Sialic Acid-Targeted Biointerface Materials and Bio-Applications
Abstract
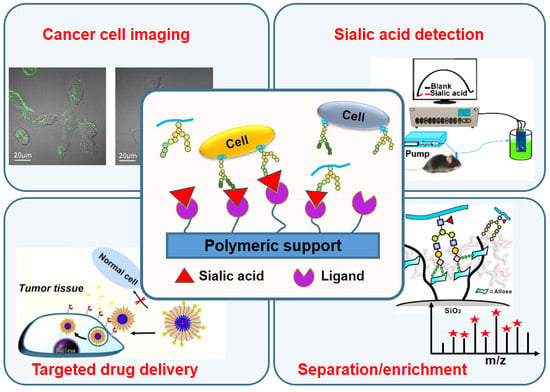
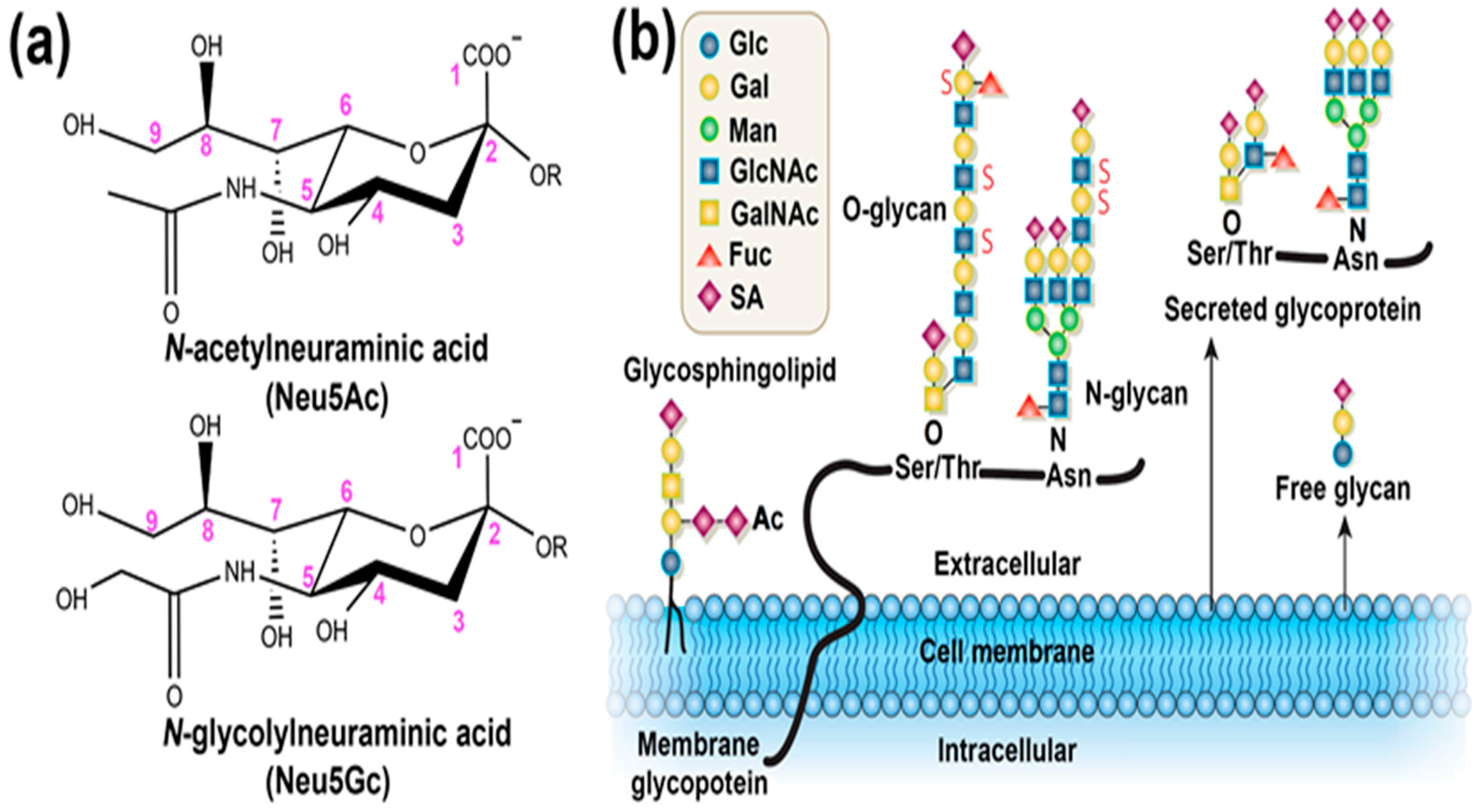
:1. Introduction
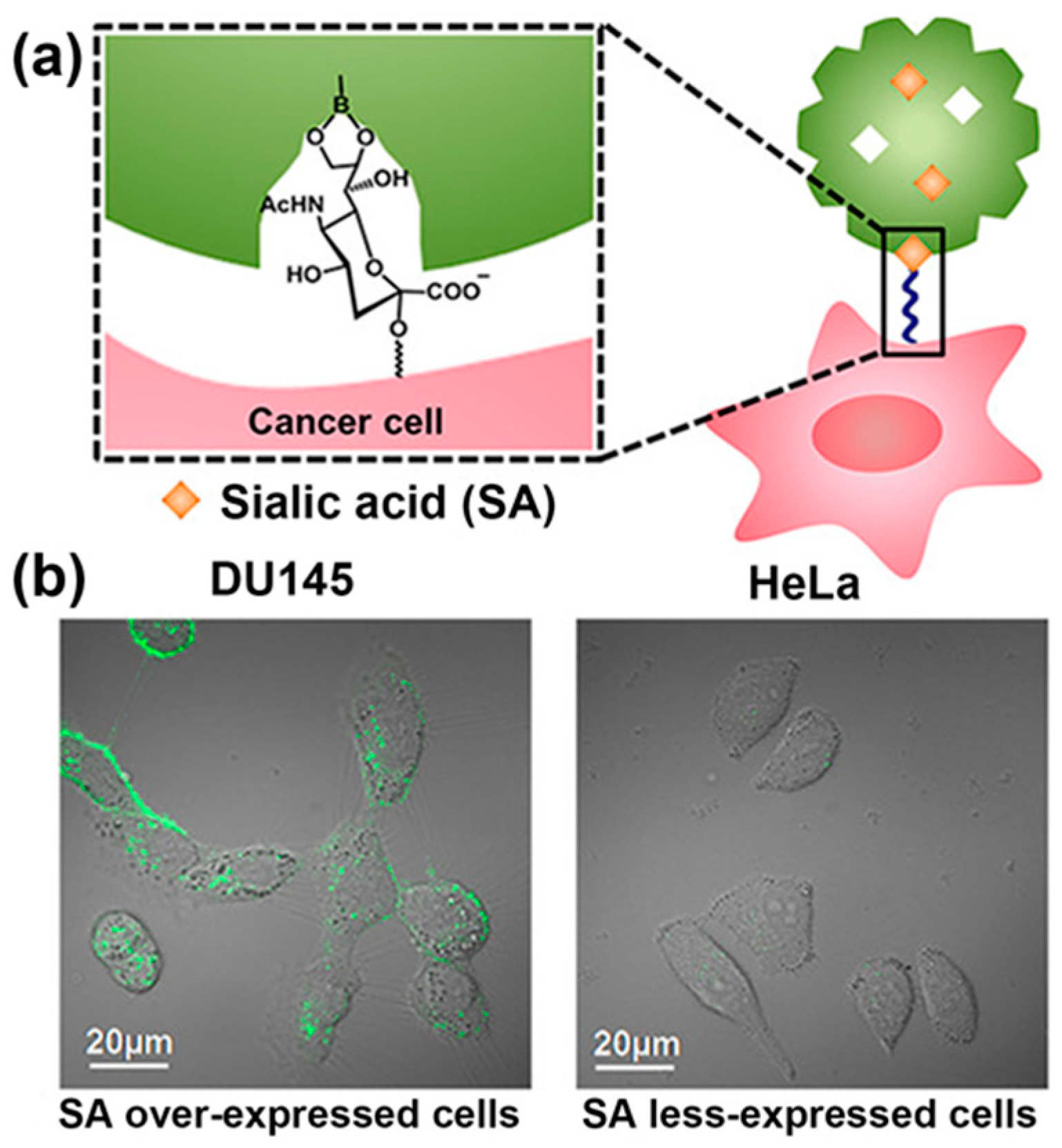
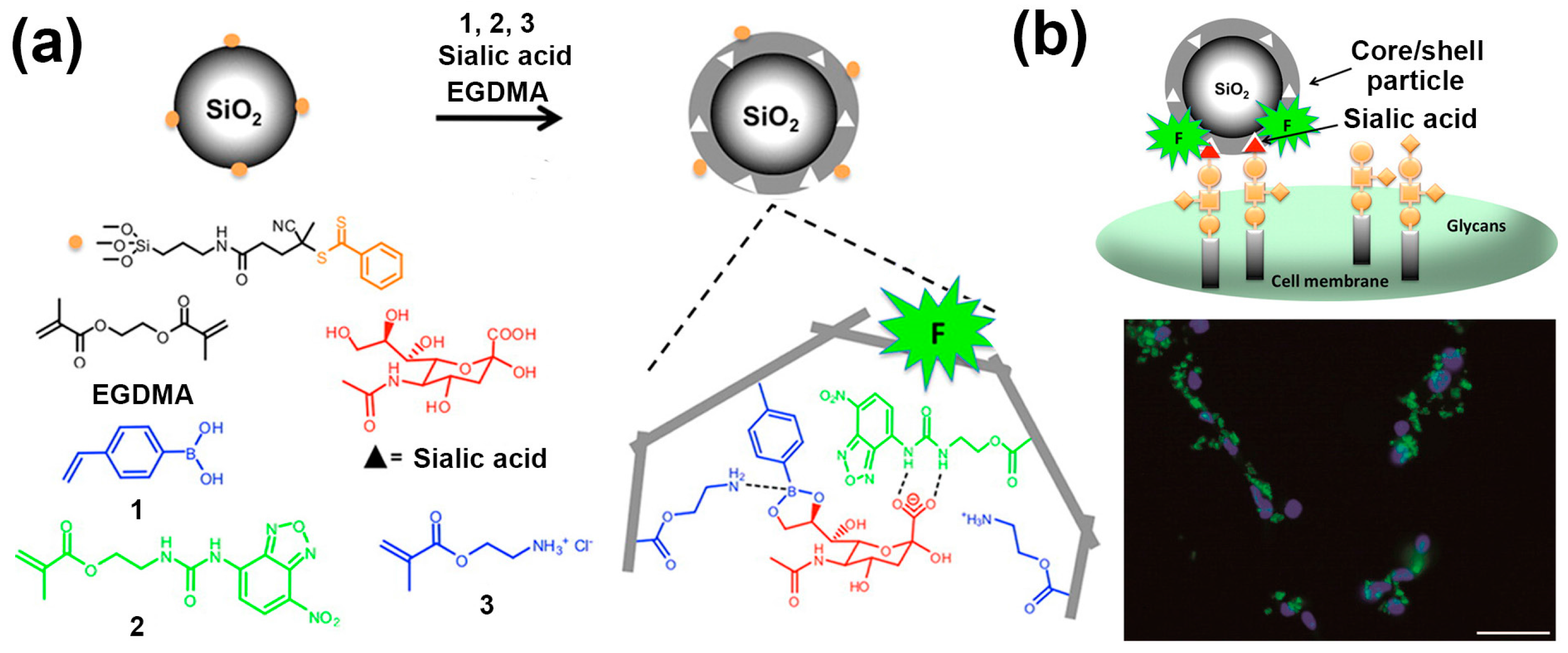
2. SA-Imprinted Particles for Cancer Cell Imaging
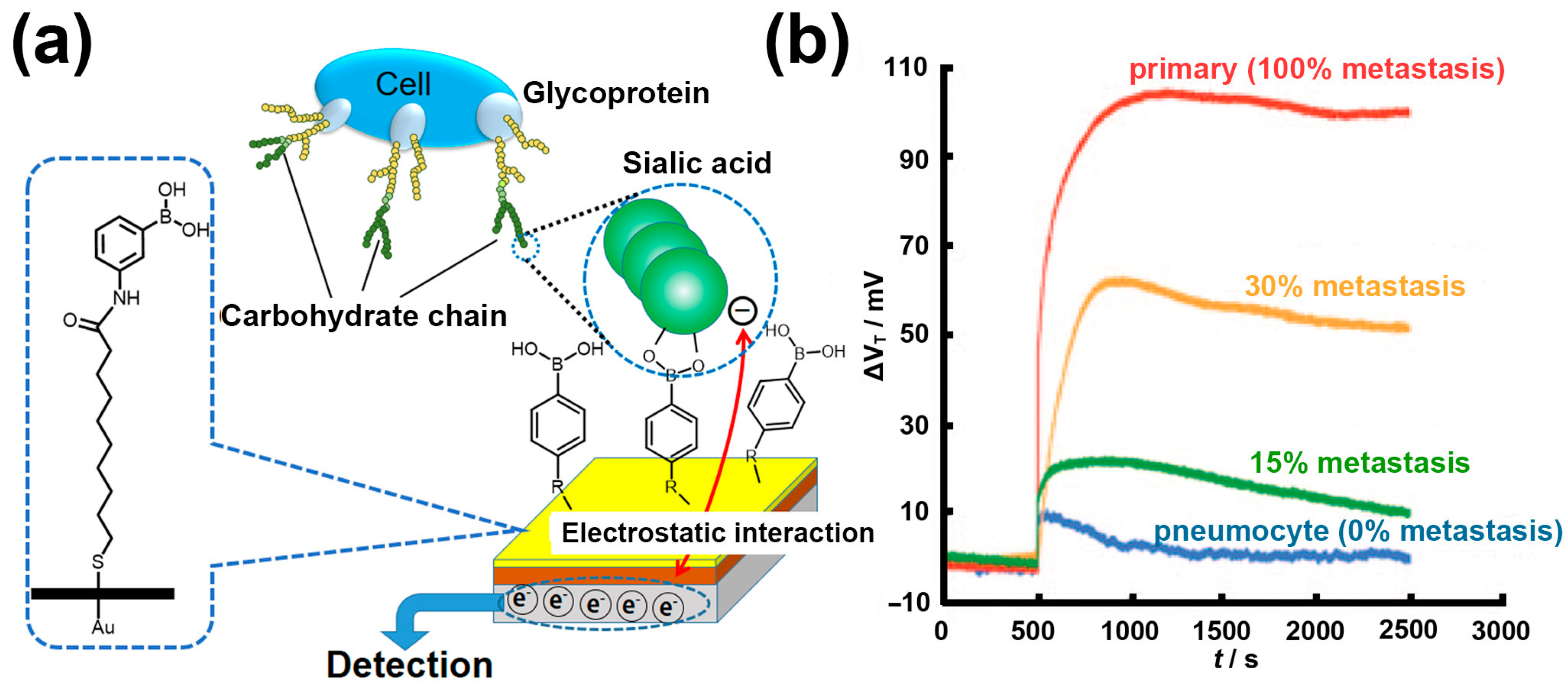
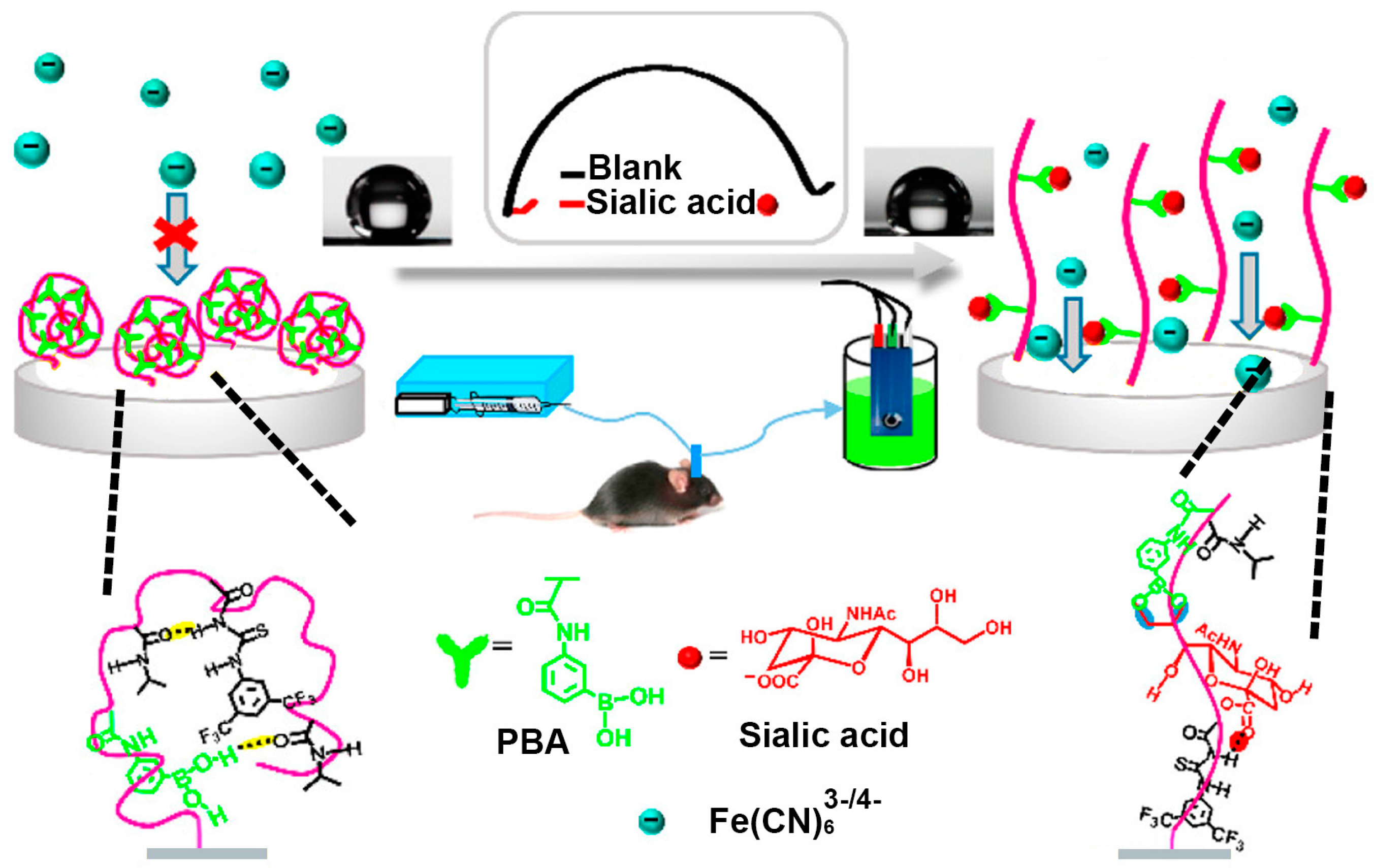
3. Boronic Acid-Modified Surfaces for Detection
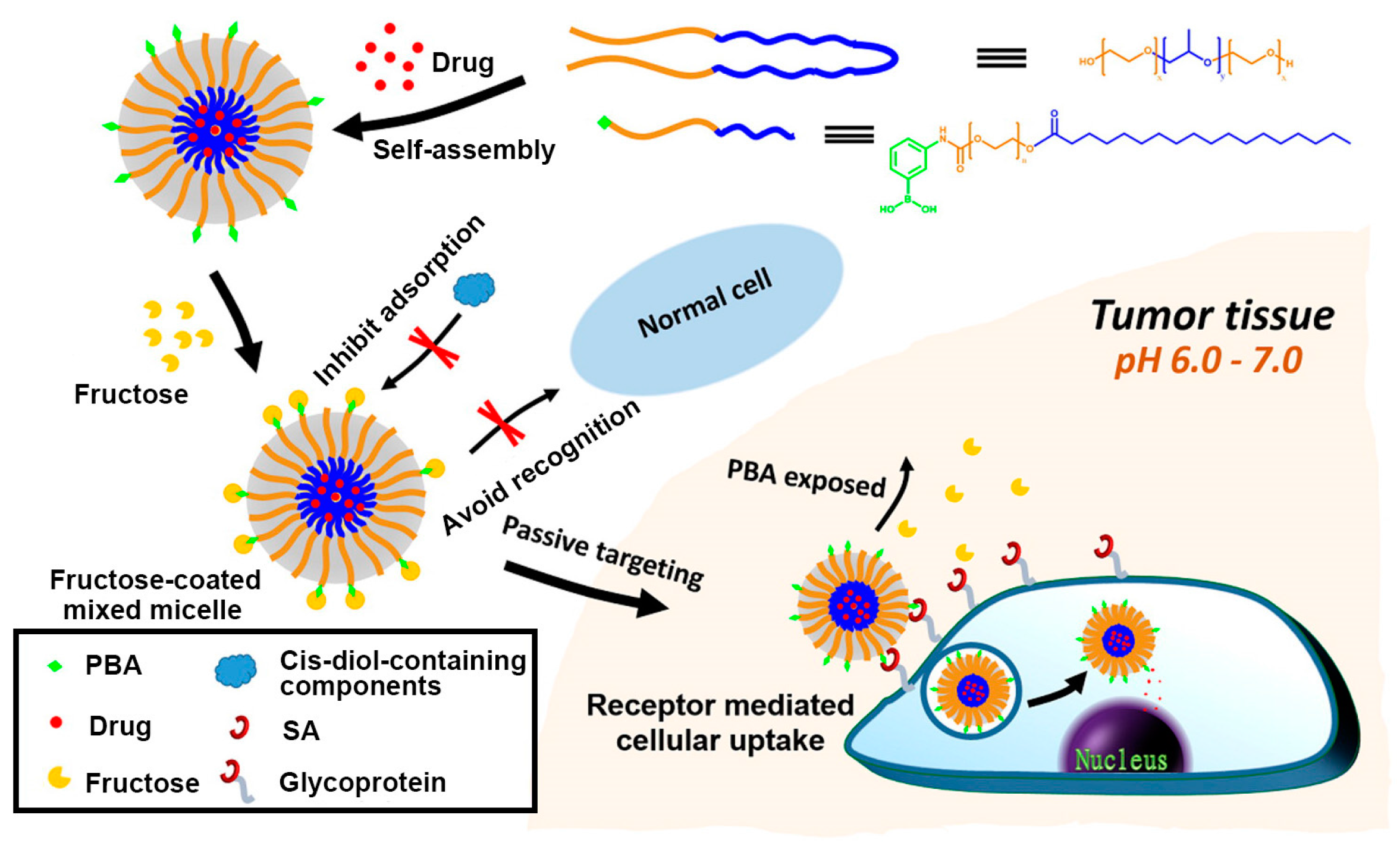
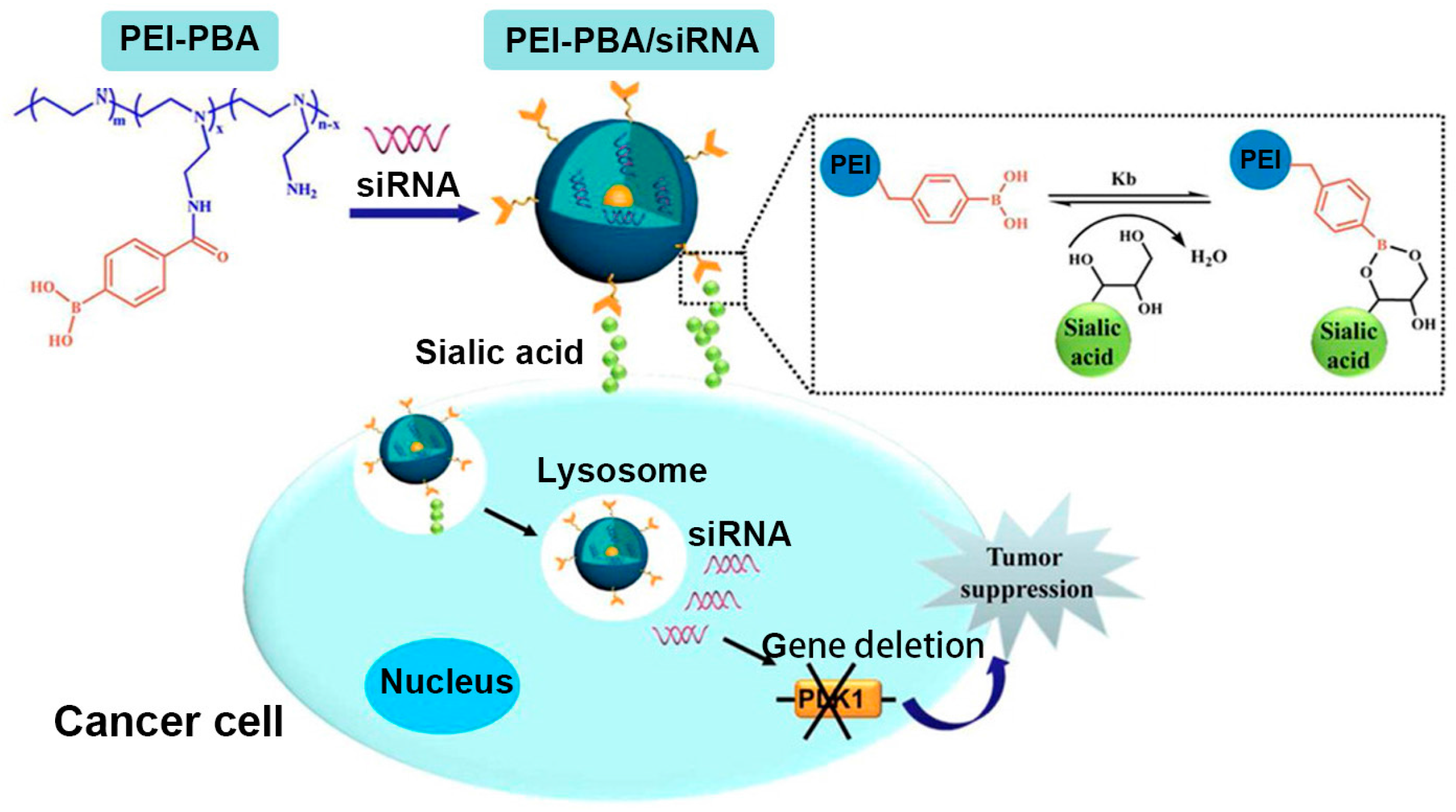
4. Polymeric Nanocarriers for Drug Delivery
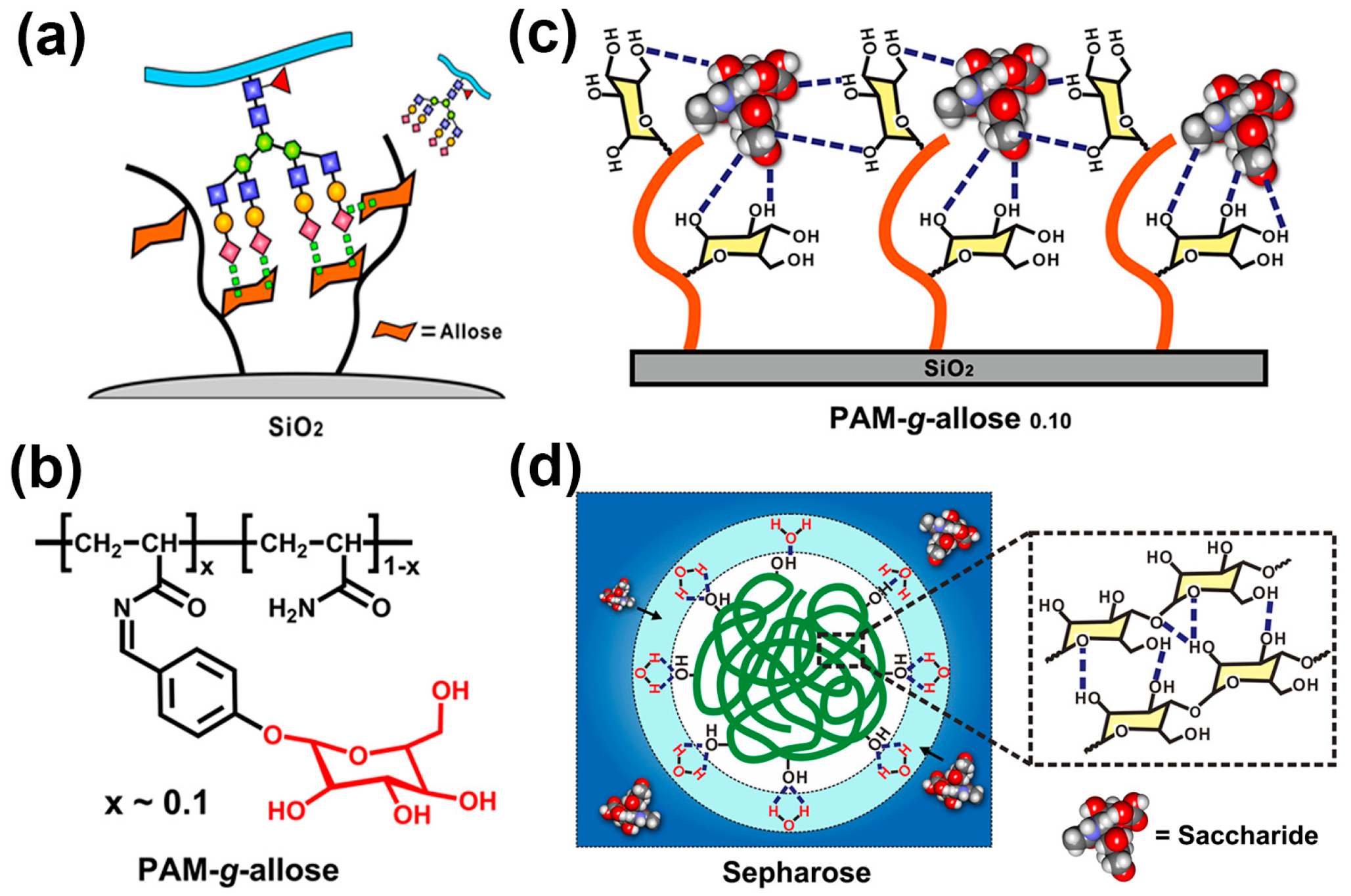
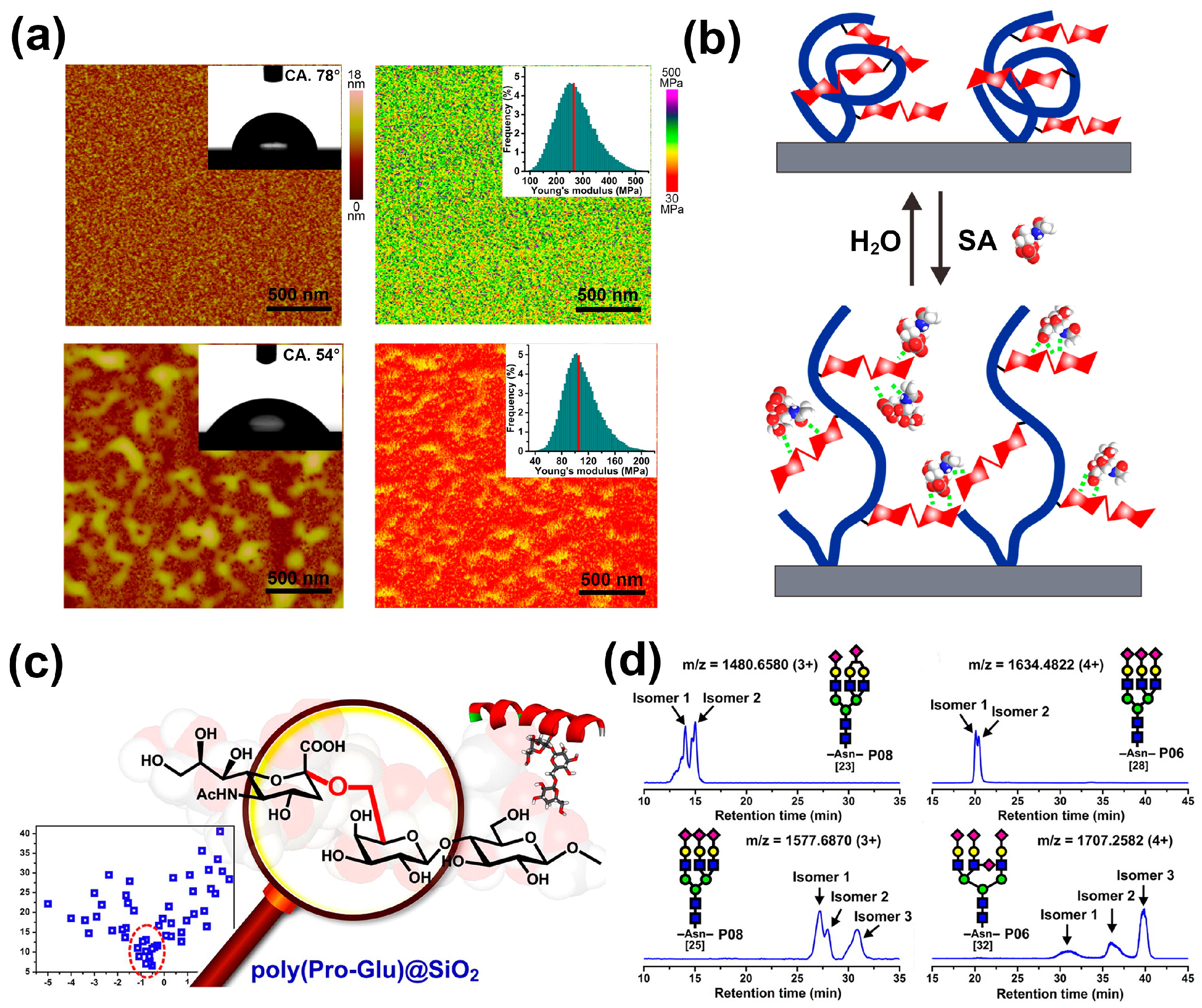
5. Bio-Inspired Materials for SA Derivatives Separation/Enrichment
6. Conclusions and Outlooks
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Blix, G.; Lindberg, E.; Odin, L.; Werner, I. Sialic acids. Nature 1955, 175, 340–341. [Google Scholar] [CrossRef] [PubMed]
- Varki, A. Glycan-based interactions involving vertebrate sialic-acid-recognizing proteins. Nature 2007, 446, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Varki, N.M.; Varki, A. Diversity in cell surface sialic acid presentations: Implications for biology and disease. Lab. Investig. 2007, 87, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Schauer, R. Sialic acids as regulators of molecular and cellular interactions. Curr. Opin. Struct. Biol. 2009, 19, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Varki, A. Sialic acids in human health and disease. Trends Mol. Med. 2008, 14, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Dall’Olio, F.; Chiricolo, M. Sialyltransferases in cancer. Glycoconj. J. 2001, 18, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Seales, E.C.; Jurado, G.A.; Brunson, B.A.; Wakefield, J.K.; Frost, A.R.; Bellis, S.L. Hypersialylation of beta1 integrins, observed in colon adenocarcinoma, may contribute to cancer progression by up-regulating cell motility. Cancer Res. 2005, 65, 4645–4652. [Google Scholar] [CrossRef] [PubMed]
- Varki, N.M.; Varki, A. Heparin inhibition of selectin-mediated interactions during the hematogenous phase of carcinoma metastasis: Rationale for clinical studies in humans. Semin. Thromb. Hemost. 2002, 28, 53–66. [Google Scholar] [CrossRef] [PubMed]
- Laubli, H.; Stevenson, J.L.; Varki, A.; Varki, N.M.; Borsig, L. L-selectin facilitation of metastasis involves temporal induction of Fut7-dependent ligands at sites of cell arrest. Cancer Res. 2006, 66, 1536–1542. [Google Scholar] [CrossRef] [PubMed]
- Park, J.J.; Lee, M. Increasing the α-2, 6-sialylation of glycoproteins may contribute to metastatic spread and therapeutic resistance in colorectal cancer. Gut Liver 2013, 7, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Costa-Nogueira, C.; Villar-Portela, S.; Cuevas, E.; Gil-Martín, E.; Fernández-Briera, A. Synthesis and expression of CDw75 antigen in human colorectal cancer. BMC Cancer 2009, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Berbec, H.; Paszkowska, A.; Siwek, B.; Gradziel, K.; Cybulski, M. Total serum sialic acid concentration as a supporting marker of malignancy in ovarian neoplasia. Eur. J. Gynaecol. Oncol. 1999, 20, 389–392. [Google Scholar] [PubMed]
- Basoglu, M.; Yildirgan, M.I.; Taysi, S.; Yilmaz, I.; Kiziltunc, A.; Balik, A.A.; Celebi, F.; Atamanalp, S.S. Levels of soluble intercellularadhesion molecule-1 and total sialic acid in serum of patients with colorectal cancer. J. Surg. Oncol. 2003, 83, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Rajpura, K.B.; Patel, P.S.; Chawda, J.G.; Shah, R.M. Clinical significance of total and lipid bound sialic acid levels in oral pre-cancerous conditions and oral cancer. J. Oral Pathol. Med. 2005, 34, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Samraj, A.N.; Pearce, O.M.T.; Laubli, H.; Crittenden, A.N.; Bergfeld, A.K.; Banda, K.; Gregg, C.J.; Bingman, A.E.; Secrest, P.; Diaz, S.L. A red meat-derived glycan promotes inflammation and cancer progression. Proc. Natl. Acad. Sci. USA 2015, 112, 542–547. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA: Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S. Sialic acids: Biomarkers in endocrinal cancers. Glycoconj. J. 2015, 32, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Szabo, Z.; Bones, J.; Guttman, A.; Glick, J.; Karger, B.L. Sialic acid speciation using capillary electrophoresis: Optimization of analyte derivatization and separation. Anal. Chem. 2012, 84, 7638–7642. [Google Scholar] [CrossRef] [PubMed]
- Maeda, E.; Kita, S.; Kinoshita, M.; Urakami, K.; Hayakawa, T.; Kakehi, K. Analysis of nonhuman N-glycans as the minor constituents in recombinant monoclonal antibody pharmaceuticals. Anal. Chem. 2012, 84, 2373–2379. [Google Scholar] [CrossRef] [PubMed]
- Jourdian, G.W.; Dean, L.; Roseman, S. The sialic acids XI. A periodate-resorcinol method for the quantitative estimation of free sialic acids and their glycosides. J. Biol. Chem. 1971, 246, 430–435. [Google Scholar] [PubMed]
- Kikkeri, R.; Padler-Karavani, V.; Diaz, S.; Verhagen, A.; Yu, H.; Cao, H.; Langereis, M.A.; De Groot, R.J.; Chen, X.; Varki, A. Quantum dot nanometal surface energy transfer based biosensing of sialic acid compositions and linkages in biological samples. Anal. Chem. 2013, 85, 3864–3870. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xiong, Y.; Qing, G.; Sun, T. Biomolecular responsive polymer materials. Prog. Chem. 2017, 29, 348–358. [Google Scholar]
- Sanjoh, M.; Miyahara, Y.; Kataoka, K.; Matsumoto, A. Phenylboronic acids-based diagnostic and therapeutic applications. Anal. Sci. 2014, 30, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Bondioli, L.; Ruozi, B.; Belletti, D.; Forni, F.; Vandelli, M.A.; Tosi, G. Sialic acid as a potential approach for the protection and targeting of nanocarriers. Expert Opin. Drug Deliv. 2011, 8, 921–937. [Google Scholar] [CrossRef] [PubMed]
- Qing, G.; Lu, Q.; Xiong, Y.; Zhang, L.; Wang, H.; Li, X.; Liang, X.; Sun, T. New opportunities and challenges of smart polymers in post-translational modification proteomics. Adv. Mater. 2017, 1604670. [Google Scholar] [CrossRef] [PubMed]
- Roy, E.; Patra, S.; Tiwari, A.; Madhuri, R.; Sharma, P.K. Introduction of selectivity and specificity to graphene using an inimitable combination of molecular imprinting and nanotechnology. Biosens. Bioelectron. 2017, 89, 234–248. [Google Scholar] [CrossRef] [PubMed]
- Kunath, S.; Panagiotopoulou, M.; Maximilien, J.; Marchyk, N.; Sanger, J.; Haupt, K. Cell and tissue imaging with molecularly imprinted polymers as plastic antibody mimics. Adv. Healthc. Mater. 2015, 4, 1322–1326. [Google Scholar] [CrossRef] [PubMed]
- Wulff, G. Molecular imprinting in cross-linked materials with the aid of molecular templates—A way towards artificial antibodies. Angew. Chem. Int. Ed. Engl. 1995, 34, 1812–1832. [Google Scholar] [CrossRef]
- Kugimiya, A.; Yoneyama, H.; Takeuchi, T. Sialic acid imprinted polymer-coated quartz crystal microbalance. Electroanalysis 2010, 12, 1322–1326. [Google Scholar] [CrossRef]
- Kugimiya, A.; Takeuchi, T. Surface plasmon resonance sensor using molecularly imprinted polymer for detection of sialic acid. Biosens. Bioelectron. 2001, 16, 1059–1062. [Google Scholar] [CrossRef]
- Liu, R.H.; Cui, Q.L.; Wang, C.; Wang, X.Y.; Yang, Y.; Li, L.D. Preparation of sialic acid-imprinted fluorescent conjugated nanoparticles and their application for targeted cancer cell imaging. ACS Appl. Mater. Interfaces 2017, 9, 3006–3015. [Google Scholar] [CrossRef] [PubMed]
- Shinde, S.; El-Schich, Z.; Malakpour, A.; Wan, W.; Dizeyi, N.; Mohammadi, R.; Rurack, K.; Wingren, A.G.; Sellergren, B. Sialic acid-imprinted fluorescent core-shell particles for selective labeling of cell surface glycans. J. Am. Chem. Soc. 2015, 137, 13908–139012. [Google Scholar] [CrossRef] [PubMed]
- El-Schich, Z.; Abdullah, M.; Shinde, S.; Dizeyi, N.; Rosen, A.; Sellergren, B.; Wingren, A.G. Different expression levels of glycans on leukemic cells—A novel screening method with molecularly imprinted polymers (MIP) targeting sialic acid. Tumor Biol. 2016, 37, 13763–13768. [Google Scholar] [CrossRef] [PubMed]
- Panagiotopoulou, M.; Kunath, S.; Medina-Rangel, P.X.; Haupt, K.; Bui, B.T.S. Fluorescent molecularly imprinted polymers as plastic antibodies for selective labeling and imaging of hyaluronan and sialic acid on fixed and living cells. Biosens. Bioelectron. 2017, 88, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Yin, D.Y.; Wang, W.J.; Shen, X.J.; Zhu, J.J.; Chen, H.Y.; Liu, Z. Targeting and imaging of cancer cells via monosaccharide-imprinted fluorescent nanoparticles. Sci. Rep. 2016, 6, 22757. [Google Scholar] [CrossRef] [PubMed]
- Hatano, K.; Miyamoto, Y.; Nonomura, N.; Kaneda, Y. Expression of gangliosides, GD1a, and sialyl paragloboside is regulated by NF-κB-dependent transcriptional control of α-2,3-sialyltransferase I, II, and VI in human castration-resistant prostate cancer cells. Int. J. Cancer 2011, 129, 1838–1847. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Cheng, L.; Wang, L.J.; Liu, H.C.; Li, L.; Wang, X.L.; Geng, M.Y. Cell surface sialic acid inhibits Cx43 gap junction functions in constructed Hela cancer cells involving in sialylated N-cadherin. Mol. Cell. Biochem. 2010, 344, 241–251. [Google Scholar] [CrossRef] [PubMed]
- Yin, D.Y.; Wang, S.S.; He, Y.J.; Liu, J.; Zhou, M.; Ouyang, J.; Liu, B.R.; Chen, H.Y.; Liu, Z. Surface-enhanced Raman scattering imaging of cancer cells and tissues via sialic acid-imprinted nanotags. Chem. Commun. 2015, 51, 17696–17699. [Google Scholar] [CrossRef] [PubMed]
- Westmark, P.R.; Gardiner, S.J.; Smith, B.D. Selective monosaccharide transport through lipid bilayers using boronic acid carriers. J. Am. Chem. Soc. 1996, 118, 11093–11100. [Google Scholar] [CrossRef]
- Bull, S.D.; Davidson, M.G.; Van den Elsen, J.M.; Fossey, J.S.; Jenkins, A.T.; Jiang, Y.B.; Kubo, Y.; Marken, F.; Sakurai, K.; Zhao, J.; James, T.D. Exploiting the reversible covalent bonding of boronic acids: Recognition, sensing, and assembly. Acc. Chem. Res. 2013, 46, 312–326. [Google Scholar] [CrossRef] [PubMed]
- Geninatti Crich, S.; Alberti, D.; Szabo, I.; Aime, S.; Djanashvili, K. MRI visualization of melanoma cells by targeting overexpressed sialic acid with a Gd(III)-dota-en-pba imaging reporter. Angew. Chem. Int. Ed. Engl. 2013, 21, 1161–1164. [Google Scholar] [CrossRef] [PubMed]
- Saito, S.; Meada, T.; Nakazumi, H.; Colyer, C.L. An application of polymer-enhanced capillary transient isotachophoresis with an emissive boronic acid functionalized squarylium dye as an on-capillary labeling agent for gram-positive bacteria. Anal. Sci. 2013, 29, 157–159. [Google Scholar] [CrossRef] [PubMed]
- Mader, H.S.; Wolfbeis, O.S. Boronic acid based probes for microdetermination of saccharides and glycosylated biomolecules. Microchim. Acta 2008, 162, 1–34. [Google Scholar] [CrossRef]
- Matsumoto, A.; Ikeda, S.; Harada, A.; Kataoka, K. Glucose-responsive polymer bearing a novel phenylborate derivative as a glucose-sensing moiety operating at physiological pH conditions. Biomacromolecules 2003, 4, 1410–1416. [Google Scholar] [CrossRef] [PubMed]
- Dowlut, M.; Hall, D.G. An improved class of sugar-binding boronic acids, soluble and capable of complexing glycosides in neutral water. J. Am. Chem. Soc. 2006, 128, 4226–4227. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, H.; Uchimura, E.; Koshina, H.; Okano, T.; Kataoka, K. Anomalous binding profile of phenylboronic acid with N-acetylneuraminic acid (Neu5Ac) in aqueous solution with varying pH. J. Am. Chem. Soc. 2003, 125, 3493–3502. [Google Scholar] [CrossRef] [PubMed]
- Djanashvili, K.; Frullano, L.; Peters, J.A. Molecular recognition of sialic acid end groups by phenylboronates. Chem. Eur. J. 2005, 11, 4010–4018. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, A.; Sato, N.; Kataoka, K.; Miyahara, Y. Noninvasive sialic acid detection at cell membrane by using phenylboronic acid modified self-assembled monolayer gold electrode. J. Am. Chem. Soc. 2009, 131, 12022–12023. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, A.; Cabral, H.; Sato, N.; Kataoka, K.; Miyahara, Y. Assessment of tumor metastasis by the direct determination of cell-membrane sialic acid expression. Angew. Chem. Int. Ed. Engl. 2010, 49, 5494–5497. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.B.; Ju, J.; Li, J.; Li, J.Y.; Qian, Q.H.; Mao, C.; Shen, J. Preparation of electrochemical cytosensor for sensitive detection of heLa cells Based on self-assembled monolayer. Electrochim. Acta 2014, 123, 511–517. [Google Scholar] [CrossRef]
- Ge, S.; Zhang, L.; Zhang, Y.; Liu, H.; Huang, J.; Yan, M.; Yu, J. Electrochemical K-562 cells sensor based on origami paper device for point-of-care testing. Talanta 2015, 145, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Dervisevic, M.; Senel, M.; Sagir, T.; Isik, S. Highly sensitive detection of cancer cells with an electrochemical cytosensor based on boronic acid functional polythiophene. Biosens. Bioelectron. 2017, 90, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Liu, J.; Liu, F.Y.; She, F.; Zheng, Q.; Tang, H.; Ma, M.; Yao, S.Z. Label-free and sensitive sialic acid biosensor based on organic electrochemical transistors. Sens. Actuators B 2017, 240, 1075–1082. [Google Scholar] [CrossRef]
- Zhang, L.; Yu, C.; Gao, R.; Niu, Y.; Li, Y.; Chen, J.; He, J. An impedimetric biosensor for the diagnosis of renal cell carcinoma based on the interaction between 3-aminophenyl boronic acid and sialic acid. Biosens. Bioelectron. 2017, 92, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.; Cao, S.; Liu, Y.; Lian, Y.; Zhu, A.; Shi, G. Rational design of a stimuli-responsive polymer electrode interface coupled with in vivo microdialysis for measurement of sialic acid in live mouse brain in Alzheimer’s Disease. ACS Sens. 2017, 394–400. [Google Scholar] [CrossRef]
- Ding, P.; Li, X.L.; Qing, G.Y.; Sun, T.L.; Liang, X.M. Disaccharide-driven macroscopic properties transition: From molecular recognition to glycopeptide enrichment. Chem. Commun. 2015, 51, 16111–16114. [Google Scholar] [CrossRef] [PubMed]
- Li, M.M.; Qing, G.Y.; Xiong, Y.T.; Lai, Y.K.; Sun, T.L. CH-π interaction driven macroscopic property transition on smart polymer surface. Sci. Rep. 2015, 5, 15742. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.P.; Peng, S.; Soo, J.C.; Kuang, M.; Chen, P.; Duan, H.W. Quantum dots with phenylboronic acid tags for specific labeling of sialic acids on living cells. Anal. Chem. 2011, 83, 1124–1130. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.T.; Zhang, P.H.; Liu, Y.M.; Abdel-Halim, E.S.; Zhu, J.J. Versatile Microfluidic Platform for the Assessment of Sialic Acid Expression on Cancer Cells Using Quantum Dots with Phenylboronic Acid Tags. ACS Appl. Mater. Interfaces 2015, 7, 14878–14884. [Google Scholar] [CrossRef] [PubMed]
- Sankoh, S.; Thammakhet, C.; Numnuam, A.; Limbut, W.; Kanatharana, P.; Thavarungkul, P. 4-mercaptophenylboronic acid functionalized gold nanoparticles for colorimetric sialic acid detection. Biosens. Bioelectron. 2016, 85, 743–750. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.J.; Yan, Y.Q.; Gao, P.Y.; Huang, S.S. Recognition of MCF-7 human breast carcinoma cells using silica-encapsulated fluorescent nanoparticles modified with aminophenylboronic acid. Microchim. Acta 2016, 183, 1–8. [Google Scholar] [CrossRef]
- Chaudhary, P.M.; Murthy, R.V.; Yadav, R.; Kikkeri, R. A rationally designed peptidomimetic biosensor for sialic acid on cell surfaces. Chem. Commun. 2015, 51, 8112–8115. [Google Scholar] [CrossRef] [PubMed]
- Du, J.Z.; Sun, T.M.; Song, W.J.; Wu, J.; Wang, J. A tumor-acidity-activated charge-conversional nanogel as an intelligent vehicle for promoted tumoral-cell uptake and drug delivery. Angew. Chem. Int. Ed. Engl. 2010, 49, 3621–3626. [Google Scholar] [CrossRef] [PubMed]
- Mishra, D.K.; Balekar, N.; Mishra, P.K. Nanoengineered strategies for siRNA delivery: From target assessment to cancer therapeutic efficacy. Drug Deliv. Transl. Res. 2017, 7, 346–358. [Google Scholar] [CrossRef] [PubMed]
- Deshayes, S.; Cabral, H.; Ishii, T.; Miura, Y.; Kobayashi, S.; Yamashita, T.; Matsumoto, A.; Miyahara, Y.; Nishiyama, N.; Kataoka, K. Phenylboronic acid-installed polymeric micelles for targeting sialylated epitopes in solid tumors. J. Am. Chem. Soc. 2013, 135, 15501–15507. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Xu, J.Q.; Yi, X.Q.; Zhang, Q.; Cheng, S.X.; Zhuo, R.X.; Li, F. pH-activated targeting drug delivery system based on the selective binding of phenylboronic acid. ACS Appl. Mater. Interfaces 2016, 8, 14845–14854. [Google Scholar] [CrossRef] [PubMed]
- Naito, M.; Ishii, T.; Matsumoto, A.; Miyata, K.; Miyahara, Y.; Kataoka, K. A phenylboronate-functionalized polyion complex micelle for ATP-triggered release of siRNA. Angew. Chem. Int. Ed. Engl. 2012, 51, 10751–10755. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, Y.M.; Kim, H.; Park, D.; Kim, J.; Kim, W.J. Phenylboronic acid-sugar grafted polymer architecture as a dual stimuli-responsive gene carrier for targeted anti-angiogenic tumor therapy. Biomaterials 2016, 75, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Ji, M.; Li, P.; Sheng, N.; Liu, L.; Pan, H.; Wang, C.; Cai, L.; Ma, Y. Sialic acid-targeted nanovectors with phenylboronic acid-grafted polyethylenimine robustly enhance siRNA-based cancer therapy. ACS Appl. Mater. Interfaces 2016, 8, 9565–9576. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.J.; Swindall, A.F.; Bellis, S.L. Regulation of the metastatic cell phenotype by sialylated glycans. Cancer Metastasis Rev. 2012, 31, 501–518. [Google Scholar] [CrossRef] [PubMed]
- Büll, C.; Stoel, M.A.; den Brok, M.H.; Adema, G.J. Sialic Acids Sweeten a Tumor’s Life. Cancer Res. 2014, 74, 3199–3204. [Google Scholar] [CrossRef] [PubMed]
- Büll, C.; Boltje, T.J.; Wassink, M.; de Graaf, A.M.A.; van Delft, F.L.; den Brok, M.H.; Adema, G.J. Targeting aberrant sialylation in cancer cells using a fluorinated sialic acid analog impairs adhesion, migration, and in vivo tumor growth. Mol. Cancer Ther. 2013, 12, 1935–1946. [Google Scholar] [CrossRef] [PubMed]
- Büll, C.; Boltje, T.J.; van Dinther, E.A.W.; Peters, T.; de Graaf, A.M.A.; Leusen, J.H.W.; Kreutz, M.; Figdor, C.G.; den Brok, M.H.; Adema, G.J. Targeted delivery of a sialic acid-blocking glycomimetic to cancer cells inhibits metastatic spread. ACS Nano. 2015, 9, 733–745. [Google Scholar] [CrossRef] [PubMed]
- He, X.M.; Aizenberg, M.; Kuksenok, O.; Zarzar, L.D.; Shastri, A.; Balazs, A.C.; Aizenberg, J. Synthetic homeostatic materials with chemo-mechano-chemical self-regulation. Nature 2012, 487, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Cummings, R.D.; Pierce, J.M. The Challenge and promise of glycomics. Chem. Biol. 2014, 21, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Handa, K.; Hakomori, S. Carbohydrate to carbohydrate interaction in development process and cancer progression. Glycoconj. J. 2012, 29, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Kojima, N.; Fenderson, B.A.; Stroud, M.R.; Goldberg, R.I.; Habermann, R.; Toyokuni, T.; Hakomori, S. Further studies on cell adhesion based on Le(x)-Le(x) interaction, with new approaches: Embryoglycan aggregation of F9 teratocarcinoma cells, and adhesion of various tumour cells based on Le(x) expression. Glycoconj. J. 1994, 11, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.J.; Nakayama, K.I.; Hikita, T.; Handa, K.; Hakomori, S.I. Epidermal growth factor receptor tyrosine kinase is modulated by GM3 interaction with N-linked GlcNAc termini of the receptor. Proc. Natl. Acad. Sci. USA 2006, 103, 18987–18991. [Google Scholar] [CrossRef] [PubMed]
- Li, X.L.; Xiong, Y.T.; Qing, G.Y.; Jiang, G.; Li, X.Q.; Sun, T.L.; Liang, X.M. Bioinspired saccharide–saccharide interaction and smart polymer for specific enrichment of sialylated glycopeptides. ACS Appl. Mater. Interfaces 2016, 8, 13294–13302. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.T.; Jiang, G.; Li, M.M.; Qing, G.Y.; Li, X.L.; Liang, X.M.; Sun, T.L. Sialic acid-responsive polymeric interface material: From molecular recognition to macroscopic property switching. Sci. Rep. 2017, 7, 40913. [Google Scholar] [CrossRef] [PubMed]
- Zielinska, D.F.; Gnad, F.; Wisniewski, J.R.; Mann, M. Precision mapping of an in vivo N-glycoproteome reveals rigid topological and sequence constraints. Cell 2010, 141, 897–907. [Google Scholar] [CrossRef] [PubMed]
- Plavina, T.; Wakshull, E.; Hancock, W.S.; Hincapie, M. Combination of abundant protein depletion and multi-lectin affinity chromatography (M-LAC) for plasma protein biomarker discovery. J. Proteome Res. 2007, 6, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Ongay, S.; Boichenko, A.; Govorukhina, N.; Bischoff, R. Glycopeptide enrichment and separation for protein glycosylation analysis. J. Sep. Sci. 2012, 35, 2341–2372. [Google Scholar] [CrossRef] [PubMed]
- Fadda, E.; Woods, R.J. Molecular simulations of carbohydrates and protein–carbohydrate interactions: Motivation, issues and prospects. Drug Discov. Today 2010, 15, 596–609. [Google Scholar] [CrossRef] [PubMed]
- Qing, G.Y.; Li, X.L.; Xiong, P.; Chen, C.; Zhan, M.M.; Liang, X.M.; Sun, T.L. Dipeptide-based carbohydrate receptors and polymers for glycopeptide enrichment and glycan discrimination. ACS Appl. Mater. Interfaces 2016, 8, 22084–22092. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiong, Y.; Li, M.; Lu, Q.; Qing, G.; Sun, T. Sialic Acid-Targeted Biointerface Materials and Bio-Applications. Polymers 2017, 9, 249. https://doi.org/10.3390/polym9070249
Xiong Y, Li M, Lu Q, Qing G, Sun T. Sialic Acid-Targeted Biointerface Materials and Bio-Applications. Polymers. 2017; 9(7):249. https://doi.org/10.3390/polym9070249
Chicago/Turabian StyleXiong, Yuting, Minmin Li, Qi Lu, Guangyan Qing, and Taolei Sun. 2017. "Sialic Acid-Targeted Biointerface Materials and Bio-Applications" Polymers 9, no. 7: 249. https://doi.org/10.3390/polym9070249