The Incidence and Genetic Diversity of Apple Mosaic Virus (ApMV) and Prune Dwarf Virus (PDV) in Prunus Species in Australia
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material and Total RNA Extraction
2.2. Reverse Transcription Polymerase Chain Reaction (RT-PCR) Amplification
2.3. Amplicon High-Throughput Sequencing (HTS) Library Preparation and Sequence Reads Analysis
2.4. Phylogenetic and Sequence Identity Analysis
3. Results
3.1. RT-PCR Detection of ApMV and PDV
3.2. Amplicon Next Generation Sequencing Data and Read Cluster Analysis
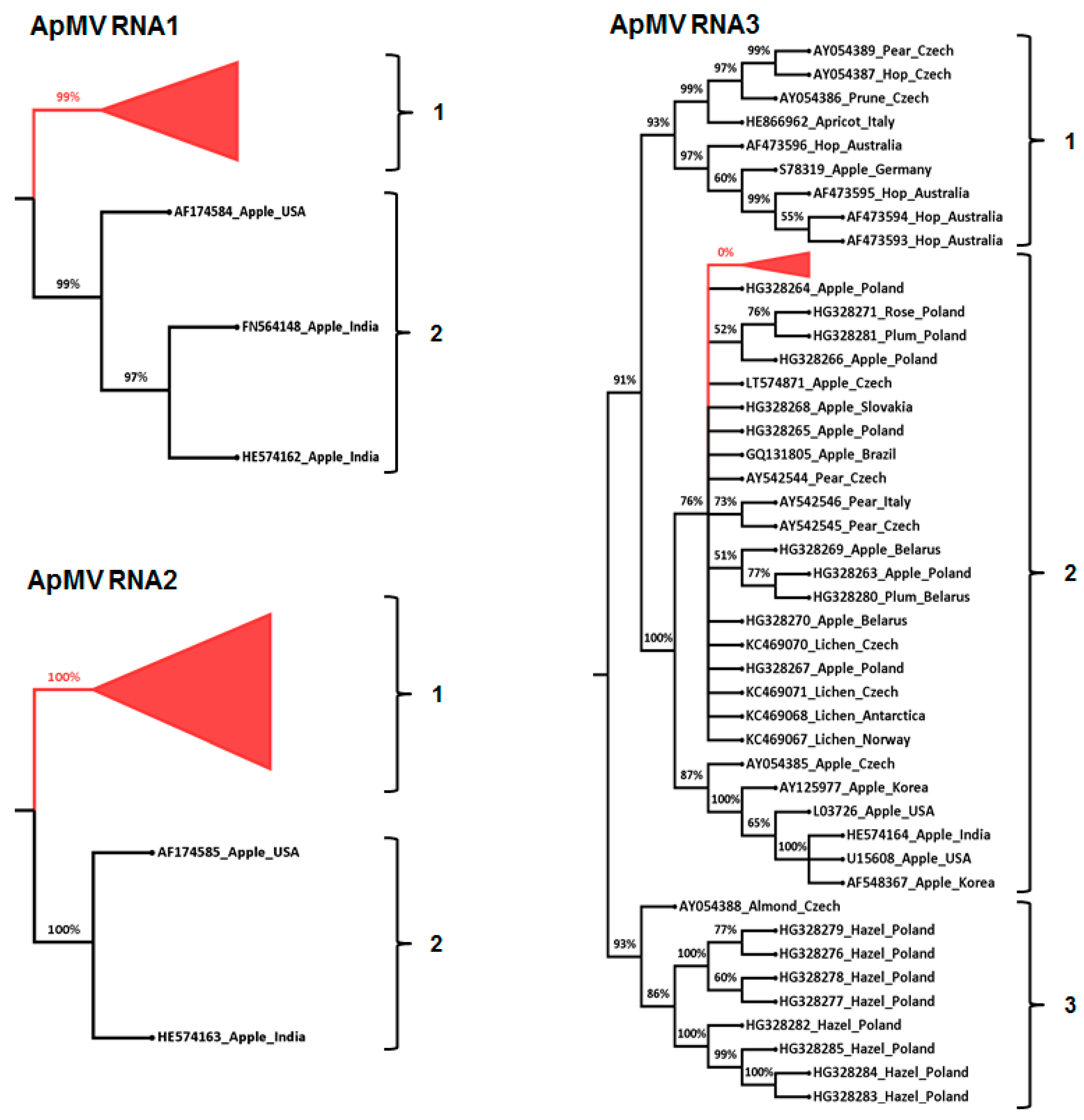
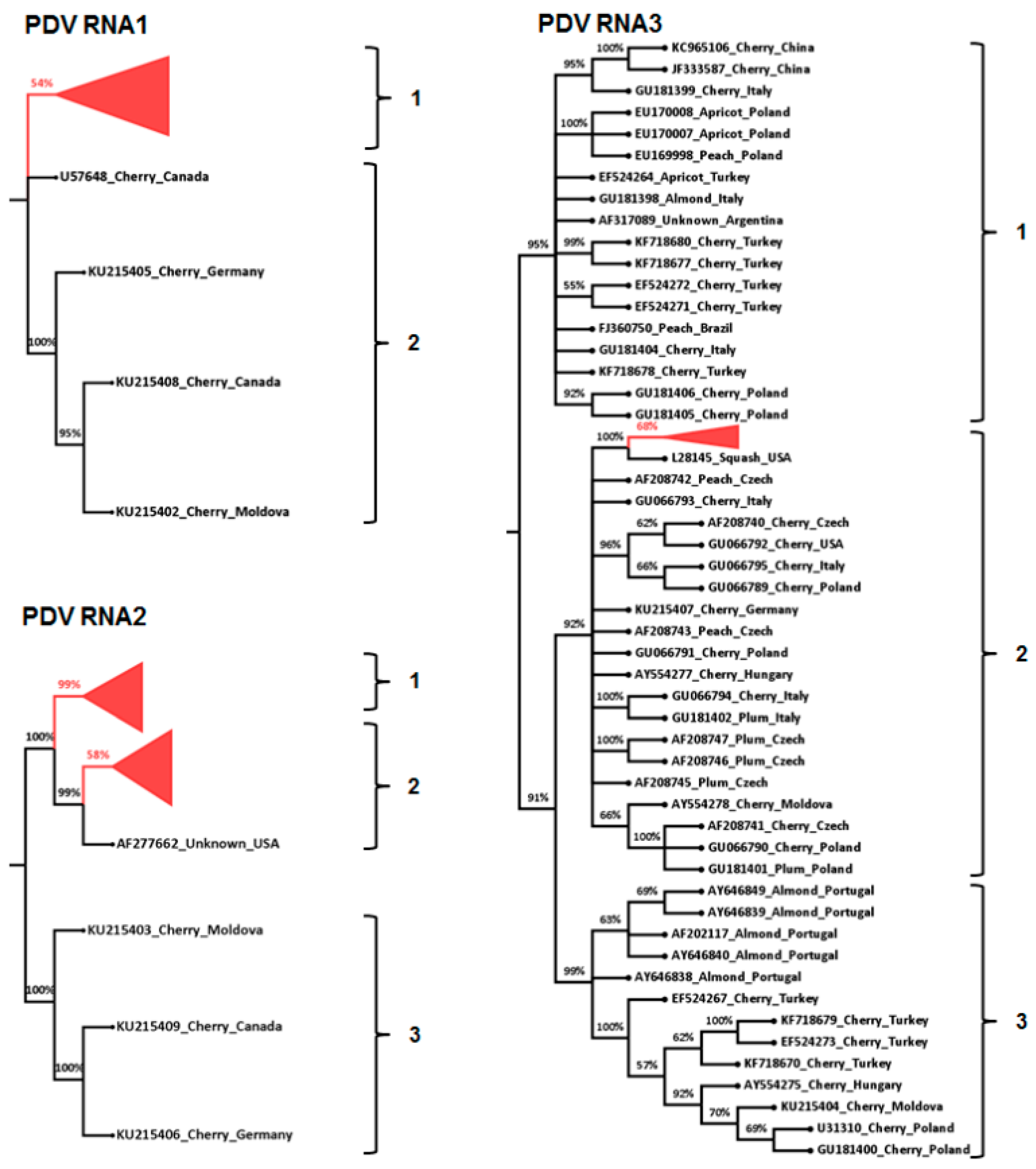
3.3. Phylogenetic and Sequence Identity Analysis
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bujarski, J.; Figlerowicz, M.; Gallittelli, D.; Roossinck, M.; Scott, S. Family bromoviridae. In Virus Taxonomy, Ninth Report of the International Committee on Taxonomy of Viruses; King, A.M.Q., Adams, M.J., Carstens, E.B., Lefkowitz, E.J., Eds.; Elsevier-Academic Press: Amsterdam, The Netherlands, 2012; Volume 9, pp. 965–976. [Google Scholar]
- Pallas, V.; Aparicio, F.; Herranz, M.; Amari, K.; Sanchez-Pina, M.; Myrta, A.; Sanchez-Navarro, J. Ilarviruses of prunus spp.: A continued concern for fruit trees. Phytopathology 2012, 102, 1108–1120. [Google Scholar] [CrossRef] [PubMed]
- Codoner, F.M.; Elena, S.F. The promiscuous evolutionary history of the family bromoviridae. J. Gen. Virol. 2008, 89, 1739–1747. [Google Scholar] [CrossRef] [PubMed]
- Shiel, P.; Berger, P. The complete nucleotide sequence of apple mosaic virus (ApMV) RNA 1 and RNA 2: ApMV is more closely related to alfalfa mosaic virus than to other ilarviruses. J. Gen. Virol. 2000, 81, 273–278. [Google Scholar] [CrossRef] [PubMed]
- Rampitsch, C.; Eastwell, K. The complete nucleotide sequenceof prune dwarf ilarvirus RNA-1. Arch. Virol. 1997, 142, 1911–1918. [Google Scholar] [CrossRef] [PubMed]
- Shiel, P.; Alrefai, R.; Domier, L.; Korban, S.; Berger, P. The complete nucleotide sequence of apple mosaic virus RNA-3. Arch. Virol. 1995, 140, 1247–1256. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, M.; Szalay-Marzsó, L.; Posnette, A. Virus, Mycoplasma and Rickettsia Diseases of Fruit Trees; Akademiai Kiado: Dordrecht, The Netherlands, 1986. [Google Scholar]
- Wood, G. Virus and Virus-Like Diseases of Pome Fruits and Stone Fruits in New Zealand; Department of Scientific and Industrial Research: Auckland, New Zealand, 1979; p. 87. [Google Scholar]
- Šutić, D.D.; Ford, R.E.; Tošić, M.T. Handbook of Plant Virus Diseases; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Martelli, G.; Savino, V. Infectious diseases of almond with special reference to the mediterranean area 1. Bull. OEPP 1997, 27, 525–534. [Google Scholar] [CrossRef]
- Çağlayan, K.; Ulubas-Serce, C.; Gazel, M.; Varveri, C. Prune dwarf virus. In Virus and Virus-Like Diseases of Pome and Stone Fruits; APS Press/American Phytopathological Society: St. Paul, MN, USA, 2011; pp. 199–205. [Google Scholar]
- Fulton, R. Apple Mosaic Virus. CMI/AAB Descriptions of Plant Viruses; Applied Biologists: Wellesbourne, UK, 1972; p. 83. [Google Scholar]
- Digiaro, M.; Savino, V.; Terlizzi, B.D. Ilarviruses in apricot and plum pollen. Acta Hortic. 1992, 309, 93–98. [Google Scholar]
- Mink, G. Pollen and seed-transmitted viruses and viroids. Annu. Rev. Phytopathol. 1993, 31, 375–402. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.P.; Ryu, K.H.; Kim, H.R.; Kim, C.S.; Lee, D.W.; Kim, J.S.; Park, M.H.; Noh, Y.M.; Choi, S.H.; Han, D.H. Cloning and phylogenetic characterization of coat protein genes of two isolates of apple mosaic virus from ‘fuji’apple. Plant Pathol. J. 2002, 18, 259–265. [Google Scholar] [CrossRef]
- Lakshmi, V.; Hallan, V.; Ram, R.; Ahmed, N.; Zaidi, A.; Varma, A. Diversity of apple mosaic virus isolates in india based on coat protein and movement protein genes. Indian J. Virol. 2011, 22, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Grimová, L.; Winkowska, L.; Ryšánek, P.; Svoboda, P.; Petrzik, K. Reflects the coat protein variability of apple mosaic virus host preference? Virus Genes 2013, 47, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Valasevich, N.; Cieślińska, M.; Kolbanova, E. Molecular characterization of apple mosaic virus isolates from apple and rose. Eur. J. Plant Pathol. 2015, 141, 839–845. [Google Scholar] [CrossRef]
- Ulubaş Serçe, Ç.; Ertunç, F.; Oeztuerk, A. Identification and genomic variability of prune dwarf virus variants infecting stone fruit trees in turkey. J. Phytopathol. 2009, 157, 298–305. [Google Scholar] [CrossRef]
- Vašková, D.; Petrzik, K.; Špak, J. Molecular variability of the capsid protein of the prune dwarf virus. Eur. J. Plant Pathol. 2000, 106, 573–580. [Google Scholar] [CrossRef]
- Predajňa, L.; Sihelská, N.; Benediková, D.; Šoltys, K.; Candresse, T.; Glasa, M. Molecular characterization of prune dwarf virus cherry isolates from slovakia shows their substantial variability and reveals recombination events in pdv RNA3. Eur. J. Plant Pathol. 2017, 147, 877–888. [Google Scholar] [CrossRef]
- Constable, F.E.; Joyce, P.A.; Rodoni, B.C. A survey of key australian pome fruit growing districts for exotic and endemic pathogens. Australas. Plant Pathol. 2007, 36, 165–172. [Google Scholar] [CrossRef]
- Stubs, L.; Smith, P. The association of prunus ringspot, prune dwarf, and dark green sunken mottle viruses in the rosetting and decline disease of peach. Crop Pasture Sci. 1971, 22, 771–785. [Google Scholar] [CrossRef]
- Greber, R.; Teakle, D.; Mink, G. Thrips-facilitated transmission of prune dwarf and prunus necrotic ringspot viruses from cherry pollen to cucumber. Plant Dis. 1992, 76, 1039–1041. [Google Scholar] [CrossRef]
- Parakh, D.; Shamloul, A.; Hadidi, A.; Waterworth, H.; Scott, S.; Howell, H.; Mink, G. Detection of prune dwarf ilarvirus from infected stone fruits using reverse transcription-polymerase chain reaction. Acta Hortic. 1994, 386, 421–430. [Google Scholar] [CrossRef]
- Petrzik, K.; Svoboda, P. Screening of apple mosaic virus in hop cultivars in the czech republic by reverse transcription-polymerase chain reaction. Acta Virol. 1997, 41, 101–103. [Google Scholar] [PubMed]
- Kinoti, W.M.; Constable, F.E.; Nancarrow, N.; Plummer, K.M.; Rodoni, B. Analysis of intra-host genetic diversity of prunus necrotic ringspot virus (PNRSV) using amplicon next generation sequencing. PLoS ONE 2017, 12, e0179284. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. Muscle: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Stamatakis, A. Raxml version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef] [PubMed]
- Andrew, R. Figtree. Available online: http://tree.bio.ed.ac.uk/software/ (accessed on 22 November 2017).
- Muhire, B.M.; Varsani, A.; Martin, D.P. Sdt: A virus classification tool based on pairwise sequence alignment and identity calculation. PLoS ONE 2014, 9, e108277. [Google Scholar] [CrossRef] [PubMed]
- Kinoti, W.M.; Constable, F.E.; Nancarrow, N.; Plummer, K.M.; Rodoni, B. Generic amplicon deep sequencing to determine ilarvirus species diversity in australian prunus. Front. Microbiol. 2017, 8, 1219. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. Mega6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef] [PubMed]
- Gümüs, M.; Paylan, I.; Matic, S.; Myrta, A.; Sipahioglu, H.; Erkan, S. Occurrence and distribution of stone fruit viruses and viroids in commercial plantings of prunus species in western anatolia, turkey. J. Plant Pathol. 2007, 89, 265–268. [Google Scholar]
- Myrta, A.; Di Terlizzi, B.; Savino, V.; Martelli, G. Virus diseases affecting the mediterranean stone fruit industry: A decade of surveys. In Virus and Virus-Like Diseases of Stone Fruits, with Particular Reference to the Mediterranean Region; Options Méditerranéennes, Série B; CIHEAM (Centre International de Hautes Etudes Agronomiques Méditerranéennes): Bari, Italy, 2003; pp. 15–23. [Google Scholar]
- Rouag, N.; Guechi, A.; Matic, S.; Myrta, A. Viruses and viroids of stone fruits in algeria. J. Plant Pathol. 2008, 90, 393–395. [Google Scholar]
- Petrzik, K. Capsid protein sequence gene analysis of apple mosaic virus infecting pears. Eur. J. Plant Pathol. 2005, 111, 355–360. [Google Scholar] [CrossRef]
- Kalinowska, E.; Mroczkowska, K.; Paduch-Cichal, E.; Chodorska, M. Genetic variability among coat protein of prune dwarf virus variants from different countries and different prunus species. Eur. J. Plant Pathol. 2014, 140, 863–868. [Google Scholar] [CrossRef]
- Cieślińska, M.; Valasevich, N. Characterization of apple mosaic virus isolates detected in hazelnut in poland. J. Plant Dis. Prot. 2016, 123, 187–192. [Google Scholar] [CrossRef]
- Boulila, M. Molecular characterization of an almond isolate of prune dwarf virus in tunisia: Putative recombination breakpoints in the partial sequences of the coat protein-encoding gene in isolates from different geographic origin. Phytopathol. Mediterr. 2010, 48, 411–421. [Google Scholar]
- Öztürk, Y.; Çevik, B. Genetic diversity in the coat protein genes of prune dwarf virus isolates from sweet cherry growing in turkey. Plant Pathol. J. 2015, 31, 41–49. [Google Scholar] [CrossRef] [PubMed]

| Location | Prunus Species | No. of Samples |
|---|---|---|
| New South Wales | Almond (P. dulcis) | 39 |
| Queensland | Apricot (P. armeniaca) | 4 |
| Nectarine (P. persica var. nucipersica) | 1 | |
| Plum (P. domestica) | 6 | |
| Peach (P. persica) | 6 | |
| Sweet Cherry (P. avium) | 2 | |
| South Australia | Nectarine (P. persica var. nucipersica) | 3 |
| Peach (P. persica) | 1 | |
| Tasmania | Apricot (P. armeniaca) | 1 |
| Almond (P. dulcis) | 1 | |
| Peach (P. persica) | 5 | |
| Plum (P. domestica) | 2 | |
| Sweet Cherry (P. avium) | 8 | |
| Victoria | Almond (P. dulcis) | 30 |
| Plum (P. domestica) | 4 | |
| Purple leaf plum (P. cerasifera) | 2 | |
| Peach (P. persica) | 12 |
| Primers | Primer Sequence (5’-3’) | RNA (Gene Target) a | Amplicon Length | Annealing Temp. | Reference |
|---|---|---|---|---|---|
| APMV-MT1 | AGTTTGTGTGATGTGAGAT | RNA1 (MT) | 222 bp | 53 °C | This study |
| APMV-MT2 | ATTTCTAAGGCGTAACTTC | ||||
| APMV-Rd1 | TCATTGGATCCCTTTGCTTC | RNA2 (RdRp) | 383 bp | 59 °C | This study |
| APMV-Rd2 | AAACTCGTCGTCCCTATCC | ||||
| APMV-CP1 | TTGCGTTAATTGCAAGTGG | RNA3 (CP) | 471 bp | 52 °C | This study |
| APMV-CP2 | TCAAAAGTTGTGTTTGGAG | ||||
| PDV-MT1 | GCGCTGACGAGACTACTA | RNA1 (MT) | 205 bp | 55 °C | This study |
| PDV-MT2 | GCGAAACTGTGTGAGGAA | ||||
| PDV-Rd1 | CGTTTCTGGAAGGAAGTGG | RNA2 (RdRp) | 382 bp | 60 °C | This study |
| PDV-Rd2 | TTGCTTCGAAATTGAACAA | ||||
| PDV-CP1 | TGTTAAGAAACAATTCCCA | RNA3 (CP) | 422 bp | 57 °C | This study |
| PDV-CP1 | GCTGAAAAGCGTTGTCATA |
| Isolate | Host | Location | Virus Detected |
|---|---|---|---|
| K73 | Almond (P. dulcis) | Victoria | ApMV |
| K74 | Plum (P. domestica) | Victoria | ApMV |
| K75 | Plum (P. domestica) | Victoria | ApMV |
| M35 | Almond (P. dulcis) | New South Wales | ApMV |
| M36 | Almond (P. dulcis) | New South Wales | ApMV |
| CNS3 | Peach (P. persica) | Victoria | PDV |
| CNS6 | Peach (P. persica) | Victoria | PDV |
| K76 | Almond (P. dulcis) | Victoria | PDV |
| NM20 | Almond (P. dulcis) | New South Wales | PDV |
| NM21 | Almond (P. dulcis) | New South Wales | PDV |
| NS3 | Peach (P. persica) | Victoria | PDV |
| NS5 | Peach (P. persica) | Victoria | PDV |
| NS7 | Peach (P. persica) | Victoria | PDV |
| NS9 | Peach (P. persica) | Victoria | PDV |
| PCH4 | Peach (P. persica) | Victoria | PDV |
| Q1 | Apricot (P. armeniaca) | Queensland | PDV |
| Q10 | Plum (P. domestica) | Queensland | PDV |
| Tas6 | Peach (P. persica) | Tasmania | PDV |
| APMV | RNA1 (Partial MT Gene: 173 bp) | RNA2 (Partial RdRp Gene: 364 bp) | RNA3 (Partial CP Gene: 452 bp) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Plant ID | No. of Reads after Trimming | No. of Sequence Variants | No. of Sequence Variants After Filter a | No. of Reads after Trimming | No. of Sequence Variants | No. of Sequence Variants after Filter | No. of Reads after Trimming | No. of Sequence Variants | No. of Sequence Variants after Filter |
| K73 | 48,587 | 1042 | 123 | 20,384 | 1114 | 186 | 34,454 | 1784 | 298 |
| K74 | 14,740 | 843 | 107 | 24,659 | 1532 | 242 | 62,852 | 2816 | 445 |
| K75 | 51,711 | 818 | 126 | 19,328 | 1422 | 229 | 55,305 | 2335 | 376 |
| M35 | 42,301 | 1516 | 141 | 16,127 | 1681 | 227 | 18,273 | 2585 | 349 |
| M36 | 15,409 | 1231 | 111 | 21,249 | 1725 | 326 | 12,589 | 3063 | 579 |
| Average | 34,550 | 1090 | 122 | 20,349 | 1495 | 242 | 36,695 | 2517 | 409 |
| PDV | RNA1 (Partial MT Gene: 193 bp) | RNA2 (Partial RdRp Gene: 358 bp) | RNA3 (Partial CP Gene: 396 bp) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Plant ID | No. of Reads after Trimming | No. of Sequence Variants | No. of Sequence Variants after Filter a | No. of Reads after Trimming | No. of Sequence Variants | No. of Sequence Variants after Filter | No. of Reads after Trimming | No. of Sequence Variants | No. of Sequence Variants after Filter |
| CNS3 | 16,020 | 1164 | 128 | 48,112 | 2101 | 270 | 56,440 | 2610 | 308 |
| CNS6 | 18,060 | 1087 | 137 | 21,083 | 2870 | 386 | 19,377 | 1055 | 134 |
| K76 | 20,460 | 1426 | 164 | 12,842 | 1409 | 185 | 16,638 | 1890 | 291 |
| NM20 | 12,330 | 725 | 103 | 12,127 | 1234 | 142 | 25,249 | 1197 | 195 |
| NM21 | 15,387 | 1043 | 121 | 16,178 | 1024 | 136 | 11,833 | 1640 | 182 |
| NS3 | 26,188 | 1027 | 116 | 17,450 | 1436 | 163 | 17,205 | 2960 | 373 |
| NS5 | 42,242 | 1191 | 137 | 25,272 | 985 | 135 | 85,141 | 3866 | 397 |
| NS7 | 10,542 | 645 | 98 | 13,794 | 1237 | 152 | 81,229 | 2928 | 342 |
| NS9 | 27,752 | 1080 | 121 | 31,725 | 1099 | 112 | 47,676 | 1916 | 251 |
| PCH4 | 47,346 | 1075 | 115 | 22,097 | 1140 | 147 | 79,293 | 2805 | 359 |
| Q1 | 30,758 | 1089 | 110 | 12,866 | 1038 | 136 | 26,150 | 1519 | 231 |
| Q10 | 11,998 | 884 | 107 | 50,068 | 1159 | 131 | 23,349 | 1650 | 193 |
| Tas6 | 21,382 | 1362 | 158 | 31,649 | 2324 | 316 | 94,629 | 3985 | 366 |
| Average | 23,113 | 1061 | 124 | 24,251 | 1466 | 185 | 44,939 | 2309 | 272 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kinoti, W.M.; Constable, F.E.; Nancarrow, N.; Plummer, K.M.; Rodoni, B. The Incidence and Genetic Diversity of Apple Mosaic Virus (ApMV) and Prune Dwarf Virus (PDV) in Prunus Species in Australia. Viruses 2018, 10, 136. https://doi.org/10.3390/v10030136
Kinoti WM, Constable FE, Nancarrow N, Plummer KM, Rodoni B. The Incidence and Genetic Diversity of Apple Mosaic Virus (ApMV) and Prune Dwarf Virus (PDV) in Prunus Species in Australia. Viruses. 2018; 10(3):136. https://doi.org/10.3390/v10030136
Chicago/Turabian StyleKinoti, Wycliff M., Fiona E. Constable, Narelle Nancarrow, Kim M. Plummer, and Brendan Rodoni. 2018. "The Incidence and Genetic Diversity of Apple Mosaic Virus (ApMV) and Prune Dwarf Virus (PDV) in Prunus Species in Australia" Viruses 10, no. 3: 136. https://doi.org/10.3390/v10030136





