1. Introduction
Chitosan is a cationic polysaccharide (obtained from the deacetylation of chitin, coming from the shells of crustaceans and insect exoskeletons) that is a suitable packaging material due to its biocompatibility, biodegradability, non-toxicity, film-forming and antimicrobial properties. This polymer is currently used in food packaging, agriculture, biomedical science and cosmetics. It can be used as a transparent film or coating to improve food quality and to extend its shelf life. Chitosan is one of the most abundant natural biopolymers with proven antimicrobial, antioxidant, and antifungal/viral properties [
1,
2,
3].
Starch is a biodegradable and thermoplastic polymer formed mainly by two polysaccharides: amylose and amylopectin. In recent research, this polymer is considered to be a promising agent for applications such as food packaging due to its biodegradability and flexibility [
3]. In this study, starch from rice was used, which is a less studied starch source. According to Jiang et al. [
4], the starch from rice is convenient for preparing chitosan-starch films because of its relatively small particle size (2–8 µm) and its hypoallergenic quality, which make it an appropriate material for food industry utilization.
On the other hand, antioxidants such as anthocyanins, betalains, thymol, carvacrol and resveratrol have attracted attention because of their recently discovered antimicrobial properties. Anthocyanins are natural, water-soluble colorants obtained from plants, flowers and fruits having orange, pink, red, violet and blue colors. Also, they have been demonstrated to be useful for the prevention of several diseases. Their stability is influenced by several factors such as pH, storage temperature, chemical structure, concentration, light, oxygen, solvents and metals [
5]. In this research, natural anthocyanins extracts obtained from cranberry (
Vaccinium oxycoccus), blueberry (
Vaccinium myrtillus) and pomegranate (
Punica granatum) were analyzed.
Betalains are pigments found in red–violet (betacyanins) or yellow (betaxanthins) plants, vegetables and fruits that play a key role in human health. Natural extracts containing betalains (betacyanins) from beetroot (
Beta vulgaris) and pitaya (
Hylocereus undatus) were investigated in this research. Beetroot contains two major pigments: betanin (a red betacyanin) and vulgaxanthine I (a yellow betaxanthin). They have antioxidant and antimicrobial properties. Pitaya is rich in betacyanins such as betanin, phyllocactin, hylocerenin and their isomers [
6]. The stability of betalains is influenced by many factors such as temperature (the most important factor), pH, water activity, light, the presence or absence of oxygen, and enzymatic action [
7].
Thymol and carvacrol are monoterpenes found in spices such as thyme and oregano. They are highly appreciated because of their antioxidant and antimicrobial activity. Owing to their phenolic nature, they are highly efficient as bactericidal agents [
8,
9]. In this study, natural extracts of thymol and carvacrol obtained from oregano (
Origanum vulgare) were evaluated.
Resveratrol is a polyphenol produced by plants such as grapes, blueberries, mulberries, cranberries and peanuts, in response to any physical damage, UV light, and fungi presence [
10]. In recent years, this compound has been the object of several studies because of its advantageous effects on human health and its antioxidant activity [
11]. In this work, resveratrol extracted from grape (
Vitis vinifera) was used for the film preparation.
The use of anthocyanins improves the performance of chitosan films in food applications according to Ge et al. [
12]. Furthermore, as stated by the results of the investigation of Berizi et al. [
13], the addition of pomegranate peel extract to a chitosan solution prevents the oxidation of fats and proteins of rainbow trout, while enhancing the chemical, antimicrobial and textural properties of a packaging film that was used to preserve the rainbow trout for a storage period of up to six months. According to Avila-Sosa et al. [
14], the combination of chitosan and oregano is becoming an appropriate material to substitute for some of the synthetic polymers used in food packaging applications. This kind of biomaterial offers desirable properties such as antimicrobial, mechanical and barrier properties.
The present article reports the synthesis and characterization of chitosan-starch films modified by the addition of cranberry, blueberry, beetroot, pomegranate, oregano, pitaya and resveratrol aqueous extracts. The influence of these natural antioxidant extracts on the properties of chitosan-starch films was evidenced through an innovative method aiming to enhance the chemical, morphological and thermal properties of the films, as well as their coloration and apparent density. The method proposed in this research is simple, effective and inexpensive because it implies the addition of organic, non-toxic and naturally available extracts, reducing costs, the use of solvents and time. These materials also presented improved antimicrobial, mechanical and optical properties as shown in a previously reported investigation [
15]. These results were correlated with those obtained in that research in order to have a more comprehensive understanding of the influence of natural extracts in the chitosan-starch blend. The overall performance of the modified films suggests that most of these materials are promising candidates for further food packaging applications. Improving these features is important because it allows the proposed materials to become more effective than unmodified chitosan-starch film against the two main causes of food decomposition: the presence of micro-organisms and the degradation of lipids.
4. Materials and Methods
4.1. Materials
The materials used for the film synthesis are detailed as follows: chitosan (medium molecular weight and a degree of deacetylation of 85%) and rice starch were purchased from Sigma-Aldrich (Toluca, Edo. Mex., Mexico), glacial acetic acid (99.9% purity) and glycerol (99.7% purity) were purchased from Fermont (Monterrey, NL, Mexico). Natural antioxidants were obtained from raw fruits: cranberry, blueberry, beetroot and pitaya, from dry oregano, commercial cranberry and pomegranate juices which were purchased in a local market. Resveratrol capsules were purchased from General Nutrition Centers (Pittsburgh, PA, USA). The extracts of blueberry, beetroot and pitaya were obtained from raw fruit pulp at 25 °C and were filtered prior to their addition in the chitosan–starch–glycerol blend. An aqueous extract of oregano was prepared at 5% (w/v) using dry oregano and water as a solvent; the solution was kept at 65 °C for 10 min, under magnetic agitation and further filtration. Commercial organic cranberry and pomegranate juices were used; the extract was filtered at 25 °C before its addition to the blend. The resveratrol solution at 5% (w/v) was prepared using water as a solvent at 25 °C, and then it was filtered. It was very important to obtain the extracts of cranberry, blueberry, beetroot, pomegranate and pitaya at 25 °C in order to preserve their antioxidants. The extracts were obtained in this manner because one of the main objectives of the study was to propose a cheap and simple method of getting the extracts, that does not involve complex leaching or extraction procedures that require using toxic solvents. The natural aqueous extracts and the film matrix solutions were compatible. For the quantification of antioxidants, Folin-Denis reagent (quality for determination of phenols) and gallic acid (97.5–102.5% titration) were purchased from Sigma-Aldrich (Toluca, Edo. Mex., Mexico), ethanol (99.9% purity) was purchased from Fermont (Monterrey, NL, Mexico) and sodium carbonate (anhydrous) was purchased from Merck (Naucalpan de Juarez, Edo. Mex., Mexico).
4.2. Synthesis of Films
The chitosan was dissolved in a glacial acetic acid solution (1%
v/
v) in order to prepare a chitosan solution at 2% (
w/
v). The starch solution (2%
w/
v) was prepared at 90 ± 2 °C for 20 min using constant stirring. The solution was kept at room temperature (25 °C approximately) for the films preparation. Each film was prepared by mixing 40 mL of chitosan solution with 40 mL of rice starch solution. After that, 0.2 mL of glycerol were added as plasticizer. Then, the extracts which contains natural antioxidants were added in three different quantities referred to the amount of the total weight of the chitosan-starch blend: 0.4, 1.6 and 4 mL (corresponding to 0.5%, 2% and 5%
v/
v, respectively). Finally, the mixture was stirred for 5–10 min and poured into a polystyrene tray [
52]. The drying process took 15 days at controlled room temperature (25 °C). The experimental design consists of twenty-two films: one control film (QS2) and twenty-one films with natural extracts (three films for each extract). The complete experimental design and the film nomenclature are presented in
Table 6. Three replications per sample were prepared.
4.3. Evaluation of Films Coloration
The evaluation of film coloration was carried out visually on day 1 and compared with that observed on day 15, in order to understand the factors involved in the coloration changes and their relationship with other film properties. Furthermore, the coloration was evaluated by means of an analyzer software Image Color Summarizer (Canada) [
53]. RGB values of each film were obtained through this software using photos taken on day 1 and day 15. During the image acquisition, each film was placed on a white background; the distance between the film and the camera was approximately 25 cm. The images were captured using a 5 megapixels camera at resolution of 480 × 800 pixels with Autofocus and CMOS image sensor, using two fluorescent lights for illumination in a closed lab room to avoid other light sources. All the images were saved and further processed using a personal computer and an image analyzer software. RGB is a color model based on additive synthesis (representation of a color by mixing of the three primary light colors: red (R), green (G) and blue (B). According to this model, red color is formed by RGB values of 255, 0, 0, while green and blue correspond to 0, 255, 0 and 0, 0, 255, respectively (monochromatic colors). The absence of color (black) is obtained when the values of RGB are 0, 0, 0. The yellow color is obtained with RGB values 255, 255, 0, the cyan color appears at RGB values 0, 255, 255 and the magenta color at RGB values 255, 0, 255 (intermediate colors). The white color is formed at the RGB maximum values 255, 255, 255 [
16].
4.4 Apparent Density
This film property was measured according to the procedure used by Ahmed et al. [
54]. Every film sample was cut in a circular shape and then it was weighed. Also, its diameter and height were measured. The apparent density (ρ) was obtained by the following Equation (1):
where
D = sample diameter
H = sample height
W = sample weight.
4.5 Fourier Transform Infrared Spectroscopy (FTIR)
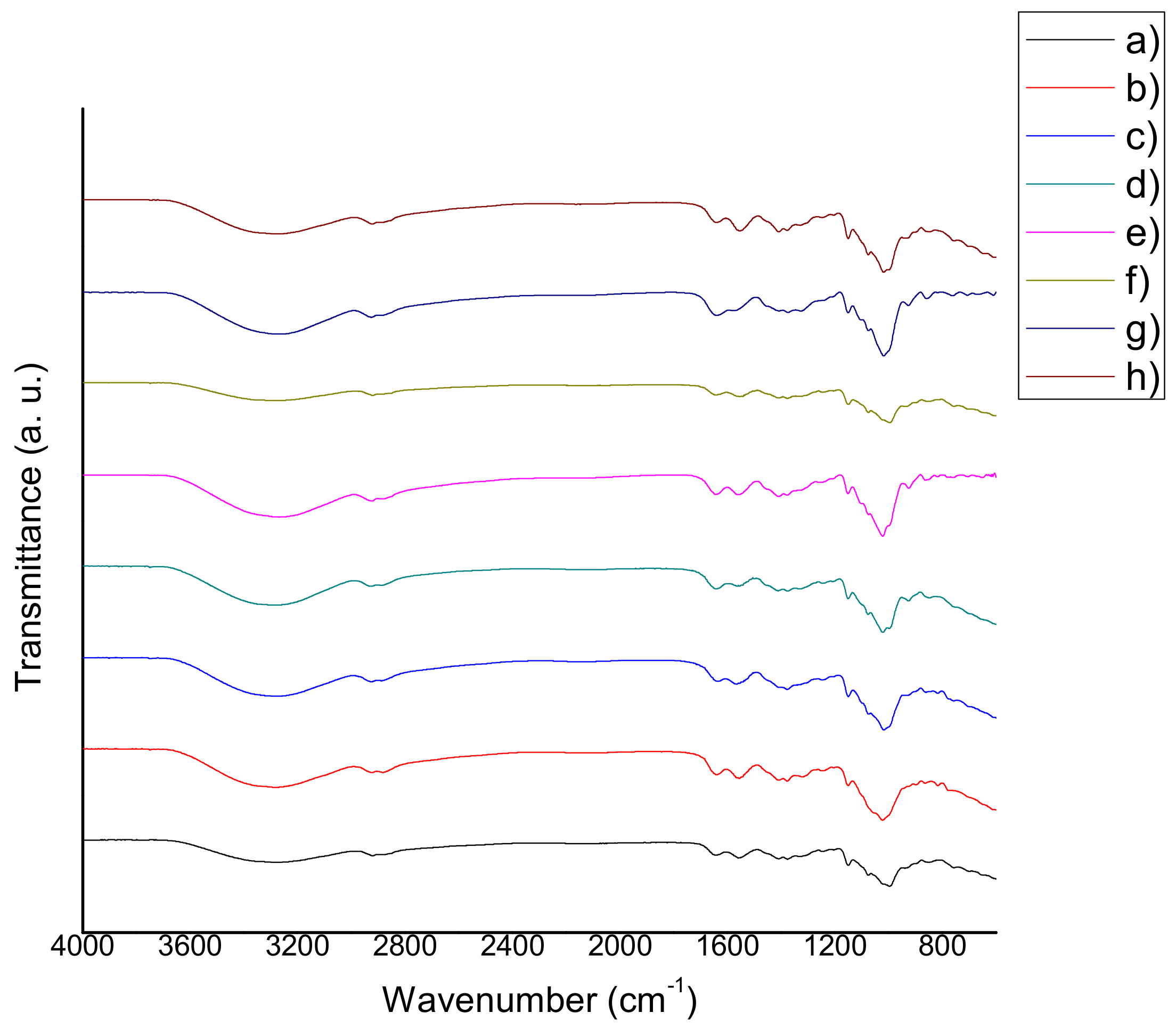
The chitosan-starch films were analyzed by means of FTIR spectroscopy equipment (Perkin Elmer Spectrum 100, Perkin Elmer, Waltham, MA, USA) in order to determine the presence of functional groups and to elucidate interactions among the film compounds. The analysis was carried out using an attenuated total reflection (ATR) method, with a total of 12 scans and a resolution of 4 cm−1. FTIR spectra were recorded from 4000 cm−1 to 600 cm−1 in the wavenumber scale.
4.6. Determination of Antioxidant Capacity of the Films
4.6.1. Extraction
Based on a modification of the method published by Adom et al. [
55], the natural antioxidants were gotten from 0.25 g of each film. Four milliliters of ethanol (99.9% purity) were added to each sample. This solvent was selected because it is compatible with the polar nature of the antioxidants. Afterwards, the samples were put in a water bath (Polystat 12050-00 Circulator, Cole-Parmer, Vernon Hills, IL, USA) at 25 °C and 90 rpm for 10 min. Then, the supernatant of each sample was centrifuged (Thermo Microlite-RF, Thermo Fisher Scientific, Waltham, MA, USA) at 19 °C and 13,000 rpm for 10 min. The procedure was repeated two times more. Finally, the extracts were stored and protected from light.
4.6.2. Qualification of Natural Antioxidants
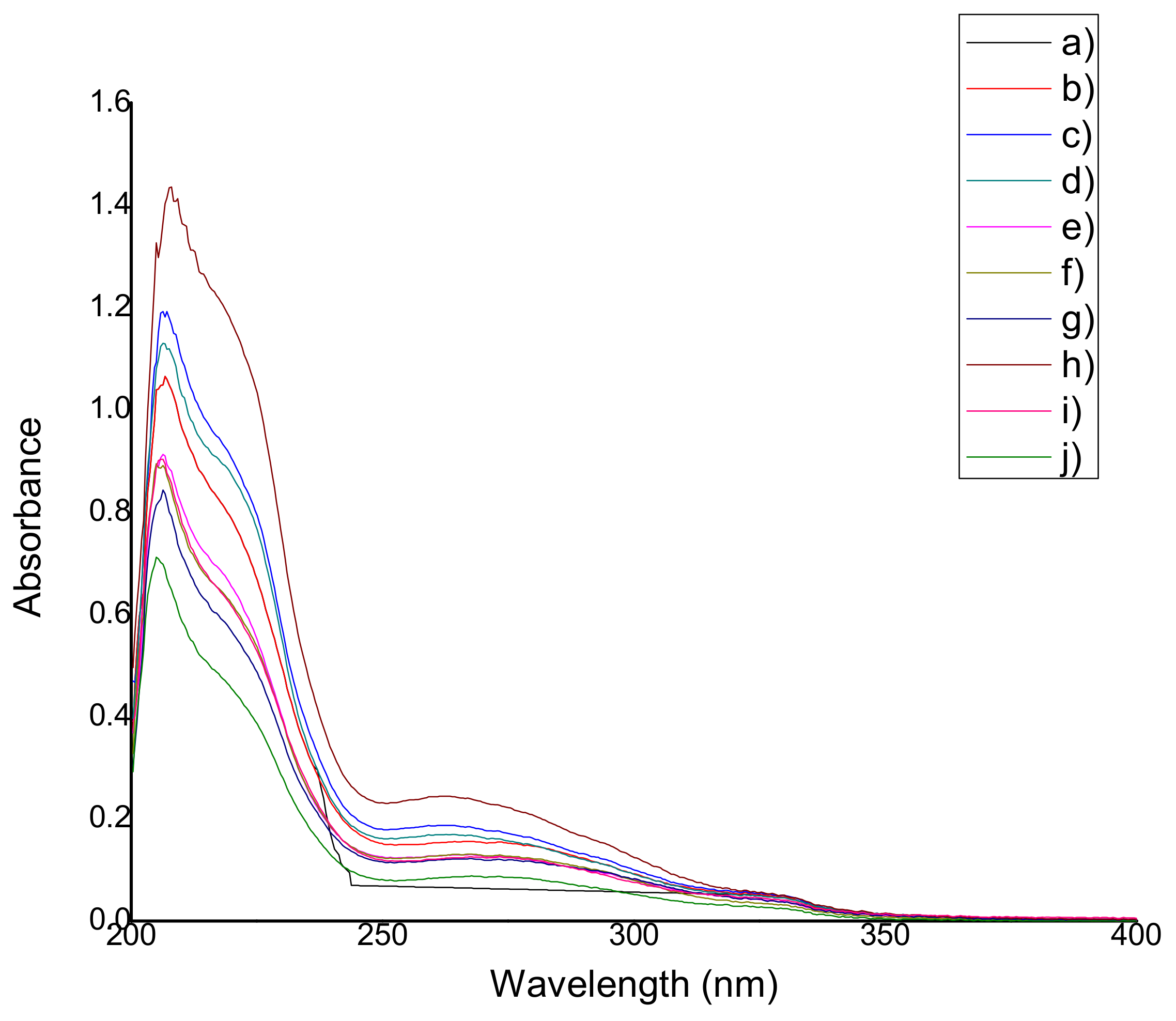
Each extract was analyzed in triplicate using a UV-Vis spectrophotometer (Cintra 303, GBC Scientific Equipment, Mexico City, Mexico) with the purpose of detecting the presence of antioxidants. UV spectra were recorded within the wavelength interval ranging from 200 nm to 400 nm. The antioxidants were identified through their characteristic peaks: anthocyanins at 280 nm according to Lee et al. [
22], thymol and carvacrol at 270 nm according to Amadio et al. [
23], betalains and resveratrol at 280–350 nm according to Jaworska et al. [
24].
4.6.3. Antioxidant Capacity (Reducing Capacity)
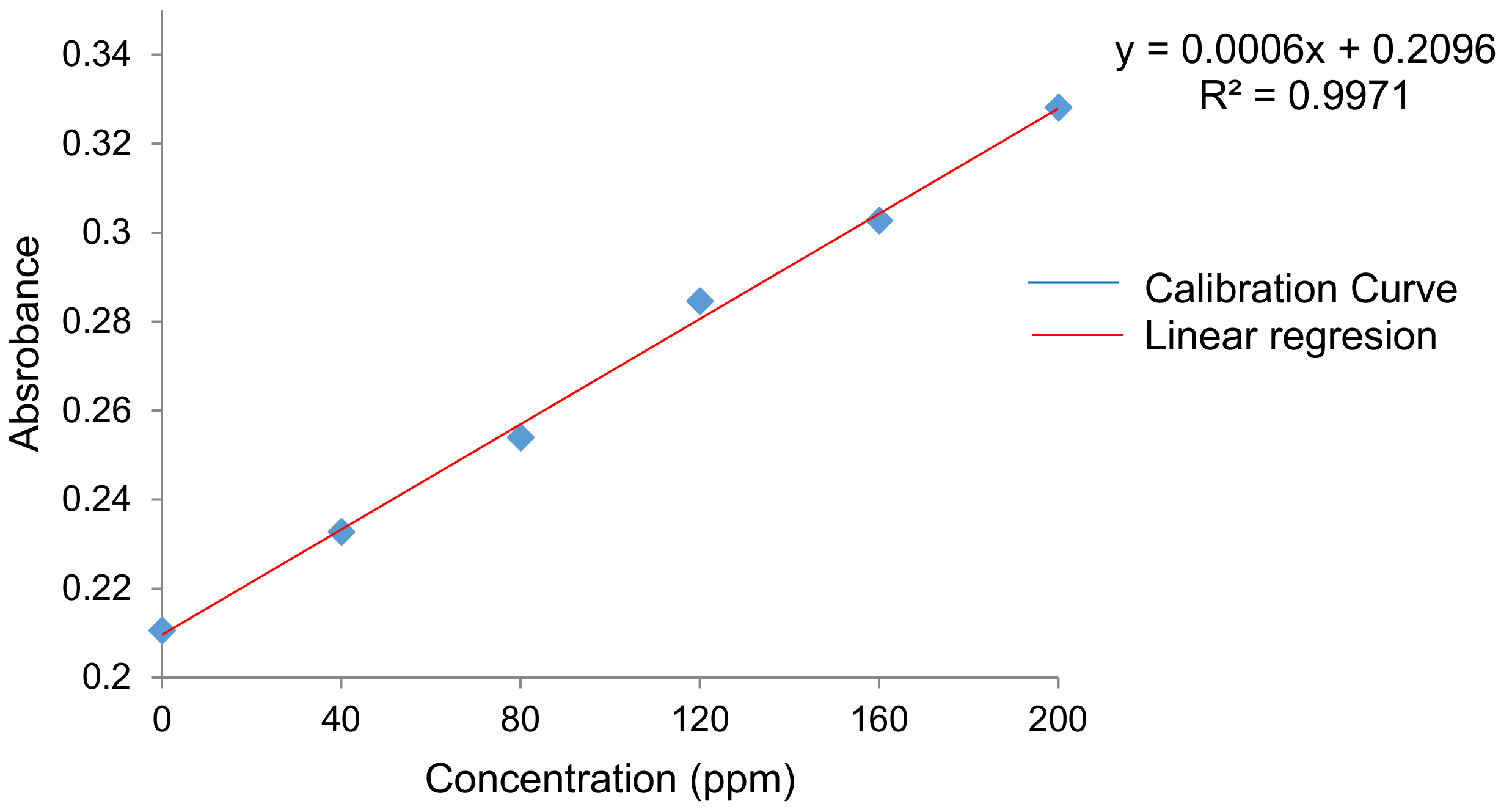
A modification of the method used by Singleton et al. [
56] was applied to determine the antioxidant capacity of each film. This modified method consisted in using 0.3 mL of film extract, 1.5 mL of Folin-Denis reagent (diluted 10 times in distilled water) and 1.2 mL of sodium carbonate (7.5%
w/
v) for analyzing each film. The samples were stirred for a few minutes, and then covered with parafilm and kept at room temperature for 30 min. Absorbance at 765 nm of each extract was measured in triplicate using a UV-Vis spectrophotometer (Cintra 303). The antioxidant capacity was expressed as mg/mg equivalent of gallic acid [
22,
57,
58,
59]. A calibration curve of gallic acid was performed using solutions (in 99.9% purity ethanol) at different concentrations (0, 40, 80, 120, 160 and 200 ppm) [
60,
61]. The antioxidant capacity was calculated through the following regression model (2):
4.7. Scanning Electronic Microscopy (SEM)
The films morphology was analyzed by means of a scanning electronic microscope (JEOL JSM 7100F, JEOL USA, Inc., Peabody, MA, USA) using magnifications of 500×, 750×, 1500× and 5000×. A carbon strip was used to hold each sample during the analysis.
4.8. Thermogravimetric Analysis (TGA)
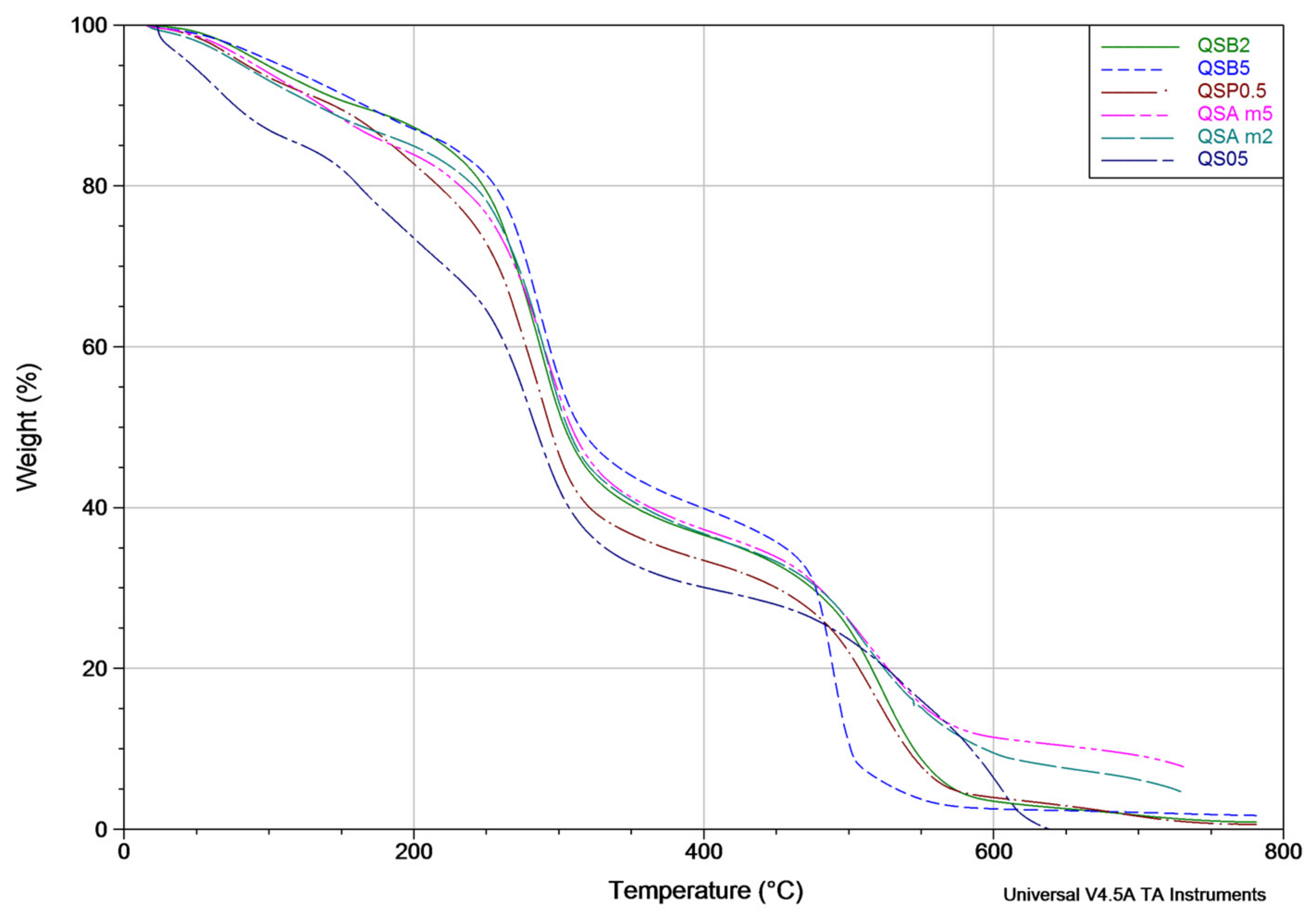
The thermal stability of the films was determined using a thermogravimetric analyzer (TA Instruments TGA Q500 V20.13 Build 39, TA Instruments, New Castle, DE, USA), under a temperature interval of 25–600 °C, a scan speed of 10 °C/min, sample weights of 15–22 mg and air as the purge gas.
4.9. Statistical Analysis
Statistical evaluation presented in
Table 1,
Table 2,
Table 3,
Table 4 and
Table 5 were performed using MS Excel (2013 version, Microsoft, Redmond, WA, USA). Multi-factor analysis of variance (ANOVA) using Minitab (16 version, Pennsylvania State University, State College, PA, USA) was used to determine if the type of extract, the type of antioxidant, the quantity of extract added to each film, or the interaction of these factors had a significant impact on the antioxidant capacity of the films. The statistically significant differences between means were evaluated using the Tukey’s test (α = 0.05).
5. Conclusions
Based on the results that were previously discussed, the addition of natural extracts gives chitosan-starch a higher apparent density values. According to the FTIR results, the only major difference among the film’ spectra is the intensity of the signals (% transmittance) and the width of the –OH band located at 3400–3300 cm−1. The FTIR spectra are similar, because both chitosan and starch have cyclic structures and multiple –OH bonds which are typical of saccharides, water, carboxylic acids, antioxidants, etc. The presence of the antioxidants in the films could be deduced by the observation of film coloration as well as the UV-Vis spectroscopy. From the results obtained in the UV-Vis analysis, it was confirmed that the films show characteristic peaks at 260–275 nm (hypochromic shifts were observed in all cases) indicating the presence of natural antioxidants in small amounts (there was no significant differences between the quantities). It is important to point out that the amount of extract (not of antioxidants) is related to antimicrobial properties. The amount of extracts does not have an influence on the film surface structure and the antimicrobial activity appears to be independent of the film surface structure. Based on the results obtained in the TGA analyses performed on some samples, a relationship between the amount of extract and the improvement in the film thermal properties was determined, and in particular, QSAm2, QSAm5 and QSO5 films proved to suffer the lowest weight losses among the studied samples, which implies that these films are the most thermally-stable and also reveals the preponderant role of anthocyanins and thymol-carvacrol.
An important finding of this study was that improving the antimicrobial activity against mesophilic aerobic and coliforms bacteria and fungi [
15] does not necessarily imply strong differences among the antioxidant capacities of the modified films. Moreover, the addition of natural extracts provided chitosan-starch films with better thermal and physical properties, making them suitable for food packaging applications, because they proved to be non-toxic, cheap and effective against micro-organisms and light. Although the type as well as the quantity of antioxidant have a significant effect on the final properties of the films, the results indicate that the influence exerted by the type of antioxidant was more substantial than that of the quantity that was added. Future research directions include a further study on the application of these films in food packaging and the sensory properties test. In addition, interesting research matter will come from looking for alternative antioxidant sources that are able to offer an optimum balance between sustainability, efficiency and applicability in terms of their antimicrobial, mechanical, optical, physical, morphological and thermal properties.