, Yan-Yun, Tao Wu, Jun-Ye Liu, Peng Gao, Kang-Chu Li, Qi-Yan Guo, Meng Yuan, Hai-Yang Lang, Li-Hua Zeng, and Guo-Zhen Guo. 2018. "1950MHz Radio Frequency Electromagnetic Radiation Inhibits Testosterone Secretion of Mouse Leydig Cells" International Journal of Environmental Research and Public Health 15, no. 1: 17. https://doi.org/10.3390/ijerph15010017




