Summertime Day-Night Differences of PM2.5 Components (Inorganic Ions, OC, EC, WSOC, WSON, HULIS, and PAHs) in Changzhou, China
Abstract
:1. Introduction
2. Experiments
2.1. Sample Collection
2.2. Chemical Analysis
3. Results and Discussions
3.1. Total Concentration and Compositions of PM2.5
3.2. Carbonaceous Species
3.2.1. OC/EC Ratios and Correlation between OC and EC
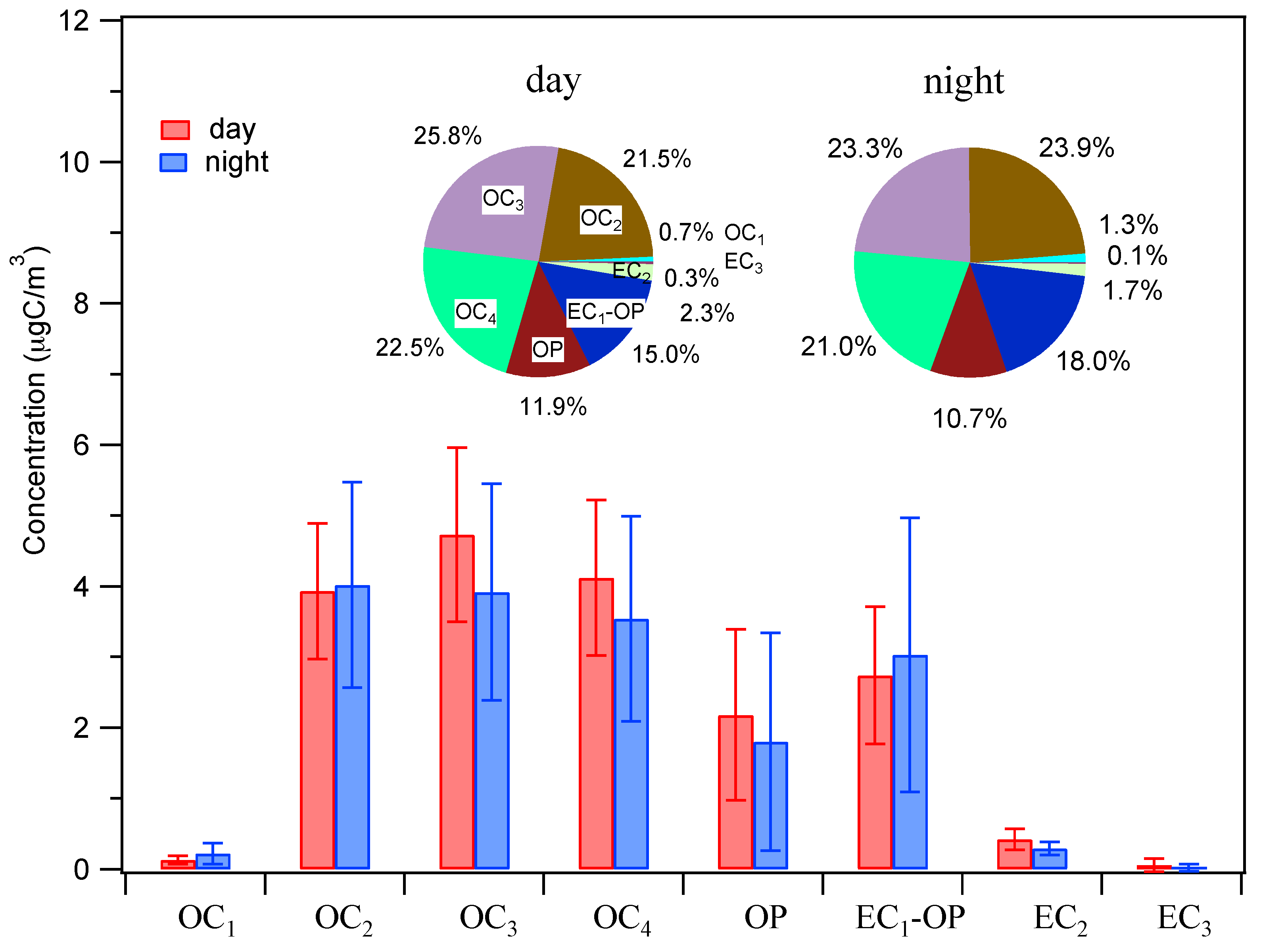
3.2.2. Characteristics of OC and EC
3.2.3. WSOC versus OC and O3
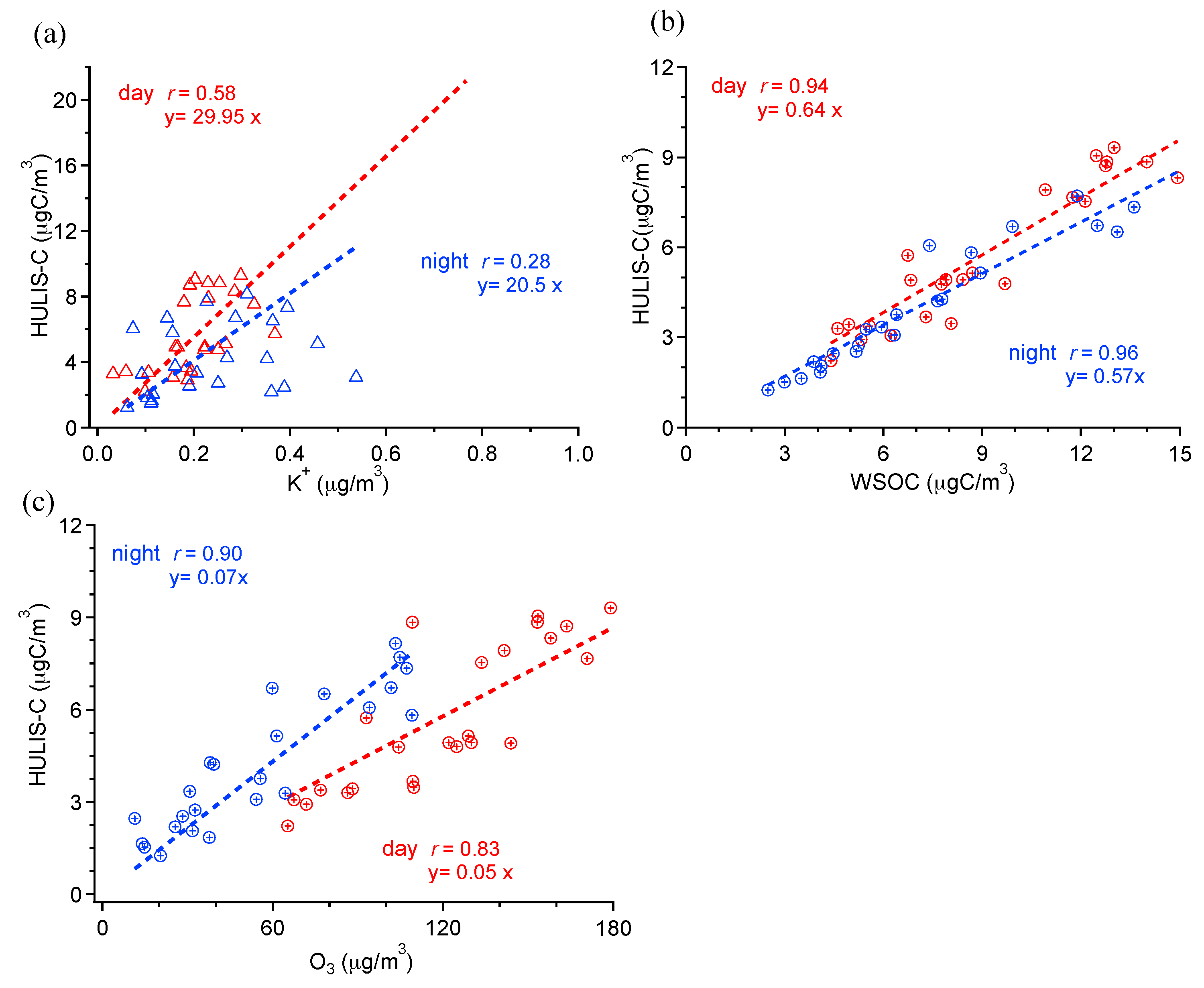
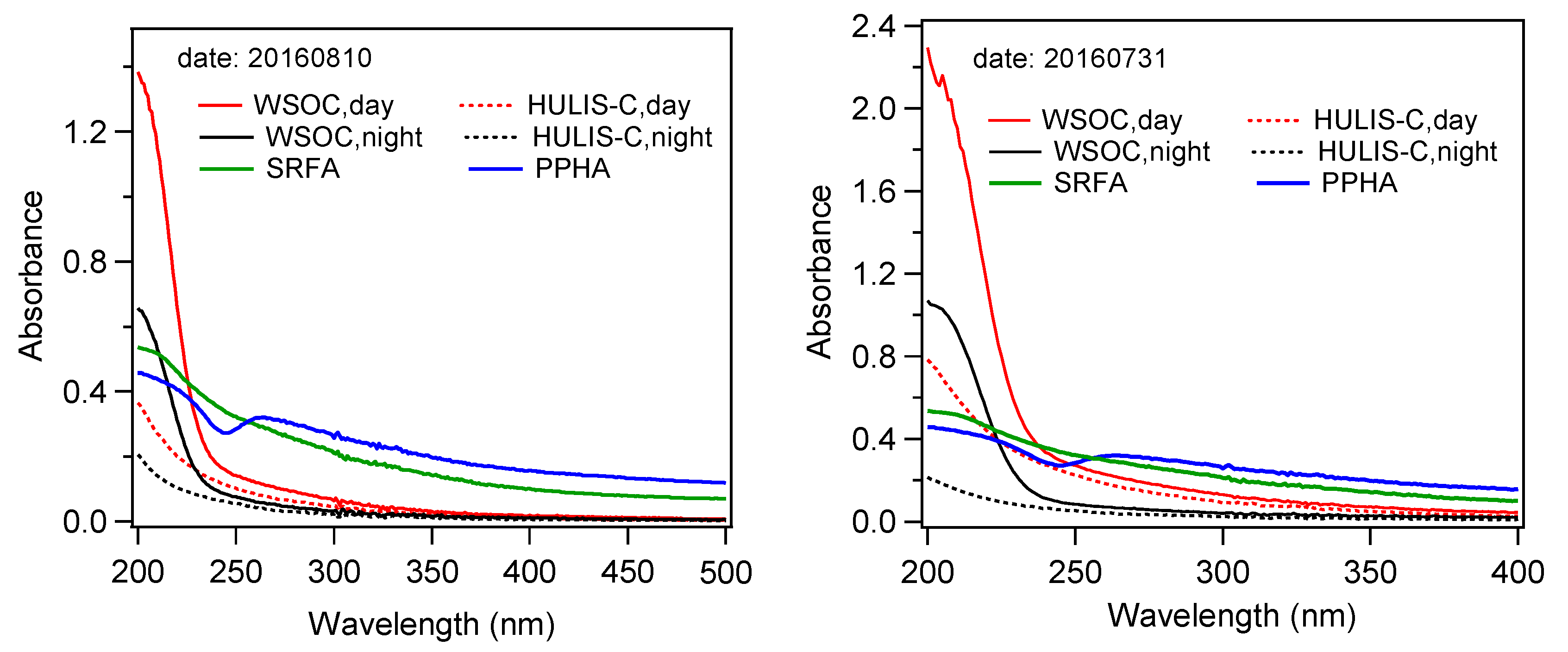
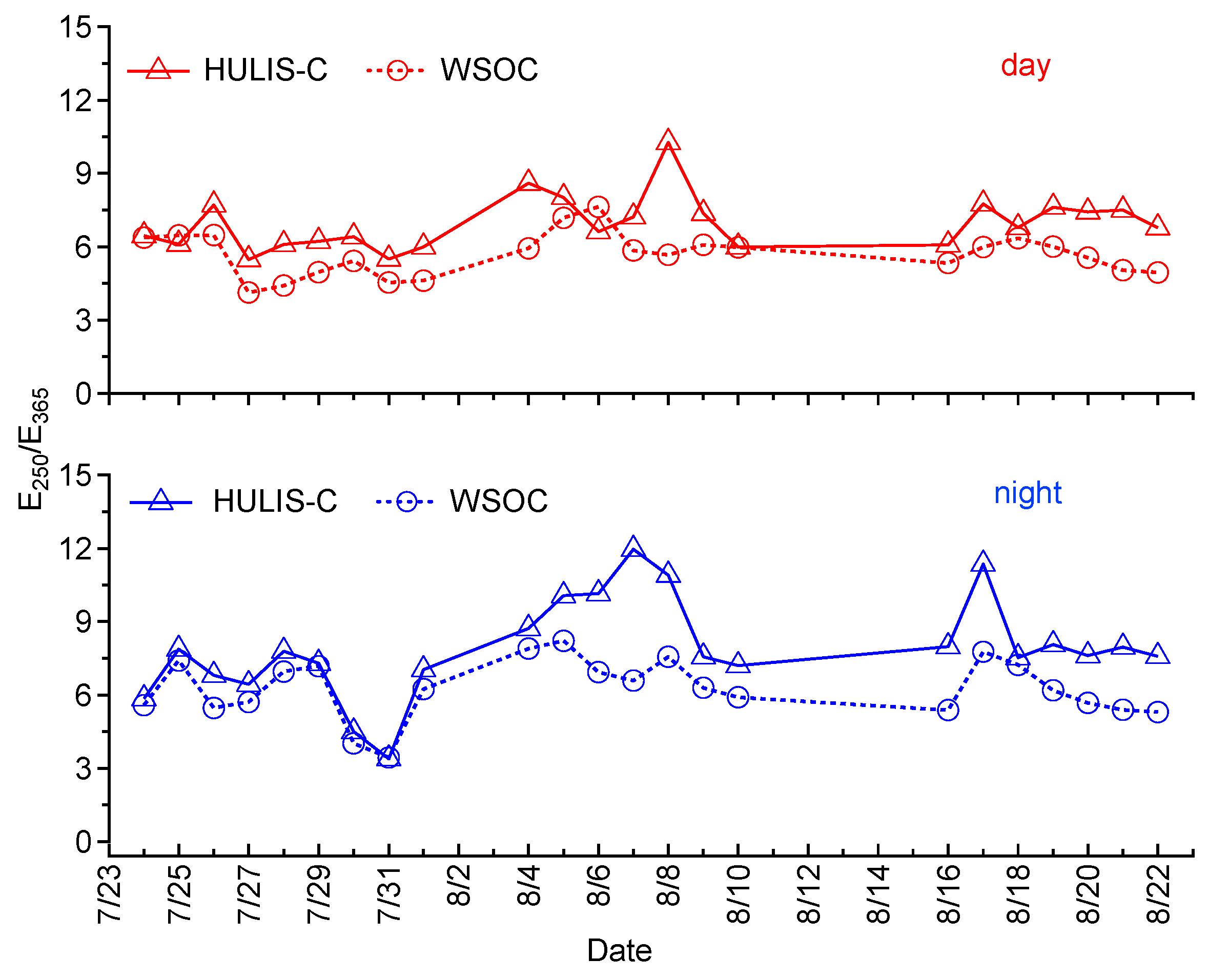
3.3. HULIS-C and Its Relationships with WSOC, K+, and O3
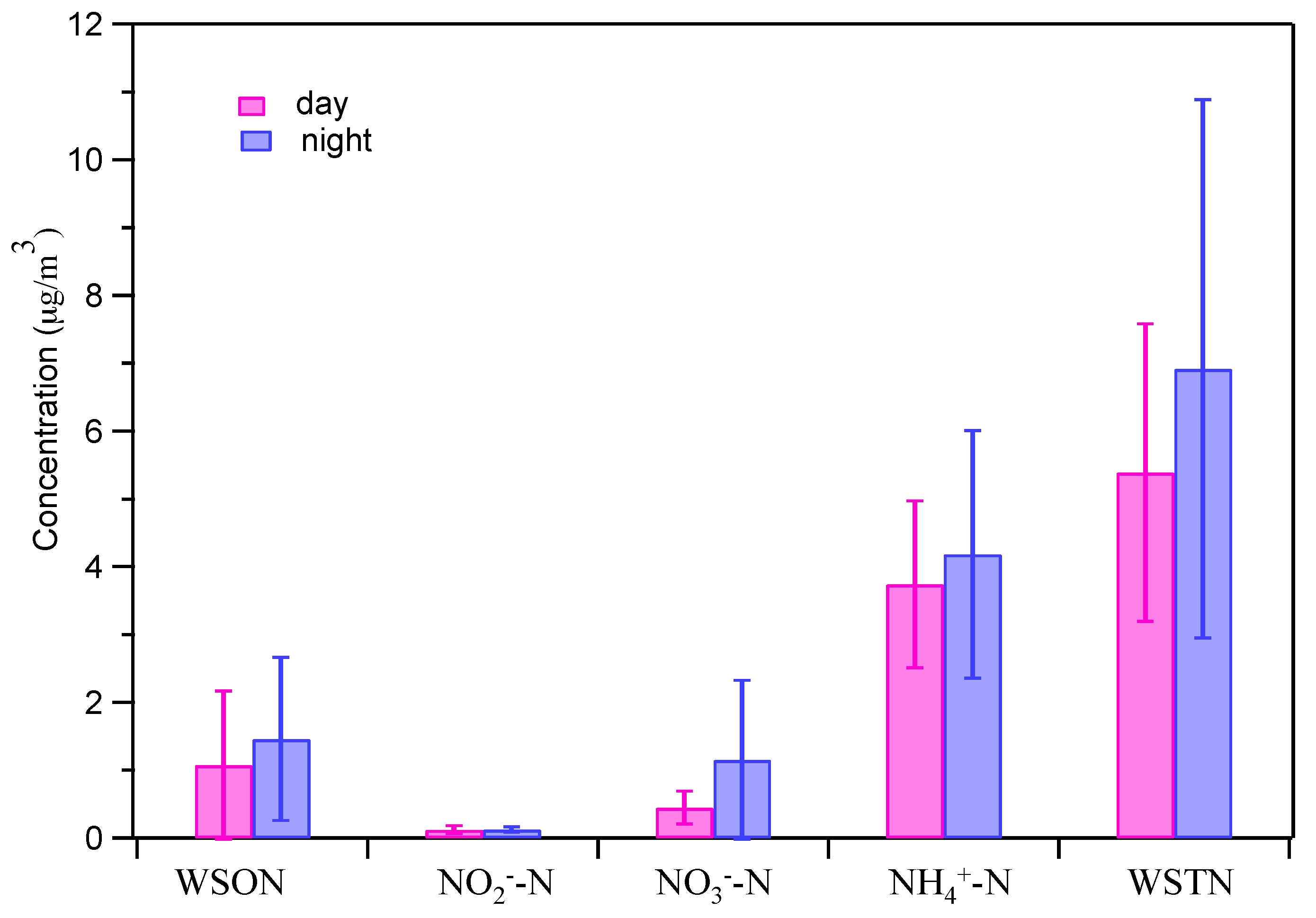
3.4. Determination and Sources of WSON and WSTN
3.5. PAHs Compounds
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sosa, B.S.; Porta, A.; Colman Lerner, J.E.; Banda Noriega, R.; Massolo, L. Human health risk due to variations in PM10–PM2.5 and associated PAHs levels. Atmos. Environ. 2017, 160, 27–35. [Google Scholar] [CrossRef]
- Cao, J.J.; Xu, H.M.; Xu, Q.; Chen, B.H.; Kan, H.D. Fine particulate matter constituents and cardiopulmonary mortality in a heavily polluted chinese city. Environ. Health Perspect. 2012, 120, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Jimenez, J.L.; Canagaratna, M.R.; Allan, J.D.; Coe, H.; Ulbrich, I.; Alfarra, M.R.; Takami, A.; Middlebrook, A.M.; Sun, Y.L. Ubiquity and dominance of oxygenated species in organic aerosols in anthropogenically-influenced northern hemisphere midlatitudes. Geophys. Res. Lett. 2007, 34, L13801. [Google Scholar] [CrossRef]
- Sun, Y.L.; Zhang, Q.; Zheng, M.; Ding, X.; Edgerton, E.S.; Wang, X. Characterization and source apportionment of water-soluble organic matter in atmospheric fine particles (PM2.5) with high-resolution aerosol mass spectrometry and GC–MS. Environ. Sci. Technol. 2011, 45, 4854–4861. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ge, X.; Chen, Y.; Shen, Y.; Zhang, Q.; Sun, Y.; Xu, J.; Ge, S.; Yu, H.; Chen, M. Highly time-resolved urban aerosol characteristics during springtime in Yangtze River Delta, China: Insights from soot particle aerosol mass spectrometry. Atmos. Chem. Phys. 2016, 16, 9109–9127. [Google Scholar] [CrossRef]
- Wang, J.; Onasch, T.B.; Ge, X.; Collier, S.; Zhang, Q.; Sun, Y.; Yu, H.; Chen, M.; Prévôt, A.S.H.; Worsnop, D.R. Observation of fullerene soot in eastern China. Environ. Sci. Technol. Lett. 2016, 3, 121–126. [Google Scholar] [CrossRef]
- Shen, R.; Schäfer, K.; Schnelle-Kreis, J.; Shao, L.; Norra, S.; Kramar, U.; Michalke, B.; Abbaszade, G.; Streibel, T.; Fricker, M.; et al. Characteristics and sources of PM in seasonal perspective—A case study from one year continuously sampling in Beijing. Atmos. Pollut. Res. 2016, 7, 235–248. [Google Scholar] [CrossRef]
- Galindo, N.; Yubero, E. Day-night variability of water-soluble ions in PM10 samples collected at a traffic site in southeastern Spain. Environ. Sci. Pollut. Res. 2017, 24, 805–812. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Shi, H.; Yu, H.; Yang, P. Inversion of nighttime PM2.5 mass concentration in Beijing based on the VIIRS day-night band. Atmosphere 2016, 7, 136. [Google Scholar] [CrossRef]
- Wang, H.; Shooter, D. Coarse-fine and day-night differences of water-soluble ions in Christchurch and Auckland, New Zealand. Atmos. Environ. 2002, 36, 3519–3529. [Google Scholar] [CrossRef]
- Shen, Z.; Cao, J.; Zhang, L.; Liu, L.; Zhang, Q.; Li, J.; Han, Y.; Zhu, C.; Zhao, Z.; Liu, S. Day-night differences and seasonal variations of chemical species in PM10 over Xi’an, Northwest China. Environ. Sci. Pollut. Res. 2014, 21, 3697–3705. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.L.; Wang, S.; Liu, C.; Zhou, B.; Wenig, M.O.; Saiz-Lopez, A. On the summertime air quality and related photochemical processes in the megacity Shanghai, China. Sci. Total Environ. 2017, 580, 974–983. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, T.B.; Du, L.; Nguyen, Q.T.; Nøjgaard, J.K.; Koch, C.B.; Nielsen, O.F.; Hallar, A.G.; Lowenthal, D.H.; Nekat, B.; Pinxteren, D. Chemical properties of HULIS from three different environments. J. Atmos. Chem. 2015, 72, 65–80. [Google Scholar] [CrossRef]
- Zheng, G.; He, K.; Duan, F.; Cheng, Y.; Ma, Y. Measurement of humic-like substances in aerosols: A review. Environ. Pollut. 2013, 181, 301–314. [Google Scholar] [CrossRef] [PubMed]
- Vione, D.; Maurino, V.; Minero, C. Photosensitised humic-like substances (HULIS) formation processes of atmospheric significance: A review. Environ. Sci. Pollut. Res. 2014, 21, 11614–11622. [Google Scholar] [CrossRef] [PubMed]
- Qiao, T.; Zhao, M.; Xiu, G.; Yu, J. Seasonal variations of water soluble composition (WSOC, Hulis and WSIIs) in PM1 and its implications on haze pollution in urban Shanghai, China. Atmos. Environ. 2015, 123, 306–314. [Google Scholar] [CrossRef]
- Graber, R.; Rudich, Y. Atmospheric HULIS: How humic-like are they? A comprehensive and critical review. Atmos. Chem. Phys. 2006, 6, 729–753. [Google Scholar] [CrossRef]
- Hawkins, L.N.; Lemire, A.N.; Galloway, M.M.; Corrigan, A.L.; Turley, J.J.; Espelien, B.M.; De Haan, D.O. Maillard chemistry in clouds and aqueous aerosol as a source of atmospheric humic-like substances. Environ. Sci. Technol. 2016, 50, 7443–7452. [Google Scholar] [CrossRef] [PubMed]
- Baduel, C.; Voisin, D.; Jaffrezo, J.L. Seasonal variations of concentrations and optical properties of water soluble HULIS collected in urban environments. Atmos. Chem. Phys. 2010, 10, 4085–4095. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.Y.; Jung, C.H.; Kim, Y.P. Estimation of optical properties for HULIS aerosols at Anmyeon Island, Korea. Atmosphere 2017, 8, 120. [Google Scholar] [CrossRef]
- Baduel, C.; Voisin, D.; Jaffrezo, J.L. Comparison of analytical methods for humic like substances (HULIS) measurements in atmospheric particles. Atmos. Chem. Phys. 2009, 9, 5949–5962. [Google Scholar] [CrossRef] [Green Version]
- Fan, X.J.; Song, J.Z.; Peng, P.A. Comparison of isolation and quantification methods to measure humic-like substances (HULIS) in atmospheric particles. Atmos. Environ. 2012, 60, 366–374. [Google Scholar] [CrossRef]
- Cape, J.N.; Cornell, S.E.; Jickells, T.D.; Nemitz, E. Organic nitrogen in the atmosphere—Where does it come from? A review of sources and methods. Atmos. Res. 2011, 102, 30–48. [Google Scholar] [CrossRef]
- Ge, X.; Wexler, A.S.; Clegg, S.L. Atmospheric amines—Part I. A review. Atmos. Environ. 2011, 45, 524–546. [Google Scholar] [CrossRef]
- Ge, X.; Wexler, A.S.; Clegg, S.L. Atmospheric amines—Part II. Thermodynamic properties and gas/particle partitioning. Atmos. Environ. 2011, 45, 561–577. [Google Scholar] [CrossRef]
- Rastogi, N.; Zhang, X.L.; Edgerton, E.S.; Ingall, E.; Weber, R.J. Filterable water-soluble organic nitrogen in fine particles over the southeastern USA during summer. Atmos. Environ. 2011, 45, 6040–6047. [Google Scholar] [CrossRef]
- Matsumoto, K.; Yamamoto, Y.; Kobayashi, H.; Kaneyasu, N.; Nakano, T. Water-soluble organic nitrogen in the ambient aerosols and its contribution to the dry deposition of fixed nitrogen species in Japan. Atmos. Environ. 2014, 95, 334–343. [Google Scholar] [CrossRef]
- Claeys, M.; Vermeylen, R.; Yasmeen, F.; Gómez-González, Y.; Chi, X.; Maenhaut, W.; Mészáros, T.; Salma, I. Chemical characterisation of humic-like substances from urban, rural and tropical biomass burning environments using liquid chromatography with UV/vis photodiode array detection and electrospray ionisation mass spectrometry. Environ. Chem. 2012, 9, 273–284. [Google Scholar] [CrossRef] [Green Version]
- Ye, Z.; Liu, J.; Gu, A.; Feng, F.; Liu, Y.; Bi, C.; Xu, J.; Li, L.; Chen, H.; Chen, Y. Chemical characterization of fine particulate matter in Changzhou, China, and source apportionment with offline aerosol mass spectrometry. Atmos. Chem. Phys. 2017, 17, 2573–2592. [Google Scholar] [CrossRef]
- Cheng, Y.; He, K.B.; Duan, F.K.; Zheng, M.; Ma, Y.L.; Tan, J.H. Positive sampling artifact of carbonaceous aerosols and its influence on the thermal-optical split of OC/EC. Atmos. Chem. Phys. 2009, 9, 7243–7256. [Google Scholar] [CrossRef]
- Ye, Z.; Li, Q.; Liu, J.; Luo, S.; Zhou, Q.; Bi, C.; Ma, S.; Chen, Y.; Chen, H.; Li, L. Investigation of submicron aerosol characteristics in Changzhou, China: Composition, source, and comparison with co-collected PM2.5. Chemosphere 2017, 183, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Chow, J.C.; Watson, J.G.; Chenc, L.W.A.; Arnott, W.P.; Moosmüller, H.; Fung, K. Equivalence of elemental carbon by thermal/optical reflectance and transmittance with different temperature protocols. Environ. Sci. Technol. 2004, 38, 4414–4422. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Li, L.; Chen, Y.; Chen, H.; Wu, D.; Wang, J.; Xie, X.; Ge, S.; Ye, Z.; Xu, J. Aerosol characteristics and sources in Yangzhou, China resolved by offline aerosol mass spectrometry and other techniques. Environ. Pollut. 2017, 225, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.; Song, J.; Peng, P. Temporal variations of the abundance and optical properties of water soluble Humic-Like Substances (HULIS) in PM2.5 at Guangzhou, China. Atmos. Res. 2016, 172–173, 8–15. [Google Scholar] [CrossRef]
- Ram, K.; Sarin, M.M. Day–night variability of EC, OC, WSOC and inorganic ions in urban environment of Indo-Gangetic Plain: Implications to secondary aerosol formation. Atmos. Environ. 2011, 45, 460–468. [Google Scholar] [CrossRef]
- Ge, X.; He, Y.; Sun, Y.; Xu, J.; Wang, J.; Shen, Y.; Chen, M. Characteristics and formation mechanisms of fine particulate nitrate in typical urban areas in China. Atmosphere 2017, 8, 62. [Google Scholar] [CrossRef]
- Ohta, S.; Okita, T.A. Chemical characterization of atmospheric aerosol in Sapporo. Atmos. Environ. 1990, 24, 815–822. [Google Scholar] [CrossRef]
- Song, J.Z.; He, L.L.; Peng, P.A.; Zhao, J.P.; Ma, S.X. Chemical and isotopic composition of humic-like substances (HULIS) in ambient aerosols in Guangzhou, South China. Aerosol Sci. Technol. 2012, 46, 533–546. [Google Scholar] [CrossRef]
- Na, K.; Sawant, A.A.; Song, C.; Cocker, D.R., III. Primary and secondary carbonaceous species in the atmosphere of Western Riverside County, California. Atmos. Environ. 2004, 38, 1345–1355. [Google Scholar] [CrossRef]
- Li, B.; Zhang, J.; Zhao, Y.; Yuan, S.; Zhao, Q.; Shen, G.; Wu, H. Seasonal variation of urban carbonaceous aerosols in a typical city Nanjing in Yangtze River Delta, China. Atmos. Environ. 2015, 106, 223–231. [Google Scholar] [CrossRef]
- Diab, J.; Streibel, T.; Cavalli, F.; Lee, S.C.; Saathoff, H.; Mamakos, A.; Chow, J.C.; Chen, L.W.A.; Watson, J.G.; Sippula, O.; et al. Hyphenation of a EC/OC thermal-optical carbon analyzer to photo-ionization time-of-flight mass spectrometry: An off-line aerosol mass spectrometric approach for characterization of primary and secondary particulate matter. Atmos. Meas. Tech. 2015, 8, 3337–3353. [Google Scholar] [CrossRef]
- Xiang, P.; Zhou, X.; Duan, J.; Tan, J.; He, K.; Yuan, C.; Ma, Y.; Zhang, Y. Chemical characteristics of water-soluble organic compounds (WSOC) in PM2.5 in Beijing, China: 2011–2012. Atmos. Res. 2017, 183, 104–112. [Google Scholar] [CrossRef]
- Fry, J.L.; Kiendler-Scharr, A.; Rollins, A.W.; Wooldridge, P.J.; Brown, S.S.; Fuchs, H.; Dubé, W.; Mensah, A.; dal Maso, M.; Tillmann, R.; et al. Organic nitrate and secondary organic aerosol yield from NO3 oxidation of β-pinene evaluated using a gas-phase kinetics/aerosol partitioning model. Atmos. Chem. Phys. 2009, 9, 1431–1449. [Google Scholar] [CrossRef]
- Monks, P.S. Gas-phase radical chemistry in the troposphere. Chem. Soc. Rev. 2005, 34, 376–395. [Google Scholar] [CrossRef] [PubMed]
- Silva, P.J.; Erupe, M.E.; Price, D.; Elias, J.; Malloy, Q.G.J.; Li, Q.; Warren, B.; Cocker, D.R., III. Trimethylamine as precursor to secondary organic aerosol formation via nitrate radical reaction in the atmosphere. Environ. Sci. Technol. 2008, 42, 4689–4696. [Google Scholar] [CrossRef] [PubMed]
- Mayol-Bracero, O.L.; Guyon, P.; Graham, B.; Roberts, G.; Andreae, M.O.; Decesari, S.; Facchini, M.C.; Fuzzi, S.; Artaxo, P. Biomass burning aerosols over Amazonia 2. Apportionment of the chemical composition and importance of the polyacidic fraction. J. Geophys. Res. 2002, 107, 8091. [Google Scholar] [CrossRef]
- Lin, P.; Engling, G.; Yu, J.Z. Humic-like substances in fresh emissions of rice straw burning and in ambient aerosols in the Pearl River Delta Region, China. Atmos. Chem. Phys. 2010, 10, 6487–6500. [Google Scholar] [CrossRef]
- Lin, P.; Huang, X.; He, L.; Yu, J. Abundance and size distribution of HULIS in ambient aerosols at a rural site in South China. J. Aerosol Sci. 2010, 41, 74–87. [Google Scholar] [CrossRef]
- Zhao, M.; Qiao, T.; Li, Y.; Tang, X.; Xiu, G.; Yu, J.Z. Temporal variations and source apportionment of Hulis-C in PM2.5 in urban Shanghai. Sci. Total Environ. 2016, 571, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Kuang, B.Y.; Lin, P.; Huang, X.H.H.; Yu, J.Z. Sources of humic-like substances in the Pearl River Delta, China: Positive matrix factorization analysis of PM2.5 major components and source markers. Atmos. Chem. Phys. 2015, 15, 1995–2008. [Google Scholar] [CrossRef]
- Tan, J.; Xiang, P.; Zhou, X.; Duan, J.; Ma, Y.; He, K.; Cheng, Y.; Yu, J.; Querol, X. Chemical characterization of humic-like substances (HULIS) in PM2.5 in Lanzhou, China. Sci. Total. Environ. 2016, 573, 1481–1490. [Google Scholar] [CrossRef] [PubMed]
- Ofner, J.; Krüger, H.U.; Grothe, H.; Schmitt-Kopplin, P.; Whitmore, K.; Zetzsch, C. Physico-chemical characterization of SOA derived from catechol and guaiacol—A model substance for the aromatic fraction of atmospheric HULIS. Atmos. Chem. Phys. 2011, 11, 1–15. [Google Scholar] [CrossRef]
- Duarte, R.M.B.O.; Pio, C.A.; Duarte, A.C. Spectroscopic study of the water-soluble organic matter isolated from atmospheric aerosols collected under different atmospheric conditions. Anal. Chim. Acta 2005, 530, 7–14. [Google Scholar] [CrossRef]
- Violaki, K.; Mihalopoulos, N. Water-soluble organic nitrogen (WSON) in size-segregated atmospheric particles over the Eastern Mediterranean. Atmos. Environ. 2010, 44, 4339–4345. [Google Scholar] [CrossRef]
- Mace, K.A. Organic nitrogen in rain and aerosol in the Eastern Mediterranean atmosphere: An association with atmospheric dust. J. Geophys. Res. Atmos. 2003, 108. [Google Scholar] [CrossRef]
- De Haan, D.O.; Hawkins, L.N.; Kononenko, J.A.; Turley, J.J.; Corrigan, A.L.; Tolbert, M.A.; Jimenez, J.L. Formation of nitrogen-containing oligomers by methylglyoxal and amines in simulated evaporating cloud droplets. Environ. Sci. Technol. 2011, 45, 984–991. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.K.Y.; Zhao, R.; Li, R.; Liggio, J.; Li, S.-M.; Abbatt, J.P.D. Formation of light absorbing organo-nitrogen species from evaporation of droplets containing glyoxal and ammonium sulfate. Environ. Sci. Technol. 2013, 47, 12819–12826. [Google Scholar] [CrossRef] [PubMed]
- Yue, S.; Ren, H.; Fan, S.; Sun, Y.; Wang, Z.; Fu, P. Springtime precipitation effects on the abundance of fluorescent biological aerosol particles and HULIS in Beijing. Sci. Rep. 2016, 6, 29618. [Google Scholar] [CrossRef] [PubMed]
- Collett, J.L.; Herckes, P.; Youngster, S.; Lee, T. Processing of atmospheric organic matter by California radiation fogs. Atmos. Res. 2008, 87, 232–241. [Google Scholar] [CrossRef]
- Ho, K.F.; Ho, S.S.H.; Huang, R.J.; Liu, S.X.; Cao, J.J.; Zhang, T.; Chuang, H.C.; Chan, C.S.; Hu, D.; Tian, L. Characteristics of water-soluble organic nitrogen in fine particulate matter in the continental area of China. Atmos. Environ. 2015, 106, 252–261. [Google Scholar] [CrossRef]
- Wu, S.; Yang, B.; Wang, X.; Hong, H.; Yuan, C. Diurnal variation of nitrated polycyclic aromatic hydrocarbons in PM10 at a roadside site in Xiamen, China. J. Environ. Sci. 2012, 24, 1767–1776. [Google Scholar] [CrossRef]
- De Pieri, S.; Arruti, A.; Huremovic, J.; Sulejmanovic, J.; Selovic, A.; Ethordevic, D.; Fernandez-Olmo, I.; Gambaro, A. PAHs in the urban air of Sarajevo: Levels, sources, day/night variation, and human inhalation risk. Environ. Monit. Assess. 2014, 186, 1409–1419. [Google Scholar] [CrossRef] [PubMed]
- Ho, K.F.; Ho, S.S.H.; Lee, S.C.; Cheng, Y.; Chow, J.C.; Watson, J.G.; Louie, P.K.K.; Tian, L. Emissions of gas- and particle-phase polycyclic aromatic hydrocarbons (PAHs) in the Shing Mun Tunnel, Hong Kong. Atmos. Environ. 2009, 43, 6343–6351. [Google Scholar] [CrossRef]



| Items | Units | Day | Night | ||
|---|---|---|---|---|---|
| Mean | Range | Mean | Range | ||
| PM2.5 | µg/m3 | 77.3 | 40.9–136.3 | 68.9 | 33.3–119.3 |
| OC | µgC/m3 | 15.1 | 7.2–20.6 | 13.5 | 6.5–25.6 |
| EC | µgC/m3 | 3.2 | 1.3–6.7 | 3.3 | 0.9–8.5 |
| OC/EC | 5.2 | 1.9–8.0 | 4.8 | 1.8–8.0 | |
| (OC + EC)/PM2.5 | % | 23.7 | 15.8–31.1 | 24.4 | 16.3–36.3 |
| WSOC | µgC/m3 | 9.1 | 4.4–14.9 | 7.4 | 2.5–15.6 |
| HULIS-C | µgC/m3 | 5.7 | 2.2–9.3 | 4.2 | 1.3–8.2 |
| WSOC/OC | % | 60.3 | 41.3–84.4 | 54.8 | 33.4–94.8 |
| HULIS-C/WSOC | % | 62.6 | 43.0–85.0 | 58.3 | 45.1–81.9 |
| ΣPAHs | ng/m3 | 2.34 | 1.47–3.94 | 3.64 | 1.73–7.76 |
| Sulfate | µg/m3 | 11.8 | 4.70–18.84 | 10.07 | 0.80–19.20 |
| Nitrate | µg/m3 | 2.0 | 0.82–5.00 | 5.11 | 0.22–21.50 |
| Ammonium | µg/m3 | 4.8 | 1.69–7.47 | 5.37 | 1.66–9.44 |
| WSON | µg/m3 | 1.08 | 0–4.71 | 1.46 | 0.01–4.07 |
| WSTN | µg/m3 | 5.39 | 2.01–10.03 | 6.92 | 1.92–16.39 |
| WSON/WSTN | % | 16.3 | 0–46.9 | 17.8 | 0–31.3 |
| Items | PM2.5 | OC | EC | ΣPAHs | WSOC | HULIS-C | WSON | WSTN | NH4+ | NO3− | SO42− | K+ | O3 | WS |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PM2.5 | 1 | 0.71 | 0.42 | 0.43 | 0.63 | 0.47 | 0.32 | 0.52 | 0.54 | 0.39 | 0.58 | 0.50 | 0.48 | −0.12 |
| OC | 0.81 | 1 | 0.40 | 0.28 | 0.88 | 0.82 | 0.40 | 0.51 | 0.50 | 0.21 | 0.55 | 0.70 | 0.74 | −0.36 |
| EC | 0.62 | 0.53 | 1 | 0.56 | 0.33 | 0.34 | 0.02 | 0.19 | 0.23 | 0.45 | 0.15 | 0.41 | 0.27 | −0.22 |
| ΣPAHs | 0.40 | 0.15 | 0.60 | 1 | 0.20 | 0.11 | 0.27 | 0.38 | 0.34 | 0.57 | 0.27 | 0.53 | 0.10 | −0.11 |
| WSOC | 0.73 | 0.86 | 0.67 | −0.11 | 1 | 0.94 | 0.68 | 0.72 | 0.65 | 0.04 | 0.70 | 0.60 | 0.84 | −0.26 |
| HULIS-C | 0.65 | 0.76 | 0.36 | −0.19 | 0.96 | 1 | 0.66 | 0.64 | 0.55 | −0.08 | 0.59 | 0.58 | 0.83 | −0.27 |
| WSON | 0.93 | 0.73 | 0.41 | 0.19 | 0.69 | 0.61 | 1 | 0.88 | 0.69 | −0.06 | 0.70 | 0.35 | 0.57 | 0.01 |
| WSTN | 0.92 | 0.69 | 0.31 | 0.19 | 0.62 | 0.54 | 0.96 | 1 | 0.94 | 0.23 | 0.91 | 0.44 | 0.63 | −0.37 |
| NH4+ | 0.93 | 0.75 | 0.40 | 0.22 | 0.69 | 0.60 | 0.95 | 0.97 | 1 | 0.26 | 0.96 | 0.23 | 0.64 | −0.13 |
| NO3− | 0.70 | 0.42 | 0.02 | 0.11 | 0.30 | 0.26 | 0.76 | 0.88 | 0.77 | 1 | 0.09 | 0.13 | −0.17 | −0.14 |
| SO42− | 0.71 | 0.76 | 0.46 | −0.19 | 0.85 | 0.76 | 0.69 | 0.69 | 0.75 | 0.46 | 1 | 0.49 | 0.72 | −0.15 |
| K+ | 0.66 | 0.55 | 0.45 | 0.55 | 0.40 | 0.28 | 0.46 | 0.60 | 0.63 | 0.48 | 0.51 | 1 | 0.47 | −0.05 |
| O3 | 0.46 | 0.55 | 0.57 | −0.32 | 0.85 | 0.90 | 0.45 | 0.36 | 0.42 | 0.08 | 0.65 | 0.15 | 1 | −0.34 |
| WS | −0.66 | −0.37 | −0.47 | −0.38 | −0.37 | −0.39 | −0.62 | −0.63 | −0.64 | −0.52 | −0.38 | −0.34 | −0.25 | 1 |
| PAH Compounds | Number of Rings | Mean Concentration (ng/m3) | Night to Day Mass Ratio | |
|---|---|---|---|---|
| Day | Night | |||
| NaP | 2-rings | 0.326 | 0.305 | 0.94 |
| Acy | 0.003 | 0.003 | 1.00 | |
| Ace | 0.012 | 0.013 | 1.08 | |
| Flu | 3-rings | 0.037 | 0.038 | 1.03 |
| Phe | 0.318 | 0.323 | 1.02 | |
| Ant | 0.025 | 0.031 | 1.24 | |
| Flua | 0.198 | 0.213 | 1.08 | |
| Pyr | 0.176 | 0.198 | 1.13 | |
| BaA | 4-rings | 0.079 | 0.117 | 1.48 |
| Chr | 0.198 | 0.260 | 1.31 | |
| BbF + BjF | 0.171 | 0.347 | 2.03 | |
| BkF | 0.095 | 0.188 | 1.98 | |
| BaP | 5-rings | 0.175 | 0.352 | 2.01 |
| BeP | 0.147 | 0.275 | 1.87 | |
| DBA | 0.028 | 0.085 | 3.04 | |
| InP | 6-rings | 0.172 | 0.398 | 2.33 |
| BghiP | 0.183 | 0.496 | 2.71 | |
| LMW-PAHs | 2-3 rings | 0.721 | 0.713 | 0.99 |
| MMW-PAHs | 4-rings | 0.651 | 0.788 | 1.21 |
| HMW-PAHs | 5-6 rings | 0.971 | 2.141 | 2.20 |
| ΣPAHs | 2.343 | 3.642 | 1.55 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, Z.; Li, Q.; Ma, S.; Zhou, Q.; Gu, Y.; Su, Y.; Chen, Y.; Chen, H.; Wang, J.; Ge, X. Summertime Day-Night Differences of PM2.5 Components (Inorganic Ions, OC, EC, WSOC, WSON, HULIS, and PAHs) in Changzhou, China. Atmosphere 2017, 8, 189. https://doi.org/10.3390/atmos8100189
Ye Z, Li Q, Ma S, Zhou Q, Gu Y, Su Y, Chen Y, Chen H, Wang J, Ge X. Summertime Day-Night Differences of PM2.5 Components (Inorganic Ions, OC, EC, WSOC, WSON, HULIS, and PAHs) in Changzhou, China. Atmosphere. 2017; 8(10):189. https://doi.org/10.3390/atmos8100189
Chicago/Turabian StyleYe, Zhaolian, Qing Li, Shuaishuai Ma, Quanfa Zhou, Yuan Gu, Yalan Su, Yanfang Chen, Hui Chen, Junfeng Wang, and Xinlei Ge. 2017. "Summertime Day-Night Differences of PM2.5 Components (Inorganic Ions, OC, EC, WSOC, WSON, HULIS, and PAHs) in Changzhou, China" Atmosphere 8, no. 10: 189. https://doi.org/10.3390/atmos8100189





