Neonatal Citrulline Supplementation and Later Exposure to a High Fructose Diet in Rats Born with a Low Birth Weight: A Preliminary Report
Abstract
:1. Introduction
2. Materials and Methods
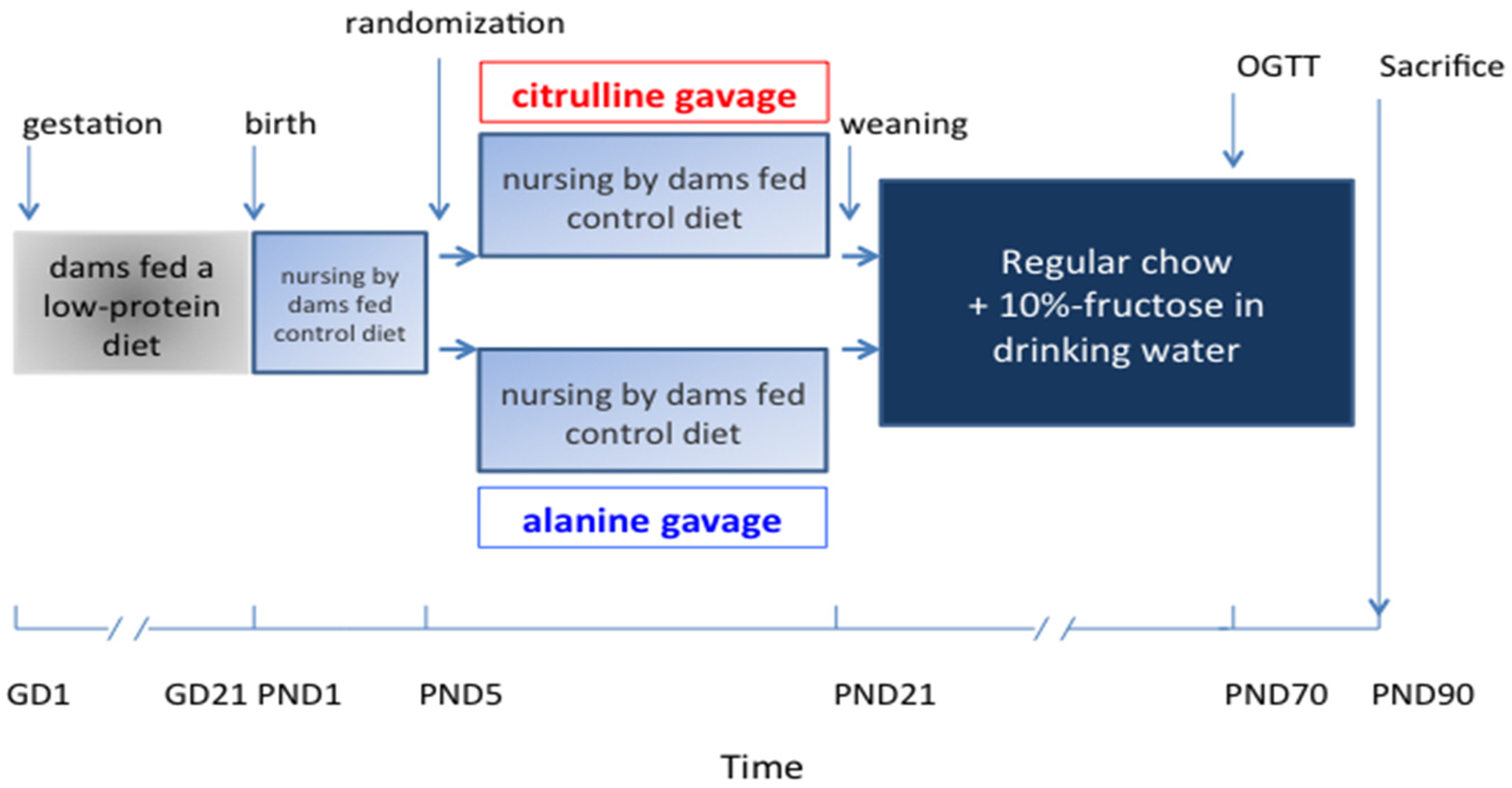
2.1. Experimental Design
2.2. Biochemical Measures
2.3. Non-Targeted Liver Phenotyping by Using Liquid Chromatography-High Resolution-Mass Spectrometry
2.4. RNA Isolation and Real-Time Polymerase Chain ReactionRT-PCR
2.5. Oral Glucose Tolerance Test
2.6. Statistical Analysis
3. Results
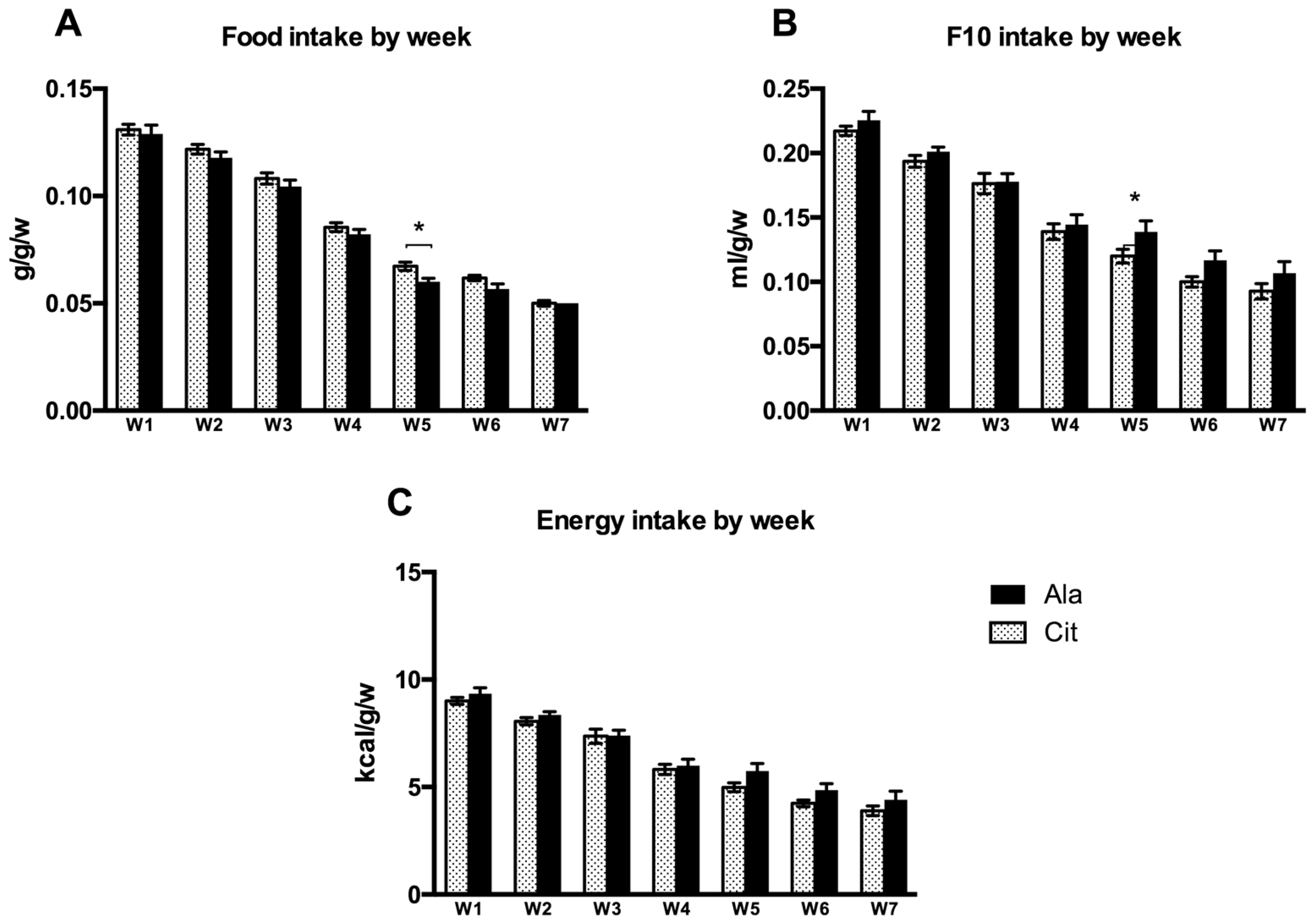
3.1. Energy Intake
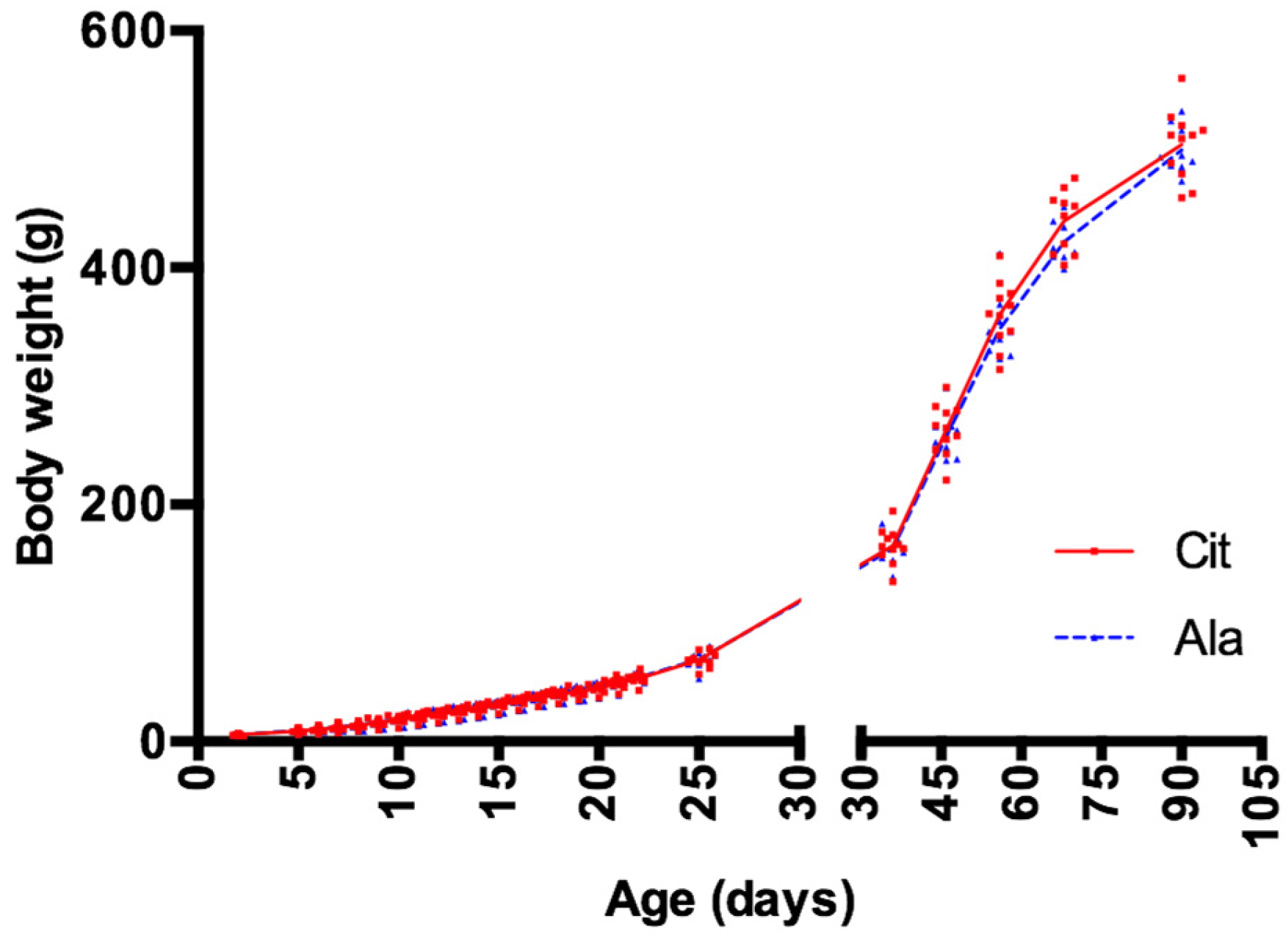
3.2. Growth and Body Composition
3.3. Glucose Metabolism (Figure 4)
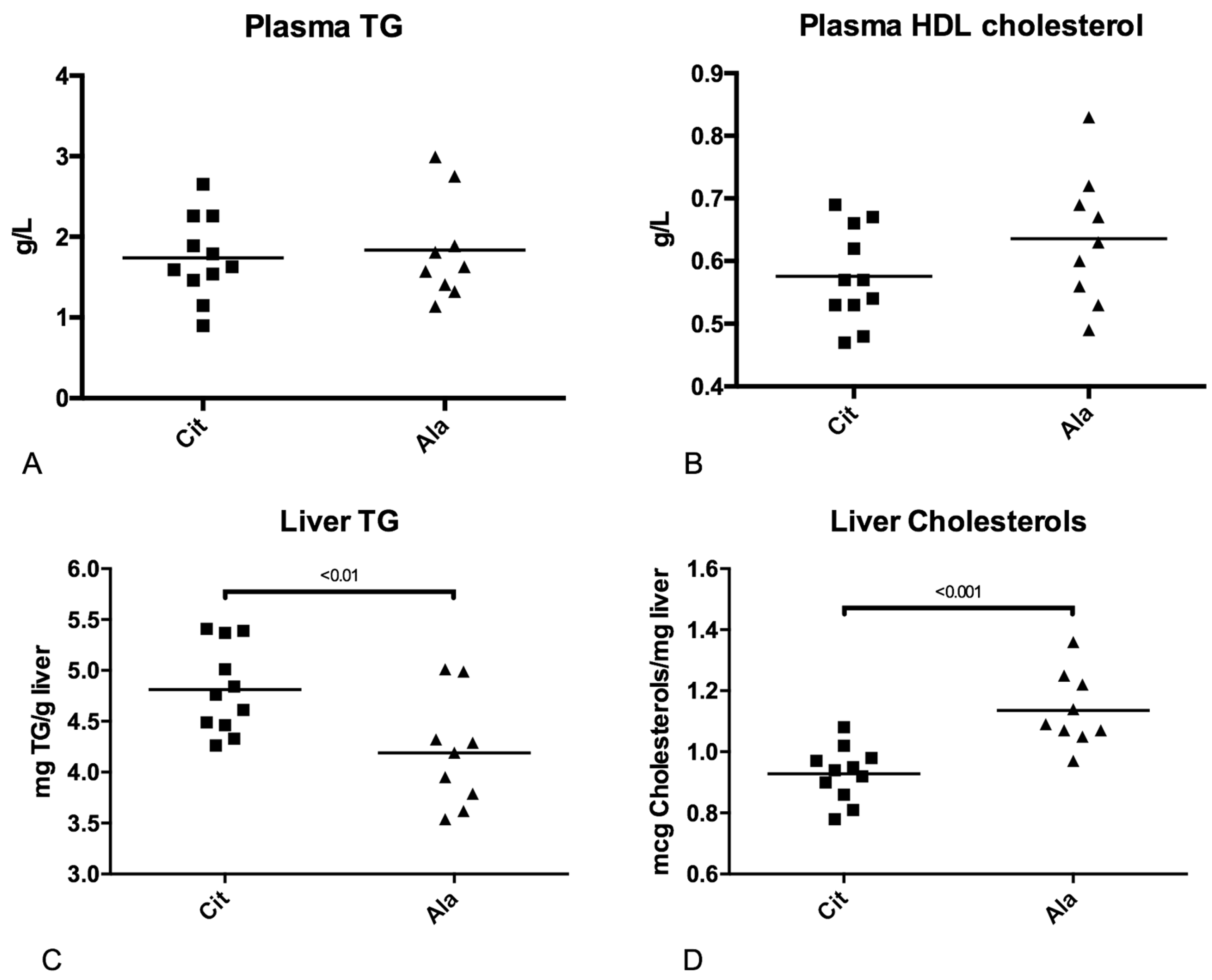
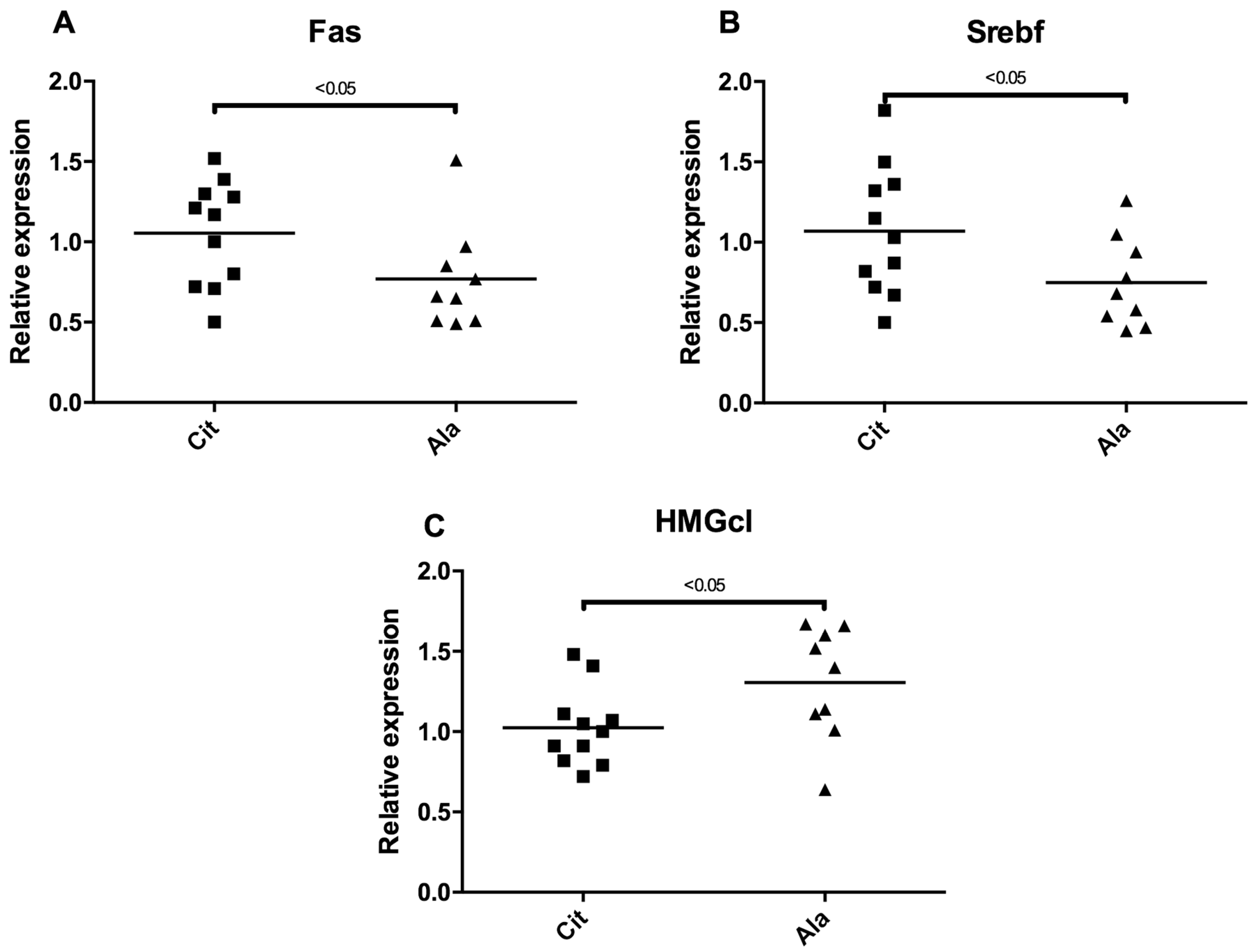
3.4. Lipid Metabolism
4. Discussion
4.1. Oral Citrulline Administration during Lactation Period Did Not Alter Pre-Weaning Growth
4.2. Oral Citrulline Administration before Weaning May Impact Liver Lipid Metabolism in the Long Run
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bernstein, I.M.; Horbar, J.D.; Badger, G.J.; Ohlsson, A.; Golan, A. Morbidity and mortality among very-low-birth-weight neonates with intrauterine growth restriction. The Vermont Oxford Network. Am. J. Obstet. Gynecol. 2000, 182, 198–206. [Google Scholar] [CrossRef]
- Godfrey, K.M.; Barker, D.J. Fetal nutrition and adult disease. Am. J. Clin. Nutr. 2000, 71, 1344S–1352S. [Google Scholar] [PubMed]
- Ehrenkranz, R.A.; Dusick, A.M.; Vohr, B.R.; Wright, L.L.; Wrage, L.A.; Poole, W.K. Growth in the neonatal intensive care unit influences neurodevelopmental and growth outcomes of extremely low birth weight infants. Pediatrics 2006, 117, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- De Curtis, M.; Rigo, J. Extrauterine growth restriction in very-low-birthweight infants. Acta Paediatr. 2004, 93, 1563–1568. [Google Scholar] [CrossRef] [PubMed]
- Stettler, N.; Stallings, V.A.; Troxel, A.B.; Zhao, J.; Schinnar, R.; Nelson, S.E.; Ziegler, E.E.; Strom, B.L. Weight gain in the first week of life and overweight in adulthood: A cohort study of European American subjects fed infant formula. Circulation 2005, 11, 1897–1903. [Google Scholar] [CrossRef] [PubMed]
- Kelishadi, R.; Mansourian, M.; Heidari-Beni, M. Association of fructose consumption and components of metabolic syndrome in human studies: A systematic review and meta-analysis. Nutrition 2014, 30, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Bursać, B.N.; Vasiljević, A.D.; Nestorović, N.M.; Veličković, N.A.; Vojnović Milutinović, D.D.; Matić, G.M.; Djordjevic, A.D. High-fructose diet leads to visceral adiposity and hypothalamic leptin resistance in male rats--do glucocorticoids play a role? J. Nutr. Biochem. 2014, 25, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Collins, J.K.; Perkins-Veazie, P.; Siddiq, M.; Dolan, K.D.; Kelly, K.A.; Heaps, C.L.; Meininger, C.J. Dietary supplementation with watermelon pomace juice enhances arginine availability and ameliorates the metabolic syndrome in Zucker diabetic fatty rats. J. Nutr. 2007, 137, 2680–2685. [Google Scholar] [PubMed]
- Jobgen, W.; Meininger, C.J.; Jobgen, S.C.; Li, P.; Lee, M.J.; Smith, S.B.; Spencer, T.E.; Fried, S.K.; Wu, G. Dietary l-arginine supplementation reduces white fat gain and enhances skeletal muscle and brown fat masses in diet-induced obese rats. J. Nutr. 2009, 139, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Mohan, S.; Patel, H.; Bolinaga, J.; Soekamto, N. AMP-activated protein kinase regulates l-arginine mediated cellular responses. Nutr. Metab. (Lond.) 2013, 10, 40. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.X.; Xu, X.; Cui, Y.; Wang, P.; Wei, X.; Yang, S.; Edin, M.L.; Zeldin, D.C.; Wang, D.W. Increased endothelial nitric-oxide synthase expression reduces hypertension and hyperinsulinemia in fructose-treated rats. J. Pharmacol. Exp. Ther. 2009, 328, 610–620. [Google Scholar] [CrossRef] [PubMed]
- El-Bassossy, H.M.; El-Fawal, R.; Fahmy, A.; Watson, M.L. Arginase inhibition alleviates hypertension in the metabolic syndrome. Br. J. Pharmacol. 2013, 169, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Piatti, P.M.; Monti, L.D.; Valsecchi, G.; Magni, F.; Setola, E.; Marchesi, F.; Galli-Kienle, M.; Pozza, G.; Alberti, K.G. Long-term oral l-arginine administration improves peripheral and hepatic insulin sensitivity in type 2 diabetic patients. Diabetes Care 2001, 24, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Windmueller, H.G.; Spaeth, A.E. Source and fate of circulating citrulline. Am. J. Physiol. 1981, 241, E473–E480. [Google Scholar] [PubMed]
- Curis, E.; Nicolis, I.; Moinard, C.; Osowska, S.; Zerrouk, N.; Bénazeth, S.; Cynober, L. Almost all about citrulline in mammals. Amino Acids 2005, 29, 177–205. [Google Scholar] [CrossRef] [PubMed]
- Rougé, C.; Des Robert, C.; Robins, A.; Le Bacquer, O.; Volteau, C.; De La Cochetière, M.F.; Darmaun, D. Manipulation of citrulline availability in humans. Am. J. Physiol. 2007, 293, G1061–G1067. [Google Scholar] [CrossRef] [PubMed]
- Hannigsberg, J.; Bourdon, A.; Tran, T.; Castellano, B.; Poupeau, G.; Parnet, P.; Winer, N.; Darmaun, D. Effets de la supplémentation orale en citrulline sur la croissance fœtale dans un modèle animal de retard de croissance intra-utérin. Nutr. Clin. Metab. 2013, 27, S36–S37. (In French) [Google Scholar] [CrossRef]
- Jegatheesan, P.; Beutheu, S.; Ventura, G.; Nubret, E.; Sarfati, G.; Bergheim, I.; De Bandt, J.P. Citrulline and Nonessential Amino Acids Prevent Fructose-Induced Nonalcoholic Fatty Liver Disease in Rats. J. Nutr. 2015, 145, 2273–2279. [Google Scholar] [CrossRef] [PubMed]
- Jegatheesan, P.; Beutheu, S.; Freese, K.; Waligora-Dupriet, A.J.; Nubret, E.; Butel, M.J.; De Bandt, J.P. Preventive effects of citrulline on Western diet-induced non-alcoholic fatty liver disease in rats. Br. J. Nutr. 2016, 116, 191–203. [Google Scholar] [CrossRef] [PubMed]
- Bourdon, A.; Parnet, P.; Nowak, C.; Tran, N.T.; Winer, N.; Darmaun, D. l-Citrulline Supplementation Enhances Fetal Growth and Protein Synthesis in Rats with Intrauterine Growth Restriction. J. Nutr. 2016, 146, 532–541. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.T.; Amarger, V.; Bourdon, A.; Misbert, E.; Grit, I.; Winer, N.; Darmaun, D. Maternal citrulline supplementation enhances placental function and fetal growth in a rat model of IUGR: Involvement of insulin-like growth factor 2 and angiogenic factors. J. Mater. Fetal Neonatal Med. 2016. [Google Scholar] [CrossRef] [PubMed]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef] [PubMed]
- R Development Core Team. R: A Language and Environment for Statistical Computing. Available online: http://www.R-project.org/ (accessed on 7 April 2017).
- Ferchaud-Roucher, V.; Croyal, M.; Krempf, M.; Ouguerram, K. Plasma lipidome characterization using UHPLC-HRMS and ion mobility of hypertriglyceridemic patients on nicotinic acid. Atherosclerosis 2015, 241, e123–e124. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Gutt, M.; Davis, C.L.; Spitzer, S.B.; Llabre, M.M.; Kumar, M.; Czarnecki, E.M.; Schneiderman, N.; Skyler, J.S.; Marks, J.B. Validation of the insulin sensitivity index (ISI(0,120)): Comparison with other measures. Diab. Res. Clin. Pract. 2000, 47, 177–184. [Google Scholar] [CrossRef]
- Martin-Agnoux, A.; Antignac, J.P.; Simard, G.; Poupeau, G.; Darmaun, D.; Parnet, P.; Alexandre-Gouabau, M.C. Time window-dependent effect of perinatal maternal protein restriction on insulin sensitivity and energy substrate oxidation in adult male offspring. Am. J. Physiol. 2014, 307, R184–R197. [Google Scholar]
- Kim, S.W.; Wu, G. Dietary arginine supplementation enhances the growth of milk-fed young pigs. J. Nutr. 2004, 134, 625–630. [Google Scholar] [PubMed]
- Yao, K.; Yin, Y.L.; Chu, W.; Liu, Z.; Deng, D.; Li, T.; Huang, R.; Zhang, J.; Tan, B.; Wang, W.; Wu, G. Dietary arginine supplementation increases mTOR signaling activity in skeletal muscle of neonatal pigs. J. Nutr. 2008, 138, 867–872. [Google Scholar] [PubMed]
- Tain, Y.L.; Hsieh, C.S.; Lin, I.C.; Chen, C.C.; Sheen, J.M.; Huang, L.T. Effects of maternal l-citrulline supplementation on renal function and blood pressure in offspring exposed to maternal caloric restriction: The impact of nitric oxide pathway. Nitric Oxide 2010, 23, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Tain, Y.L.; Lee, C.T.; Huang, L.T. Long-term effects of maternal citrulline supplementation on renal transcriptome prevention of nitric oxide depletion-related programmed hypertension: The impact of gene-nutrient interactions. Int. J. Mol. Sci. 2014, 15, 23255–23268. [Google Scholar] [CrossRef] [PubMed]
- Tain, Y.L.; Sheen, J.M.; Chen, C.C.; Yu, H.R.; Tiao, M.M.; Kuo, H.C.; Huang, L.T. Maternal citrulline supplementation prevents prenatal dexamethasone-induced programmed hypertension. Free Radic. Res. 2014, 48, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Dekker, M.J.; Baker, C.; Naples, M.; Samsoondar, J.; Zhang, R.; Qiu, W.; Sacco, J.; Adeli, K. Inhibition of sphingolipid synthesis improves dyslipidemia in the diet-induced hamster model of insulin resistance: Evidence for the role of sphingosine and sphinganine in hepatic VLDL-apoB100 overproduction. Atherosclerosis 2013, 228, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Watt, M.J.; Barnett, A.C.; Bruce, C.R.; Schenk, S.; Horowitz, J.F.; Hoy, A.J. Regulation of plasma ceramide levels with fatty acid oversupply: Evidence that the liver detects and secretes de novo synthesised ceramide. Diabetologia 2012, 55, 2741–2746. [Google Scholar] [CrossRef] [PubMed]
- Wakil, S.J.; Abu-Elheiga, L.A. Fatty acid metabolism: Target for metabolic syndrome. J. Lipid Res. 2009, 50, S138–S143. [Google Scholar] [CrossRef] [PubMed]
- Capel, F.; Chabrier, G.; Pitois, E.; Rigaudière, J.P.; Le Plenier, S.; Durand, C.; Jouve, C.; de Bandt, J.P.; Cynober, L.; Moinard, C.; et al. Combining citrulline with atorvastatin preserves glucose homeostasis in a murine model of diet-induced obesity. Br. J. Pharmacol. 2015, 172, 4996–5008. [Google Scholar] [CrossRef] [PubMed]
- Massa, N.M.; Silva, A.S.; de Oliveira, C.V.; Costa, M.J.; Persuhn, D.C.; Barbosa, C.V.; Gonçalves, M.D. Supplementation with watermelon extract reduces total cholesterol and LDL cholesterol in adults with dyslipidemia under the influence of the MTHFR C677T polymorphism. J. Am. Coll. Nutr. 2016, 35, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Jegatheesan, P.; Beutheu, S.; Ventura, G.; Sarfati, G.; Nubret, E.; Kapel, N.; Waligora-Dupriet, A.J.; Bergheim, I.; Cynober, L.; De-Bandt, J.P. Effect of specific amino acids on hepatic lipid metabolism in fructose-induced non-alcoholic fatty liver disease. Clin. Nutr. 2016, 35, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Ilan, Y. Compounds of the sphingomyelin-ceramide-glycosphingolipid pathways as secondary messenger molecules: New targets for novel therapies for fatty liver disease and insulin resistance. Am. J. Physiol. 2016, 310, G1102–G1117. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Polyunsaturated fatty acids and inflammation. Biochem. Soc. Trans. 2005, 33, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Banni, S.; Di Marzo, V. Effect of dietary fat on endocannabinoids and related mediators: Consequences on energy homeostasis, inflammation and mood. Mol. Nutr. Food Res. 2010, 54, 82–92. [Google Scholar] [CrossRef] [PubMed]
- Rossmeisl, M.; Jilkova, Z.M.; Kuda, O.; Jelenik, T.; Medrikova, D.; Stankova, B.; Kristinsson, B.; Haraldsson, G.G.; Svensen, H.; Stoknes, I.; et al. Metabolic effects of n-3 PUFA as phospholipids are superior to triglycerides in mice fed a high-fat diet: Possible role of endocannabinoids. PLoS ONE 2012, 7, e38834. [Google Scholar] [CrossRef] [PubMed]
- Petry, C.J.; Dorling, M.W.; Pawlak, D.B.; Ozanne, S.E.; Hales, C.N. Diabetes in old male offspring of rat dams fed a reduced protein diet. Int. J. Exp. Diabetes Res. 2001, 2, 139–143. [Google Scholar] [CrossRef] [PubMed]


| Tissue | Citrulline Group (n = 11) | Alanine Group (n = 9) | |
|---|---|---|---|
| (mean ± SEM) | (mean ± SEM) | ||
| White adipose tissue (WAT) (intra-abdominal) | 21.19 ± 1.4 g | 21.63 ± 1.8 g | NS |
| Intra-abdominal WAT/total WAT ratio | 0.67 ± 0.02 | 0.67 ± 0.02 | NS |
| Estimated lean body mass/body weight ratio | 0.94 ± 0.00 | 0.94 ± 0.00 | NS |
| Liver mass/body weight ratio | 0.035 ± 0.001 | 0.036 ± 0.002 | NS |
| Lipid Class | Mode | Marker | Adduct | Citrulline Mean | Alanine Mean | p-Value—Unpaired t Test with Welch’s Correction |
|---|---|---|---|---|---|---|
| Phosphatidylcholines | ESI+ | PC(32:0) PC(16:0/16:0) | (M + H)+ | 2.885 | 3.266 | 0.0444 |
| ESI+ | PC(34:1) PC(16:0/18:1) | (M + H)+ | 4.8434 | 5.3756 | 0.0137 | |
| ESI+ | PC(38:5) PC(18:2/20:3) | (M + H)+ | 0.0574 | 0.0342 | 0.0227 | |
| Phosphatidylethanolamines | ESI− | PE(36:2) PE(18:0/18:2) | (M – H)− | 3.6840 | 0.0000 | <0.0001 |
| ESI− | PE(36:4) PE(16:0/20:4) | (M − H)− | 11.4000 | 0.0000 | <0.0001 | |
| ESI− | PE(36:4) PE(18:1/18:3) | (M − H)− | 1.8010 | 0.0000 | <0.0001 | |
| ESI− | PE(38:4) PE(18:0/20:4) | (M − H)− | 13.6400 | 0.0000 | <0.0001 | |
| ESI+ | PE (38:5) PE(18:1/20:4) | (M + H)+ | 1.3860 | 1.5560 | 0.0098 | |
| ESI− | PE(38:6) PE(18:2/20:4) | (M − H)− | 0.5036 | 0.0000 | <0.0001 | |
| ESI+ | PE(38:6) PE(16:0/22:6) | (M + H)+ | 0.8097 | 0.8858 | 0.0575 | |
| ESI+ | PE(40:6) PE(18:0/22:6) | (M + H)+ | 0.4043 | 0.4734 | 0.0043 | |
| ESI+ | PE(40:6) PE(20:2/20:4) | (M + H)+ | 0.0136 | 0.0202 | 0.0085 | |
| ESI+ | PE(40:7) PE(18:1/22:6) | (M + H)+ | 0.1834 | 0.2133 | 0.0624 | |
| Phosphatidylinositol | ESI− | PI(34:2) PI(16:0/18:2) | (M − H)− | 1.4680 | 0.0000 | <0.0001 |
| Phosphatidylserine | ESI− | PS(38:4) PS(18:0/20:4) | (M − H)− | 5.8520 | 0.0000 | <0.0001 |
| Sphingomyeline | ESI+ | SM(d18:1/24:1) | (M + H)+ | 2.6510 | 3.0650 | 0.0075 |
| Ceramides | ESI− | Cer(d(18:1/20:0) | (M + HC − O2)− | 0.2765 | 1.670 | <0.0001 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran, N.-T.; Alexandre-Gouabau, M.-C.; Pagniez, A.; Ouguerram, K.; Boquien, C.-Y.; Winer, N.; Darmaun, D. Neonatal Citrulline Supplementation and Later Exposure to a High Fructose Diet in Rats Born with a Low Birth Weight: A Preliminary Report. Nutrients 2017, 9, 375. https://doi.org/10.3390/nu9040375
Tran N-T, Alexandre-Gouabau M-C, Pagniez A, Ouguerram K, Boquien C-Y, Winer N, Darmaun D. Neonatal Citrulline Supplementation and Later Exposure to a High Fructose Diet in Rats Born with a Low Birth Weight: A Preliminary Report. Nutrients. 2017; 9(4):375. https://doi.org/10.3390/nu9040375
Chicago/Turabian StyleTran, Nhat-Thang, Marie-Cécile Alexandre-Gouabau, Anthony Pagniez, Khadija Ouguerram, Clair-Yves Boquien, Norbert Winer, and Dominique Darmaun. 2017. "Neonatal Citrulline Supplementation and Later Exposure to a High Fructose Diet in Rats Born with a Low Birth Weight: A Preliminary Report" Nutrients 9, no. 4: 375. https://doi.org/10.3390/nu9040375






