Upconversion Nanophosphor-Involved Molecularly Imprinted Fluorescent Polymers for Sensitive and Specific Recognition of Sterigmatocystin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Materials
2.2. Instrumentation
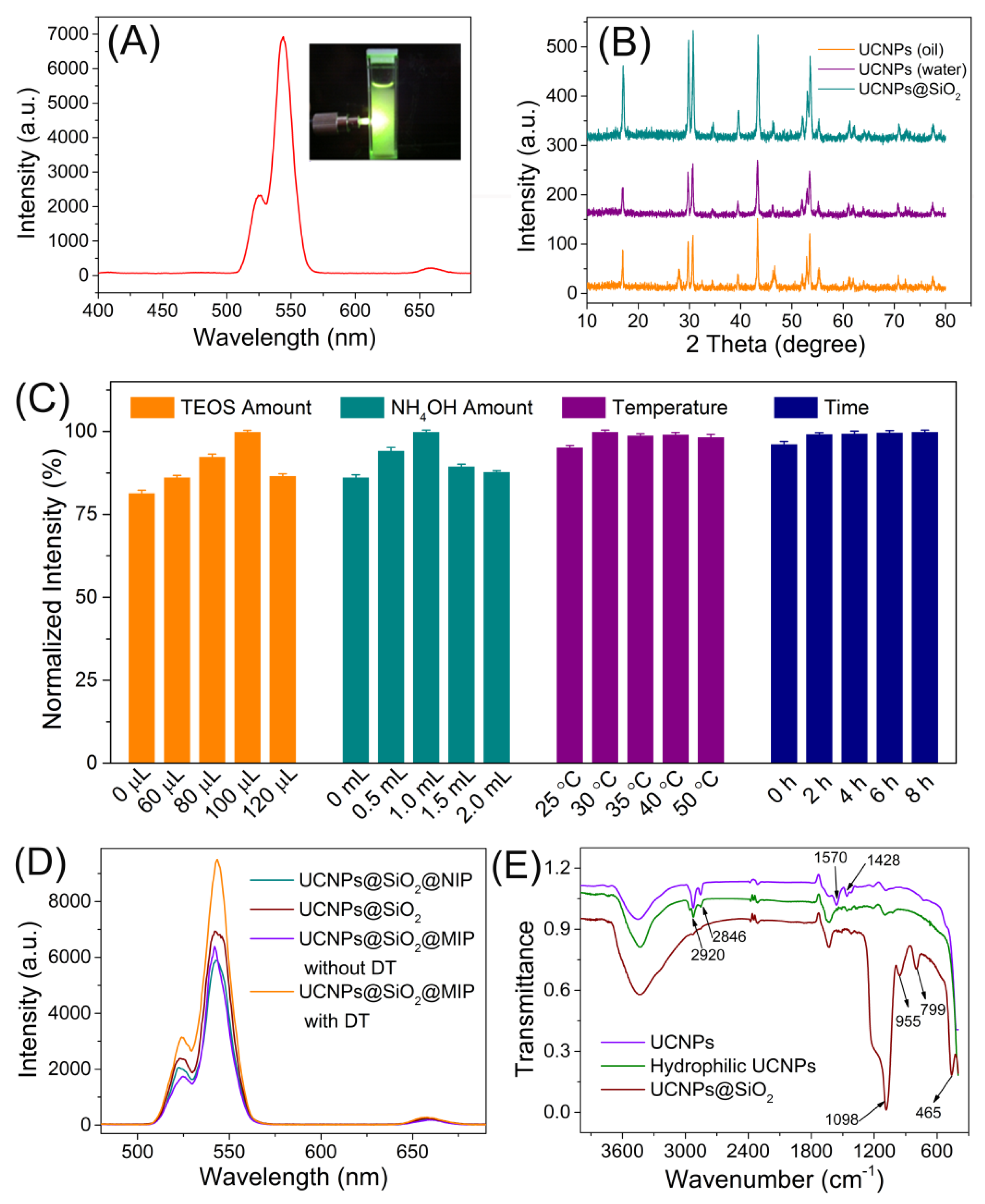
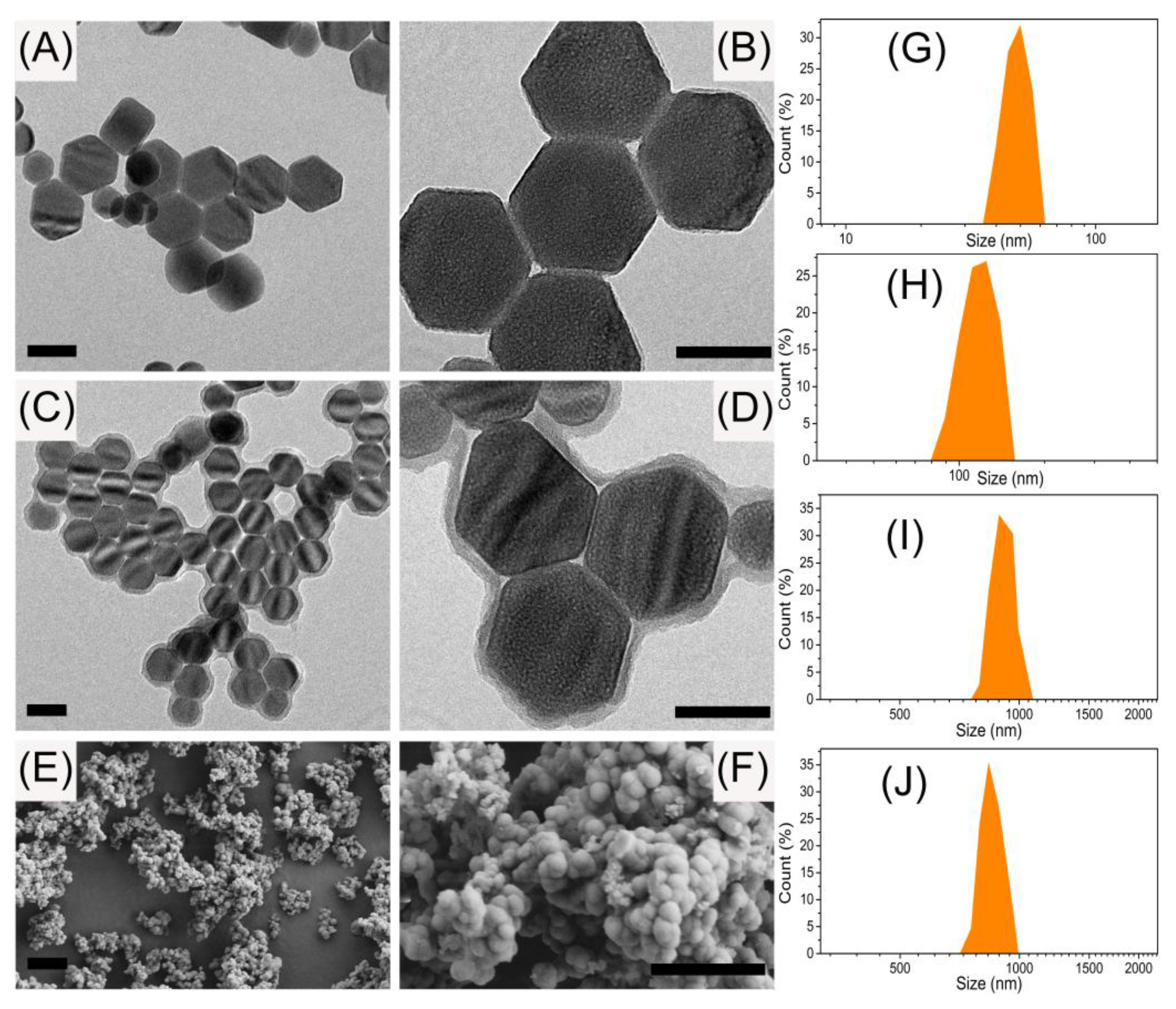
2.3. Preparation of UCNPs
2.4. Preparation of UCNPs@SiO2
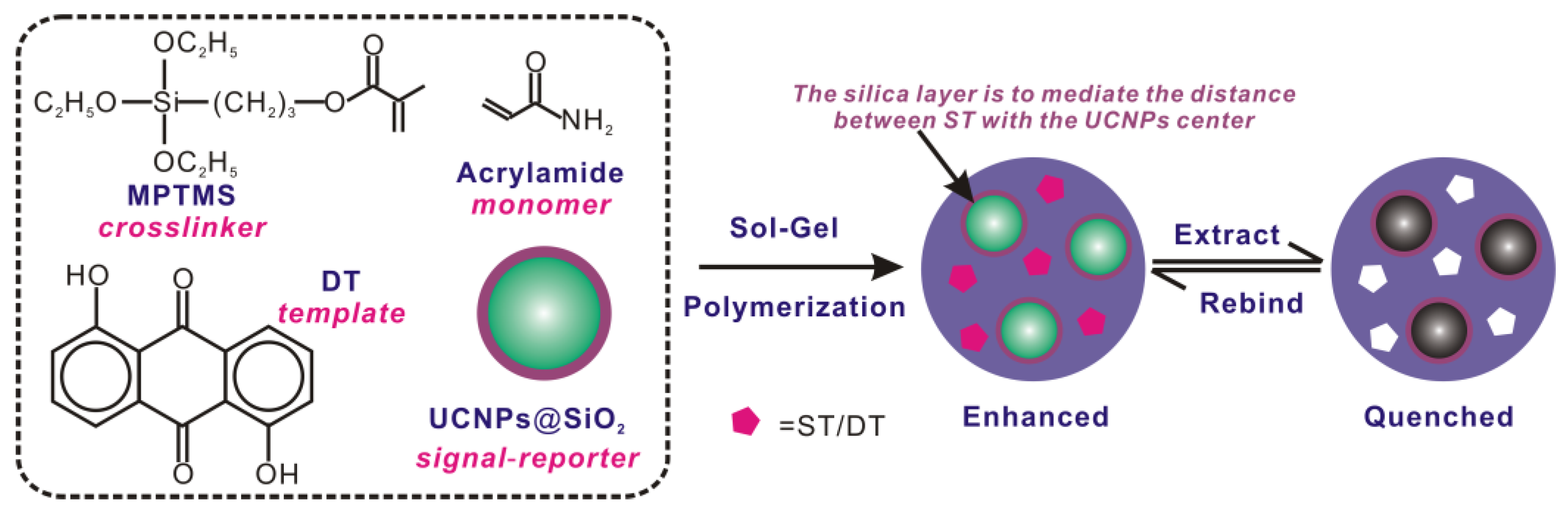
2.5. Preparation of UCNPs@SiO2 MIPs
2.6. Imprinting Procedures
3. Results and Discussion
3.1. Preparation and Characterization of the UCNPs@SiO2@MIP Materials
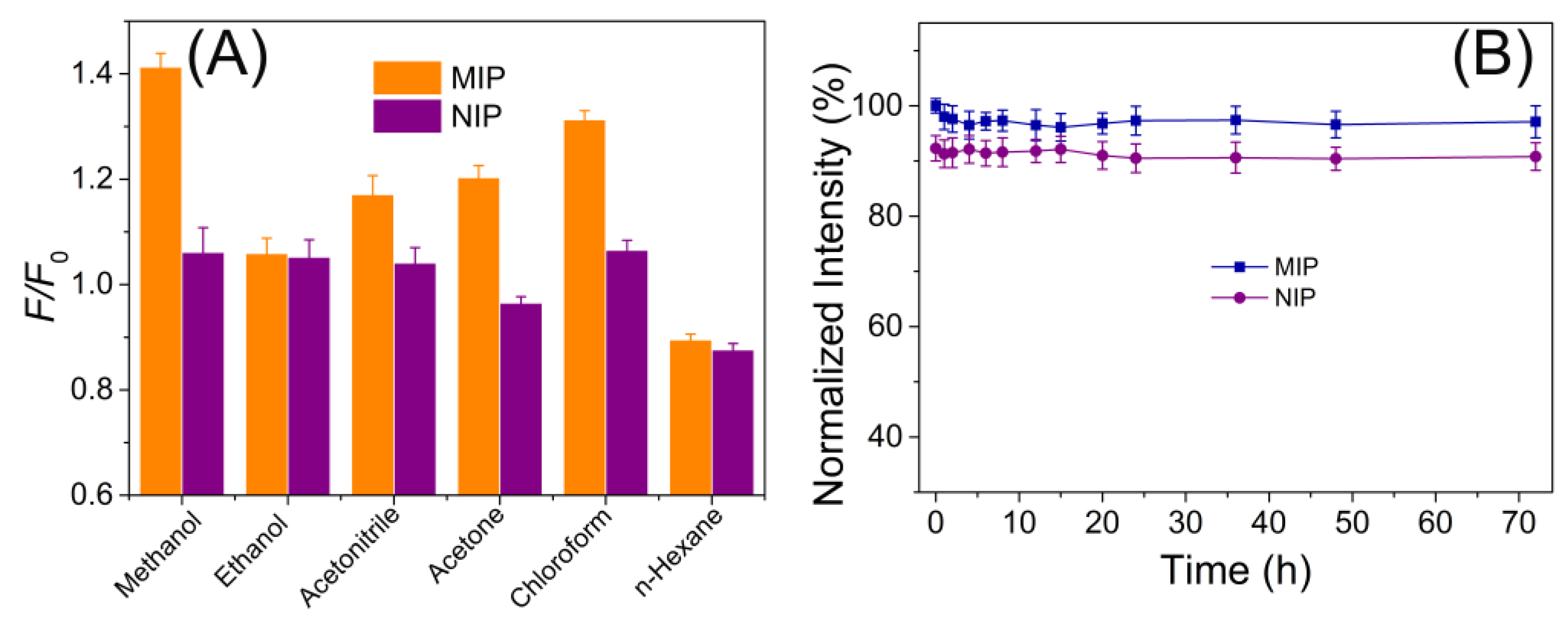
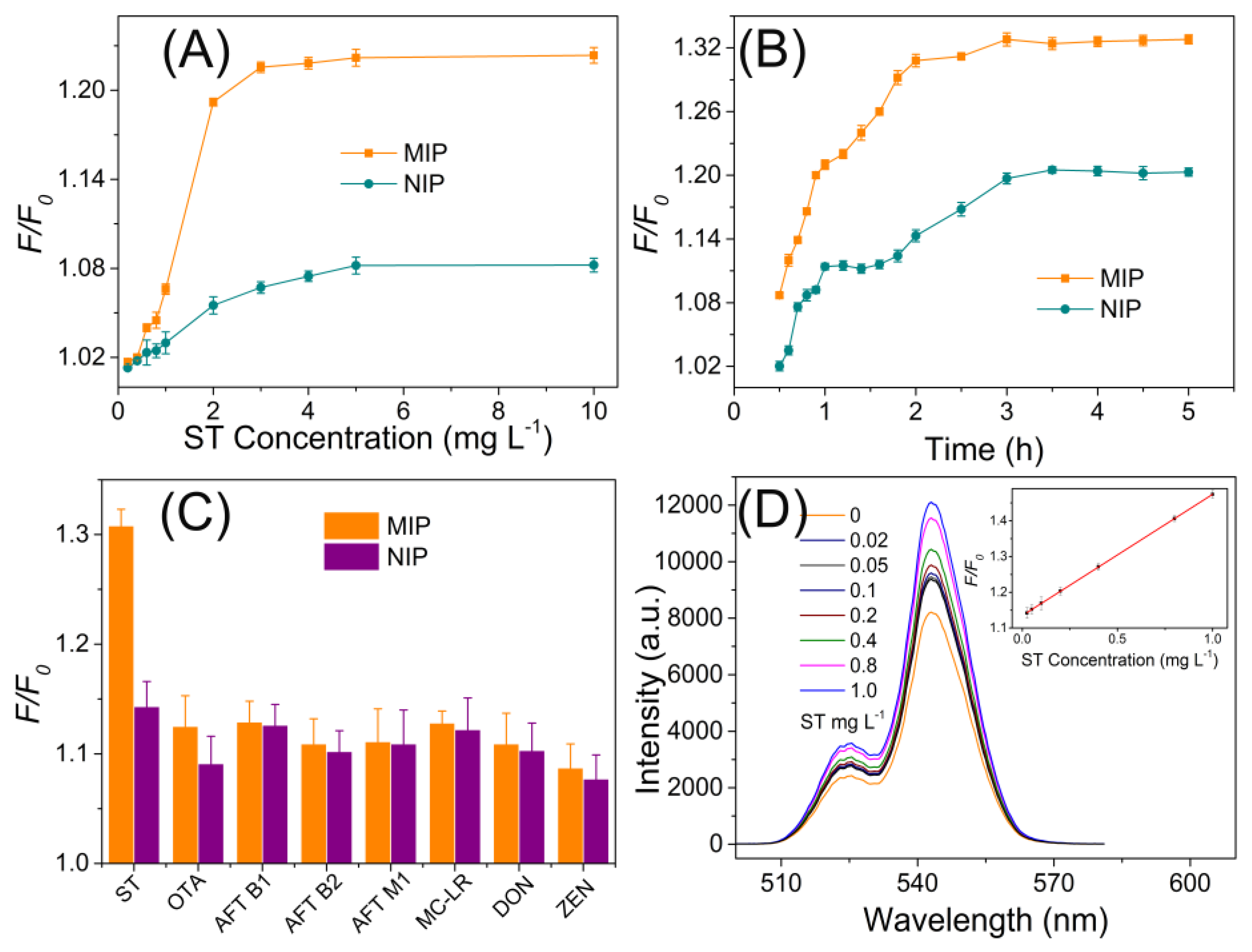
3.2. MIPs Recognition of ST
3.3. MIPs Performance for ST
3.4. Real Sample Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, H.; Yi, J.H.; Velado, D.; Yu, Y.Y.; Zhou, S.Q. Immobilization of Carbon Dots in Molecularly Imprinted Microgels for Optical Sensing of Glucose at Physiological pH. ACS Appl. Mater. Interfaces 2015, 7, 15735–15745. [Google Scholar] [CrossRef] [PubMed]
- Zor, E.; Morales-Narvaez, E.; Zamora-Galvez, A.; Bingol, H.; Ersoz, M.; Merkoci, A. Graphene quantum dots-based photoluminescent sensor: A multifunctional composite for pesticide detection. ACS Appl. Mater. Interfaces 2015, 7, 20272–20279. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.Y.; Liu, H.J.; Li, M.; Xing, Y.; Ren, X.Q. Surface molecular imprinting on CdTe quantum dots for fluorescence sensing of 4-nitrophenol. Anal. Methods 2016, 8, 2226–2232. [Google Scholar] [CrossRef]
- Panagiotopoulou, M.; Salinas, Y.; Beyazit, S.; Kunath, S.; Duma, L.; Prost, E.; Mayes, A.G.; Resmini, M.; Bui, B.T.S.; Haupt, K. Molecularly imprinted polymer coated quantum dots for multiplexed cell targeting and imaging. Angew. Chem. Int. Ed. 2016, 55, 8244–8248. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.X.; Liu, L.; He, Y.H.; He, Q.H.; Ma, H. Quantum-dots-encoded-microbeads based molecularly imprinted polymer. Biosens. Bioelectron. 2016, 77, 886–893. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.H.; Chen, L.G. Quantum dots coated with molecularly imprinted polymer as fluorescence probe for detection of cyphenothrin. Biosens. Bioelectron. 2015, 64, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Uzun, L.; Turner, A.P.F. Molecularly-imprinted polymer sensors: Realising their potential. Biosens. Bioelectron. 2016, 76, 131–144. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Y.; Yu, J.L.; Kang, Q.; Shen, D.Z.; Li, J.H.; Chen, L.X. Molecular imprinting ratiometric fluorescence sensor for highly selective and sensitive detection of phycocyanin. Biosens. Bioelectron. 2016, 77, 624–630. [Google Scholar] [CrossRef] [PubMed]
- Asman, S.; Mohamad, S.; Sarih, N.M. Effects of RAFT agent on the selective approach of molecularly imprinted polymers. Polymers 2015, 7, 484–503. [Google Scholar] [CrossRef]
- Huang, L.; Li, M.; Wu, D.; Ma, X.; Wu, Z.; Xiang, S.; Chen, S. Molecularly imprinted nanofiber film for sensitive sensing 2,4,6-tribromophenol. Polymers 2016, 8, 222. [Google Scholar] [CrossRef]
- Yusof, N.A.; Ab Rahman, S.K.; Hussein, M.Z.; Ibrahim, N.A. Preparation and characterization of molecularly imprinted polymer as SPE sorbent for melamine isolation. Polymers 2013, 5, 1215–1228. [Google Scholar] [CrossRef]
- Liu, H.L.; Wu, D.; Zhou, K.W.; Wang, J.; Sun, B.G. Development and applications of molecularly imprinted polymers based on hydrophobic CdSe/ZnS quantum dots for optosensing of N-epsilon-carboxymethyllysine in foods. Food Chem. 2016, 211, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.L.; Chen, X.M.; Mu, L.; Wang, J.; Sun, B.G. Application of quantum dot-molecularly imprinted polymer core-shell particles sensitized with graphene for optosensing of n-epsilon-carboxymethyllysine in dairy products. J. Agric. Food Chem. 2016, 64, 4801–4806. [Google Scholar] [CrossRef] [PubMed]
- Hao, T.F.; Wei, X.; Nie, Y.J.; Xu, Y.Q.; Yan, Y.S.; Zhou, Z.P. An eco-friendly molecularly imprinted fluorescence composite material based on carbon dots for fluorescent detection of 4-nitrophenol. Microchim. Acta 2016, 183, 2197–2203. [Google Scholar] [CrossRef]
- Guo, T.; Deng, Q.L.; Fang, G.Z.; Liu, C.C.; Huang, X.; Wang, S. Molecularly imprinted upconversion nanoparticles for highly selective and sensitive sensing of Cytochrome c. Biosens. Bioelectron. 2015, 74, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.J.; Pan, M.F.; Fang, G.Z.; He, X.L.; Yang, Y.K.; Dai, J.; Wang, S. Molecularly imprinted quartz crystal microbalance sensor based on poly(o-aminothiophenol) membrane and Au nanoparticles for ractopamine determination. Biosens. Bioelectron. 2014, 51, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.L.; Fang, G.Z.; Wang, S. Molecularly imprinted optosensing material based on hydrophobic CdSe quantum dots via a reverse microemulsion for specific recognition of ractopamine. Biosens. Bioelectron. 2014, 55, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.K.; Fang, G.Z.; Wang, X.M.; Liu, G.Y.; Wang, S. Imprinting of molecular recognition sites combined with pi-donor-acceptor interactions using bis-aniline-crosslinked Au-CdSe/ZnS nanoparticles array on electrodes: Development of electrochemiluminescence sensor for the ultrasensitive and selective detection of 2-methyl-4-chlorophenoxyacetic acid. Biosens. Bioelectron. 2016, 77, 1134–1143. [Google Scholar] [PubMed]
- Qian, K.; Fang, G.Z.; Wang, S. Highly sensitive and selective novel core-shell molecularly imprinted polymer based on NaYF4: Yb3+, Er3+ upconversion fluorescent nanorods. RSC Adv. 2013, 3, 3825–3828. [Google Scholar] [CrossRef]
- Liu, H.L.; Fang, G.Z.; Li, C.M.; Pan, M.F.; Liu, C.C.; Fan, C.; Wang, S. Molecularly imprinted polymer on ionic liquid-modified CdSe/ZnS quantum dots for the highly selective and sensitive optosensing of tocopherol. J. Mater. Chem. 2012, 22, 19882–19887. [Google Scholar] [CrossRef]
- Liu, H.L.; Liu, D.R.; Fang, G.Z.; Liu, F.F.; Liu, C.C.; Yang, Y.K.; Wang, S.O. A novel dual-function molecularly imprinted polymer on CdTe/ZnS quantum dots for highly selective and sensitive determination of ractopamine. Anal. Chim. Acta 2013, 762, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Bouzigues, C.; Gacoin, T.; Alexandrou, A. Biological applications of rare-earth based nanoparticles. ACS Nano 2011, 5, 8488–8505. [Google Scholar] [CrossRef] [PubMed]
- Haase, M.; Schäfer, H. Upconverting nanoparticles. Angew. Chem. Int. Ed. 2011, 50, 5808–5829. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Liu, Z.; Li, F.Y. Upconversion nanophosphors for small-animal imaging. Chem. Soc. Rev. 2012, 41, 1323–1349. [Google Scholar] [CrossRef] [PubMed]
- Gnach, A.; Bednarkiewicz, A. Lanthanide-doped up-converting nanoparticles: Merits and challenges. Nano Today 2012, 7, 532–563. [Google Scholar] [CrossRef]
- Chatterjee, D.K.; Gnanasammandhan, M.K.; Zhang, Y. Small upconverting fluorescent nanoparticles for biomedical applications. Small 2010, 6, 2781–2795. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Tong, Y. Lanthanide ion-based luminescent nanomaterials for bioimaging. Trac-Trends Anal. Chem. 2012, 39, 60–71. [Google Scholar] [CrossRef]
- Wang, Y.; Shen, P.; Li, C.; Wang, Y.; Liu, Z. Upconversion fluorescence resonance energy transfer based biosensor for ultrasensitive detection of matrix metalloproteinase-2 in blood. Anal. Chem. 2012, 84, 1466–1473. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Duan, N.; Ma, X.; Xia, Y.; Wang, H.; Wang, Z.; Zhang, Q. Multiplexed fluorescence resonance energy transfer aptasensor between upconversion nanoparticles and graphene oxide for the simultaneous determination of mycotoxins. Anal. Chem. 2012, 84, 6263–6270. [Google Scholar] [CrossRef] [PubMed]
- Song, K.; Kong, X.; Liu, X.; Zhang, Y.; Zeng, Q.; Tu, L.; Shi, Z.; Zhang, H. Aptamer optical biosensor without bio-breakage using upconversion nanoparticles as donors. Chem. Commun. 2012, 48, 1156–1158. [Google Scholar] [CrossRef] [PubMed]
- Tu, D.; Liu, L.; Ju, Q.; Liu, Y.; Zhu, H.; Li, R.; Chen, X. Time-resolved FRET biosensor based on amine-functionalized lanthanide-doped NaYF4 nanocrystals. Angew. Chem. Int. Ed. 2011, 50, 6306–6310. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.L.; Yuan, Y.X.; Zhang, S.M.; Wang, Y.H.; Liu, Z.H. Biosensing platform based on fluorescence resonance energy transfer from upconverting nanocrystals to graphene oxide. Angew. Chem. Int. Ed. 2011, 50, 6851–6854. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Xu, L.; Ma, W.; Wu, X.; Sun, M.; Kuang, H.; Wang, L.; Kotov, N.A.; Xu, C. Dual-mode ultrasensitive quantification of microrna in living cells by chiroplasmonic nanopyramids self-assembled from gold and upconversion nanoparticles. J. Am. Chem. Soc. 2016, 138, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Kang, N.; Lv, J.; Zhou, Z.; Zhao, Q.; Ma, L.; Chen, Z.; Ren, L.; Nie, L. Deep photoacoustic/luminescence/magnetic resonance multimodal imaging in living subjects using high-efficiency upconversion nanocomposites. Adv. Mater. 2016, 28, 6411–6419. [Google Scholar] [CrossRef] [PubMed]
- Tian, G.; Gu, Z.; Zhou, L.; Yin, W.; Liu, X.; Yan, L.; Jin, S.; Ren, W.; Xing, G.; Li, S.; et al. Mn2+ dopant-controlled synthesis of NaYF4:Yb/Er upconversion nanoparticles for in vivo imaging and drug delivery. Adv. Mater. 2012, 24, 1226–1231. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Wang, J.; Xing, L.; Shen, H.; Lian, W.; Yi, L.; Zhang, D.; Yang, H.; Liu, J.; Zhang, X. Sterigmatocystin-induced checkpoint adaptation depends on Chk1 in immortalized human gastric epithelial cells in vitro. Arch. Toxicol. 2017, 91, 259–270. [Google Scholar] [CrossRef] [PubMed]
- Krol, E.S. Metabolic detoxication pathways for sterigmatocystin in primary tracheal epithelial cells: Structural identification of glutathione adducts. Chem. Res. Toxicol. 2011, 24, 1339–1340. [Google Scholar] [CrossRef] [PubMed]
- Wegner, S.; Bauer, J.I.; Dietrich, R.; Maertlbauer, E.; Usleber, E.; Gottschalk, C.; Gross, M. A highly specific competitive direct enzyme immunoassay for sterigmatocystin as a tool for rapid immunochemotaxonomic differentiation of mycotoxigenic Aspergillus species. Lett. Appl. Microbiol. 2017, 64, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Li, P.; Wu, H.; Zhang, Q.; Ma, F.; Zhang, Z.; Ding, X.; Wang, H. An ultra-sensitive monoclonal antibody-based competitive enzyme immunoassay for sterigmatocystin in cereal and oil products. PLoS ONE 2014, 9, e106415. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Jiang, L.; Ge, L.; Chen, M.; Geng, C.; Yang, G.; Li, Q.; Ji, F.; Yan, Q.; Zou, Y.; et al. Sterigmatocystin-induced oxidative DNA damage in human liver-derived cell line through lysosomal damage. Toxicol. In Vitro 2015, 29, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, Z.; Molnar, A.P.; Fejes, B.; Novak, L.; Karaffa, L.; Keller, N.P.; Fekete, E. Growth-phase sterigmatocystin formation on lactose is mediated via low specific growth rates in aspergillus nidulans. Toxins 2016, 8, 354. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.H.; Fang, G.Z.; Pan, M.F.; Wang, X.F.; Wang, S. One-pot synthesis of carbon dots-embedded molecularly imprinted polymer for specific recognition of sterigmatocystin in grains. Biosens. Bioelectron. 2016, 77, 950–956. [Google Scholar] [CrossRef] [PubMed]
- Zheng, R.; Xu, H.; Wang, W.; Zhan, R.; Chen, W. Simultaneous determination of aflatoxin B-1, B-2, G(1), G(2), ochratoxin A, and sterigmatocystin in traditional Chinese medicines by LC-MS-MS. Anal. Bioanal. Chem. 2014, 406, 3031–3039. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.Z.; Goto, T. Determination of sterigmatocystin in grain using gas chromatography-mass spectrometry with an on-column injector. Mycotoxin Res. 2015, 31, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Ok, H.E.; Tian, F.; Hong, E.Y.; Paek, O.; Kim, S.-H.; Kim, D.; Chun, H.S. Harmonized collaborative validation of aflatoxins and sterigmatocystin in white rice and sorghum by liquid chromatography coupled to tandem mass spectrometry. Toxins 2016, 8, 371. [Google Scholar] [CrossRef] [PubMed]
- Luan, L.; Chen, N.; Han, Z.; Liu, X.; Zheng, Y.; Wu, Y. Simultaneous determination of aflatoxin B-1, aflatoxin B-2, mycophenolic acid and sterigmatocystin in grape pomace by UHPLC-MS/MS. World Mycotoxin J. 2014, 7, 121–129. [Google Scholar] [CrossRef]
- Zhang, F.; Wan, Y.; Yu, T.; Zhang, F.; Shi, Y.; Xie, S.; Li, Y.; Xu, L.; Tu, B.; Zhao, D. Uniform Nanostructured Arrays of Sodium Rare-Earth Fluorides for Highly Efficient Multicolor Upconversion Luminescence. Angew. Chem. Int. Ed. 2007, 46, 7976–7979. [Google Scholar] [CrossRef] [PubMed]
- Stöber, W.; Fink, A.; Bohn, E. Controlled growth of monodisperse silica spheres in the micron size range. J. Colloid Interf. Sci. 1968, 26, 62–69. [Google Scholar] [CrossRef]
- Priyam, A.; Idris, N.M.; Zhang, Y. Gold Nanoshell Coated NaYF4 Nanoparticles for Simultaneously Enhanced Upconversion Fluorescence and Darkfield Imaging. J. Mater. Chem. 2012, 22, 960–965. [Google Scholar] [CrossRef]
- Schietinger, S.; Aichele, T.; Wang, H.Q.; Nann, T.; Benson, O. Plasmon-Enhanced Upconversion in Single NaYF4:Yb3+/Er3+ Codoped Nanocrystals. Nano Lett. 2010, 10, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Yuan, P.Y.; Lee, Y.H.; Gnanasammandhan, M.K.; Guan, Z.P.; Zhang, Y.; Xu, Q.H. Plasmon Enhanced Upconversion Luminescence of NaYF4:Yb, Er@SiO2@Ag Core-Shell Nanocomposites for Cell Imaging. Nanoscale 2012, 4, 5132–5137. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Braun, G.B.; Shi, Y.F.; Zhang, Y.C.; Sun, X.H.; Reich, N.O.; Zhao, D.Y.; Stucky, G. Fabrication of Ag@SiO2@Y2O3:Er Nanostructures for Bioimaging: Tuning of the Upconversion Fluorescence with Silver Nanoparticles. J. Am. Chem. Soc. 2010, 132, 2850–2851. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Fang, A.; Li, H.; Zhang, Y.; Yao, S. Enzymatic-induced upconversion photoinduced electron transfer for sensing tyrosine in human serum. Biosens. Bioelectron. 2016, 77, 957–962. [Google Scholar] [CrossRef] [PubMed]
- Gunnlaugsson, T.; Bichell, B.; Nolan, C. A novel fluorescent photoinduced electron transfer (PET) sensor for lithium. Tetrahedron Lett. 2002, 43, 4989–4992. [Google Scholar] [CrossRef]
| Polymer | DT (mmol) | AM (mmol) | MPTMS (mmol) | AIBN (mg) | Imprinting factor | |
|---|---|---|---|---|---|---|
| MIP1/NIP1 | 1.0 | 3.0 | 6.0 | 10.0 | 1.4506 | |
| MIP2/NIP2 | 1.0 | 3.0 | 8.0 | 10.0 | 1.2874 | |
| MIP3/NIP3 | 1.0 | 4.0 | 4.0 | 10.0 | - | |
| MIP4/NIP4 | 1.0 | 4.0 | 6.0 | 10.0 | 3.4876 | |
| MIP5/NIP5 | 1.0 | 4.0 | 8.0 | 10.0 | 3.2404 | |
| MIP6/NIP6 | 1.0 | 5.0 | 4.0 | 10.0 | 2.8362 | |
| MIP7/NIP7 | 1.0 | 5.0 | 6.0 | 10.0 | 2.7018 | |
| MIP8/NIP8 | 1.0 | 5.0 | 8.0 | 10.0 | 1.9183 | |
| Polymer | DT (mmol) | Temperature (°C) | Time (h) | AIBN (mg) | Imprinting factor |
|---|---|---|---|---|---|
| MIP10/NIP10 | 1.0 | 50 | 18 | 10.0 | 1.1238 |
| MIP3/NIP3 | 1.0 | 60 | 18 | 10.0 | 3.4876 |
| MIP15/NIP15 | 1.0 | 60 | 12 | 10.0 | - |
| MIP16/NIP16 | 1.0 | 60 | 24 | 10.0 | - |
| MIP17/NIP17 | 1.0 | 70 | 18 | 10.0 | 1.0782 |
| Method | Detection limit (μg·L−1) | Linear range (μg·L−1) | Reference |
|---|---|---|---|
| ELISA | 0.36 | - | [39] |
| Fluorescence | 19 | 50–2000 | [42] |
| LC-MS | 0.025 | NA | [43] |
| GC-MS | 3 | 10–150 | [44] |
| LC-MS | 3 | - | [45] |
| LC-MS | 3 | - | [46] |
| Fluorescence | 13 | 20–1000 | This work |
| Samples | Spiked (μg·kg−1) | FL intensity | Determined (μg·kg−1) | Recovery (%) | RSD (%) | |
|---|---|---|---|---|---|---|
| Initial | Sampling | |||||
| Rice | 10 | 8120 | 9321 | 8.6 ± 0.2 | 86.0 | 2.3 |
| 20 | 8120 | 9378 | 17.9 ± 0.5 | 89.5 | 2.8 | |
| 50 | 8120 | 9419 | 44.4 ± 0.8 | 88.8 | 1.8 | |
| 100 | 8120 | 9581 | 83.3.2 ± 1.3 | 83.3 | 1.6 | |
| 200 | 8120 | 9987 | 168.0 ± 2.9 | 84.0 | 1.7 | |
| Maize | 10 | 7856 | 9018 | 8.8 ± 0.2 | 88.0 | 2.3 |
| 20 | 7856 | 9087 | 18.2 ± 0.4 | 91.0 | 2.2 | |
| 50 | 7856 | 9127 | 42.8 ± 3.7 | 85.6 | 4.0 | |
| 100 | 7856 | 9301 | 82.1 ± 1.0 | 82.1 | 1.2 | |
| 200 | 7856 | 9741 | 182.6 ± 3.1 | 87.8 | 1.7 | |
| Soybean | 10 | 7980 | 9158 | 8.8 ± 0.3 | 88.0 | 3.4 |
| 20 | 7980 | 9207 | 17.7 ± 0.4 | 88.5 | 2.2 | |
| 50 | 7980 | 9261 | 40.3 ± 2.4 | 80.6 | 5.9 | |
| 100 | 7980 | 9466 | 86.3 ± 1.8 | 86.3 | 2.1 | |
| 200 | 7980 | 9879 | 178.4 ± 2.3 | 89.2 | 1.3 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.-M.; Cao, F.-Z.; Fang, G.-Z.; Wang, S. Upconversion Nanophosphor-Involved Molecularly Imprinted Fluorescent Polymers for Sensitive and Specific Recognition of Sterigmatocystin. Polymers 2017, 9, 299. https://doi.org/10.3390/polym9070299
Liu J-M, Cao F-Z, Fang G-Z, Wang S. Upconversion Nanophosphor-Involved Molecularly Imprinted Fluorescent Polymers for Sensitive and Specific Recognition of Sterigmatocystin. Polymers. 2017; 9(7):299. https://doi.org/10.3390/polym9070299
Chicago/Turabian StyleLiu, Jing-Min, Feng-Zhen Cao, Guo-Zhen Fang, and Shuo Wang. 2017. "Upconversion Nanophosphor-Involved Molecularly Imprinted Fluorescent Polymers for Sensitive and Specific Recognition of Sterigmatocystin" Polymers 9, no. 7: 299. https://doi.org/10.3390/polym9070299





