Human Coronaviruses: Insights into Environmental Resistance and Its Influence on the Development of New Antiseptic Strategies
Abstract
:1. Introduction
3. HCoVs: Enveloped, but not that Fragile
3.1. Survival Under Different Conditions of Humidity and Temperature
| HCoV 229E | Type 1-Poliovirus, Sabin strain | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Relative humidity | 20 °C | 6 °C | 20 °C | 6 °C | ||||||
| 15 min | 24 h | 72 h | 6 days | 15 min | 24 h | 15 min | 24 h | 15 min | 24 h | |
| 30% | 87% | 65% | >50% | n.d. | 91% | 65% | 0% | 0% | n.d. | n.d. |
| 50% | 90.9% | 75% | >50% | 20% | 96.5% | 80% | 0% | 0% | n.d. | n.d. |
| 80% | 55% | 3% | 0% | n.d. | 104.8% | 86% | 90% | 30% | n.d. | n.d. |
3.2. Suspension vs. Desiccation
3.4. Survival in Biological Fluids
4. Antisepsis-Disinfection: An Efficient Weapon, with Room for Improvement
4.1. How Prevention Measures Halted the Propagation of SARS-CoV
4.2 What is Antisepsis-Disinfection and how do we Evaluate its Efficiency?
4.4. International Standardization Context
4.5. Sensitivity of HCoVs to Antiseptics-Disinfectants
4.5.1. Sensitivity of “Classic” HCoVs (other than SARS-CoV) to Antiseptics-Disinfectants
| Tested antiseptics-disinfectants | Concentration (%) - (pH at used concentration) | HCoV 229E | Type 3-parainfluenzavirus | Type B-Coxsackievirus | Type 5-Adenovirus |
|---|---|---|---|---|---|
| Enveloped | Enveloped | Non-enveloped | Non-enveloped | ||
| Halogenous compounds | |||||
| Sodium hypochlorite | 0.01 (8.0) | No | No | No | No |
| 0.10 (9.4) | Yes | Yes | No | No | |
| 0.50 (11.0) | Yes | Yes | Yes | Yes | |
| Chloramine T | 0.01 (7.0) | No | Yes | No | No |
| 0.10 (8.0) | Yes | No | No | No | |
| 0.30 (8.0) | Yes | Yes | Yes | Yes | |
| Sodium hypochlorite and potassium bromide | 0.01 (10.0) | No | No | No | No |
| 0.05 (11.5) | Yes | Yes | No | No | |
| 0.10 (12.0) | Yes | Yes | No | No | |
| Povidone-iodine | 10.0 (3.0) (1% available iodine) | Yes | Yes | No | No |
| Ethanol | 70.0 (4.0) | Yes | Yes | No | Yes |
| Glutaraldehyde | 2.0 (7.0) | Yes | Yes | Yes | Yes |
| Quaternary ammonium compounds | |||||
| n-alkyl-dimethylbenzyl chloride | 0.04 (6.0) | No | No | No | No |
| n-alkyl-dimethylbenzyl chloride | 0.04 (1.0) | Yes | Yes | Yes | Yes |
| + HCl | 7.00 | ||||
| n-alkyl-dimethylbenzyl chloride | 0.04 (5.0) | Yes | Yes | No | Yes |
| + ethanol | 70.0 | ||||
| n-alkyl-dimethylbenzyl chloride | 0.04 (11.0) | Yes | Yes | No | Yes |
| + sodium metasilicate | 0.5 | ||||
| Chlorhexidine gluconate | 0.008 (5.0) | No | Yes | No | No |
| + cetrimide | 0.08 | ||||
| Chlorhexidine gluconate | 0.05 (4.5) | Yes | Yes | No | Yes |
| + cetrimide | 0.50 | ||||
| + ethanol | 70.0 | ||||
| Phenolic compounds | |||||
| o-phenylphenol | 0.02 (9.0) | No | No | No | No |
| + o-benzyl-chlorophenol | 0.03 | ||||
| + p-tert-amylphenol | 0.01 | ||||
| o-phenylphenol | 0.02 (9.0) | Yes | Yes | No | No |
| + o-benzyl-chlorophenol l | 0.03 | ||||
| + p-tert-amylpheno | 0.01 | ||||
| + SDS | 0.60 | ||||
| o-phenylphenol | 0.02 (9.0) | Yes | Yes | No | Yes |
| + o-benzyl-chlorophenol | 0.03 | ||||
| + p-tert-amylphenol | 0.01 | ||||
| + ethanol | 70.0 | ||||
| Sodium o-benzyl-p-chlorophenate | 0.50 (13.0) | Yes | Yes | Yes | Yes |
| +Sodium dodecyl sulfate | 0.60 |
4.5.2. SARS-CoV Sensitivity to Antiseptics-Disinfectants
| Tested formulations | Contact times | Minimal reduction factor (log10) |
|---|---|---|
| 100% 2-propanol | 30 s | ≥ 3.31 |
| 70% 2-propanol | 30 s | ≥ 3.31 |
| 78% ethanol | 30 s | ≥ 5.01 |
| 45% 2-propanol, 30% 1-propanol | 30 s | ≥ 2.78 |
| Wine vinegar | 60 s | ≥ 3.0 |
| 0.7% formaldehyde | 2 min | ≥ 3.01 |
| 1.0% formaldehyde | 2 min | ≥ 3.01 |
| 0.5% glutardialdehyde | 2 min | ≥ 4.01 |
| 26% glucoprotamin | 2 min | ≥ 1.68 |
| Concentration of active ingredients of the tested commercial formulations | MHV | TGEV |
|---|---|---|
| Bleach (6% sodium hypochlorite – use dilution: 1:100, ≈ 600 mg/mL) | No | No |
| 9.09% o-phenylphenol, 7.66% p-tertiary amylphenol | No | No |
| 0.55% ortho-phthalaldehyde | No | No |
| 70% ethanol | Yes | Yes |
| 62% ethanol | No | Yes |
| 71% ethanol | No | Yes |
5. Conclusions
Acknowledgments
Conflict of Interest
References
- ICTV (International Committee on Taxonomy on Viruses). Virus Taxonomy: 2011 Release (current). Available online: http://ictvonline.org/virusTaxonomy.asp?version=2011 (Accessed on 24 September 2012).
- Almeida, J.D.; Tyrrell, D.A. The morphology of three previously uncharacterized human respiratory viruses that grow in organ culture. J. Gen. Virol. 1967, 1, 175–178. [Google Scholar] [CrossRef]
- Bradburne, A.F.; Bynoe, M.L.; Tyrrell, D.A. Effects of a "new" human respiratory virus in volunteers. Br. Med. J. 1967, 3, 767–769. [Google Scholar] [CrossRef]
- Hamre, D.; Procknow, J.J. A new virus isolated from the human respiratory tract. Proc. Soc. Exp. Biol. Med. 1966, 121, 190–193. [Google Scholar]
- McIntosh, K.; Dees, J.H.; Becker, W.B.; Kapikian, A.Z.; Chanock, R.M. Recovery in tracheal organ cultures of novel viruses from patients with respiratory disease. Proc. Natl. Acad. Sci. USA 1967, 57, 933–940. [Google Scholar] [CrossRef]
- Larson, H.E.; Reed, S.E.; Tyrrell, D.A. Isolation of rhinoviruses and coronaviruses from 38 colds in adults. J. Med. Virol. 1980, 5, 221–229. [Google Scholar] [CrossRef]
- Esper, F.; Weibel, C.; Ferguson, D.; Landry, M.L.; Kahn, J.S. Evidence of a novel human coronavirus that is associated with respiratory tract disease in infants and young children. J. Infect. Dis. 2005, 191, 492–498. [Google Scholar] [CrossRef]
- Fouchier, R.A.; Hartwig, N.G.; Bestebroer, T.M.; Niemeyer, B.; de Jong, J.C.; Simon, J.H.; Osterhaus, A.D. A previously undescribed coronavirus associated with respiratory disease in humans. Proc. Natl. Acad. Sci. USA 2004, 101, 6212–6216. [Google Scholar]
- Van der Hoek, L.; Pyrc, K.; Jebbink, M.F.; Vermeulen-Oost, W.; Berkhout, R.J.; Wolthers, K.C.; Wertheim-van Dillen, P.M.; Kaandorp, J.; Spaargaren, J.; Berkhout, B. Identification of a new human coronavirus. Nat. Med. 2004, 10, 368–373. [Google Scholar]
- Woo, P.C.; Lau, S.K.; Chu, C.M.; Chan, K.H.; Tsoi, H.W.; Huang, Y.; Wong, B.H.; Poon, R.W.; Cai, J.J.; Luk, W.K.; et al. Characterization and complete genome sequence of a novel coronavirus, coronavirus HKU1, from patients with pneumonia. J. Virol. 2005, 79, 884–895. [Google Scholar]
- Drosten, C.; Gunther, S.; Preiser, W.; van der Werf, S.; Brodt, H.R.; Becker, S.; Rabenau, H.; Panning, M.; Kolesnikova, L.; Fouchier, R.A.; et al. Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N. Engl. J. Med. 2003, 348, 1967–1976. [Google Scholar] [CrossRef]
- Ksiazek, T.G.; Erdman, D.; Goldsmith, C.S.; Zaki, S.R.; Peret, T.; Emery, S.; Tong, S.; Urbani, C.; Comer, J.A.; Lim, W.; et al. A novel coronavirus associated with severe acute respiratory syndrome. N. Engl. J. Med. 2003, 348, 1953–1966. [Google Scholar] [CrossRef]
- Peiris, J.S.; Lai, S.T.; Poon, L.L.; Guan, Y.; Yam, L.Y.; Lim, W.; Nicholls, J.; Yee, W.K.; Yan, W.W.; Cheung, M.T.; et al. Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet 2003, 361, 1319–1325. [Google Scholar]
- WHO multicentre collaborative network for Severe Acute Respiratory Syndrome diagnosis. A multicentre collaboration to investigate the cause of severe acute respiratory syndrome. Lancet 2003, 361, 1730–1733. [CrossRef]
- Arden, K.E.; Nissen, M.D.; Sloots, T.P.; Mackay, I.M. New human coronavirus, HCoV-NL63, associated with severe lower respiratory tract disease in Australia. J. Med. Virol. 2005, 75, 455–462. [Google Scholar] [CrossRef]
- Bastien, N.; Anderson, K.; Hart, L.; Van Caeseele, P.; Brandt, K.; Milley, D.; Hatchette, T.; Weiss, E.C.; Li, Y. Human coronavirus NL63 infection in Canada. J. Infect. Dis. 2005, 191, 503–506. [Google Scholar] [CrossRef]
- Vabret, A.; Mourez, T.; Dina, J.; van der Hoek, L.; Gouarin, S.; Petitjean, J.; Brouard, J.; Freymuth, F. Human coronavirus NL63, France. Emerg. Infect. Dis. 2005, 11, 1225–1229. [Google Scholar] [CrossRef]
- Van der Hoek, L.; Sure, K.; Ihorst, G.; Stang, A.; Pyrc, K.; Jebbink, M.F.; Petersen, G.; Forster, J.; Berkhout, B.; Uberla, K. Croup is associated with the novel coronavirus NL63. PLoS Med. 2005, 2, e240. [Google Scholar] [CrossRef] [Green Version]
- Gerna, G.; Percivalle, E.; Sarasini, A.; Campanini, G.; Piralla, A.; Rovida, F.; Genini, E.; Marchi, A.; Baldanti, F. Human respiratory coronavirus HKU1 versus other coronavirus infections in Italian hospitalised patients. J. Clin. Virol. 2007, 38, 244–250. [Google Scholar] [CrossRef]
- Sloots, T.P.; McErlean, P.; Speicher, D.J.; Arden, K.E.; Nissen, M.D.; Mackay, I.M. Evidence of human coronavirus HKU1 and human bocavirus in Australian children. J. Clin. Virol. 2006, 35, 99–102. [Google Scholar] [CrossRef]
- Chiu, S.S.; Chan, K.H.; Chu, K.W.; Kwan, S.W.; Guan, Y.; Poon, L.L.; Peiris, J.S. Human coronavirus NL63 infection and other coronavirus infections in children hospitalized with acute respiratory disease in Hong Kong, China. Clin. Infect. Dis. 2005, 40, 1721–1729. [Google Scholar] [CrossRef] [Green Version]
- Kon, M.; Watanabe, K.; Tazawa, T.; Tamura, T.; Tsukagoshi, H.; Noda, M.; Kimura, H.; Mizuta, K. Detection of human coronavirus NL63 and OC43 in children with acute respiratory infections in Niigata, Japan, between 2010 and 2011. Jpn. J. Infect. Dis. 2012, 65, 270–272. [Google Scholar] [CrossRef]
- Gerna, G.; Campanini, G.; Rovida, F.; Percivalle, E.; Sarasini, A.; Marchi, A.; Baldanti, F. Genetic variability of human coronavirus OC43-, 229E-, and NL63-like strains and their association with lower respiratory tract infections of hospitalized infants and immunocompromised patients. J. Med. Virol. 2006, 78, 938–949. [Google Scholar] [CrossRef]
- Esposito, S.; Bosis, S.; Niesters, H.G.; Tremolati, E.; Begliatti, E.; Rognoni, A.; Tagliabue, C.; Principi, N.; Osterhaus, A.D. Impact of human coronavirus infections in otherwise healthy children who attended an emergency department. J. Med. Virol. 2006, 78, 1609–1615. [Google Scholar] [CrossRef]
- Vabret, A.; Dina, J.; Gouarin, S.; Petitjean, J.; Tripey, V.; Brouard, J.; Freymuth, F. Human (non-severe acute respiratory syndrome) coronavirus infections in hospitalised children in France. J. Paediatr. Child. Health. 2008, 44, 176–181. [Google Scholar] [CrossRef]
- Talbot, H.K.; Shepherd, B.E.; Crowe, J.E., Jr.; Griffin, M.R.; Edwards, K.M.; Podsiad, A.B.; Tollefson, S.J.; Wright, P.F.; Williams, J.V. The pediatric burden of human coronaviruses evaluated for twenty years. Pediatr. Infect. Dis. J. 2009, 28, 682–687. [Google Scholar] [CrossRef]
- Vabret, A.; Mourez, T.; Gouarin, S.; Petitjean, J.; Freymuth, F. An outbreak of coronavirus OC43 respiratory infection in Normandy, France. Clin. Infect. Dis. 2003, 36, 985–989. [Google Scholar] [CrossRef]
- Van Elden, L.J.; van Loon, A.M.; van Alphen, F.; Hendriksen, K.A.; Hoepelman, A.I.; van Kraaij, M.G.; Oosterheert, J.J.; Schipper, P.; Schuurman, R.; Nijhuis, M. Frequent detection of human coronaviruses in clinical specimens from patients with respiratory tract infection by use of a novel real-time reverse-transcriptase polymerase chain reaction. J. Infect. Dis. 2004, 189, 652–657. [Google Scholar] [CrossRef]
- Riski, H.; Hovi, T. Coronavirus infections of man associated with diseases other than the common cold. J. Med. Virol. 1980, 6, 259–265. [Google Scholar] [CrossRef]
- Talbot, H.K.; Crowe, J.E., Jr.; Edwards, K.M.; Griffin, M.R.; Zhu, Y.; Weinberg, G.A.; Szilagyi, P.G.; Hall, C.B.; Podsiad, A.B.; Iwane, M.; et al. Coronavirus infection and hospitalizations for acute respiratory illness in young children. J. Med. Virol. 2009, 81, 853–856. [Google Scholar] [CrossRef]
- Woo, P.C.; Lau, S.K.; Tsoi, H.W.; Huang, Y.; Poon, R.W.; Chu, C.M.; Lee, R.A.; Luk, W.K.; Wong, G.K.; Wong, B.H.; et al. Clinical and molecular epidemiological features of coronavirus HKU1-associated community-acquired pneumonia. J. Infect. Dis. 2005, 192, 1898–1907. [Google Scholar] [CrossRef] [Green Version]
- Gagneur, A.; Sizun, J.; Vallet, S.; Legr, M.C.; Picard, B.; Talbot, P.J. Coronavirus-related nosocomial viral respiratory infections in a neonatal and paediatric intensive care unit: a prospective study. J. Hosp. Infect. 2002, 51, 59–64. [Google Scholar] [CrossRef]
- Sizun, J.; Soupre, D.; Legrand, M.C.; Giroux, J.D.; Rubio, S.; Cauvin, J.M.; Chastel, C.; Alix, D.; de Parscau, L. Neonatal nosocomial respiratory infection with coronavirus: a prospective study in a neonatal intensive care unit. Acta Paediatr. 1995, 84, 617–620. [Google Scholar] [CrossRef]
- Falsey, A.R.; Walsh, E.E.; Hayden, F.G. Rhinovirus and coronavirus infection-associated hospitalizations among older adults. J. Infect. Dis. 2002, 185, 1338–1341. [Google Scholar] [CrossRef]
- Nicholson, K.G.; Kent, J.; Hammersley, V.; Cancio, E. Acute viral infections of upper respiratory tract in elderly people living in the community: comparative, prospective, population based study of disease burden. BMJ 1997, 315, 1060–1064. [Google Scholar] [CrossRef]
- Pene, F.; Merlat, A.; Vabret, A.; Rozenberg, F.; Buzyn, A.; Dreyfus, F.; Cariou, A.; Freymuth, F.; Lebon, P. Coronavirus 229E-related pneumonia in immunocompromised patients. Clin. Infect. Dis. 2003, 37, 929–932. [Google Scholar] [CrossRef]
- Folz, R.J.; Elkordy, M.A. Coronavirus pneumonia following autologous bone marrow transplantation for breast cancer. Chest 1999, 115, 901–905. [Google Scholar] [CrossRef]
- Chany, C.; Moscovici, O.; Lebon, P.; Rousset, S. Association of coronavirus infection with neonatal necrotizing enterocolitis. Pediatrics 1982, 69, 209–214. [Google Scholar]
- Vabret, A.; Dina, J.; Gouarin, S.; Petitjean, J.; Corbet, S.; Freymuth, F. Detection of the new human coronavirus HKU1: a report of 6 cases. Clin. Infect. Dis. 2006, 42, 634–639. [Google Scholar] [CrossRef]
- Zhang, X.M.; Herbst, W.; Kousoulas, K.G.; Storz, J. Biological and genetic characterization of a hemagglutinating coronavirus isolated from a diarrhoeic child. J. Med. Virol. 1994, 44, 152–161. [Google Scholar] [CrossRef]
- Arbour, N.; Cote, G.; Lachance, C.; Tardieu, M.; Cashman, N.R.; Talbot, P.J. Acute and persistent infection of human neural cell lines by human coronavirus OC43. J. Virol. 1999, 73, 3338–3350. [Google Scholar]
- Arbour, N.; Ekande, S.; Cote, G.; Lachance, C.; Chagnon, F.; Tardieu, M.; Cashman, N.R.; Talbot, P.J. Persistent infection of human oligodendrocytic and neuroglial cell lines by human coronavirus 229E. J. Virol. 1999, 73, 3326–3337. [Google Scholar]
- Bonavia, A.; Arbour, N.; Yong, V.W.; Talbot, P.J. Infection of primary cultures of human neural cells by human coronaviruses 229E and OC43. J. Virol. 1997, 71, 800–806. [Google Scholar]
- Arbour, N.; Day, R.; Newcombe, J.; Talbot, P.J. Neuroinvasion by human respiratory coronaviruses. J. Virol. 2000, 74, 8913–8921. [Google Scholar] [CrossRef]
- Murray, R.S.; Brown, B.; Brian, D.; Cabirac, G.F. Detection of coronavirus RNA and antigen in multiple sclerosis brain. Ann. Neurol. 1992, 31, 525–533. [Google Scholar] [CrossRef]
- Stewart, J.N.; Mounir, S.; Talbot, P.J. Human coronavirus gene expression in the brains of multiple sclerosis patients. Virology 1992, 191, 502–505. [Google Scholar] [CrossRef]
- St-Jean, J.R.; Jacomy, H.; Desforges, M.; Vabret, A.; Freymuth, F.; Talbot, P.J. Human respiratory coronavirus OC43: genetic stability and neuroinvasion. J. Virol. 2004, 78, 8824–8834. [Google Scholar] [CrossRef]
- Riski, H.; Hovi, T.; Frick, M.H. Carditis associated with coronavirus infection. Lancet 1980, 2, 100–101. [Google Scholar]
- Hsu, L.Y.; Lee, C.C.; Green, J.A.; Ang, B.; Paton, N.I.; Lee, L.; Villacian, J.S.; Lim, P.L.; Earnest, A.; Leo, Y.S. Severe acute respiratory syndrome (SARS) in Singapore: clinical features of index patient and initial contacts. Emerg. Infect. Dis. 2003, 9, 713–717. [Google Scholar] [CrossRef]
- Poutanen, S.M.; Low, D.E.; Henry, B.; Finkelstein, S.; Rose, D.; Green, K.; Tellier, R.; Draker, R.; Adachi, D.; Ayers, M.; et al. Identification of severe acute respiratory syndrome in Canada. N. Engl. J. Med. 2003, 348, 1995–2005. [Google Scholar] [CrossRef]
- WHO (World Health Organization). Summary of probable SARS cases with onset of illness from 1 November 2002 to 31 July. Available online: http://www.who.int/csr/sars/country/table2004_04_21/en/index.html (Accessed on 21 February 2010).
- Lee, N.; Hui, D.; Wu, A.; Chan, P.; Cameron, P.; Joynt, G.M.; Ahuja, A.; Yung, M.Y.; Leung, C.B.; To, K.F.; et al. A major outbreak of severe acute respiratory syndrome in Hong Kong. N. Engl. J. Med. 2003, 348, 1986–1994. [Google Scholar] [CrossRef]
- Varia, M.; Wilson, S.; Sarwal, S.; McGeer, A.; Gournis, E.; Galanis, E.; Henry, B. Investigation of a nosocomial outbreak of severe acute respiratory syndrome (SARS) in Toronto, Canada. CMAJ 2003, 169, 285–292. [Google Scholar]
- Zhao, Z.; Zhang, F.; Xu, M.; Huang, K.; Zhong, W.; Cai, W.; Yin, Z.; Huang, S.; Deng, Z.; Wei, M.; et al. Description and clinical treatment of an early outbreak of severe acute respiratory syndrome (SARS) in Guangzhou, PR China. J. Med. Microbiol. 2003, 52, 715–720. [Google Scholar] [CrossRef]
- Booth, C.M.; Matukas, L.M.; Tomlinson, G.A.; Rachlis, A.R.; Rose, D.B.; Dwosh, H.A.; Walmsley, S.L.; Mazzulli, T.; Avendano, M.; Derkach, P.; et al. Clinical features and short-term outcomes of 144 patients with SARS in the greater Toronto area. JAMA 2003, 289, 2801–2809. [Google Scholar] [CrossRef]
- Donnelly, C.A.; Ghani, A.C.; Leung, G.M.; Hedley, A.J.; Fraser, C.; Riley, S.; Abu-Raddad, L.J.; Ho, L.M.; Thach, T.Q.; Chau, P.; et al. Epidemiological determinants of spread of causal agent of severe acute respiratory syndrome in Hong Kong. Lancet 2003, 361, 1761–1766. [Google Scholar]
- Peiris, J.S.; Chu, C.M.; Cheng, V.C.; Chan, K.S.; Hung, I.F.; Poon, L.L.; Law, K.I.; Tang, B.S.; Hon, T.Y.; Chan, C.S.; et al. Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. Lancet 2003, 361, 1767–1772. [Google Scholar]
- Peiris, J.S.; Yuen, K.Y.; Osterhaus, A.D.; Stohr, K. The severe acute respiratory syndrome. N. Engl. J. Med. 2003, 349, 2431–2441. [Google Scholar] [CrossRef]
- WHO (World Health Organization). Consensus document on the epidemiology of severe acute respiratory syndrome (SARS). Available online: http://www.who.int/csr/sars/en/WHOconsensus.pdf (Accessed on 25-09-12).
- Hamming, I.; Timens, W.; Bulthuis, M.L.; Lely, A.T.; Navis, G.J.; van Goor, H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004, 203, 631–637. [Google Scholar] [CrossRef]
- Li, W.; Moore, M.J.; Vasilieva, N.; Sui, J.; Wong, S.K.; Berne, M.A.; Somasundaran, M.; Sullivan, J.L.; Luzuriaga, K.; Greenough, T.C.; et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 2003, 426, 450–454. [Google Scholar] [CrossRef]
- Dwosh, H.A.; Hong, H.H.; Austgarden, D.; Herman, S.; Schabas, R. Identification and containment of an outbreak of SARS in a community hospital. CMAJ 2003, 168, 1415–1420. [Google Scholar]
- Ng, S.K. Possible role of an animal vector in the SARS outbreak at Amoy Gardens. Lancet 2003, 362, 570–572. [Google Scholar] [CrossRef]
- Vijgen, L.; Keyaerts, E.; Moes, E.; Thoelen, I.; Wollants, E.; Lemey, P.; Vandamme, A.M.; Van Ranst, M. Complete genomic sequence of human coronavirus OC43: molecular clock analysis suggests a relatively recent zoonotic coronavirus transmission event. J. Virol. 2005, 79, 1595–1604. [Google Scholar]
- Lau, S.K.; Woo, P.C.; Li, K.S.; Huang, Y.; Tsoi, H.W.; Wong, B.H.; Wong, S.S.; Leung, S.Y.; Chan, K.H.; Yuen, K.Y. Severe acute respiratory syndrome coronavirus-like virus in Chinese horseshoe bats. Proc. Natl. Acad. Sci. USA 2005, 102, 14040–14045. [Google Scholar]
- Li, W.; Shi, Z.; Yu, M.; Ren, W.; Smith, C.; Epstein, J.H.; Wang, H.; Crameri, G.; Hu, Z.; Zhang, H.; et al. Bats are natural reservoirs of SARS-like coronaviruses. Science 2005, 310, 676–679. [Google Scholar] [CrossRef]
- Poon, L.L.; Chu, D.K.; Chan, K.H.; Wong, O.K.; Ellis, T.M.; Leung, Y.H.; Lau, S.K.; Woo, P.C.; Suen, K.Y.; Yuen, K.Y.; et al. Identification of a novel coronavirus in bats. J. Virol. 2005, 79, 2001–2009. [Google Scholar]
- Woo, P.C.; Lau, S.K.; Li, K.S.; Poon, R.W.; Wong, B.H.; Tsoi, H.W.; Yip, B.C.; Huang, Y.; Chan, K.H.; Yuen, K.Y. Molecular diversity of coronaviruses in bats. Virology 2006, 351, 180–187. [Google Scholar] [CrossRef]
- Kan, B.; Wang, M.; Jing, H.; Xu, H.; Jiang, X.; Yan, M.; Liang, W.; Zheng, H.; Wan, K.; Liu, Q.; et al. Molecular evolution analysis and geographic investigation of severe acute respiratory syndrome coronavirus-like virus in palm civets at an animal market and on farms. J. Virol. 2005, 79, 11892–11900. [Google Scholar] [CrossRef]
- Cinatl, J., Jr.; Michaelis, M.; Morgenstern, B.; Doerr, H.W. High-dose hydrocortisone reduces expression of the pro-inflammatory chemokines CXCL8 and CXCL10 in SARS coronavirus-infected intestinal cells. Int. J. Mol. Med. 2005, 15, 323–327. [Google Scholar]
- Cinatl, J.; Morgenstern, B.; Bauer, G.; Chandra, P.; Rabenau, H.; Doerr, H.W. Glycyrrhizin, an active component of liquorice roots, and replication of SARS-associated coronavirus. Lancet 2003, 361, 2045–2046. [Google Scholar] [CrossRef]
- Morgenstern, B.; Michaelis, M.; Baer, P.C.; Doerr, H.W.; Cinatl, J., Jr. Ribavirin and interferon-beta synergistically inhibit SARS-associated coronavirus replication in animal and human cell lines. Biochem. Biophys. Res. Commun. 2005, 326, 905–908. [Google Scholar] [CrossRef]
- Mazzulli, T.; Farcas, G.A.; Poutanen, S.M.; Willey, B.M.; Low, D.E.; Butany, J.; Asa, S.L.; Kain, K.C. Severe acute respiratory syndrome-associated coronavirus in lung tissue. Emerg. Infect. Dis. 2004, 10, 20–24. [Google Scholar] [CrossRef]
- Yamamoto, N.; Yang, R.; Yoshinaka, Y.; Amari, S.; Nakano, T.; Cinatl, J.; Rabenau, H.; Doerr, H.W.; Hunsmann, G.; Otaka, A.; et al. HIV protease inhibitor nelfinavir inhibits replication of SARS-associated coronavirus. Biochem. Biophys. Res. Commun. 2004, 318, 719–725. [Google Scholar] [CrossRef]
- Cinatl, J., Jr.; Michaelis, M.; Hoever, G.; Preiser, W.; Doerr, H.W. Development of antiviral therapy for severe acute respiratory syndrome. Antiviral Res. 2005, 66, 81–97. [Google Scholar] [CrossRef]
- Haagmans, B.L.; Osterhaus, A.D. Coronaviruses and their therapy. Antiviral Res. 2006, 71, 397–403. [Google Scholar] [CrossRef]
- Bisht, H.; Roberts, A.; Vogel, L.; Bukreyev, A.; Collins, P.L.; Murphy, B.R.; Subbarao, K.; Moss, B. Severe acute respiratory syndrome coronavirus spike protein expressed by attenuated vaccinia virus protectively immunizes mice. Proc. Natl. Acad. Sci. USA. 2004, 101, 6641–6646. [Google Scholar]
- Kapadia, S.U.; Rose, J.K.; Lamirande, E.; Vogel, L.; Subbarao, K.; Roberts, A. Long-term protection from SARS coronavirus infection conferred by a single immunization with an attenuated VSV-based vaccine. Virology 2005, 340, 174–182. [Google Scholar] [CrossRef]
- Chen, Z.; Zhang, L.; Qin, C.; Ba, L.; Yi, C.E.; Zhang, F.; Wei, Q.; He, T.; Yu, W.; Yu, J.; et al. Recombinant modified vaccinia virus Ankara expressing the spike glycoprotein of severe acute respiratory syndrome coronavirus induces protective neutralizing antibodies primarily targeting the receptor binding region. J. Virol. 2005, 79, 2678–2688. [Google Scholar]
- Bukreyev, A.; Lamirande, E.W.; Buchholz, U.J.; Vogel, L.N.; Elkins, W.R.; St Claire, M.; Murphy, B.R.; Subbarao, K.; Collins, P.L. Mucosal immunisation of African green monkeys (Cercopithecus aethiops) with an attenuated parainfluenza virus expressing the SARS coronavirus spike protein for the prevention of SARS. Lancet 2004, 363, 2122–2127. [Google Scholar]
- Ishii, K.; Hasegawa, H.; Nagata, N.; Mizutani, T.; Morikawa, S.; Suzuki, T.; Taguchi, F.; Tashiro, M.; Takemori, T.; Miyamura, T.; et al. Induction of protective immunity against severe acute respiratory syndrome coronavirus (SARS-CoV) infection using highly attenuated recombinant vaccinia virus DIs. Virology 2006, 351, 368–380. [Google Scholar] [CrossRef]
- See, R.H.; Zakhartchouk, A.N.; Petric, M.; Lawrence, D.J.; Mok, C.P.; Hogan, R.J.; Rowe, T.; Zitzow, L.A.; Karunakaran, K.P.; Hitt, M.M.; et al. Comparative evaluation of two severe acute respiratory syndrome (SARS) vaccine candidates in mice challenged with SARS coronavirus. J. Gen. Virol. 2006, 87, 641–650. [Google Scholar] [CrossRef]
- Czub, M.; Weingartl, H.; Czub, S.; He, R.; Cao, J. Evaluation of modified vaccinia virus Ankara based recombinant SARS vaccine in ferrets. Vaccine 2005, 23, 2273–2279. [Google Scholar] [CrossRef]
- He, Y.; Li, J.; Heck, S.; Lustigman, S.; Jiang, S. Antigenic and immunogenic characterization of recombinant baculovirus-expressed severe acute respiratory syndrome coronavirus spike protein: implication for vaccine design. J. Virol. 2006, 80, 5757–5767. [Google Scholar] [CrossRef]
- Zhou, Z.; Post, P.; Chubet, R.; Holtz, K.; McPherson, C.; Petric, M.; Cox, M. A recombinant baculovirus-expressed S glycoprotein vaccine elicits high titers of SARS-associated coronavirus (SARS-CoV) neutralizing antibodies in mice. Vaccine 2006, 24, 3624–3631. [Google Scholar] [CrossRef]
- He, Y.; Zhou, Y.; Siddiqui, P.; Jiang, S. Inactivated SARS-CoV vaccine elicits high titers of spike protein-specific antibodies that block receptor binding and virus entry. Biochem. Biophys. Res. Commun. 2004, 325, 445–452. [Google Scholar] [CrossRef]
- Spruth, M.; Kistner, O.; Savidis-Dacho, H.; Hitter, E.; Crowe, B.; Gerencer, M.; Bruhl, P.; Grillberger, L.; Reiter, M.; Tauer, C.; et al. A double-inactivated whole virus candidate SARS coronavirus vaccine stimulates neutralising and protective antibody responses. Vaccine 2006, 24, 652–661. [Google Scholar]
- Tang, L.; Zhu, Q.; Qin, E.; Yu, M.; Ding, Z.; Shi, H.; Cheng, X.; Wang, C.; Chang, G.; Fang, F.; et al. Inactivated SARS-CoV vaccine prepared from whole virus induces a high level of neutralizing antibodies in BALB/c mice. DNA Cell Biol. 2004, 23, 391–394. [Google Scholar] [CrossRef]
- Xiong, S.; Wang, Y.F.; Zhang, M.Y.; Liu, X.J.; Zhang, C.H.; Liu, S.S.; Qian, C.W.; Li, J.X.; Lu, J.H.; Wan, Z.Y.; et al. Immunogenicity of SARS inactivated vaccine in BALB/c mice. Immunol. Lett. 2004, 95, 139–143. [Google Scholar] [CrossRef]
- Takasuka, N.; Fujii, H.; Takahashi, Y.; Kasai, M.; Morikawa, S.; Itamura, S.; Ishii, K.; Sakaguchi, M.; Ohnishi, K.; Ohshima, M.; et al. A subcutaneously injected UV-inactivated SARS coronavirus vaccine elicits systemic humoral immunity in mice. Int. Immunology 2004, 16, 1423–1430. [Google Scholar] [CrossRef]
- Stadler, K.; Roberts, A.; Becker, S.; Vogel, L.; Eickmann, M.; Kolesnikova, L.; Klenk, H.D.; Murphy, B.; Rappuoli, R.; Abrignani, S.; et al. SARS vaccine protective in mice. Emerg. Infect. Dis. 2005, 11, 1312–1314. [Google Scholar]
- Woo, P.C.; Lau, S.K.; Tsoi, H.W.; Chen, Z.W.; Wong, B.H.; Zhang, L.; Chan, J.K.; Wong, L.P.; He, W.; Ma, C.; et al. SARS coronavirus spike polypeptide DNA vaccine priming with recombinant spike polypeptide from Escherichia coli as booster induces high titer of neutralizing antibody against SARS coronavirus. Vaccine 2005, 23, 4959–4968. [Google Scholar] [CrossRef]
- Yang, Z.Y.; Kong, W.P.; Huang, Y.; Roberts, A.; Murphy, B.R.; Subbarao, K.; Nabel, G.J. A DNA vaccine induces SARS coronavirus neutralization and protective immunity in mice. Nature 2004, 428, 561–564. [Google Scholar]
- Zhao, P.; Ke, J.S.; Qin, Z.L.; Ren, H.; Zhao, L.J.; Yu, J.G.; Gao, J.; Zhu, S.Y.; Qi, Z.T. DNA vaccine of SARS-Cov S gene induces antibody response in mice. Acta Biochim. Biophys. Sin. 2004, 36, 37–41. [Google Scholar] [CrossRef]
- Wang, Z.; Yuan, Z.; Matsumoto, M.; Hengge, U.R.; Chang, Y.F. Immune responses with DNA vaccines encoded different gene fragments of severe acute respiratory syndrome coronavirus in BALB/c mice. Biochem. Biophys. Res. Commun. 2005, 327, 130–135. [Google Scholar] [CrossRef]
- Perlman, S.; Dandekar, A.A. Immunopathogenesis of coronavirus infections: implications for SARS. Nat. Rev. Immunol. 2005, 5, 917–927. [Google Scholar] [CrossRef]
- Bolles, M.; Deming, D.; Long, K.; Agnihothram, S.; Whitmore, A.; Ferris, M.; Funkhouser, W.; Gralinski, L.; Totura, A.; Heise, M.; et al. A double-inactivated severe acute respiratory syndrome coronavirus vaccine provides incomplete protection in mice and induces increased eosinophilic proinflammatory pulmonary response upon challenge. J. Virol. 2011, 85, 12201–12215. [Google Scholar] [CrossRef]
- Deming, D.; Sheahan, T.; Heise, M.; Yount, B.; Davis, N.; Sims, A.; Suthar, M.; Harkema, J.; Whitmore, A.; Pickles, R.; et al. Vaccine efficacy in senescent mice challenged with recombinant SARS-CoV bearing epidemic and zoonotic spike variants. PLoS Med. 2006, 3, e525. [Google Scholar] [CrossRef]
- Lokugamage, K.G.; Yoshikawa-Iwata, N.; Ito, N.; Watts, D.M.; Wyde, P.R.; Wang, N.; Newman, P.; Kent Tseng, C.T.; Peters, C.J.; Makino, S. Chimeric coronavirus-like particles carrying severe acute respiratory syndrome coronavirus (SCoV) S protein protect mice against challenge with SCoV. Vaccine 2008, 26, 797–808. [Google Scholar]
- Yasui, F.; Kai, C.; Kitabatake, M.; Inoue, S.; Yoneda, M.; Yokochi, S.; Kase, R.; Sekiguchi, S.; Morita, K.; Hishima, T.; et al. Prior immunization with severe acute respiratory syndrome (SARS)-associated coronavirus (SARS-CoV) nucleocapsid protein causes severe pneumonia in mice infected with SARS-CoV. J. Immunol. 2008, 181, 6337–6348. [Google Scholar]
- Tseng, C.T.; Sbrana, E.; Iwata-Yoshikawa, N.; Newman, P.C.; Garron, T.; Atmar, R.L.; Peters, C.J.; Couch, R.B. Immunization with SARS coronavirus vaccines leads to pulmonary immunopathology on challenge with the SARS virus. PLoS ONE. 2012, 7, e35421. [Google Scholar]
- Olsen, S.J.; Chang, H.L.; Cheung, T.Y.; Tang, A.F.; Fisk, T.L.; Ooi, S.P.; Kuo, H.W.; Jiang, D.D.; Chen, K.T.; Lando, J.; et al. Transmission of the severe acute respiratory syndrome on aircraft. N. Engl. J. Med. 2003, 349, 2416–2422. [Google Scholar] [CrossRef]
- Lee, S.H. The SARS epidemic in Hong Kong. J. Epidemiol. Community Health. 2003, 57, 652–654. [Google Scholar] [CrossRef]
- Ijaz, M.K.; Brunner, A.H.; Sattar, S.A.; Nair, R.C.; Johnson-Lussenburg, C.M. Survival characteristics of airborne human coronavirus 229E. J. Gen. Virol. 1985, 66 ( Pt 12). [Google Scholar]
- Rabenau, H.F.; Cinatl, J.; Morgenstern, B.; Bauer, G.; Preiser, W.; Doerr, H.W. Stability and inactivation of SARS coronavirus. Med. Microbiol. Immunol. 2005, 194, 1–6. [Google Scholar] [CrossRef]
- Duan, S.M.; Zhao, X.S.; Wen, R.F.; Huang, J.J.; Pi, G.H.; Zhang, S.X.; Han, J.; Bi, S.L.; Ruan, L.; Dong, X.P. Stability of SARS coronavirus in human specimens and environment and its sensitivity to heating and UV irradiation. Biomed. Environ. Sci. 2003, 16, 246–255. [Google Scholar]
- Sizun, J.; Yu, M.W.; Talbot, P.J. Survival of human coronaviruses 229E and OC43 in suspension and after drying on surfaces: a possible source of hospital-acquired infections. J. Hosp. Infect. 2000, 46, 55–60. [Google Scholar] [CrossRef]
- Lai, M.Y.; Cheng, P.K.; Lim, W.W. Survival of severe acute respiratory syndrome coronavirus. Clin. Infect. Dis. 2005, 41, e67–71. [Google Scholar] [CrossRef]
- Chen, Y.C.; Huang, L.M.; Chan, C.C.; Su, C.P.; Chang, S.C.; Chang, Y.Y.; Chen, M.L.; Hung, C.C.; Chen, W.J.; Lin, F.Y.; et al. SARS in hospital emergency room. Emerg. Infect. Dis. 2004, 10, 782–788. [Google Scholar]
- Dowell, S.F.; Simmerman, J.M.; Erdman, D.D.; Wu, J.S.; Chaovavanich, A.; Javadi, M.; Yang, J.Y.; Anderson, L.J.; Tong, S.; Ho, M.S. Severe acute respiratory syndrome coronavirus on hospital surfaces. Clin. Infect. Dis. 2004, 39, 652–657. [Google Scholar] [CrossRef]
- Casanova, L.; Rutala, W.A.; Weber, D.J.; Sobsey, M.D. Survival of surrogate coronaviruses in water. Water Res. 2009, 43, 1893–1898. [Google Scholar] [CrossRef]
- Lamarre, A.; Talbot, P.J. Effect of pH and temperature on the infectivity of human coronavirus 229E. Can. J. Microbiol. 1989, 35, 972–974. [Google Scholar]
- Sturman, L.S.; Ricard, C.S.; Holmes, K.V. Conformational change of the coronavirus peplomer glycoprotein at pH 8.0 and 37 degrees C correlates with virus aggregation and virus-induced cell fusion. J. Virol. 1990, 64, 3042–3050. [Google Scholar]
- Daniel, C.; Talbot, P.J. Physico-chemical properties of murine hepatitis virus, strain A 59. Brief report. Arch. Virol. 1987, 96, 241–248. [Google Scholar] [CrossRef]
- Pocock, D.H.; Garwes, D.J. The influence of pH on the growth and stability of transmissible gastroenteritis virus in vitro. Arch. Virol. 1975, 49, 239–247. [Google Scholar] [CrossRef]
- Pratelli, A. Canine coronavirus inactivation with physical and chemical agents. Vet. J. 2008, 177, 71–79. [Google Scholar] [CrossRef]
- WHO (World Health Organization). First data on stability and resistance of SARS coronavirus compiled by members of WHO laboratory network. Available online: http://www.who.int/csr/sars/survival_2003_05_04/en/index.html (Accessed on 16 February 2010).
- Lipsitch, M.; Cohen, T.; Cooper, B.; Robins, J.M.; Ma, S.; James, L.; Gopalakrishna, G.; Chew, S.K.; Tan, C.C.; Samore, M.H.; et al. Transmission dynamics and control of severe acute respiratory syndrome. Science 2003, 300, 1966–1970. [Google Scholar]
- Riley, S.; Fraser, C.; Donnelly, C.A.; Ghani, A.C.; Abu-Raddad, L.J.; Hedley, A.J.; Leung, G.M.; Ho, L.M.; Lam, T.H.; Thach, T.Q.; et al. Transmission dynamics of the etiological agent of SARS in Hong Kong: impact of public health interventions. Science 2003, 300, 1961–1966. [Google Scholar]
- Seto, W.H.; Tsang, D.; Yung, R.W.; Ching, T.Y.; Ng, T.K.; Ho, M.; Ho, L.M.; Peiris, J.S. Effectiveness of precautions against droplets and contact in prevention of nosocomial transmission of severe acute respiratory syndrome (SARS). Lancet 2003, 361, 1519–1520. [Google Scholar]
- WHO (World Health Organization). Global surveillance for Severe Acute Respiratory Syndrome (SARS). Wkly. Epidemiol. Rec. 2003, 78, 100–109.
- AFNOR, Chemical disinfectants and- Virucidal quantitative suspension test for chemical disinfectants and antiseptics used in human medicine - Test method and requirements (phase 2, step 1). NF EN 14476+A1. 2007.
- ASTM, Standard Test Method for Efficacy of Virucidal Agents Intended for Inanimate Environmental Surfaces. E1053-97 (last reapproval in 2002). 1997.
- ASTM, Standard Test Method for Efficacy of Antimicrobial Agents Against Viruses in Suspension. E1052-96 (last reapproval in 2002), 1996.
- ASTM, Standard Test Method for Neutralization of Virucidal Agents in Virucidal Efficacy Evaluations. E1482-04. 2004.
- ASTM (American Society for Testing and Materials), Standard Test Method for Determining the Virus-Eliminating Effectiveness of Liquid Hygienic Handwash and Handrub Agents Using the Fingerpads of Adult Volunteers. E1838-02. 2002.
- ASTM (American Society for Testing and Materials), Standard Test Method for Evaluation of Hygienic Handwash and Handrub Formulations for Virus-Eliminating Activity Using the Entire Hand. E2011-09. 2009.
- Sattar, S.A.; Springthorpe, V.S.; Karim, Y.; Loro, P. Chemical disinfection of non-porous inanimate surfaces experimentally contaminated with four human pathogenic viruses. Epidemiol. Infect. 1989, 102, 493–505. [Google Scholar] [CrossRef]
- Kariwa, H.; Fujii, N.; Takashima, I. Inactivation of SARS coronavirus by means of povidone-iodine, physical conditions and chemical reagents. Dermatology. 2006, 212 Suppl 1, 119–123. [Google Scholar]
- Geller, C.; Fontanay, S.; Finance, C.; Duval, R.E. A new Sephadex-based method for removing microbicidal and cytotoxic residues when testing antiseptics against viruses: Experiments with a human coronavirus as a model. J. Virol. Methods. 2009, 159, 217–226. [Google Scholar] [CrossRef]
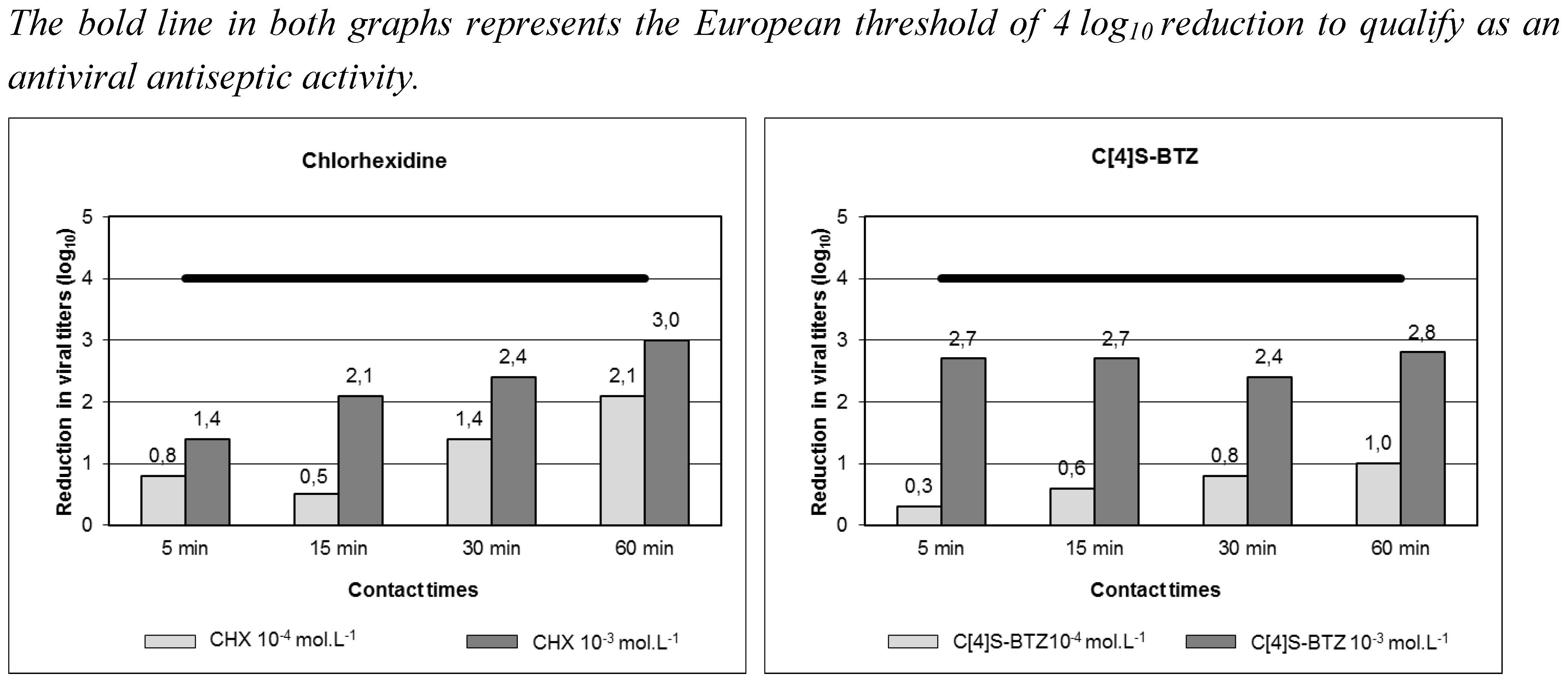
- Geller, C.; Fontanay, S.; Mourer, M.; Dibama, H.M.; Regnouf-de-Vains, J.B.; Finance, C.; Duval, R.E. Antiseptic properties of two calix[4]arenes derivatives on the human coronavirus 229E. Antiviral Res. 2010, 88, 343–346. [Google Scholar] [CrossRef]
- Rabenau, H.F.; Kampf, G.; Cinatl, J.; Doerr, H.W. Efficacy of various disinfectants against SARS coronavirus. J. Hosp. Infect. 2005, 61, 107–111. [Google Scholar] [CrossRef]
- Hulkower, R.L.; Casanova, L.M.; Rutala, W.A.; Weber, D.J.; Sobsey, M.D. Inactivation of surrogate coronaviruses on hard surfaces by health care germicides. Am. J. Infect. Control. 2011, 39, 401–407. [Google Scholar] [CrossRef]
- Dellanno, C.; Vega, Q.; Boesenberg, D. The antiviral action of common household disinfectants and antiseptics against murine hepatitis virus, a potential surrogate for SARS coronavirus. Am. J. Infect. Control. 2009, 37, 649–652. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Geller, C.; Varbanov, M.; Duval, R.E. Human Coronaviruses: Insights into Environmental Resistance and Its Influence on the Development of New Antiseptic Strategies. Viruses 2012, 4, 3044-3068. https://doi.org/10.3390/v4113044
Geller C, Varbanov M, Duval RE. Human Coronaviruses: Insights into Environmental Resistance and Its Influence on the Development of New Antiseptic Strategies. Viruses. 2012; 4(11):3044-3068. https://doi.org/10.3390/v4113044
Chicago/Turabian StyleGeller, Chloé, Mihayl Varbanov, and Raphaël E. Duval. 2012. "Human Coronaviruses: Insights into Environmental Resistance and Its Influence on the Development of New Antiseptic Strategies" Viruses 4, no. 11: 3044-3068. https://doi.org/10.3390/v4113044