Biocompatibility of Collagen Membranes Assessed by Culturing Human J111 Macrophage Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Confocal microscopy
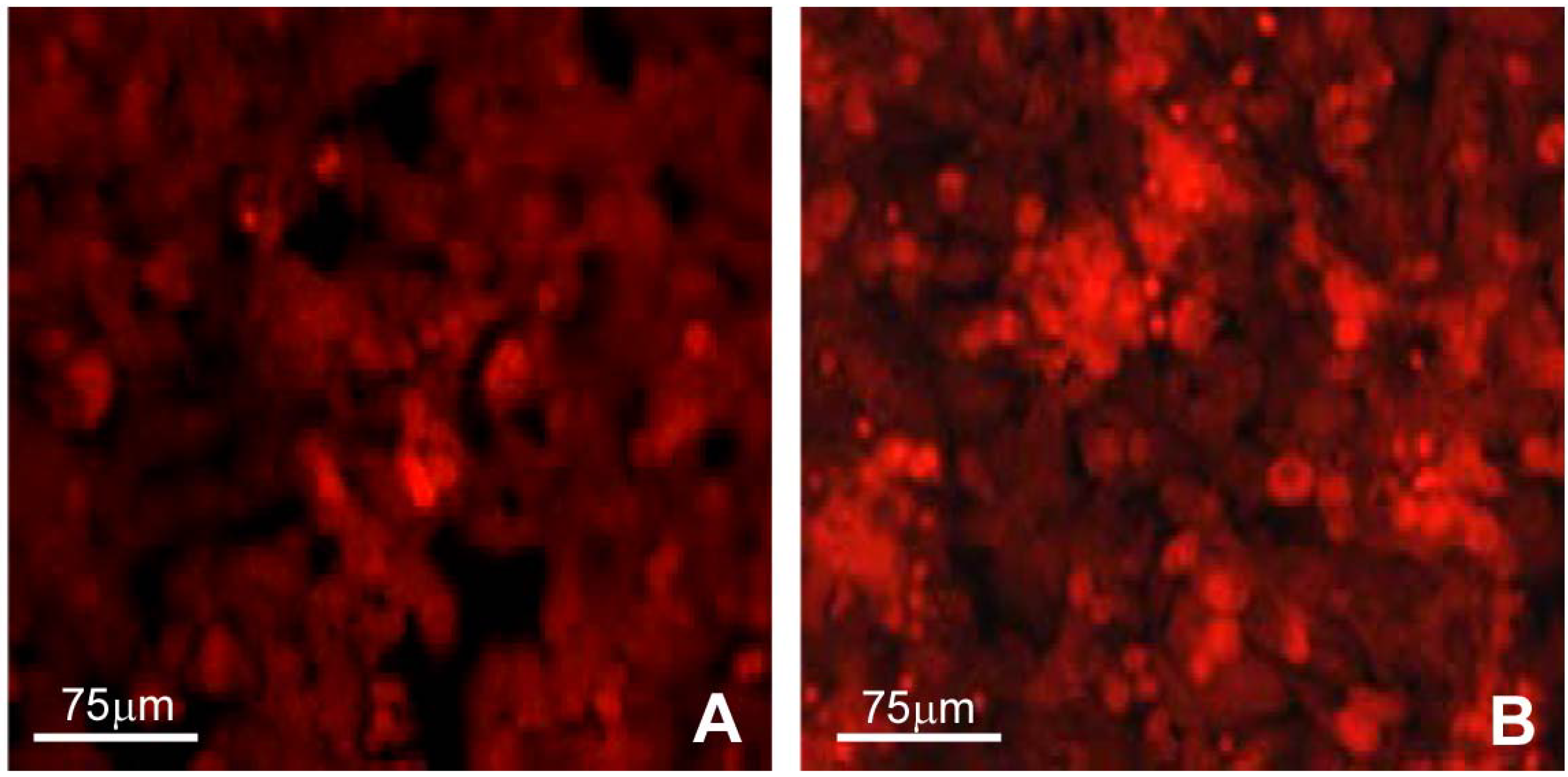
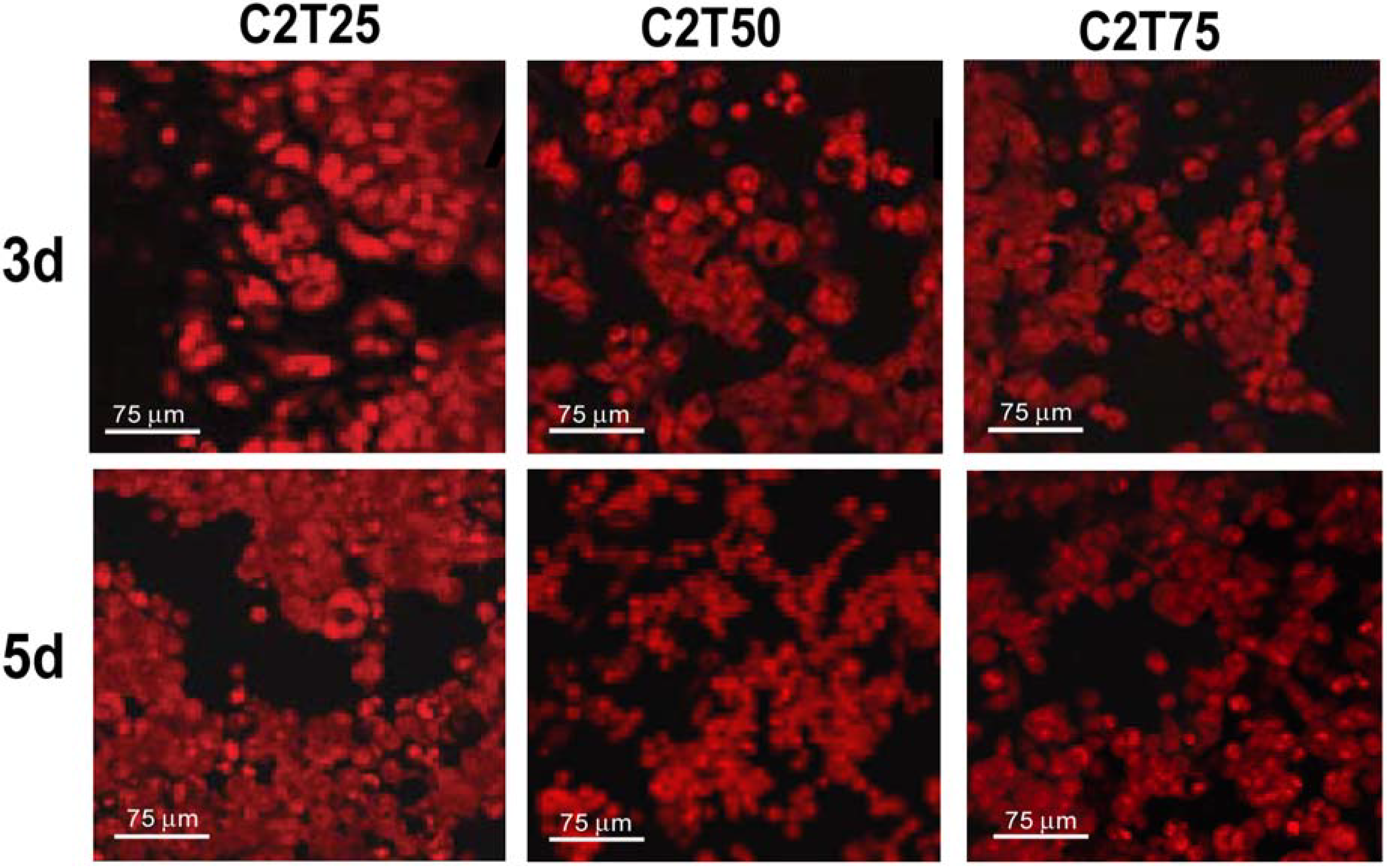

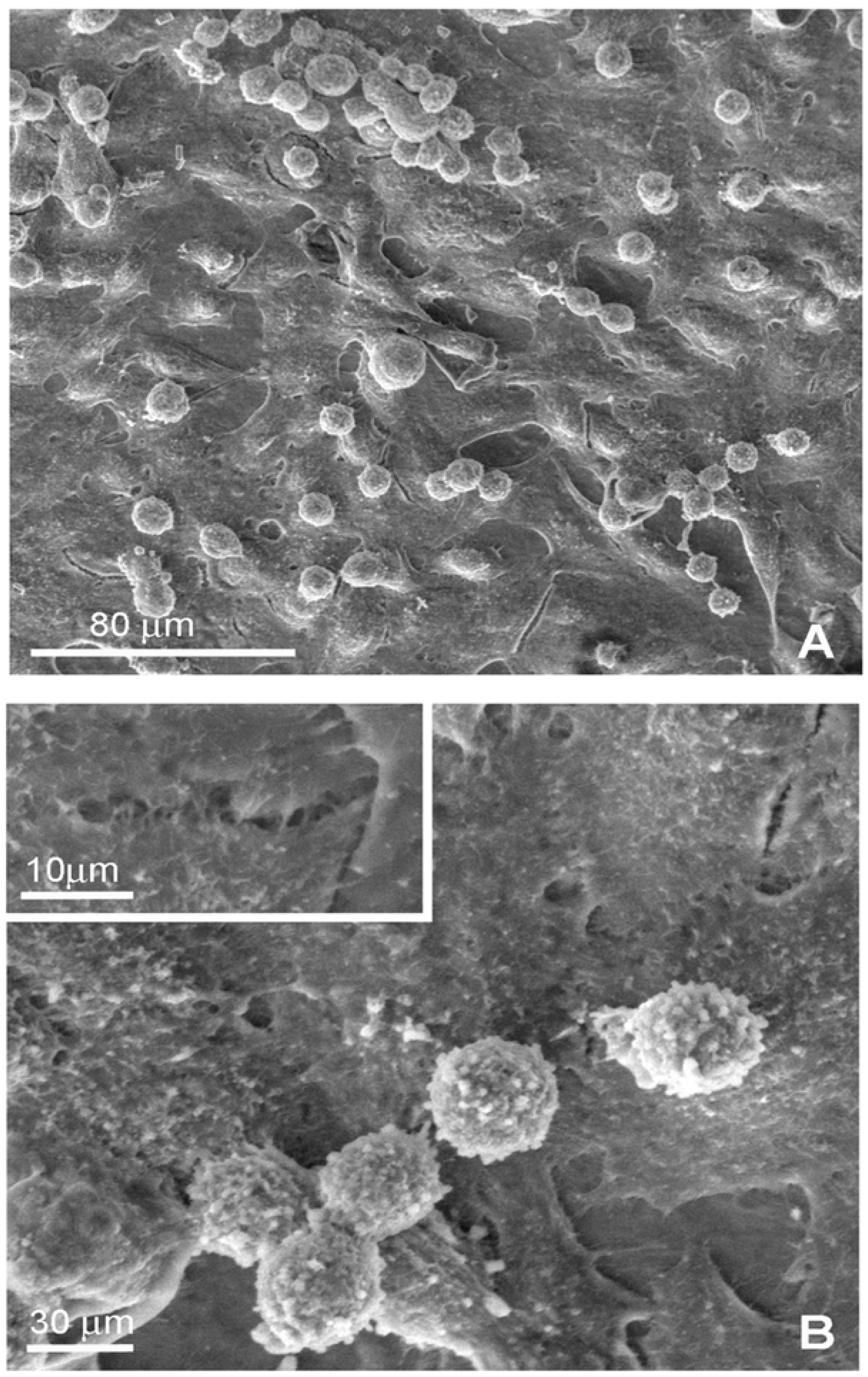
2.2. Environmental Scanning Electron Microscopy (ESEM)

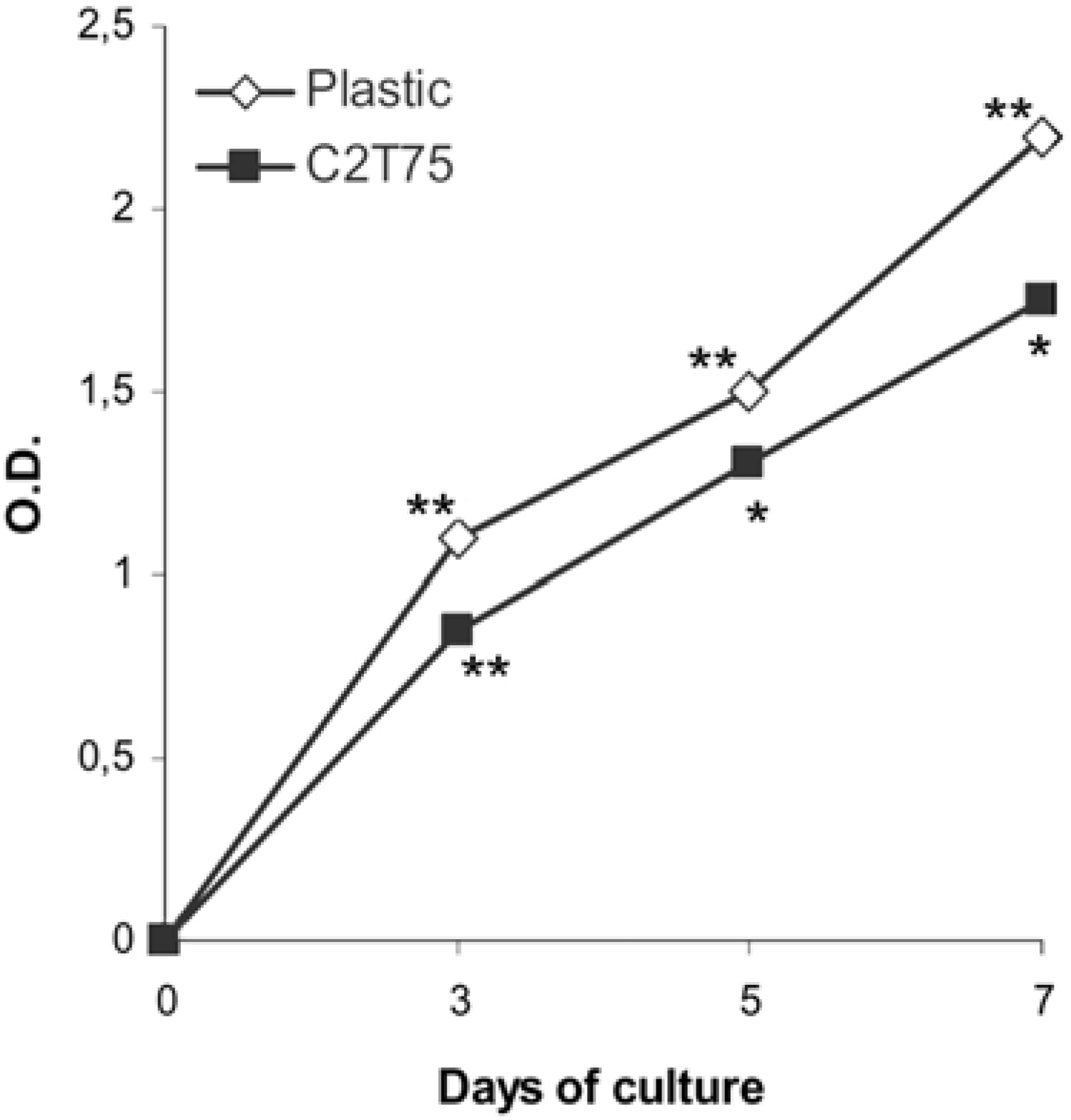
2.3. Macrophage proliferation
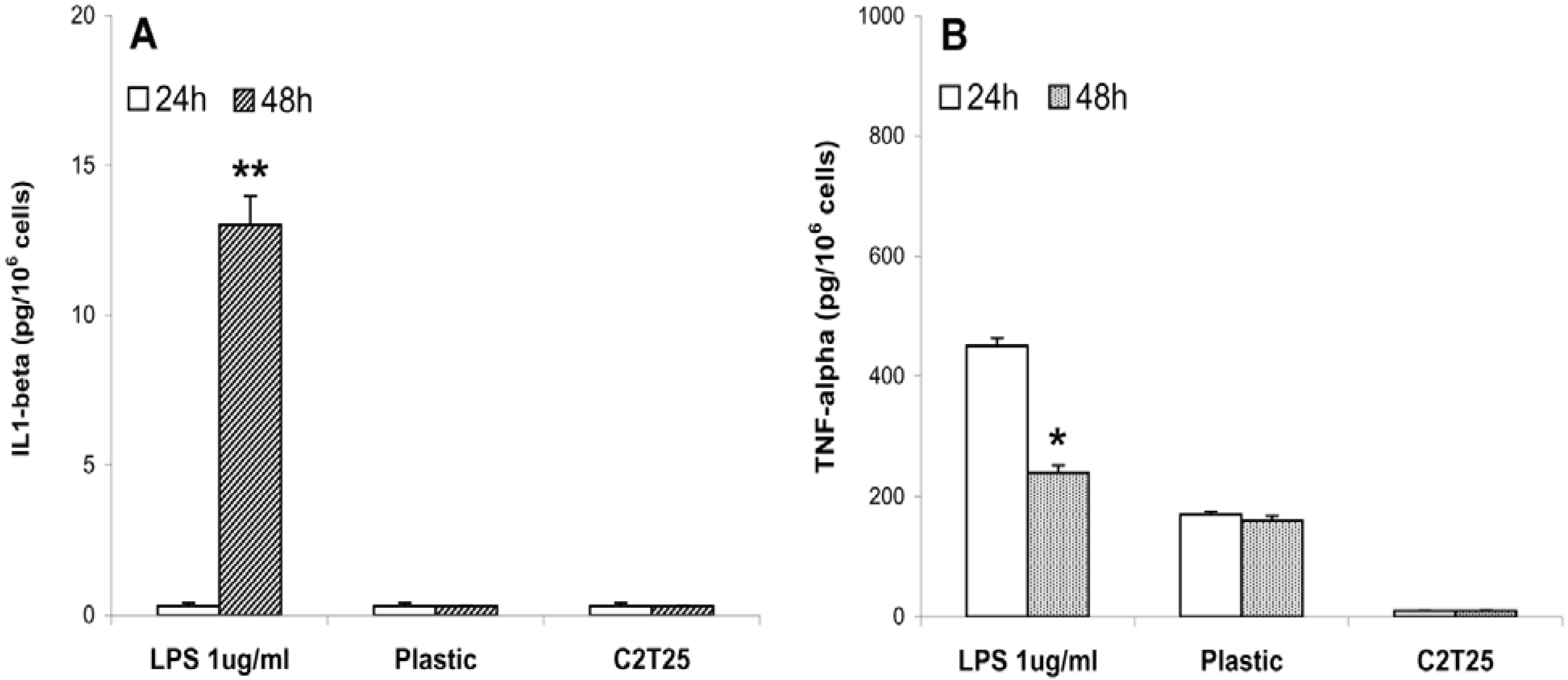
2.4. Cytokine production
3. Experimental Section
3.1. Preparation of collagen membranes
3.2. Macrophage culture
3.3. Fluorescence optical and confocal microscopy
3.4. Environmental Scanning Electron Microscopy (ESEM)
3.5. MTT assay
3.6. Cytokine production
4. Conclusions
References
- Badylak, S.F. The extracellular matrix as a scaffold for tissue reconstruction. Semin. Cell Dev. Biol. 2002, 13, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Van der Rest, M.; Garrone, R. Collagen family of protein. FASEB J. 1991, 5, 2814–2823. [Google Scholar]
- Rao, K.P. Recent developments of collagen-based materials for medical applications and drug delivery systems. J. Biomater. Sci. Polym. 1995, 7, 623–645. [Google Scholar]
- Lee, C.H.; Sigla, A.; Lee, Y. Biomedical application of collagen. Int. J. Pharm. 2001, 221, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Stone, K.R.; Steadman, J.R.; Rodkey, W.G.; Li, S.T. Regeneration of meniscal cartilagine with use of a collagen scaffold. Analysis of preliminary data. J. Bone Joint Surg. Am. 1997, 79, 1770–1777. [Google Scholar] [PubMed]
- Hillmann, G.; Gebert, A.; Geurtsen, W. Matrix expression and proliferation of primary gingival fibroblasts in a three-dimensional cell culture model. J. Cell Sci. 1999, 122, 2823–2832. [Google Scholar]
- Fuss, M.; Ehlers, E.M.; Russlies, M.; Rohwedel, J.; Behrens, P. Characteristics of human chondrocytes, osteoblasts and fibroblasts seeded onto a type I/III collagen sponge under different culture conditions. A light, scanning and transmission electron microscopy study. Ann. Anat. 2000, 182, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Bilir, A.; Aybar, B.; Tanrikulu, S.H.; Issever, H.; Tuna, S. Biocompatibility of different barrier membranes in cultures of human CRL 11372 osteoblast-like cells: An immunohistochemical study. Clin. Oral Implants Res. 2007, 18, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, J. C. Living skin equivalents and their application in wound healing. Clin. Podiatri. Med. Surg. 1998, 15, 129–137. [Google Scholar]
- Lamme, E.N.; van Leeuwen, R.T.; Jonker, A.; van Marle, J.; Middelkoop, E. Living skin substitutes: Survival and function of fibroblasts seeded in a dermal substitute in experimental wounds. J. Invest. Dermatol. 1998, 111, 989–995. [Google Scholar] [CrossRef] [PubMed]
- Mansbridge, J.; Liu, K.; Patch, R.; Symons, K.; Pinney, E. Three-dimensional fibroblast culture implant for the treatment of diabetic foot ulcers: Metabolic activity and therapeutic range. Tissue Eng. 1998, 4, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Machens, H.G.; Berger, A.C.; Mailaender, P. Bioartificial skin. Cells Tissues Organs 2000, 167, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Daamen, W.F.; van Moerkerk, H.T.; Hafmans, T.; Buttafoco, L.; Poot, A.A.; Veerkamp, J.H.; van Kuppevelt, T.H. Preparation and evaluation of molecularly-defined collagen–elastin–glycosaminoglycan scaffolds for tissue engineering. Biomaterials 2003, 24, 4001–4009. [Google Scholar] [CrossRef]
- Gibson, A.J.; McDonnel, S.M.; Price, A.J. Matrix-induced autologous chondrocyte implantation. Oper. Tech. Orthop. 2006, 16, 262–265. [Google Scholar] [CrossRef]
- Abelow, S.P.; Guillen, P.; Ramos, T. Arthroscopic technique for matrix-induces autologous chondrocyte implatation for the treatment of large chondral defects in the knee and ankle. Oper. Tech. Orthop. 2006, 16, 257–261. [Google Scholar]
- Gigante, A.; Bevilacqua, C.; Ricevuto, A.; Mattioli-Belmonte, M.; Greco, F. Membrane-seeded autologous chondrocytes: cell viability and characterization at surgery. Knee Surg. Sports Traumatol. Arthrosc. 2007, 15, 88–92. [Google Scholar] [CrossRef]
- Gigante, A.; Moschini, A.; Verdenelli, A.; Del Torto, M.; Ulisse, S.; de Palma, L. Open versus percutaneous repair in the treatment of acute Ahilles tendon rupture: A randomized prospective study. Knee Surg. Sports Traumatol. Arthrosc. 2008, 16, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Gigante, A.; Enea, D.; Greco, F.; Bait, C.; Denti, M.; Schonhuber, H.; Volpi, P. Distal realignment and patellar autologous chondrocyte implantation: Mid-term results in a selected population. Knee Surg Sports Traumatol. Arthrosc. 2009, 17, 2–10. [Google Scholar] [CrossRef]
- Sosio, C.; Boschetti, F.; Bevilacqua, C.; Mangiavini, L.; Scotti, C.; Buragas, M.S.; Diressi, S.; Fraschini, G.; Gigante, A.; Peretti, G.M. Effect of blood on the morphological, biochemical and biomechanical properties of engineered cartilage. Knee Surg. Sports Traumatol. Arthrosc. 2007, 15, 1251–1257. [Google Scholar] [CrossRef] [PubMed]
- Singer, A.J; Clark, R.A. Cutaneous wound healing. N. Engl. J. Med. 1999, 341, 738–746. [Google Scholar] [CrossRef]
- Sefton, M.V.; Woodhouse, K.A. Tissue engineering. J. Cutan. Med. Surg. 1998, Suppl 1, 18–23. [Google Scholar]
- Schor, S.L.; Allen, T.D.; Harrison, C.J. Cell migration through three-dimensional gels of native collagen fibres: Collagenolytic activity is not required for the migration of two permanent cell lines. J. Cell Sci. 1980, 46, 171–186. [Google Scholar] [PubMed]
- Mio, T.; Adachi, Y.; Romberger, D.J.; Ertl, R.F.; Rennard, S.I. Regulation of fibroblast proliferation in three-dimensional collagen gel matrix. In Vitro Cell Dev. Biol. Anim. 1996, 32, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.J.; Rhodes, R.K. Preparation and characterization of the different type of collagen. In Methods in Enzymology; Cunningham, L.W., Frederiksen, D.W., Eds.; Academic Press: New York, NY, USA, 1982; Chapter 82; pp. 33–64. [Google Scholar]
- Bianchini, P.; Parma, B. Immunological safety evaluation of a horse collagen haemostatic pad. Arzneim-Forsch. 2001, 51, 414–419. [Google Scholar]
- Croce, M.A.; Silvestri, C.; Guerra, D.; Carnevali, E.; Boraldi, F.; Tiozzo, R.; Parma, B. Adhesion and proliferation of human dermal fibroblast on collagen matrix. J. Biomater. Appl. 2004, 18, 209–222. [Google Scholar]
- Kawamura, A. Fluorescent Antibody Techniques and Their Application; University of Tokyo Press: Tokyo, Japan, 1969. [Google Scholar]
- Wang, L.J.; Colella, R.; Roisen, F.J. Ganglioside GM1 alters neuronal morphology by modulating the association of map2 with microtubules and actin filaments. Brain Res. Dev. 1998, 105, 227–239. [Google Scholar] [CrossRef]
- Mosmann, T. A rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Ciapetti, G.; Granchi, D.; Stea, S.; Savarino, L.; Verri, E.; Gori, A.; Salvioli, F.; Montanaro, L. Cytotoxicity testing of matrials with limited in vivo exposure is affected by the duration of cell-material contact. J. Biomed. Mater. Res. 1998, 42, 485–490. [Google Scholar] [CrossRef] [PubMed]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Aruta, C.G.; Croce, M.A.; Quaglino, D.; Guerra, D.; Tiozzo, R. Biocompatibility of Collagen Membranes Assessed by Culturing Human J111 Macrophage Cells. Materials 2009, 2, 945-957. https://doi.org/10.3390/ma2030945
Aruta CG, Croce MA, Quaglino D, Guerra D, Tiozzo R. Biocompatibility of Collagen Membranes Assessed by Culturing Human J111 Macrophage Cells. Materials. 2009; 2(3):945-957. https://doi.org/10.3390/ma2030945
Chicago/Turabian StyleAruta, Claudia Gaetana, Maria Antonietta Croce, Daniela Quaglino, Deanna Guerra, and Roberta Tiozzo. 2009. "Biocompatibility of Collagen Membranes Assessed by Culturing Human J111 Macrophage Cells" Materials 2, no. 3: 945-957. https://doi.org/10.3390/ma2030945



