Deleterious Effects of Mycotoxin Combinations Involving Ochratoxin A
Abstract
:1.Introduction
2. Food Contamination with Mycotoxin Mixtures Involving OTA
| Sample/No (Country) | Mycotoxin | Contamination (%) | Range (μg·L−1 or µg·kg−1) | Co-occurrence | Reference | |||
|---|---|---|---|---|---|---|---|---|
| Beer/106 (25 EU)a | AF | Nd | - | Two toxins | [35] | |||
| DON | 66 | <0.5–18.6 | 41.5% | |||||
| FB1 | 96 | <0.1–30.3 | Three toxins | |||||
| FB2 | 57 | <0.1–3.9 | 42.4% | |||||
| OTA | 66 | <0.002–0.189 | ||||||
| Spices/105 (Turkey) | AF | 21.1–79.2 | 0.13–37.38 | 4.3%–62.5% | [36] | |||
| OTA | 17.4–75 (6.7% > EU limit for both toxins) | 0.06–98.2 | ||||||
| Cereals/110 Turkey | AF | 24.5 | 0.022–0.233 | 14.6% | [37] | |||
| OTA | 43.6 | 0.066–1.125 | ||||||
| Baby milk/62 (Turkey) | AFM1 | 8 | 0.06–0.022 | 1.6% | [38] | |||
| OTA | 19,4 | 0.017–0.184 | ||||||
| Food b/345 (Italy) | AF | 5 (1.2% > EU limit) | 0.33–70.69 | Not specified | [39] | |||
| OTA | 17.6 | 2.76–23.7 | ||||||
| Pasta/27 (Italy) | AFB1 | Nd | Not specified | [40] | ||||
| DON | 81.5 (26% > EU limit) | 35.1–450.0 | ||||||
| OTA | 96.3 | 0.2–0.52 | ||||||
| Breakfast Cereals/46 (Spain) | AF | 19 | 0.04 c | Two toxins 28% | [41] | |||
| OTA | 5 | 0.03 | ||||||
| ZEA | 43 | 2.87 | ||||||
| Barley/123 (Spain) | AF | 100 | 0.15 c | Two or three | [31] | |||
| OTA | 58 | 0.06 | toxins 80% | |||||
| ZEA | 39 | 0.84 | ||||||
| Wheat/37 Oat bran/30 (Spain) | DON | 62/17 (total of 19% > EU limit) | 1308/230c | Two toxins 10.5% Three toxins 4.5% | [42] | |||
| OTA | 30/20 | 1.1/0.3 | ||||||
| ZEA | 13/17 | 8/8 | ||||||
| Cereals and cereal-based food/265 (Mediterranean region) d | AFs | 10 (8% AFB1 > EU limit) | 4.2–66.7 | Two toxins 14%> two toxins 18% | [43] | |||
| BEA | 10 | 2.4–844 | ||||||
| DAS | 2.8 | 6.4–97 | ||||||
| DON | 4.5 | 63.2–296 | ||||||
| FB1 | 3 | <LOQ-186 | ||||||
| FB2 | 3.3 | <LOQ-176 | ||||||
| HT-2 | 4.5 | <LOQ-87 | ||||||
| NIV | 50 | 100–903 | ||||||
| OTA | 1.8 (All > EU limit) | 75–112 | ||||||
| T-2 | 5 | 12.9–78.4 | ||||||
| Fermented meat/90 (Croatia) | AFB1 | 10 | <1.0–3.0 | Not specified | [44] | |||
| OTA | 64.4 | <0.05–7.83 | ||||||
| CIT | 4.4 | <1.0–1.0 | ||||||
3.Toxicity of Mycotoxin Combinations Involving OTA
| Experimental model | Mycotoxin combination/treatment | Effect (Interactions) | References |
|---|---|---|---|
| Opossum kidney cells | OTA + CIT (0.5–50 µM): | [76] | |
| concentrated OTA < concentrated CIT; | ↓Cytotoxicity (AN) | ||
| concentrated OTA ≤ concentrated CIT | ↑Cytotoxicity (A) | ||
| OTA (10 µM) + CIT (50 µM) | ↑DNA adducts | ||
| Human kidney cells (HK2) | OTA + CIT | ↑DNA adducts ↑COX2 Inhibition of CYP 3A4 expression | [77] |
| Porcine urinary bladder cells | OTA (0.001–1 μM) + CIT (0.001–1 μM)/24 h | No effect on viability | [80] |
| V79 cells | OTA (1–10 μM) + CIT (1–10 μM)/24 h | No effect on viability | [80] |
| PK15 cells | OTA (6 and 10 µM)+ CIT (30 and 50 μM)/24 h | ↑cytotoxicity (A) | [66] |
| Human proximal tubule cells | OTA (25 and 50 nm·L−1) + CIT (0.25 and 1 μmol·L−1)/24 h | No effect on caspase-3 activation | [82] |
| OTA (25 and 50 nmol·L−1) + CIT (2.5 and 5 μmol·L−1) | ↓caspase-3 (AN) | ||
| OTA (25 and 50 nmol·L−1) + CIT (7.5 and 15 μmol·L−1) | ↑;caspase-3 (A) | ||
| PK15 cells | OTA (30 and 50 μM) + CIT (6 and 10 μM)/12 and 24 h | ↑apoptosis (S) ↑cytosolic calcium level ↓micronucleus rate (AN) ↑nuclear buds (A) | [66] |
| Vero cells | OTA (12.5 and 25 μM) + CIT (60 μM)/12 h | ↓cell viability (S) ↑MDA level (S) ↑Hsp 70 expression | [63] |
| Rat | OTA (0.125 mg·kg−1 21 d, p.o.) + CIT (20 mg·kg−1 p.o.) | ↑plasma MDA level ↑plasma GSH level ↑hOGG1 tail intensity in liver and kidney | [83] |
| Rat | OTA (26 μg·kg−1 feed) + CIT (100 μg·kg−1 feed) for 21 d | ↑kidney OTA-DNA adduct formation (S) | [76] |
| S. typhimurium TA102 | OTA (12.3–1000.0 μg/plate) + CIT (3.0–250.0 μg/plate) | No increased mutagenicity | [77] |
| Chicken embryos | OTA (0.03–0.5 μg/embryo) + CIT (4 μg/embryo) | No increased teratogenicity | [85] |
| Pregnant rats | OTA (1 mg·kg−1) + CIT (30 mg·kg−1)/1 × s.c. | ↑fetal malformations | [86] |
| Chicken | OTA (0.5 mg·kg−1 b.m. for 2 d, i.m.) + CIT (25 mg·kg−1 infusion) | No effect on diuresis | [87] |
| Broiler chicks | OTA (3.0 mg·kg−1 feed) + CIT (300 mg·kg−1 feed) 21 d | No effect on plasma constituents | [88] |
| Rabbits | OTA (0.75 mg·kg−1 feed) + CIT (15 mg·kg−1 feed) 60 d | ↑ ultrastructural kidney changes | [89] |
| Dogs | OTA (0.1 and 0.2 mg·kg−1 b.m) p.o. + CIT (5 kg−1 b.m)/i.p. 14 d | Kidney necrosis Ulceration of intestinal mucosa ↑mortality | [90] |
| Human PBM | OTA (0.0038–12.5 mg·L−1) + mixture PA + CIT + FB1 (0.038–125 mg·L−1)/24 h | No effect on metabolic activity (AN) | [95] |
| Chickens | OTA 0.1 mg·mL−1 + PA 6 mg·mL−1/gastric intubation 20 and 28 d | ↑mortality (S) | [98] |
| Chickens | OTA (0.13–0.8 mg·kg−1 feed) + PA (1–2 mg·kg−1 feed)/6–10 weeks | ↓body weight, ↑ degenerative changes in the kidney, liver and lymphoid organs (S) | [103] |
| Mice | OTA (10 mg·kg−1 feed) + PA (40 mg·kg−1 feed)/10 d | ↑mortality, acute multifocal toxic tubular nephrosis (S) | [99] |
| Pigs | OTA (0.09–0.79 mg·kg−1 feed) + PA (1–9 mg·kg−1 feed)/3–5 months | ↑degenerations in kidney proximal tubules and proliferation in the interstitium (S) | [104] |
| Human PBM | OTA (0.0038–12.5 mg·L−1) + mixture CIT + FB1 (0.038–125 mg·L−1)/24 h | ↑cytotoxicity (S) | [95] |
| C6 glioma cells, Caco-2 and Vero cells | OTA (10 µM) + FB1 (5, 25 and 50 µM)/24–72 h | ↑cytotoxicity (S) | [116] |
| PK15 cells | OTA (0.05, 0.5 and 5 µg·mL−1) + FB1 (0.05, 0.5 and 5 µg·mL−1)/24 h | ↑cytotoxicity, lipid peroxidation and micronuclei (AD); ↓ glutathione level (AD), ↑caspase-3 (S) | [117,118,121] |
| Turkey poults | OTA (3 mg·kg−1 feed) + FB1 (300 mg·kg−1 feed)/3 weeks | ↓body weight, ↑AST, ALT, creatinine (S); serum triglycerides (AN) | [119] |
| Rabbits | OTA (2 mg·kg−1 feed) + FB1 (10 mg·kg−1 feed)/45 d | ↑ALP (AD); ↑AST, ALT (LAD) | [120] |
| Rats | OTA (5 ng·kg−1, 0.05, 0.5 mg·kg−1) + FB1 (200 ng·kg−1, 0.05, 0.5 mg·kg−1)/p.o. 15 d | ↑lipid peroxidation, protein carbonyls, DNA damage in the kidneys (S); ↓ catalase (S); ↑DNA adducts (S) | [8,112,113] |
| Pigs | OTA (0.5 mg·kg−1 feed) + FB1 (10 mg·kg−1 feed)/35–49 d | ↑kidney damage, AST, ALT, creatinine (S); ↓ antibody titer against the Morbus Aujeszky (S) | [122] |
| Vero cells | OTA + AFB1 (5–50 µM)/24 hOTA (1 µM) + AFB1 (1–20 µM)OTA (1–20 µM) + AFB1 (1 µM) | ↑cytotoxicity (A) ↑DNA damage, p53 ↓bcl-2 | [129] |
| HepG2 cells | OTA 1–200 µM + AFB1 (100 or 150 µM)/24 h | ↑cytotoxicity (AD); ↓ DNA damage (AN) | [130] |
| Rats | Single doses OTA 0.5 mg·kg−1 + AFB1 0.25 mg·kg−1 p.o. | No interactions in acute liver toxicity, kidney or immunological organs damage | [133] |
3.1.Combined Toxicity of OTA and CIT
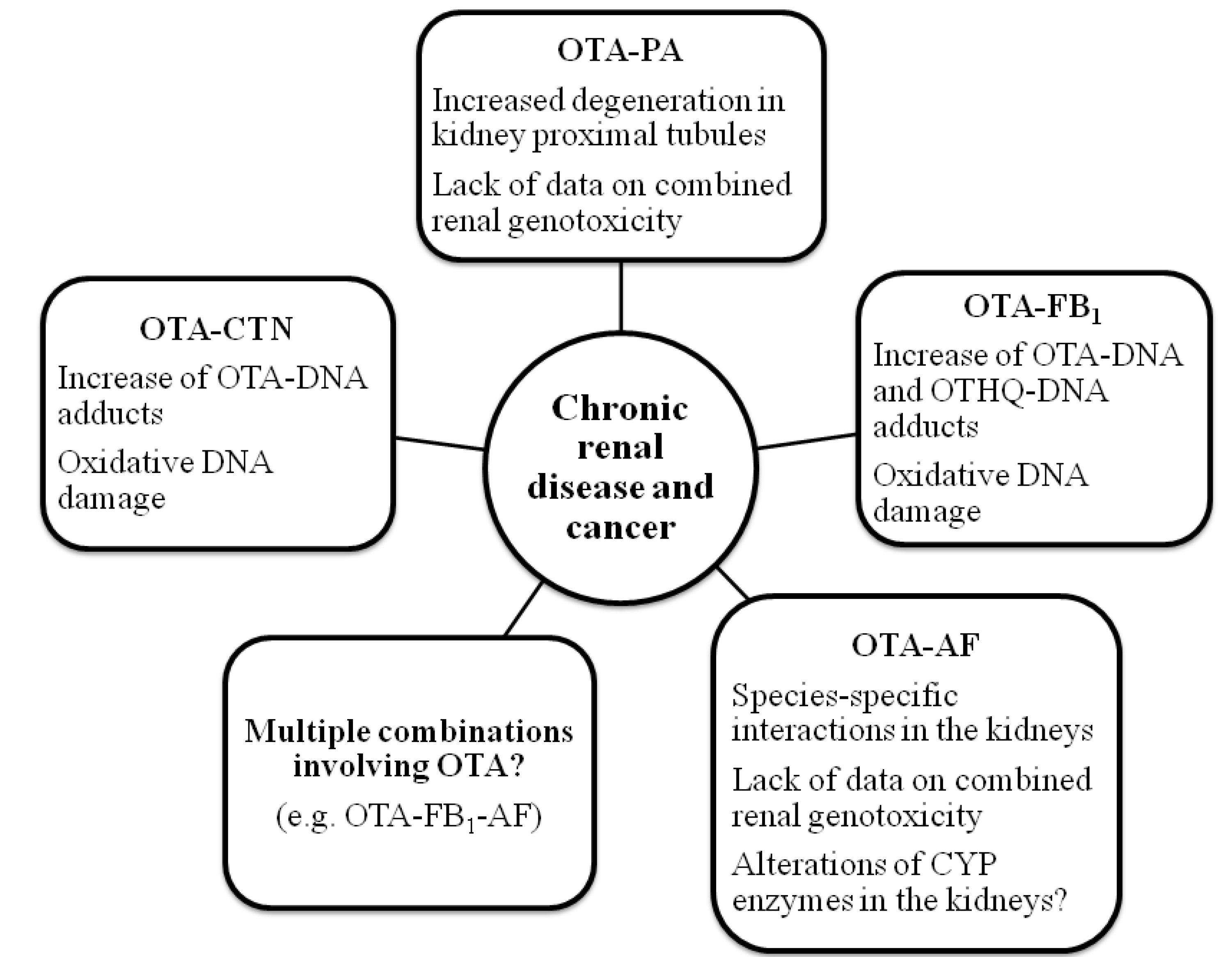
3.2.Combined Toxicity of OTA and PA
3.3.Combined Toxicity of OTA and FB1
3.4.Combined Toxicity of OTA and AF
4. Conclusions
Conflict of Interest
References
- Commission regulation (EC) no. 1881/2006 of 19 December 2006 on setting maximum levels for certain contaminants in foodstuffs amended by Commission regulations no. 1126/2007 of 28 September 2007, no. 105/2010 of 5 February 2010 and no. 165/2010 of 26 February 2010. pp. 1–26. Available online: http://www.eur-lex.europa.eu (accessed on 29 September 2013).
- O’Brien, E.; Heussner, A.H.; Dietrich, D.R. Species-, sex-, and cell type-specific effects of ochratoxin A and B. Toxicol. Sci. 2001, 63, 256–264. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer (IARC). Ochratoxin A. In Some Naturally Occurring Substances: Food Items and Constituents, Heterocyclic Aromatic Amines and Mycotoxins. IARC Monogr. Eval. Carcinog. Risks Hum. 1993, 56, 489–521.
- Duarte, S.C.; Pena, A.; Lino, C.M. A review on ochratoxin A occurrence and effects of processing of cereal and cereal derived food products. Food Microbiol. 2010, 27, 187–198. [Google Scholar] [CrossRef]
- Peraica, M.; Flajs, D.; Domijan, A.M.; Ivić, D.; Cvjetković, B. Ochratoxin A contamination of food from Croatia. Toxins 2010, 2, 2098–2105. [Google Scholar]
- Peraica, M.; Domijan, A.M.; Miletić-Medved, M.; Fuchs, R. The involvement of mycotoxins in the development of endemic nephropathy. Wien. Klin. Wochenschr. 2008, 120, 402–407. [Google Scholar] [CrossRef]
- Pfohl-Leszkowicz, A.; Tozlovanu, M.; Manderville, R.A.; Peraica, M.; Castegnaro, M.; Stefanovic, V. New molecular and field evidences for the implication of mycotoxins but not aristolochic acid in human nephropathy and urinary tract tumor. Mol. Nutr. Food Res. 2007, 51, 131–146. [Google Scholar]
- Pfohl-Leszkowicz, A. Ochratoxin A and aristolochic acid in the nephropathies and associated urothelial tract tumours development. Arh. Hig. Rada Toksikol. 2009, 60, 465–483. [Google Scholar]
- Reddy, L.; Bhoola, K. Ochratoxins-food contaminants: Impact on human health. Toxins 2010, 2, 771–779. [Google Scholar] [CrossRef]
- Domijan, A.M.; Peraica, M.; Cvjetković, B.; Turčin, S.; Jurjević, Ž.; Cvjetković, B. Mold contamination and co-occurrence of mycotoxins in corn grain in Croatia. Acta Pharm. 2005, 55, 349–356. [Google Scholar]
- Šegvić Klarić, M.; Cvetnić, Z.; Pepeljnjak, S.; Kosalec, I. Co-occurrence of aflatoxins, ochratoxin A, fumonisins, and zearalenone in cereals and feed, determined by competitive direct enzyme-linked immunosorbent assay and thin-layer chromatography. Arh. Hig. Rada Toksikol. 2009, 60, 427–434. [Google Scholar]
- Stoev, S.D.; Dutton, M.; Njobeh, P.; Mosonik, J.; Steenkamp, P. Mycotoxic nephropathy in Bulgarian pigs and chickens: complex aetiology and similarity to Balkan endemic nephropathy. Food Addit. Contam. A 2010, 27, 72–88. [Google Scholar] [CrossRef]
- Cabañes, F.J.; Accensi, F.; Bragulat, M.R.; Abarca, M.L.; Castellá, G.; Minguez, S.; Pons, A. What is the source of ochratoxin A in wine? Int. J. Food Microbiol. 2002, 79, 213–215. [Google Scholar]
- Joint FAO/WHO Expert Committee on Food Additives (JECFA), Safety Evaluation of Certain Food Additives and Contaminants; WHO Food Additives Series 59; WHO: Geneva, Switzerland, 2008.
- Paterson, R.R.M.; Lima, N. How will climate change affect mycotoxins in food? Food Res. Int. 2010, 43, 1902–1914. [Google Scholar] [CrossRef] [Green Version]
- Bragulat, M.R.; Martínez, E.; Castellá, G.; Cabañes, F.J. Ochratoxin A and citrinin producing species of the genus Penicillium from feedstuffs. Int. J. Food Microbiol. 2008, 126, 43–48. [Google Scholar]
- Frisvad, J.C.; Frank, J.M.; Houbraken, J.A.M.P.; Kuijpers, A.F.A.; Samson, R.A. New ochratoxin A producing species of Aspergillus section. Circumdati. Stud. Mycol. 2004, 50, 23–43. [Google Scholar]
- Stoev, S.D.; Hald, B.; Mantle, P. Porcine nephropathy in Bulgaria: A progressive syndrome of complex of uncertain (mycotoxin) etiology. Vet. Res. 1998, 142, 190–194. [Google Scholar]
- Frisvad, J.C.; Smedsgaard, J.; Samson, R.A.; Larsen, T.O.; Thrane, U. Fumonisin B2 production by Aspergillus niger. J. Agr. Food Chem. 2007, 55, 9727–9732. [Google Scholar] [CrossRef]
- Frisvad, J.C.; Larsen, T.O.; Thrane, U.; Meijer, M.; Varga, J.; Samson, R.A.; Nielsen, K.F. Fumonisin and ochratoxin production in industrial Aspergillus niger strains. PloS One 2011, 6, 1–6. [Google Scholar]
- Susca, A.; Proctor, R.H.; Mule, G.; Stea, G.; Ritieni, A.; Logrieco, A.; Moretti, A. Correlation of mycotoxin fumonisin B2 production and presence of the fumonisin biosynthetic gene fum8 in Aspergillus niger from grape. J. Agric. Food Chem. 2010, 58, 9266–9272. [Google Scholar]
- Paterson, R.R.M.; Lima, N. Further mycotoxin effects from climate change. Food Res. Int. 2011, 44, 2555–2566. [Google Scholar] [Green Version]
- Soares, C.; Calado, T.; Venâncio, A. Mycotoxin production by Aspergillus niger aggregate strains isolated from harvested maize in three Portuguese regions. Rev. Iberoam. Micol. 2013, 30, 9–13. [Google Scholar] [CrossRef]
- Pepeljnjak, S.; Šegvić Klarić, M. «Suspects» in etiology of endemic nephropathy: Aristolochic acid versus mycotoxins. Toxins 2010, 2, 1414–1427. [Google Scholar]
- Šegvić Klarić, M. Adverse effects of combined mycotoxins. Arh. Hig. Rada Toksikol. 2012, 63, 519–530. [Google Scholar]
- Jurjević, Ž.; Solfrizzo, M.; Cvjetković, B.; Avantaggiato, G.; Visconti, A. Ochratoxin A and fumonisins (B1 and B2) in corn from Balkan nephropathy endemic and non endemic areas of Croatia. Mycot. Res. 1999, 15, 67–80. [Google Scholar]
- Vrabcheva, T.; Usleber, E.; Dietrich, R.; Maärtlbauer, E. Co-occurrence of ochratoxin A and citrinin in cereals from bulgarian villages with a history of Balkan endemic nephropathy. J. Agric. Food Chem. 2000, 48, 2483–2488. [Google Scholar] [CrossRef]
- Puntarić, D.; Bošnir, J.; Šmit, Z.; Škes, I.; Baklaić, Ž. Ochratoxin A in corn and wheat: Geographical association with endemic nephropathy. Croat. Med. J. 2001, 42, 175–180. [Google Scholar]
- Domijan, A.M.; Peraica, M.; Žlender, V.; Cvjetković, B.; Jurjević, Ž.; Topolovec-Pintarić, S.; Ivić, D. Seed-borne fungi and ochratoxin A contamination of dry beans (Phaseolus vulgaris L.) in the Republic of Croatia. Food Chem. Toxicol. 2005, 43, 427–432. [Google Scholar] [CrossRef]
- Streit, E.; Schatzmayr, G.; Tassis, P.; Tzika, E.; Marin, D.; Taranu, I.; Tabuc, C.; Nicolau, A.; Aprodu, I.; Puel, O.; et al. Current situation of mycotoxin contamination and co-occurrence in animal feed-focus on Europe. Toxins 2012, 4, 788–809. [Google Scholar] [CrossRef]
- Ibáńez-Vea, M.; González-Peńas, E.; Lizarraga, E.; López de Cerain, A.L. Co-occurrence of aflatoxins, ochratoxin A and zearalenone in barley from a northern region of Spain. Food Chem. 2012, 132, 35–42. [Google Scholar]
- Ibáñez-Vea, M.; González-Peñas, E.; Lizarraga, E.; López de Cerain, A.L. Co-occurrence of mycotoxins in Spanish barley: A statistical overview. Food Control 2012, 28, 295–298. [Google Scholar]
- Anfossi, L.; Baggiani, C.; Giovannoli, C.; D’Arco, G.; Passini, C.; Giraudi, G. Occurrence of aflatoxin M1 in Italian cheese: Results of a survey conducted in 2010 and correlation with manufacturing, production season, milking animals, and maturation of cheese. Food Control 2012, 25, 125–130. [Google Scholar]
- Domaćinović, M.; Ćosić, J.; Klapec, T.; Peraica, M.; Mitak, M.; Gross-Bošković, A.; Žarković, K. Frequency and level of animal feed contamination by mycotoxins in Croatia. In Proceedings of Krmiva 2013, Opatija, Croatia, 5–7 June 2013; pp. 29–30.
- Bertuzzi, T.; Rastelli, S.; Mulazzi, A.; Donadini, G.; Pietri, A. Mycotoxin occurrence in beer produced in several European countries. Food Control 2011, 22, 2059–2064. [Google Scholar] [CrossRef]
- Ozbey, F.; Kabak, B. Natural co-occurrence of aflatoxins and ochratoxin A in spices. Food Control 2012, 28, 354–361. [Google Scholar] [CrossRef]
- Kabak, B. Determination of aflatoxins and ochratoxin A in retail cereal products from Turkey by high performance liquid chromatography with fluorescence detection. Food Control 2012, 28, 1–6. [Google Scholar] [CrossRef]
- Kabak, B. Aflatoxin M1 and ochratoxin A in baby formulae in Turkey: Occurrence and safety evaluation. Food Control 2012, 26, 182–187. [Google Scholar] [CrossRef]
- Imperato, R.; Campone, L.; Piccinelli, A.L.; Veneziano, A.; Rastrelli, L. Survey of aflatoxins and ochratoxin a contamination in food products imported in Italy. Food Control 2011, 22, 1905–1910. [Google Scholar] [CrossRef]
- Raiola, A.; Meca, G.; Mañes, J.; Ritieni, A. Bioaccessibility of deoxynivalenol and its natural co-occurrence with ochratoxin A and aflatoxin B1 in Italian commercial pasta. Food Addit. Contam. 2012, 50, 280–287. [Google Scholar]
- Ibáńez-Vea, M.; González-Peńas, E.; Lizarraga, E.; López de Cerain, A.L. Co-occurrence of aflatoxins, ochratoxin A and zearalenone in breakfast cereals from spanish market. Food Control 2011, 22, 1949–1955. [Google Scholar] [CrossRef]
- Vidal, A.; Marin, S.; Ramos, A.J.; Cano-Sancho, G.; Sanchis, V. Determination of aflatoxins, deoxynivalenol, ochratoxin A and zearalenone in wheat and oat based bran supplements sold in the Spanish market. Food Chem. Toxicol. 2013, 53, 133–138. [Google Scholar]
- Serrano, A.B.; Font, G.; Ruiz, M.J.; Ferrer, E. Co-occurrence and risk assessment of mycotoxins in food and diet from Mediterranean area. Food Chem. 2012, 135, 423–429. [Google Scholar] [CrossRef]
- Markov, K.; Pleadin, J.; Bevardi, M.; Vahčić, N.; Sokolić-Mihalak, D.; Frece, J. Natural occurrence of aflatoxin B1, ochratoxin A and citrinin in Croatian fermented meat products. Food Control 2013, 34, 312–317. [Google Scholar] [CrossRef]
- Pepeljnjak, S.; Blažević, N. Contamination with molds and occurrence of ochratoxin A in smoked meat products from endemic nephropathy region of Yugoslavia. In Proceedings of Vth International IUPAC Symposium on Mycotoxins and Phycotoxins, Vienna, Austria, 1–3 September 1982; Pfannhauser, W., Czedick-Eysenberg, P.B., Eds.; pp. 102–105.
- Sørensen, L.M.; Mogensen, J.; Nielsen, K.F. Simultaneous determination of ochratoxin A, mycophenolic acid and fumonisin B2 in meat products. Anal. Bioanal. Chem. 2010, 398, 1535–1542. [Google Scholar]
- Speijers, G.J.A.; Speijers, M.H.M. Combined toxic effect of mycotoxins. Toxicol. Lett. 2004, 153, 91–98. [Google Scholar] [CrossRef]
- Grenier, B.; Oswald, I.P. Mycotoxin co-contamination of food and feed: meta-analysis of publications describing toxicological interactions. World Mycotoxin J. 2011, 4, 285–313. [Google Scholar] [CrossRef]
- Pfohl-Leszkowicz, A.; Petkova-Bocharova, T.; Chernozemsky, I.N.; Castegnaro, M. Balkan endemic nephropathy and associated urinary tract tumors: A review on aetiological causes and the potential role of mycotoxins. Food Addit. Contam. 2002, 19, 282–302. [Google Scholar] [CrossRef]
- Pfohl-Leszkowicz, A.; Manderville, R.A. Ochratoxin A: An overview on toxicity and carcinogenicity in animals and humans. Mol. Nutr. Food Res. 2007, 51, 61–99. [Google Scholar] [CrossRef]
- Pfohl-Leszkowicz, A.; Manderville, R.A. An update on direct genotoxicity as molecular mechanism of ochratoxin A carcinogenicity. Chem. Res. Toxicol. 2012, 25, 252–262. [Google Scholar] [CrossRef]
- Vettorazzi, A.; van Delft, J.; López de Cerain, A. A review on ochratoxin A transcriptomic studies. Food Chem. Toxicol. 2013, 59, 766–783. [Google Scholar] [CrossRef]
- Akman, S.A.; Adams, M.; Case, D.; Park, G.; Manderville, R.A. Mutagenicity of ochratoxin A and its hydroquinone metabolite in the SupF gene of the mutation reporter plasmid Ps189. Toxins 2012, 4, 267–280. [Google Scholar] [CrossRef]
- Hadjeba-Medjdoub, K.; Tozlovanu, M.; Pfohl-Leszkowicz, A.; Frenette, C.; Paugh, R.J.; Manderville, R.A. Structure-activity relationships imply different mechanisms of action for ochratoxin A-mediated cytotoxicity and genotoxicity. Chem. Res. Toxicol. 2012, 25, 181–190. [Google Scholar] [CrossRef]
- Hibi, D.; Suzuki, Y.; Ishii, Y.; Jin, M.; Watanabe, M.; Sugita-Konishi, Y.; Yanai, T.; Nohmi, T.; Nishikawa, A.; Umemura, T. Site-specific in vivo mutagenicity int he kidney of gpt delta ratsgiven carcinogenic dose of ochratoxin A. Toxicol. Sci. 2011, 122, 406–414. [Google Scholar] [CrossRef]
- Hibi, D.; Kajima, A.; Kuroda, K.; Suzuki, Y.; Jin, M.; Nakajima, M.; Sugita-Konishi, Y.; Yanai, T.; Nohmi, T.; Nishikawa, A.; et al. Molecular mechanisms underlaying ochratoxin A-induced genotoxicity: Global gene expression analysis suggests induction of DNA double-strand breaks and cell cycle progression. J. Toxicol. Sci. 2013, 38, 57–69. [Google Scholar] [CrossRef]
- Mantle, P.G.; Faucet-Marquis, V.; Manderville, R.A.; Squillaci, B.; Pfohl-Leszkowicz, A. Structures of covalent adducts between DNA and ochratoxin A: A new factor in debate about genotoxicity and human risk assessment. Chem. Res. Toxicol. 2010, 23, 89–98. [Google Scholar] [CrossRef]
- Hadjeba-Medjdoub, K. Risk Assesment of Multiexposure to Mycotoxins and Detoxification Strategies Using Yeast by Product Enriched in Glutathione and Selenomethione. Master’s Thesis, University of Toulouse, Toulouse, France, June 2012. [Google Scholar]
- IARC. Some traditional herbal medicines, some mycotoxins, naphthalene and styrene. IARC Monogr. Eval. Carcinog. Risks Hum. 2002, 82, 301–366.
- Hadjeba-Medjdoub, K.; Faucet-Marquis, V.; Tozlovanu, M.; Peraica, M.; Manderville, R.A.; Pfohl-Leszkowicz, A. Synergistic effect of three nephrotoxic and carcinogenic mycotoxins (citrinin, fumonisin, ochratoxin A) on human kidney cells viability and genotoxicity. In Power of Fungi and Mycotoxins in Health and Disease, Proceedings of Power of Fungi and Mycotoxins in Health and Disease, Primošten, Croatia, 19–22 October 2011; Antolović, R., Miličević, T., Eds.; Croatian Mycrobiological Society: Zagreb, Croatia, 2011; p. 57. [Google Scholar]
- Hetherington, A.C.; Raistrick, H. Studies on biochemistry of microorganisms. Par XIV. On the production and chemical constitution of a new yellow coloring matter, citrinin produced from glucose by Penicillium citrininum Thom. Phil. Trans. Roy. Soc. Lond. B Biol. Sci. 1931, 220, 269–296. [Google Scholar] [CrossRef]
- Raistrick, H.; Smith, G. Antibacterial substances from moulds. Chem. Ind. London 1941, 6, 828–830. [Google Scholar]
- Bouslimi, A.; Ouannes, Z.; Golli, E.E.; Bouaziz, C.; Hassen, W.; Bacha, H. Cytotoxicity and oxidative damage in kidney cells exposed to the mycotoxins ochratoxin A and citrinin: Individual and combined effects. Toxicol. Mechan. Meth. 2008, 18, 341–349. [Google Scholar] [CrossRef]
- Chagas, G.M.; Campello, A.P.; Klüppel, M.L. Mechanism of citrinin-induced dysfunction of mitochondria. I. Effects on respiration enzyme activities and membrane potential of renal cortical mitochondria. J. Appl. Toxicol. 1992, 12, 123–129. [Google Scholar] [CrossRef]
- Chagas, G.M.; Olivera, M.A.; Campello, A.P.; Klüppel, M.L. Mechanism of citrinin-induced dysfunction of mitochondria. IV. Effect on Ca2+ transport. Cell Biochem. Funct. 1995, 13, 53–59. [Google Scholar]
- Klarić, M.S.; Želježić, D.; Rumora, L.; Peraica, M.; Pepeljnjak, S.; Domijan, A.M. A potential role of calcium in apoptosis and aberrant chromatin forms in porcine kidney PK15 cells induced by individual and combined ochratoxin A and citrinin. Arch. Toxicol. 2012, 86, 97–107. [Google Scholar] [CrossRef]
- Yu, F.Y.; Liao, Y.C.; Chang, C.H.; Liu, B.H. Citrinin induces apoptosis in HL-60 cells via activation of the mitochondrial pathway. Toxicol. Lett. 2006, 161, 143–151. [Google Scholar] [CrossRef]
- Jeswal, P. Citrinin-induced chromosomal abnormalities in the bone-marrow cells of Mus musculus. Cytobiosis 1996, 86, 29–33. [Google Scholar]
- Martin, W.; Lorkowsky, G.; Creppy, E.E.; Dirheimer, G.; Roschenthaler, R. Action of citrinin on bacteria chromosomal and plasmid DNA in vivo and in vitro. Appl. Environ. Microbiol. 1986, 52, 1273–1279. [Google Scholar]
- Knasmüller, S.; Cavin, C.; Chakraborty, A.; Darroudi, F.; Majer, B.J.; Huber, W.W.; Erlich, V.A. Structurally related mycotoxin ochratoxin A, ochratoxin B, and citrinin differ in their genotoxic activities and in their mode of action in human-derived liver (HepG2) cells: Implications for risk assessment. Nutr. Cancer 2004, 50, 190–197. [Google Scholar]
- Dömnez-Altuntas, H.; Dumlupinar, G.; Imamoglu, N.; Hamurcu, Z.; Liman, B.C. Effects of the mycotoxin citrinin on micronucleus formation in a cytokinesis-block genotoxicity assay in cultured human lymphocytes. J. Appl. Toxicol. 2007, 27, 337–341. [Google Scholar]
- Pfeiffer, E.; Gross, K.; Metzler, M. Aneuploidogenic and clastogenic potential of mycotoxins citrinin and patulin. Carcinogenesis 1998, 19, 1313–1318. [Google Scholar]
- Thust, R.; Kneist, S. Activity of citrinin mebabolized by rat and human microsome fraction in clastogenicity and SCE assay on Chinese hamster V78-E cells. Mutat. Res. 1979, 67, 321–330. [Google Scholar] [CrossRef]
- Liu, B.H.; Yu, F.Y.; Wu, T.S.; Li, S.Y.; Su, M.C.; Wang, M.C.; Shih, S.M. Evaluation of genotoxic risk on oxidative DNA damage in mammalian cells exposed to mycotoxins, patulin and citrinin. Toxicol. Appl. Pharmacol. 2003, 191, 255–263. [Google Scholar] [CrossRef]
- Flajs, D.; Želježić, D.; Mladinić, M.; Peraica, M. Effects of citrinin treatment on oxidative stress in rat kidney. Toxicol. Lett. 2010, 196 (Suppl. 17), S239. [Google Scholar]
- Pfohl-Leszkowicz, A.; Molinié, A.; Tozlovanu, M.; Manderville, R.A. Combined toxic effects of ochratoxin A and citrinin, in vivo and in vitro. In Food Contaminants: Mycotoxins and Food Allergens; Siantar, D.P., Trucksess, MW., Scott, P.M., Herman, EM., Eds.; Oxford University Press: New York, NY, USA, 2008; pp. 56–79. [Google Scholar]
- Manderville, R.A.; Pfohl-Leszkowicz, A. (2008) Bioactivation and DNA Adduction as a Rationale for Ochratoxin A Carcinogenesis. World Mycotoxin J. 2008, 1, 357–367. [Google Scholar] [CrossRef]
- Würgler, F.E.; Friedrich, U.; Schlatter, J. Lack of mutagenicity of ochratoxin A and B, citrinin, patulin and cnestine in Salmonella typhimurium TA102. Mutat. Res. 1991, 261, 209–216. [Google Scholar] [CrossRef]
- IARC. Citrinin. In Some Naturally Occurring and Synthetic Food Components, Coumarins and Ultraviolet Radiation. IARC Monogr. Eval. Carcinog. Risks Hum. 1998, 40, 67.
- Föllman, W.; Lebrun, S.; Kullik, B.; Koch, M.; Römer, C.; Golka, K. Cytotoxicity of ochratoxin A and citrinin in different cell types in vitro. Mycotox. Res. 2000, 16, 123–126. [Google Scholar] [CrossRef]
- Heussner, A.H.; O’Brien, E.; Haehnlein, J.; Biester, M.A.; Dietrich, D.R. Comparison of interactive cytotoxic effects of selected mycotoxins on renal cells. Toxicol. Sci. 2004, 78 (S-1), 89. [Google Scholar]
- Knecht, A.; Schwerdt, G.; Gekle, M.; Humpf, H.-U. Combinatory effects of citrinin and ochratoxin A in immortalized human proximal tubule cells. Mycotoxin Res. 2005, 21, 176–181. [Google Scholar] [CrossRef]
- Flajs, D.; Mladinić, M; Želježić, D.; Peraica, M. Citrinin potentiates ochratoxin A toxicity. Toxicol. Lett. 2011, 205 (Suppl. 28), S220–S221. [Google Scholar]
- Bouslimi, A.; Bouaziz, C.; Ayed-Boussema, I.; Hassen, W.; Bacha, H. Individual and cobined effects of ochratoxin A and citrinin on viability and DNA fragmentation in cultured Vero cells and on chromosome abberations in mice bone marrow cells. Toxicology 2008, 251, 1–7. [Google Scholar] [CrossRef]
- Vesela, D.; Vesely, D.; Jelinek, R. Toxic effect of ochratoxin A and citrinin, alone and in combination, on chicken embryos. Appl. Environ. Microbiol. 1983, 45, 91–93. [Google Scholar]
- Mayura, K.; Parker, R.; Berndt, W.O.; Phillips, T.D. Effect of simultaneous prenatal exposure to ochratoxin A and citrinin in the rat. J. Toxicol. Environ. Health 1984, 13, 553–561. [Google Scholar] [CrossRef]
- Glahn, R.P.; Wiederman, R.F.; Evangelisti, J.W. Effects of ochratoxin A alone and in combination with citrinin on kidney function of single comb white leghorn pullets. Poult. Sci. 1988, 67, 1034–1042. [Google Scholar] [CrossRef]
- Manning, R.O.; Brown, T.P.; Wyatt, R.D.; Fletcher, O.J. The individual and combined effects of citrinin and ochratoxin A in broiler chicks. Avian Dis. 1985, 29, 986–997. [Google Scholar] [CrossRef]
- Kumar, M.; Dwivedi, P.; Sharma, A.K.; Singh, N.S., Patil. Ochratoxin A and citrinin nephrotoxicity in New Zealand White rabbits: An ultrastructural assessment. Mycopathologia 2007, 163, 21–30. [Google Scholar] [CrossRef]
- Kitchen, D.N.; Carlton, W.W.; Tuite, J. Ochratoxin A and citrinin induced nephrosis in beagle dogsl. II. Pathology. Vet. Pathol. 1997, 14, 261–272. [Google Scholar] [CrossRef]
- Ciegler, A.; Detroy, R.W.; Lillehoj, L.B. Patulin, penicillic acid, and other carcinogenic lactones. Microb. Toxins 1971, 6, 409–434. [Google Scholar]
- Dickens, F.; Jones, H.E.H. Carcinogenic activity of a series of reactive lactones and related substances. Br. J. Cancer. 1961, 15, 85–100. [Google Scholar] [CrossRef]
- Umeda, M.; Yamamoto, T.; Saito, M. DNA-strand breakage of HeLa cells induced by several mycotoxins. Jpn. J. Exp. Med. 1972, 42, 527–539. [Google Scholar]
- IARC. Penicillic acid. In Some Naturally Occurring Substances. IARC Monogr. Eval. Carcinog. Risks Hum. 1998, 10, 21.
- Stoev, S.D.; Denev, S.; Dutton, M.; Nkosi, B. Cytotoxic effect of some mycotoxins and their combinations on human peripheral blood mononuclear cells as measured by MTT assay. Open Toxinol. J. 2009, 2, 1–8. [Google Scholar] [CrossRef]
- Huff, W.E.; Hamilton, P.B.; Ciegler, A. Evaluation of penicillic acid for toxicity in broiler chickens. Poult. Sci. 1980, 59, 1203–1207. [Google Scholar]
- Chan, P.K.; Hayes, A.W. Effect of penicillic acid on biliary excretion of indocyanin green in the mouse and rat. J. Toxicol. Environ. Heal. 1981, 7, 169–179. [Google Scholar] [CrossRef]
- Kubena, L.F.; Phillips, T.D.; Witzel, D.A.; Heidelbaugh, N.D. Toxicity of ochratoxin A and penicillic acid to chicks. Bull. Environ. Contam. Toxicol. 1984, 32, 711–716. [Google Scholar] [CrossRef]
- Shepherd, E.C.; Phillips, T.D.; Joiner, G.N.; Kubena, L.F.; Heidelbaugh, N.D. Ochratoxin A and penicillic acid interaction in mice. J. Environ. Sci. Health 1981, B16, 557–573. [Google Scholar]
- Parker, R.; Phillips, T.; Kubena, L.; Russell, L.H.; Heidelbaugh, N.D. Inhibition of pancreatic carboxypeptidase A: A possible mechanism of interaction between penicillic acid and ochratoxin A. J. Environ. Sci. Health 1982, B17, 77–91. [Google Scholar]
- Stoev, S.D.; Denev, S.; Dutton, M.; Njobeh, P.; Mosonik, J.; Steenkamp, P.; Petkov, I. Complex etiology and pathology of mycotoxic nephropathy in South African pigs. Mycotox. Res. 2010, 26, 31–46. [Google Scholar]
- Stoev, S.D.; Daskalov, H.; Radić, B.; Domijan, A.; Peraica, M. Spontaneous mycotoxin nephropathy in Bulgarian chickens with unclarified mycotoxin aetiology. Vet. Res. 2002, 33, 83–94. [Google Scholar] [CrossRef]
- Stoev, S.D.; Vitanov, S.; Anguelov, G.; Petkova-Bocharova, T.; Creppy, E.E. Experimental mycotoxic nephropathy in pigs provoked by a mouldy diet containing ochratoxin A and penicillic acid. Vet. Res. Commun. 2001, 25, 205–223. [Google Scholar]
- Stoev, S.D.; Stefanov, M.; Denev, S.; Radić, B.; Domijan, A.; Peraica, M. Experimental mycotoxicosis in chickens induced by ochratoxin A and penicillic acid and intervention by natural plant extracts. Vet. Res. Commun. 2004, 28, 727–746. [Google Scholar]
- Micco, C.; Miraglia, M.; Onori, R.; Libanori, A.; Brera, C.; Mantovani, A.; Macri, C. Effect of combined exposure to ochratoxin A and penicillic acid on residues and toxicity in broilers. La Ravista della Societa Italiana di Scienza dell’Allimentazione 1991, 20, 101–108. [Google Scholar]
- Müller, S.; Dekant, W.; Mally, A. Fumonisin B1 and the kidney: Modes of action for renal tumor formation by fumonisin B1 in rodents. Food Chem. Toxicol. 2012, 50, 3833–3846. [Google Scholar]
- National Toxicology Program (NTP), Toxicology and Carcinogenesis Studies of Fumonisin B1 (CAS No.116355-83-0) in F344/N Rats and B6C3F1 Mice (Feed Studies); National Toxicology Program Technical Reports; National Institute of Environmental Health Sciences: Research Triangle Park, NC, USA, 2001; pp. 1–352.
- Ueno, Y.; Iijima, K.; Wang, S.-D.; Sugiura, Y.; Sekijima, M.; Tanaka, T.; Chen, C.; Yu, S.Z. Fumonisins as a possible contributory risk factor for primary liver cancer: A 3-year study of corn harvested in Haimen, China, by HPLC and ELISA. Food Chem. Toxicol. 1997, 35, 1143–1150. [Google Scholar] [CrossRef]
- Knasmüeller, S.; Bresgen, N.; Kassie, F.; Mersch-Sundermann, V.; Gelderblom, W.; Zoehrer, E.; Eckl, P.M. Genotoxic effects of three Fusarium mycotoxins, fumonisin B1, moniliformin and vomitoxin in bacteria and in primary culturesof rat hepatocytes. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 1997, 391, 39–48. [Google Scholar] [CrossRef]
- Gelderblom, W.C.; Semple, E.; Marasas, W.F.; Farber, E. The cancer-initiating potential of the fumonisin B mycotoxins. Carcinogenesis 1992, 13, 433–437. [Google Scholar]
- Norred, W.P.; Plattner, R.D.; Vesonder, R.F.; Bacon, C.W.; Voss, K.A. Effects of selected secondary metabolites of Fusarium moniliforme on unscheduled synthesis of DNA by rat primary hepatocytes. Food Chem. Toxicol. 1992, 30, 233–237. [Google Scholar] [CrossRef]
- Domijan, A.M.; Peraica, M.; Vrdoljak, A.L.; Radić, B.; Žlender, V.; Fuchs, R. The involvement of oxidative stress in ochratoxin A and fumonisin B1 toxicity in rats. Mol. Nutr. Food Res. 2007, 51, 1147–1151. [Google Scholar] [CrossRef]
- Domijan, A.M.; Želježić, D.; Kopjar, N.; Peraica, M. Standard and Fpg-modified comet assay in kidney cells of ochratoxin A- and fumonisin B1-treated rats. Toxicology 2006, 222, 53–59. [Google Scholar] [CrossRef]
- Pinelli, E.; Poux, N.; Garren, L.; Castegnaro, M.; Pipy, B.; Miller, J.D.; Pfohl-Leszkowicz, A. Activation of Mitogen-activated protein kinase by fumonisin B1 stimulates the arachidonic acid cascade and cAMP production. Carcinogenesis 1999, 20, 1683–1688. [Google Scholar]
- Poux, N.; Pinelli, E.; Castegnaro, M.; Miller, D.J.; Pfohl-Leszkowicz, A. Effects of fumonisin B1 on cell signal transduction pathways: Main role of MAPKs. In Mycotoxins and Phycotoxins in Perspective at the Turn of the Millenium, Proceedings of Xth International IUPAC Symposium on Mycotoxins and Phycotoxins, Guarujá, Brasil, 21–25 May 2000; Koe, W.J., Samson, R.A., Gilbert, J., Sabino, M., Eds.; International Union of Pure and Applied Chemistry (IUPAC): Research Triangle Park, NC, USA, 2001; pp. 251–257. [Google Scholar]
- Creppy, E.E.; Chirappa, P.; Baudrimont, I.; Borracci, P.; Moukha, S.; Carratu, M.R. Synergistic effects of fumonisn B1 and ochratoxin A: Are in vitro cytotoxicity data predictive of in vivo acute toxicity? Toxicology 2004, 201, 115–123. [Google Scholar]
- Klarić, M.S.; Pepeljnjak, S.; Domijan, A.M.; Petrik, J. Lipid peroxidation and glutathione levels in porcine kidney PK15 cells after individual and combined treatment with fumonisin B1, beauvericin and ochratoxin A. Basic Clin. Pharmacol. Toxicol. 2007, 100, 157–164. [Google Scholar] [CrossRef]
- Klarić, M.S.; Rumora, L.; Ljubanović, D.; Pepeljnjak, S. Cytotoxicity and apoptosis induced by fumonisin B1, beauvericin and ochratoxin A in porcine kidney PK15 cells: Effects of individual and combined treatment. Arch. Toxicol. 2008, 82, 247–255. [Google Scholar]
- Kubena, L.F.; Edrington, T.S.; Harvey, R.B.; Phillips, T.D.; Sarr, A.B.; Rottinghaus, G.E. Individual and combined effects of fumonisin B1 present in Fusarium moniliforme culture material and diacetoxyscirpenol or ochratoxin A in turkey poults. Poult. Sci. 1997, 76, 256–264. [Google Scholar]
- Sivakumar, G.; Dwivedi, P.; Sharma, A.K.; Kumar, M.; Nimalesan, S. Fumonisin B1 and ochratoxin A induced biochemical changes in young male New Zealand White rabbits. Indian J. Vet. Pathol. 2009, 33, 30–34. [Google Scholar]
- Klarić, M.S.; Pepeljnjak, S.; Rozgaj, R. Genotoxicity of fumonisin B1, beauvericin and ochratoxin A in porcine kidney PK15 cells: Effects of individual and combined treatment. Croat. Chem. Acta 2008, 81, 139–146. [Google Scholar]
- Stoev, S.D.; Gundasheva, D.; Zarkov, I.; Mircheva, T.; Zapryanova, D.; Denev, S.; Mitev, Y.; Daskalov, H.; Dutton, M.; Mwanza, M.; et al. Experimental mycotoxic nephropathy in pigs provoked by a mouldy diet containing ochratoxin A and fumonisin B1. Exp. Toxicol. Pathol. 2012, 64, 733–741. [Google Scholar] [CrossRef]
- Bedard, L.L.; Massey, T.E. Aflatoxin B1-induced DNA damage and its repair. Cancer Lett. 2006, 241, 174–183. [Google Scholar]
- Eaton, D.L.; Gallagher, E.P. Mechanisms of aflatoxin carcinogenesis. Annu. Rev. Pharmacol. Toxicol. 1994, 34, 135–172. [Google Scholar] [CrossRef]
- Wu, H.C.; Santella, R. The role of aflatoxins in hepatocellular carcinoma. Hepat. Mon. 2012, 12, e7238. [Google Scholar]
- IARC. Aflatoxins. In Some Traditional Herbal Medicines, Some Mycotoxins, Naphthalene and Styrene. IARC Monogr. Eval. Carcinog. Risks Hum. 2002, 82, 1–556.
- Galtier, P.; Meissonnier, G.; Laffitte, J.; Oswald, I.P.; Loiseau, N. Molecular interactions between mycotoxins and liver enzymes involved in drug metabolism in rodents and farm animals. Krmiva 2008, 50, 205–213. [Google Scholar]
- Yunus, A.W.; Razzazi-Fazeli, E.; Böhm, J. Aflatoxin B1 in affecting broiler’s performance, immunity, and gastrointestinal tract: A review of history and contemporary issues. Toxins 2011, 3, 566–590. [Google Scholar] [CrossRef]
- Golli-Bennour, E.E.; Kouidhi, B.; Bouslimi, A.; Abid-Essefi, S.; Hassen, W.; Bacha, H. Cytotoxicity and genotoxicity induced by aflatoxin B1, ochratoxin A, and their combination in cultured Vero cells. J. Biochem. Mol. Toxicol. 2010, 24, 42–50. [Google Scholar] [CrossRef]
- Corcuera, L.A.; Arbillaga, L.; Vettorazzi, A.; Azqueta, A.; de Cerain, L. Ochratoxin A reduces aflatoxin B1 induced DNA damage detected by the comet assay in Hep G2 cells. Food Chem. Toxicol. 2011, 49, 2883–2889. [Google Scholar] [CrossRef]
- Huff, W.E.; Doerr, J.A. Synergism between aflatoxin and ochratoxin A in broiler chickens. Poult. Sci. 1981, 60, 550–555. [Google Scholar] [CrossRef]
- Sakhare, P.S.; Harne, S.D.; Kalorey, D.R.; Warke, S.R.; Bhandarkar, A.G.; Kurkure, N.V. Effect of Toxiroak® polyherbal feed supplement during induced aflatoxicosis, ochratoxicosis and combined mycotoxicoses in broilers. Vet. Arh. 2007, 2, 129–146. [Google Scholar]
- Corcuera, L.A.; Vettorazzi, A.; Arbillaga, L.; González-Peńas, E.; López de Cerain, A. An approach to the toxicity and toxicokinetics of aflatoxin B1 and ochratoxin A after simultaneous oral administration to fasted F344 rats. Food Chem. Toxicol. 2012, 50, 3440–3446. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Klarić, M.Š.; Rašić, D.; Peraica, M. Deleterious Effects of Mycotoxin Combinations Involving Ochratoxin A. Toxins 2013, 5, 1965-1987. https://doi.org/10.3390/toxins5111965
Klarić MŠ, Rašić D, Peraica M. Deleterious Effects of Mycotoxin Combinations Involving Ochratoxin A. Toxins. 2013; 5(11):1965-1987. https://doi.org/10.3390/toxins5111965
Chicago/Turabian StyleKlarić, Maja Šegvić, Dubravka Rašić, and Maja Peraica. 2013. "Deleterious Effects of Mycotoxin Combinations Involving Ochratoxin A" Toxins 5, no. 11: 1965-1987. https://doi.org/10.3390/toxins5111965






