Orally Delivered Scorpion Antimicrobial Peptides Exhibit Activity against Pea Aphid (Acyrthosiphon pisum) and Its Bacterial Symbionts
Abstract
:1. Introduction
2. Results
2.1. Effect of AMP Treatments on Aphid Survival
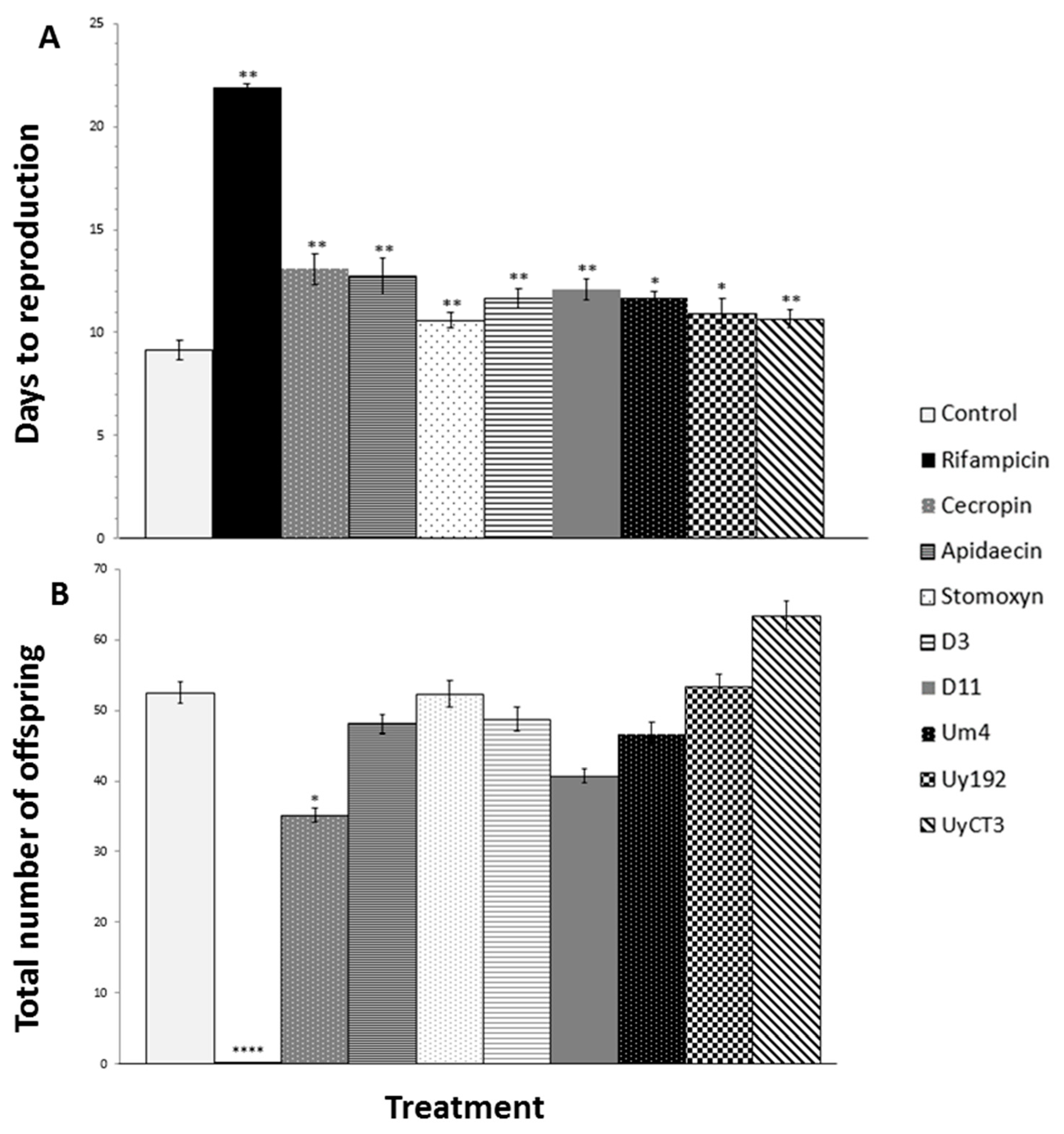
2.2. Effect of AMP Treatments on Aphid Reproduction
2.3. Effect of AMPs on Bacterial Growth In Vitro
2.4. qPCR-Based Quantification of Bacterial Symbionts in Treated Aphids
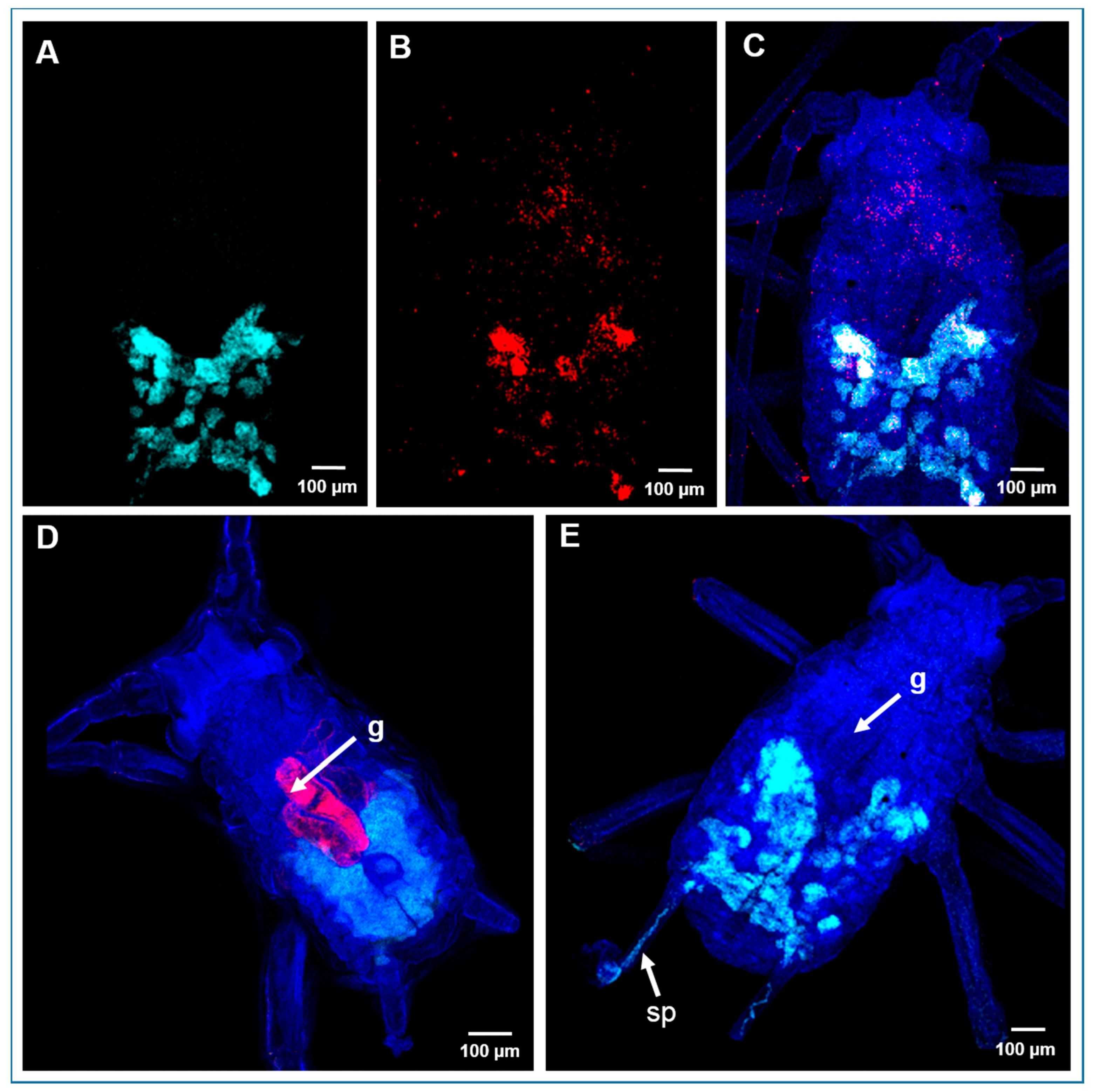
2.5. Localization of Bacterial Symbionts in A. pisum by Fluorescence In Situ Hybridization
3. Discussion
4. Materials and Methods
4.1. Antimicrobial Peptides
4.2. Aphids and the Detection of Bacterial Symbionts
4.3. Aphid Feeding with AMPs
4.4. In Vitro Activity of Scorpion and Insect AMPs against Serratia Symbiotica CWBI-2.3
4.5. Relative Quantification of Bacterial Symbionts In Vivo
4.6. Localization of Bacterial Symbionts In Vivo by FISH
4.7. Data Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Van Emden, H.F.; Harrington, R. Aphids as Crop Pests; CABI: Wallingford, UK, 2007. [Google Scholar]
- Moran, N.A. Old and new symbiotic partners in lachnine aphids. Environ. Microbiol. 2017, 19, 7. [Google Scholar] [CrossRef] [PubMed]
- Akman Gündüz, E.; Douglas, A.E. Symbiotic bacteria enable insect to use a nutritionally inadequate diet. Proc. Biol. Sci. 2009, 276, 987–991. [Google Scholar] [CrossRef] [PubMed]
- Skaljac, M. Bacterial symbionts of aphids (Hemiptera: Aphididae). In Biology and Ecology of Aphids; CRC Press: Boca Raton, FL, USA, 2016; pp. 100–125. [Google Scholar]
- Oliver, K.M.; Degnan, P.H.; Burke, G.R.; Moran, N.A. Facultative symbionts in aphids and the horizontal transfer of ecologically important traits. Annu. Rev. Entomol. 2010, 55, 247–266. [Google Scholar] [CrossRef] [PubMed]
- Oliver, K.M.; Smith, A.H.; Russell, J.A. Defensive symbiosis in the real world—Advancing ecological studies of heritable, protective bacteria in aphids and beyond. Funct. Ecol. 2014, 28, 341–355. [Google Scholar] [CrossRef]
- Russell, J.A.; Moran, N.A. Costs and benefits of symbiont infection in aphids: Variation among symbionts and across temperatures. Proc. Biol. Sci. 2006, 273, 603–610. [Google Scholar] [CrossRef] [PubMed]
- The International Aphid Genomics Consortium. Genome sequence of the pea aphid Acyrthosiphon pisum. PLoS Biol. 2010, 8, e1000313. [Google Scholar]
- Altincicek, B.; Gross, J.; Vilcinskas, A. Wounding—Mediated gene expression and accelerated viviparous reproduction of the pea aphid Acyrthosiphon pisum. Insect Mol. Biol. 2008, 17, 711–716. [Google Scholar] [CrossRef] [PubMed]
- Gerardo, N.M.; Altincicek, B.; Anselme, C.; Atamian, H.; Barribeau, S.M.; de Vos, M.; Duncan, E.J.; Evans, J.D.; Gabaldon, T.; Ghanim, M.; et al. Immunity and other defenses in pea aphids, Acyrthosiphon pisum. Genome Biol. 2010, 11, R21. [Google Scholar] [CrossRef] [PubMed]
- Laughton, A.M.; Garcia, J.R.; Gerardo, N.M. Condition-dependent alteration of cellular immunity by secondary symbionts in the pea aphid, Acyrthosiphon pisum. J. Insect Physiol. 2016, 86, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Shigenobu, S.; Stern, D.L. Aphids evolved novel secreted proteins for symbiosis with bacterial endosymbiont. Proc. Biol. Sci. 2013, 280, 20121952. [Google Scholar] [CrossRef] [PubMed]
- Laughton, A.M.; Garcia, J.R.; Altincicek, B.; Strand, M.R.; Gerardo, N.M. Characterisation of immune responses in the pea aphid, acyrthosiphon pisum. J. Insect Physiol. 2011, 57, 830–839. [Google Scholar] [CrossRef] [PubMed]
- Vilcinskas, A. Evolutionary plasticity of insect immunity. J. Insect Physiol. 2013, 59, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Koga, R.; Tsuchida, T.; Fukatsu, T. Changing partners in an obligate symbiosis: A facultative endosymbiont can compensate for loss of the essential endosymbiont Buchnera in an aphid. Proc. Biol. Sci. 2003, 270, 2543–2550. [Google Scholar] [CrossRef] [PubMed]
- Koga, R.; Tsuchida, T.; Sakurai, M.; Fukatsu, T. Selective elimination of aphid endosymbionts: Effects of antibiotic dose and host genotype, and fitness consequences. FEMS Microbiol. Ecol. 2007, 60, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Moran, N.A.; Yun, Y. Experimental replacement of an obligate insect symbiont. Proc. Natl. Acad. Sci. USA 2015, 112, 2093–2096. [Google Scholar] [CrossRef] [PubMed]
- Cristofoletti, P.T.; Ribeiro, A.F.; Deraison, C.; Rahbe, Y.; Terra, W.R. Midgut adaptation and digestive enzyme distribution in a phloem feeding insect, the pea aphid Acyrthosiphon pisum. J. Insect Physiol. 2003, 49, 11–24. [Google Scholar] [CrossRef]
- Ortiz, E.; Gurrola, G.B.; Schwartz, E.F.; Possani, L.D. Scorpion venom components as potential candidates for drug development. Toxicon 2015, 93, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Almaaytah, A.; Albalas, Q. Scorpion venom peptides with no disulfide bridges: A review. Peptides 2014, 51, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Luna-Ramírez, K.; Sani, M.-A.; Silva-Sanchez, J.; Jiménez-Vargas, J.M.; Reyna-Flores, F.; Winkel, K.D.; Wright, C.E.; Possani, L.D.; Separovic, F. Membrane interactions and biological activity of antimicrobial peptides from australian scorpion. Biochim. Biophys. Acta 2014, 1838, 2140–2148. [Google Scholar] [CrossRef] [PubMed]
- Harrison, P.L.; Abdel-Rahman, M.A.; Miller, K.; Strong, P.N. Antimicrobial peptides from scorpion venoms. Toxicon 2014, 88, 115–137. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.C.; Corzo, G.; Hahin, R. Scorpion venom peptides without disulfide bridges. IUBMB Life 2005, 57, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Luna-Ramírez, K.; Quintero-Hernández, V.; Vargas-Jaimes, L.; Batista, C.V.F.; Winkel, K.D.; Possani, L.D. Characterization of the venom from the australian scorpion Urodacus yaschenkoi: Molecular mass analysis of components, cdna sequences and peptides with antimicrobial activity. Toxicon 2013, 63, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Luna-Ramírez, K.; Bartok, A.; Restano-Cassulini, R.; Quintero-Hernández, V.; Coronas, F.I.V.; Christensen, J.; Wright, C.E.; Panyi, G.; Possani, L.D. Structure, molecular modeling, and function of the novel potassium channel blocker urotoxin isolated from the venom of the Australian scorpion Urodacus yaschenkoi. Mol. Pharmacol. 2014, 86, 28–41. [Google Scholar] [CrossRef] [PubMed]
- Luna-Ramirez, K.; Quintero-Hernandez, V.; Juarez-Gonzalez, V.R.; Possani, L.D. Whole transcriptome of the venom gland from Urodacus yaschenkoi scorpion. PLoS ONE 2015, 10, e0127883. [Google Scholar] [CrossRef] [PubMed]
- Sunagar, K.; Undheim, E.A.B.; Chan, A.H.C.; Koludarov, I.; Muñoz-Gómez, S.A.; Antunes, A.; Fry, B.G. Evolution stings: The origin and diversification of scorpion toxin peptide scaffolds. Toxins 2013, 5, 2456–2487. [Google Scholar] [CrossRef] [PubMed]
- Luna-Ramirez, K.; Tonk, M.; Rahnamaeian, M.; Vilcinskas, A. Bioactivity of natural and engineered antimicrobial peptides from venom of the scorpions Urodacus yaschenkoi and U. manicatus. Toxins 2017, 9, 22. [Google Scholar] [CrossRef] [PubMed]
- Sabri, A.; Leroy, P.; Haubruge, E.; Hance, T.; Frere, I.; Destain, J.; Thonart, P. Isolation, pure culture and characterization of Serratia symbiotica sp. Nov., the r-type of secondary endosymbiont of the black bean aphid aphis fabae. Int. J. Syst. Evol. Microbiol. 2011, 61, 2081–2088. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, T.L. The elimination of intracellular microorganisms from insects: An analysis of antibiotic-treatment in the pea aphid (Acyrthosiphon pisum). Comp. Biochem. Physiol. Part A 1998, 119, 871–881. [Google Scholar] [CrossRef]
- Keymanesh, K.; Soltani, S.; Sardari, S. Application of antimicrobial peptides in agriculture and food industry. World J. Microbiol. Biotechnol. 2009, 25, 933–944. [Google Scholar] [CrossRef]
- Will, T.; Vilcinskas, A. Aphid-proof plants: Biotechnology-based approaches for aphid control. In Yellow Biotechnology II: Insect Biotechnology in Plant Protection and Industry; Vilcinskas, A., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 179–203. [Google Scholar]
- Langen, G.; Imani, J.; Altincicek, B.; Kieseritzky, G.; Kogel, K.H.; Vilcinskas, A. Transgenic expression of gallerimycin, a novel antifungal insect defensin from the greater wax moth Galleria mellonella, confers resistance to pathogenic fungi in tobacco. Biol. Chem. 2006, 387, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Rahnamaeian, M.; Langen, G.; Imani, J.; Khalifa, W.; Altincicek, B.; von Wettstein, D.; Kogel, K.H.; Vilcinskas, A. Insect peptide metchnikowin confers on barley a selective capacity for resistance to fungal ascomycetes pathogens. J. Exp. Bot. 2009, 60, 4105–4114. [Google Scholar] [CrossRef] [PubMed]
- Sani, M.A.; Separovic, F. How membrane-active peptides get into lipid membranes. Acc. Chem. Res. 2016, 49, 1130–1138. [Google Scholar] [CrossRef] [PubMed]
- Kuhn-Nentwig, L. Antimicrobial and cytolytic peptides of venomous arthropods. Cell. Mol. Life Sci. 2003, 60, 2651–2668. [Google Scholar] [CrossRef] [PubMed]
- Boulanger, N.; Munks, R.J.; Hamilton, J.V.; Vovelle, F.; Brun, R.; Lehane, M.J.; Bulet, P. Epithelial innate immunity. A novel antimicrobial peptide with antiparasitic activity in the blood-sucking insect Stomoxys calcitrans. J. Biol. Chem. 2002, 277, 49921–49926. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, J.A.; Reichhart, J.-M. Drosophila innate immunity: An evolutionary perspective. Nat. Immunol. 2002, 3, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Rees, J.A.; Moniatte, M.; Bulet, P. Novel antibacterial peptides isolated from a european bumblebee, Bombus pascuorum (hymenoptera, apoidea). Insect Biochem. Mol. Biol. 1997, 27, 413–422. [Google Scholar] [CrossRef]
- Steiner, H.; Hultmark, D.; Engstrom, A.; Bennich, H.; Boman, H.G. Sequence and specificity of two antibacterial proteins involved in insect immunity. Nature 1981, 292, 246–248. [Google Scholar] [CrossRef] [PubMed]
- Casteels, P.; Ampe, C.; Jacobs, F.; Vaeck, M.; Tempst, P. Apidaecins: Antibacterial peptides from honeybees. EMBO J. 1989, 8, 2387–2391. [Google Scholar] [PubMed]
- Mylonakis, E.; Podsiadlowski, L.; Muhammed, M.; Vilcinskas, A. Diversity, evolution and medical applications of insect antimicrobial peptides. Philos. Trans. R. Soc. B 2016, 371. [Google Scholar] [CrossRef] [PubMed]
- Rahnamaeian, M.; Cytryńska, M.; Zdybicka-Barabas, A.; Vilcinskas, A. The functional interaction between abaecin and pore-forming peptides indicates a general mechanism of antibacterial potentiation. Peptides 2016, 78, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Foray, V.; Grigorescu, A.S.; Sabri, A.; Haubruge, E.; Lognay, G.; Francis, F.; Fauconnier, M.L.; Hance, T.; Thonart, P. Whole-genome sequence of Serratia symbiotica strain CWBI-2.3t, a free-living symbiont of the black bean aphid Aphis fabae. Genome Announc. 2014, 2, e00767-14. [Google Scholar] [CrossRef] [PubMed]
- Ratzka, C.; Gross, R.; Feldhaar, H. Endosymbiont tolerance and control within insect hosts. Insects 2012, 3, 553–572. [Google Scholar] [CrossRef] [PubMed]
- Charles, H.; Balmand, S.; Lamelas, A.; Cottret, L.; Pérez-Brocal, V.; Burdin, B.; Latorre, A.; Febvay, G.; Colella, S.; Calevro, F.; et al. A genomic reappraisal of symbiotic function in the aphid Buchnera symbiosis: Reduced transporter sets and variable membrane organisations. PLoS ONE 2011, 6, e29096. [Google Scholar] [CrossRef] [PubMed]
- Koga, R.; Meng, X.Y.; Tsuchida, T.; Fukatsu, T. Cellular mechanism for selective vertical transmission of an obligate insect symbiont at the bacteriocyte-embryo interface. Proc. Natl. Acad. Sci. USA 2012, 109, E1230–E1237. [Google Scholar] [CrossRef] [PubMed]
- Lamelas, A.; Gosalbes, M.J.; Manzano-Marín, A.; Peretó, J.; Moya, A.; Latorre, A. Serratia symbiotica from the aphid Cinara cedri: A missing link from facultative to obligate insect endosymbiont. PLoS Genet. 2011, 7, e1002357. [Google Scholar] [CrossRef] [PubMed]
- Santos-Garcia, D.; Silva, F.J.; Moya, A.; Latorre, A. No exception to the rule: Candidatus Portiera aleyrodidarum cell wall revisited. FEMS Microbiol. Lett. 2014, 360, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Le-Feuvre, R.R.; Ramírez, C.C.; Olea, N.; Meza-Basso, L. Effect of the antimicrobial peptide indolicidin on the green peach aphid Myzus persicae (Sulzer). J. Appl. Entomol. 2007, 131, 71–75. [Google Scholar] [CrossRef]
- Selsted, M.E.; Novotny, M.J.; Morris, W.L.; Tang, Y.Q.; Smith, W.; Cullor, J.S. Indolicidin, a novel bactericidal tridecapeptide amide from neutrophils. J. Biol. Chem. 1992, 267, 4292–4295. [Google Scholar] [PubMed]
- Baumann, P.; Moran, N.A.; Baumann, L.C. Bacteriocyte-associated endosymbionts of insects. In The Prokaryotes: Prokaryotic Biology and Symbiotic Associations; Rosenberg, E., DeLong, E.F., Lory, S., Stackebrandt, E., Thompson, F., Eds.; Springer: Berlin/Heidelberg, Germany, 2000; pp. 465–496. [Google Scholar]
- King, G.F. Insecticidal polypeptides from spider venom. Ind. Bioprocess. 2007, 29, 4. [Google Scholar]
- King, G.F.; Hardy, M.C. Spider-venom peptides: Structure, pharmacology, and potential for control of insect pests. Annu. Rev. Entomol. 2013, 58, 475–496. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, E.; Possani, L.D. The unfulfilled promises of scorpion insectotoxins. J. Venom. Anim. Toxins Incl. Trop. Dis. 2015, 21, 16. [Google Scholar] [CrossRef] [PubMed]
- Windley, M.J.; Herzig, V.; Dziemborowicz, S.A.; Hardy, M.C.; King, G.F.; Nicholson, G.M. Spider-venom peptides as bioinsecticides. Toxins 2012, 4, 191–227. [Google Scholar] [CrossRef] [PubMed]
- Will, T.; Schmidtberg, H.; Skaljac, M.; Vilcinskas, A. Heat shock protein 83 plays pleiotropic roles in embryogenesis, longevity, and fecundity of the pea aphid Acyrthosiphon pisum. Dev. Genes Evol. 2017, 227, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, Y.; Hosokawa, T.; Serracin, M.; Tulgetske, G.M.; Miller, T.A.; Fukatsu, T. Bacterial symbionts of a devastating coffee plant pest, the stinkbug Antestiopsis thunbergii (Hemiptera: Pentatomidae). Appl. Environ. Microbiol. 2014, 80, 3769–3775. [Google Scholar] [CrossRef] [PubMed]
- Tsuchida, T.; Koga, R.; Shibao, H.; Matsumoto, T.; Fukatsu, T. Diversity and geographic distribution of secondary endosymbiotic bacteria in natural populations of the pea aphid, Acyrthosiphon pisum. Mol. Ecol. 2002, 11, 2123–2135. [Google Scholar] [CrossRef] [PubMed]
- Shahjahan, R.M.; Hughes, K.J.; Leopold, R.A.; DeVault, J.D. Lower incubation temperature increases yield of insect genomic DNA isolated by the ctab method. Biotechniques 1995, 19, 332–334. [Google Scholar] [PubMed]
- Fukatsu, T.; Nikoh, N. Two intracellular symbiotic bacteria from the mulberry psyllid anomoneura mori (insecta, homoptera). Appl. Environ. Microbiol. 1998, 64, 3599–3606. [Google Scholar] [PubMed]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Febvay, G.; Delobel, B.; Rahbé, Y. Influence of the amino acid balance on the improvement of an artificial diet for a biotype of Acyrthosiphon pisum (homoptera: Aphididae). Can. J. Zool. 1988, 66, 2449–2453. [Google Scholar] [CrossRef]
- Sadeghi, A.; Van Damme, E.J.; Smagghe, G. Evaluation of the susceptibility of the pea aphid, Acyrthosiphon pisum, to a selection of novel biorational insecticides using an artificial diet. J. Insect Sci. (Online) 2009, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Will, T.; Vilcinskas, A. The structural sheath protein of aphids is required for phloem feeding. Insect Biochem. Mol. Biol. 2015, 57, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Sapountzis, P.; Duport, G.; Balmand, S.; Gaget, K.; Jaubert-Possamai, S.; Febvay, G.; Charles, H.; Rahbe, Y.; Colella, S.; Calevro, F. New insight into the RNA interference response against cathepsin-l gene in the pea aphid, Acyrthosiphon pisum: Molting or gut phenotypes specifically induced by injection or feeding treatments. Insect Biochem. Mol. Biol. 2014, 51, 20–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
- Kliot, A.; Kontsedalov, S.; Lebedev, G.; Brumin, M.; Cathrin, P.B.; Marubayashi, J.M.; Skaljac, M.; Belausov, E.; Czosnek, H.; Ghanim, M. Fluorescence in situ hybridizations (fish) for the localization of viruses and endosymbiotic bacteria in plant and insect tissues. J. Vis. Exp. 2014. [Google Scholar] [CrossRef] [PubMed]
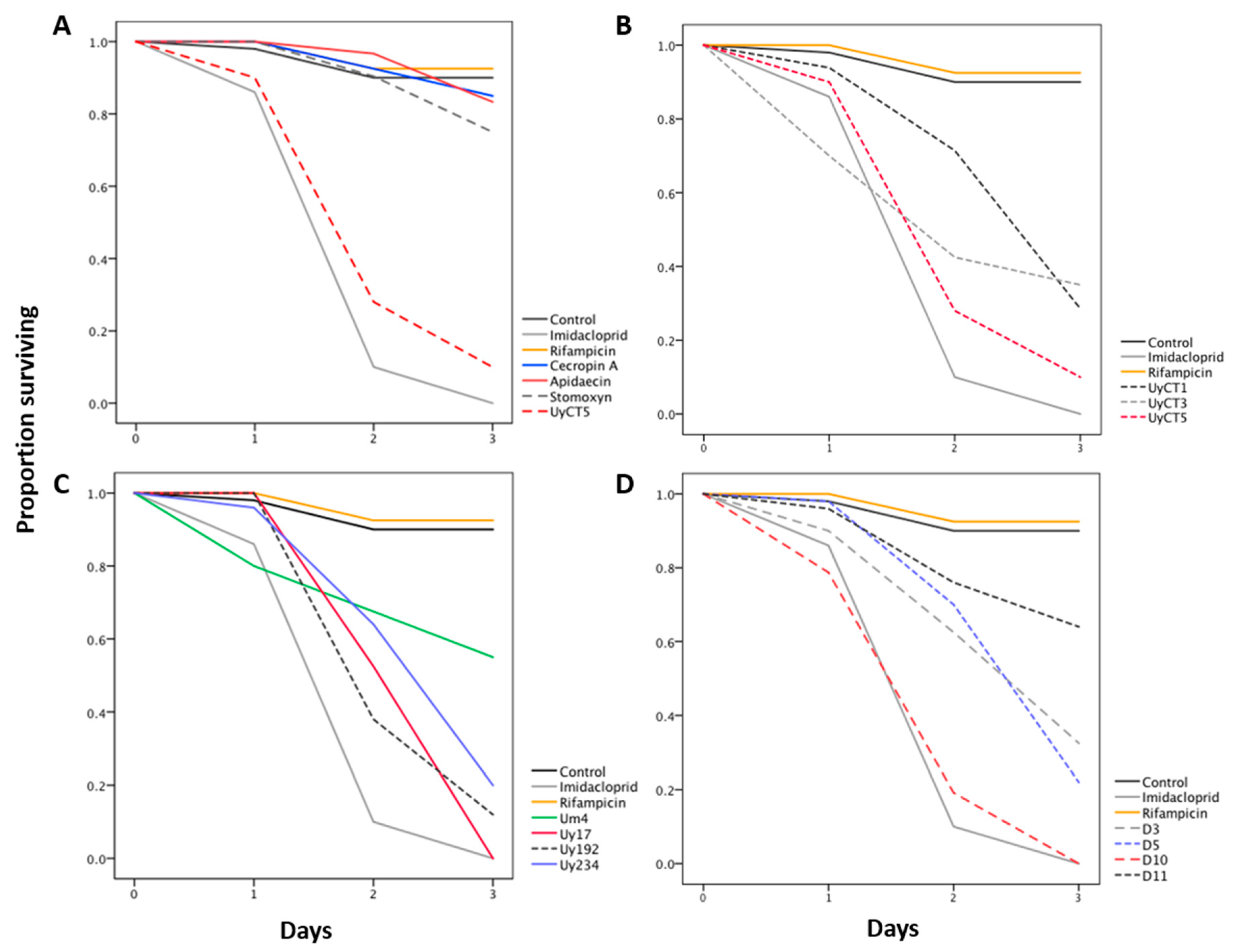

| Treatment | Concentration (µg/mL) | % Survival | Significance ˄ | |
|---|---|---|---|---|
| Insecticide | Imidacloprid | 5 | 0 | **** |
| Antibiotic | Rifampicin | 50 | 92.5 | ns |
| Scorpion AMPs | UyCT1 | 50 | 96 | ns |
| 250 | 76 | ns | ||
| 500 | 28.6 | **** | ||
| UyCT3 | 50 | 70 | * | |
| 250 | 52.5 | **** | ||
| 500 | 35 | **** | ||
| UyCT5 | 50 | 68 | ** | |
| 250 | 27.5 | **** | ||
| 500 | 10 | **** | ||
| Uy17 | 50 | 75 | ns | |
| 250 | 4 | **** | ||
| 500 | 0 | **** | ||
| Uy192 | 50 | 82 | ns | |
| 250 | 57.5 | **** | ||
| 500 | 12 | **** | ||
| Uy234 | 50 | 85 | ns | |
| 250 | 0 | **** | ||
| 500 | 20 | **** | ||
| Um4 | 50 | 85 | ns | |
| 250 | 90 | ns | ||
| 500 | 55 | **** | ||
| D3 | 50 | 72.5 | * | |
| 250 | 72.5 | * | ||
| 500 | 32.5 | **** | ||
| D5 | 50 | 83.7 | ns | |
| 250 | 42.5 | **** | ||
| 500 | 22 | **** | ||
| D10 | 50 | 86 | ns | |
| 250 | 0 | **** | ||
| 500 | 0 | **** | ||
| D11 | 50 | 86.7 | ns | |
| 250 | 57.1 | ** | ||
| 500 | 64 | ** | ||
| Insect AMPs | Apidaecin | 50 | 100 | ns |
| 250 | 90 | |||
| 500 | 83.3 | |||
| Cecropin A | 50 | 80 | ||
| 250 | 83.3 | |||
| 500 | 85 | |||
| Stomoxyn | 50 | 68 | ||
| 250 | 68 | |||
| 500 | 75 | |||
| Compounds | MIC (In Vitro) (µg/mL) | |
|---|---|---|
| Scorpion AMPs | UyCT1 | 125 |
| UyCT3 | 125 | |
| UyCT5 | 125 | |
| Uy17 | 250 | |
| Uy192 | 500 | |
| Uy234 | >500 | |
| Um4 | >500 | |
| D3 | 250 | |
| D5 | 500 | |
| D10 | >500 | |
| D11 | >500 | |
| Insect AMPs | Apidaecin | >500 |
| Cecropin A | >500 | |
| Stomoxyn | >500 | |
| Antibiotics | Rifampicin | 50 |
| Compounds | Sequence or Chemical Formula | |
|---|---|---|
| Scorpion AMPs | UyCT1 | GFWGKLWEGVKNAI |
| UyCT3 | ILSAIWSGIKSLF | |
| UyCT5 | IWSAIWSGIKGLL | |
| Uy17 | ILSAIWSGIKGLL | |
| Uy192 | FLSTIWNGIKGLL | |
| Uy234 | FPFLLSLIPSAISAIKRL | |
| Um4 | FFSALLSGIKSLF | |
| D3 | LWGKLWEGVKSLI | |
| D5 | GFWGKLLEGVKKAI | |
| D10 | FPFLKLSLKIPKSAIKSAIKRL | |
| D11 | GFWGKLWEGVKNAIKKK | |
| Insect AMPs | Apidaecin | GNRPVYIPPPRPPHPRL |
| Cecropin A | KWKL FKKIEKVGQN IRDGIIKAGPAVAVVGQATQIA | |
| Stomoxyn | RGFRKH FNKLVKKVKH TISETAHVAKDTAVIAGSGA AVVAAT | |
| Antibiotic | Rifampicin | C43H58N4O12 (CAS number 13292-46-1) |
| Insecticide | Imidacloprid | C9H10ClN5O2 (CAS number 138261-41-3) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luna-Ramirez, K.; Skaljac, M.; Grotmann, J.; Kirfel, P.; Vilcinskas, A. Orally Delivered Scorpion Antimicrobial Peptides Exhibit Activity against Pea Aphid (Acyrthosiphon pisum) and Its Bacterial Symbionts. Toxins 2017, 9, 261. https://doi.org/10.3390/toxins9090261
Luna-Ramirez K, Skaljac M, Grotmann J, Kirfel P, Vilcinskas A. Orally Delivered Scorpion Antimicrobial Peptides Exhibit Activity against Pea Aphid (Acyrthosiphon pisum) and Its Bacterial Symbionts. Toxins. 2017; 9(9):261. https://doi.org/10.3390/toxins9090261
Chicago/Turabian StyleLuna-Ramirez, Karen, Marisa Skaljac, Jens Grotmann, Phillipp Kirfel, and Andreas Vilcinskas. 2017. "Orally Delivered Scorpion Antimicrobial Peptides Exhibit Activity against Pea Aphid (Acyrthosiphon pisum) and Its Bacterial Symbionts" Toxins 9, no. 9: 261. https://doi.org/10.3390/toxins9090261





