Application of Pyrolysis Gas Chromatography/Mass Spectrometry in Lacquer Research: A Review
Abstract
:1. Introduction
2. Pyrolysis-Gas Chromatography/Mass Spectrometry
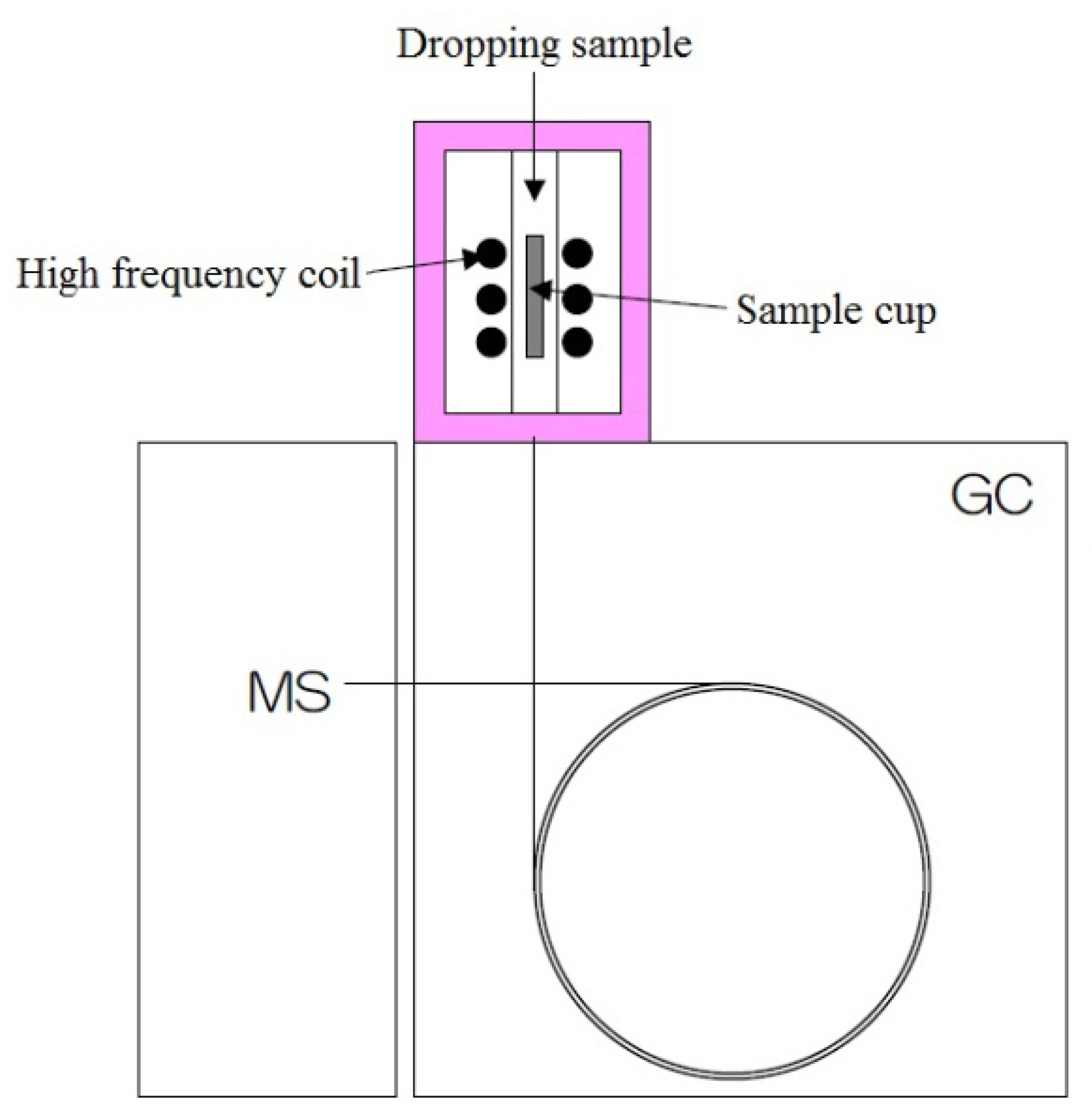
2.1. Single-Shot Py-GC/MS
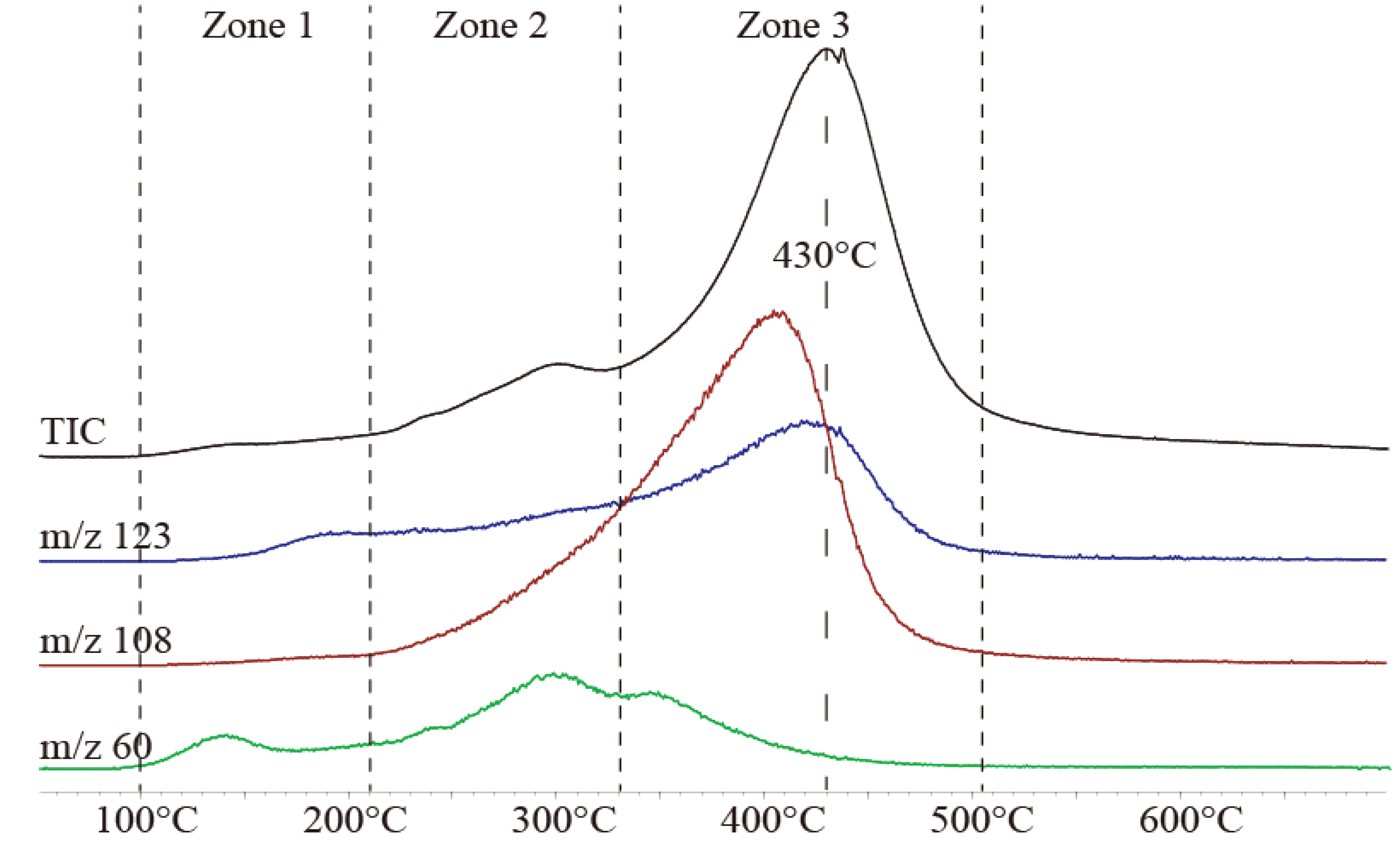
2.2. Evolved Gas Analysis
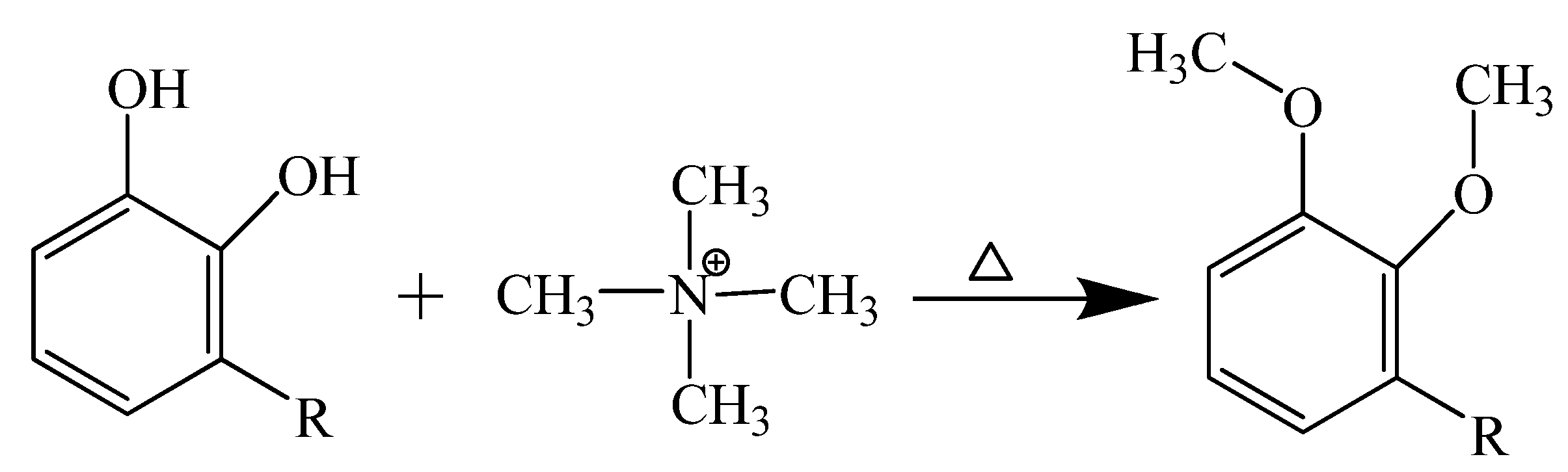
2.3. Double-Shot Pyrolysis
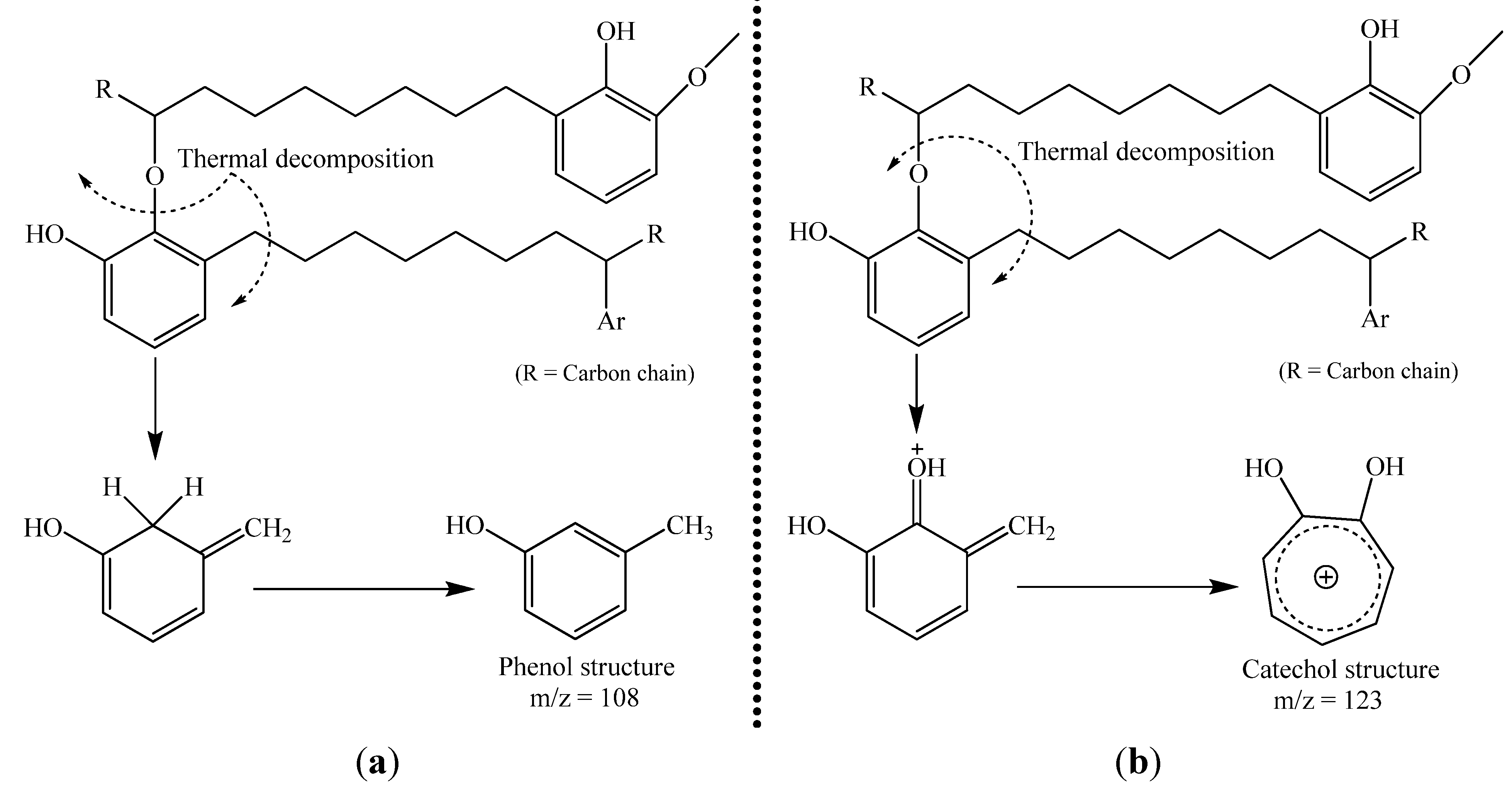
3. Mechanism of Lacquer Drying

4. Identification of Lacquer Species by Py-GC/MS
5. Application of Py-GC/MS to Ancient Lacquer Wares
6. Conclusions
Conflicts of Interest
References
- Ishimatsu, S. On a chemical investigation of Japan liquor or urushi. Series-3. Mem. Proc. Manch. Lit. Philos. Soc. 1882, 7, 249–251. [Google Scholar]
- Burmester, A. Far eastern lacquers: Classification by pyrolysis mass spectrometry. Archaeometry 1983, 25, 45–58. [Google Scholar] [CrossRef]
- Shedrinsky, A.M. Application of analysis to problems in art and archaeology: A review. J. Anal. Appl. Pyrolysis 1989, 15, 393–412. [Google Scholar] [CrossRef]
- Niimura, N.; Miyakoshi, T.; Onodera, J.; Higuchi, T. Thermal degradation of the lacquer film by pyrolysis gas chromatography mass spectrometry. Chem. Soc. Jpn. 1995, 9, 724–729. [Google Scholar]
- Niimura, N.; Miyakoshi, T.; Onodera, J.; Higuchi, T. Characterization of Rhus vernicifera and Rhus succedanea lacquer films and their pyrolysis mechanisms studied using two-stage pyrolysis-gas chromatography/mass spectrometry. J. Anal. Appl. Pyrolysis 1996, 37, 199–209. [Google Scholar] [CrossRef]
- Niimura, N.; Miyakoshi, T.; Onodera, J.; Higuchi, T. Structural studies of Melanorrhoea usitate lacquer film using two-stage pyrolysis/gas chromatography/mass spectrometry. Rapid Commun. Mass Spectrom. 1996, 10, 1719–1724. [Google Scholar] [CrossRef]
- Wampler, T.P. Pyrolysis, Gas Chromatography; Oxford, PA, USA, 2005; pp. 81–95. [Google Scholar]
- Sobeih, K.L.; Baron, M.; Gonzalez-Rodriguez, J. Recent trends and developments in pyrolysis-gas chromatography. J. Chromatogr. A 2008, 1186, 51–66. [Google Scholar] [CrossRef] [Green Version]
- Tsuge, S.; Ohtani, H.; Watanabe, C. Pyrolysis-GC/MS Data Book of Synthetic Polymers, 1st ed.; Elsevier: Oxford, UK, 2011. [Google Scholar]
- Herreraa, M.; Matuschek, G. Fast identification of polymer additives by pyrolysis-gas chromatography/mass spectrometry. J. Anal. Appl. Pyrolysis 2003, 70, 35–42. [Google Scholar] [CrossRef]
- Arii, T. Complex thermal analysis: Evolved gas analysis-mass spectrometry EGA-MS. Rigaku Janaru 2008, 39, 17–25. (in Japanese). [Google Scholar]
- Arii, T. Evolved gas analysis-mass spectrometry (EGA-MS) using skimmer interface system equipped with pressure control function. J. Mass Spectrom. Soc. Jpn. 2005, 53, 211–216. [Google Scholar] [CrossRef]
- Tsugoshi, T.; Furukawa, M.; Ohashi, M.; Iida, Y. Comparison of capillary and skimmer interfaces in evolved gas analysis-mass spectrometry (EGA-MS) with regard to impurities in ceramic raw materials. J. Therm. Anal. Calor. 2001, 64, 1127–1132. [Google Scholar] [CrossRef]
- Matuschek, G.; Kettrup, A.; Prior, A. EGA/MS investigations on the thermal degradation of diammoniumhexachloroplatinate. J. Therm. Anal. Calorim. 1999, 56, 471–477. [Google Scholar] [CrossRef]
- Urakami, K.; Higashi, A.; Umemoto, K.; Godo, M.; Watanabe, C.; Hashimoto, K. Compositional analysis of copoly(dl-lactic/glycolic acid) (PLGA) by pyrolysis-gas chromatography/mass spectrometry combined with one-step thermally assisted hydrolysis and methylation in the presence of tetramethylammonium hydroxide. Chem. Pharm. Bull. 2001, 49, 203–205. [Google Scholar] [CrossRef]
- Fukushima, M.; Yamamoto, M.; Komai, T.; Yamamoto, K. Studies of structural alterations of humic acids from conifer bark residue during composting by pyrolysis-gas chromatography/mass spectrometry using tetramethylammonium hydroxide (TMAH-py-GC/MS). J. Anal. Appl. Pyrolysis 2009, 86, 200–206. [Google Scholar] [CrossRef]
- Araki, H.; Tatarazako, N.; Kishi, K.; Kuroda, K. Evaluation of bioaccumulation potential of 3,4,5-trichloroguaiacol in a zooplankton (Daphnia magna) by pyrolysis-GC/MS in the presence of tetramethylammonium hydroxide (TMAH). J. Anal. Appl. Pyrolysis 2000, 55, 69–80. [Google Scholar] [CrossRef]
- Oshima, R.; Yamauchi, Y.; Watanabe, C.; Kumanotani, J. Enzymic oxidative coupling of urushiol in sap of the lac tree, Rhus vernicifera. J. Org. Chem. 1985, 50, 2613–2621. [Google Scholar] [CrossRef]
- Takei, R.; Lu, R.; Miyakoshi, T. Dimer structures and laccase-catalyzed polymerization mechanism of laccol in fresh Rhus succedanea lacquer sap. Int. J. Polym. Anal. Charact. 2013, 18, 1–12. [Google Scholar] [CrossRef]
- Lu, R.; Kanamori, D.; Miyakoshi, T. Characterization of thitsiol dimer structures from Melanorrhoea usitata with laccase catalyst by NMR spectroscopy. Int. J. Polym. Anal. Charact. 2011, 16, 86–94. [Google Scholar] [CrossRef]
- Nagase, K.; Kamiya, Y.; Kimura, T.; Hodumi, K.; Miyakoshi, T. The relationship between the change of progress time in the urushi liquid by the enzymic polymerization and the natural drying property occurring under a low humidity environment. Nippon Kagaku Kaishi 2001, 10, 587–593. (in Japanese). [Google Scholar]
- Nagase, K.; Kamiya, Y.; Hodumi, K.; Miyakoshi, T. The relationship between the change in urushiol by repetitive “KUROME” of the urushi liquid and natural drying property occurring in a low humidity environment. Nippon Kagaku Kaishi 2002, 3, 377–384. (in Japanese). [Google Scholar]
- Nagase, K.; Lu, R.; Miyakoshi, T. Studies on the fast drying hybrid urushi in low humidity environment. Chem. Lett. 2004, 33, 90–91. [Google Scholar] [CrossRef]
- Kumanotani, J. Super-durable structure of Oriental lacquer films. Polym. Mater. 1988, 59, 278–282. (in Japanese). [Google Scholar]
- Kumanotani, J. Urushi (Oriental lacquer)—A natural aesthetic durable and future-promising coating. Prog. Org. Coat. 1995, 26, 163–195. [Google Scholar] [CrossRef]
- Lambert, J.; Frye, J.; Carriveau, G. The structure of Oriental lacquer by solid state nuclear magnetic resonance spectroscopy. Archaeometry 1991, 33, 87–93. [Google Scholar] [CrossRef]
- Niimura, N.; Miyakoshi, T. Characterization of natural resin films and identification of ancient coating. J. Mass Spectrom. Soc. Jpn. 2003, 51, 439–457. [Google Scholar]
- Lu, R.; Kamiya, Y.; Miyakoshi, T. Preparation and characterization of Melanorrhoea usitata lacquer film based on pyrolysis-gas chromatography/mass spectrometry. J. Anal. Appl. Pyrolysis 2007, 78, 172–179. [Google Scholar] [CrossRef]
- Lu, R.; Kamiya, Y.; Wan, Y.-Y.; Honda, T.; Miyakoshi, T. Synthesis of Rhus succedanea lacquer film and analysis by pyrolysis-gas chromatography/mass spectrometry. J. Anal. Appl. Pyrolysis 2007, 78, 117–124. [Google Scholar] [CrossRef]
- Kamiya, Y.; Niimura, N.; Miyakoshi, T. Evaluation of synthesized lacquer films using pyrolysis-gas chromatography/mass spectrometry. Bull. Chem. Soc. Jpn. 2000, 73, 2621–2626. [Google Scholar] [CrossRef]
- Niimura, N.; Miyakoshi, T.; Iijima, Y. Characterization of synthesized lacquer analogue films by two-stage pyrolysis gas chromatography/mass spectrometry and X-ray photoelectron spectroscopy. Anal. Sci. 2001, 17 (Suppl.), i155–i158. [Google Scholar] [CrossRef]
- Kamiya, Y.; Satto, W.; Miyakoshi, T. Synthesis and identification of laccol components from Rhus succedanea lacquer sap. J. Oleo Sci. 2002, 51, 473–483. [Google Scholar] [CrossRef]
- Lu, R.; Kamiya, Y.; Miyakoshi, T. Characterization of lipid components of Melanorrhoea usitata lacquer sap. Talanta 2007, 71, 1536–1540. [Google Scholar] [CrossRef]
- Kamiya, Y.; Miyakoshi, T. Synthesis of urushiol components and analysis of urushi sap from Rhus vernicifera. J. Oleo Sci. 2001, 50, 865–874. [Google Scholar] [CrossRef]
- Chiavari, G.; Mazzeo, R. Characterisation of paint layers in Chinese archaelogical relics by pyrolysis-GC-MS. Chromatographia 1999, 49, 268–272. [Google Scholar] [CrossRef]
- Niimura, N.; Miyakoshi, T.; Onodera, J.; Higuchi, T. Identification of ancient lacquer film using two-stage pyrolysis-gas chromatography/mass spectrometry. Archaeometry 1999, 41, 137–149. [Google Scholar] [CrossRef]
- Buzzini, P.; Stoecklein, W. Paints, varnishes, and lacquers. Forensic Sci. 2005, 453–464. [Google Scholar]
- Raffaelly, L.; Pons, E.; Lacoudre, N.; Bleton, J.; Duy, S.; Tchapla, A. Characterisation physico-chimique de laques de Mongolie du Ier siecle ap. J.-C. ArcheoSciences 2005, 29, 69–81. [Google Scholar]
- Kamiya, Y.; Lu, R.; Kumamoto, T.; Honnda, T.; Miyakoshi, T. Deterioration of surface structure of lacquer films due to ultraviolet irradiation. Surf. Inter. Anal. 2006, 38, 1311–1315. [Google Scholar] [CrossRef]
- He, L.; Maiqian, N.; Chiavari, G.; Mazzeo, R. Analytical characterization of binding medium used in ancient Chinese artworks by pyrolysis-gas chromatography/mass spectrometry. Microchem. J. 2007, 85, 347–353. [Google Scholar] [CrossRef]
- Domenech-Carbo, M.T. Novel analytical methods for characterising binding media and protective coatings in artworks. Anal. Chim. Acta 2008, 621, 109–139. [Google Scholar] [CrossRef]
- Lu, R.; Miyakoshi, T. Meiji University: Kawasaki-shi, Japan., Unpublished work. 2012.
- Wan, Y.-Y.; Lu, R.; Du, Y.-M.; Honda, T.; Miyakoshi, T. Does Donglan lacquer tree belong to Rhus vernicifera species? Int. J. Biol. Macromol. 2007, 41, 497–503. [Google Scholar] [CrossRef]
- Frade, J.C.; Ribeiro, I.; Graca, J.; Vasconcelos, T.; Rodrigues, J. Chemotaxonomic application of Py-GC/MS: Identification of lacquer trees. J. Anal. Appl. Pyrolysis 2010, 89, 117–121. [Google Scholar] [CrossRef]
- Lu, R.; Kamiya, Y.; Miyakoshi, T. Applied analysis of lacquer films based on pyrolysis-gas chromatography/mass spectrometry. Talanta 2006, 70, 370–376. [Google Scholar] [CrossRef]
- Lu, R.; Ma, X.-M.; Kamiya, Y.; Honda, T.; Kamiya, Y.; Okamoto, A.; Miyakoshi, T. Identification of Ryukyu lacquerware by pyrolysis-gas chromatography/mass spectrometry. J. Anal. Appl. Pyrolysis 2007, 80, 101–110. [Google Scholar] [CrossRef]
- Honda, T.; Lu, R.; Kitano, N.; Kamiya, Y.; Miyakoshi, T. Applied analysis and identification of ancient lacquer based on pyrolysis-gas chromatography/mass spectrometry. J. Appl. Polym. Sci. 2010, 118, 897–901. [Google Scholar]
- Heginbotham, A.; Khanjian, H.; Rivenc, R.; Schilling, M. A procedure for the efficient and simultaneous analysis of asian and european lacquers in furniture of mixed origin. ICOM Comm. Conserv. 2008, 2, 608–616. [Google Scholar]
- Frade, J.C.; Ribeiro, M.I.; Graca, J.; Rodrigues, J. Applying pyrolysis-gas chromatography/mass spectrometry to the identification of Oriental lacquers: Study of two lacquered shields. Anal. Bioanal. Chem. 2009, 395, 2167–2174. [Google Scholar] [CrossRef]
- Niimura, N. Determination of the type of lacquer on East Asian lacquer ware. Int. J. Mass Spectrom. 2009, 284, 93–94. [Google Scholar] [CrossRef]
- Pitthard, V.; Wei, S.; Miklin-Kniefacz, S.; Stanek, S.; Griesser, M.; Schreiner, M. Scientific investigations of antique lacquers from a 17th-century Japanese ornamental cabinet. Archaeometry 2010, 52, 1044–1056. [Google Scholar]
- Wei, S.; Pintus, V.; Pitthard, V.; Schreiner, M.; Song, G. Analytical characterization of lacquer objects excavated from a Chu tomb in China. J. Archaeol. Sci. 2011, 38, 2667–2674. [Google Scholar]
- Le Ho, A.S.; Regert, M.; Marescot, O.; Duhamel, C.; Langlois, J.; Miyakoshi, T.; Genty, C.; Sablier, M. Molecular criteria for discriminating museum Asian lacquerware from different vegetal origins by pyrolysis gas chromatography/mass spectrometry. Anal. Chim. Acta 2012, 710, 9–16. [Google Scholar] [CrossRef]
- Niimura, N.; Miyakoshi, T. Structural study of oriental lacquer films during the hardening process. Talanta 2006, 70, 146–152. [Google Scholar] [CrossRef]
- Wan, Y.-Y.; Lu, R.; Akiyama, K.; Miyakoshi, T.; Du, Y.-M. Enzymatic synthesis of bioactive compounds by Rhus laccase from Chinese vernicifera. Sci. China Ser. B Chem. 2007, 50, 179–172. [Google Scholar] [CrossRef]
- Yoshida, T.; Lu, R.; Han, S.; Hattori, K.; Katsuta, T.; Takada, K.; Sugimoto, K.; Funaoka, M. Laccase-catalyzed polymerization of lignocatechol and affinity on proteins of resulting polymers. J. Polym. Sci. A Polym. Chem. 2009, 47, 824–832. [Google Scholar] [CrossRef]
- Kamiya, Y.; Taketa, S.; Watanabe, C.; Miyakoshi, T. Characterization of volatile products from raw lacquer film during ultraviolet irradiation using on-line micro ultraviolet pyrolysis-GC/MS. Bunseki Kagaku 2011, 60, 269–274. (in Japanese). [Google Scholar] [CrossRef]
- Tsukagoshi, M.; Kitahara, Y.; Takahashi, S.; Tsugoshi, T.; Fuji, T. Characterization of Japanese lacquer liquid and films by means of evolved gas analysis-ion attachment mass spectrometry. Anal. Methods 2011, 3, 1943–1947. [Google Scholar] [CrossRef]
- Niimura, N. Thermogravimetry-linked scan mass spectrometry—Detection of urushiol from an East Asian lacquer film. Therm. Acta 2012, 532, 164–167. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ma, X.-M.; Lu, R.; Miyakoshi, T. Application of Pyrolysis Gas Chromatography/Mass Spectrometry in Lacquer Research: A Review. Polymers 2014, 6, 132-144. https://doi.org/10.3390/polym6010132
Ma X-M, Lu R, Miyakoshi T. Application of Pyrolysis Gas Chromatography/Mass Spectrometry in Lacquer Research: A Review. Polymers. 2014; 6(1):132-144. https://doi.org/10.3390/polym6010132
Chicago/Turabian StyleMa, Xiao-Ming, Rong Lu, and Tetsuo Miyakoshi. 2014. "Application of Pyrolysis Gas Chromatography/Mass Spectrometry in Lacquer Research: A Review" Polymers 6, no. 1: 132-144. https://doi.org/10.3390/polym6010132



