Acute Effects of Polyphenols from Cranberries and Grape Seeds on Endothelial Function and Performance in Elite Athletes
Abstract
:1. Introduction
2. Subjects and Methods
2.1. Population
2.2. Nutritional Habits
2.3. Study Designs
2.3.1. Study 1: Polyphenols and FMD in Elite Athletes
2.3.2. Study 2: Polyphenols and Performance
2.4. Statistical Analyses
3. Results
| Variable | Study 1 | Study 2 |
|---|---|---|
| Number of subjects | 8 | 12 |
| Females (N) | 4 | 2 |
| Age (years) | 23.4 ± 7.7 | 22.0 ± 2.4 |
| Weight (kg) | 70.8 ± 6.7 | 75.7 ± 10.4 |
| Height (m) | 1.75 ± 0.08 | 1.79 ± 0.07 |
| BMI (kg/m2) | 23.2 ± 0.8 | 23.6 ± 1.9 |
| Energy (kcal) | 3931 ± 1347 | 3139 ± 735 |
| % from carbohydrates | 57 ± 7 | 55 ± 6 |
| % from protein | 16 ± 2 | 17 ± 3 |
| % from fat | 26 ± 5 | 28 ± 6 |

3.1. Study 1- Polyphenols and FMD
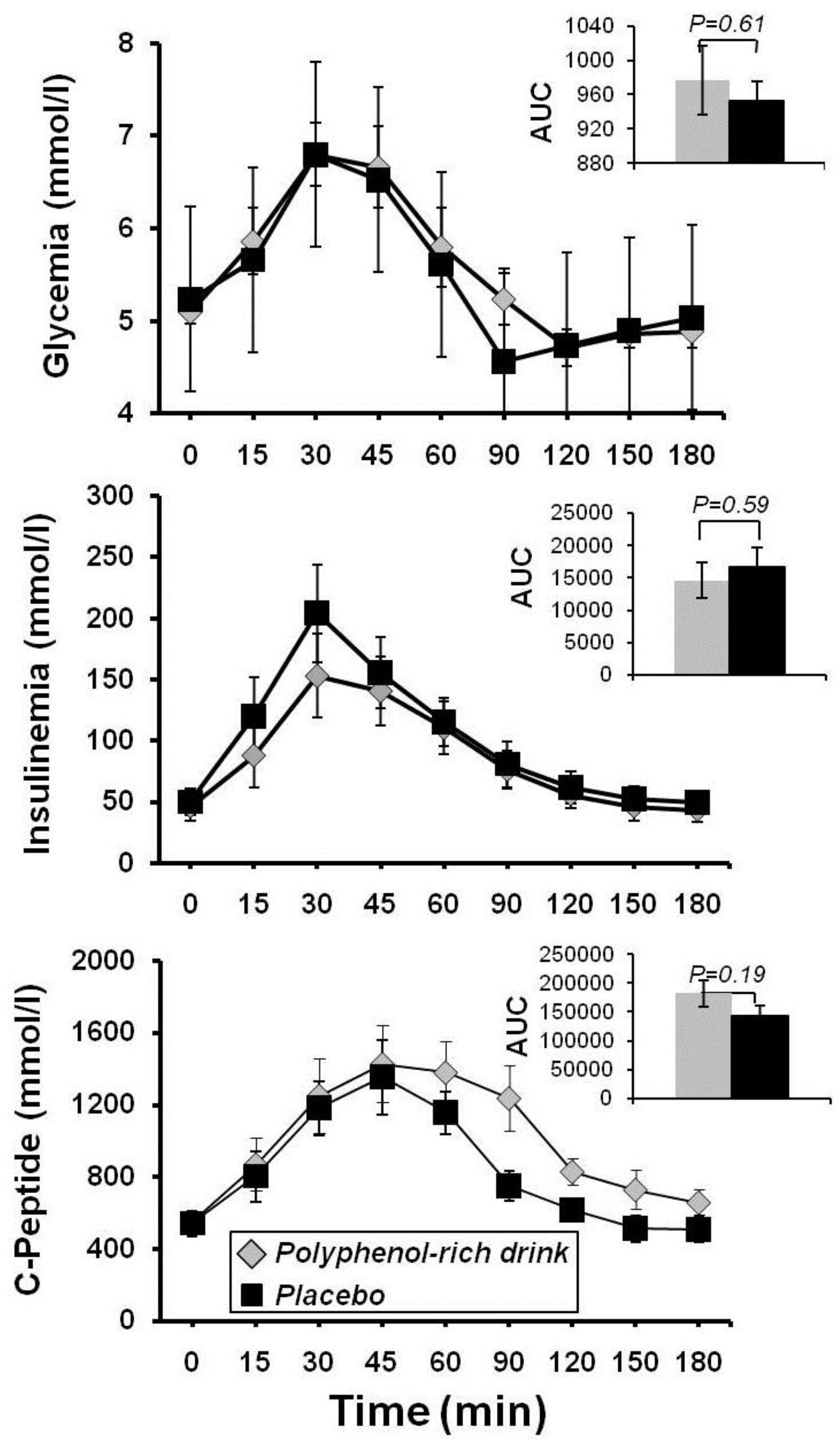
3.2. Study 2- Polyphenols and Performance
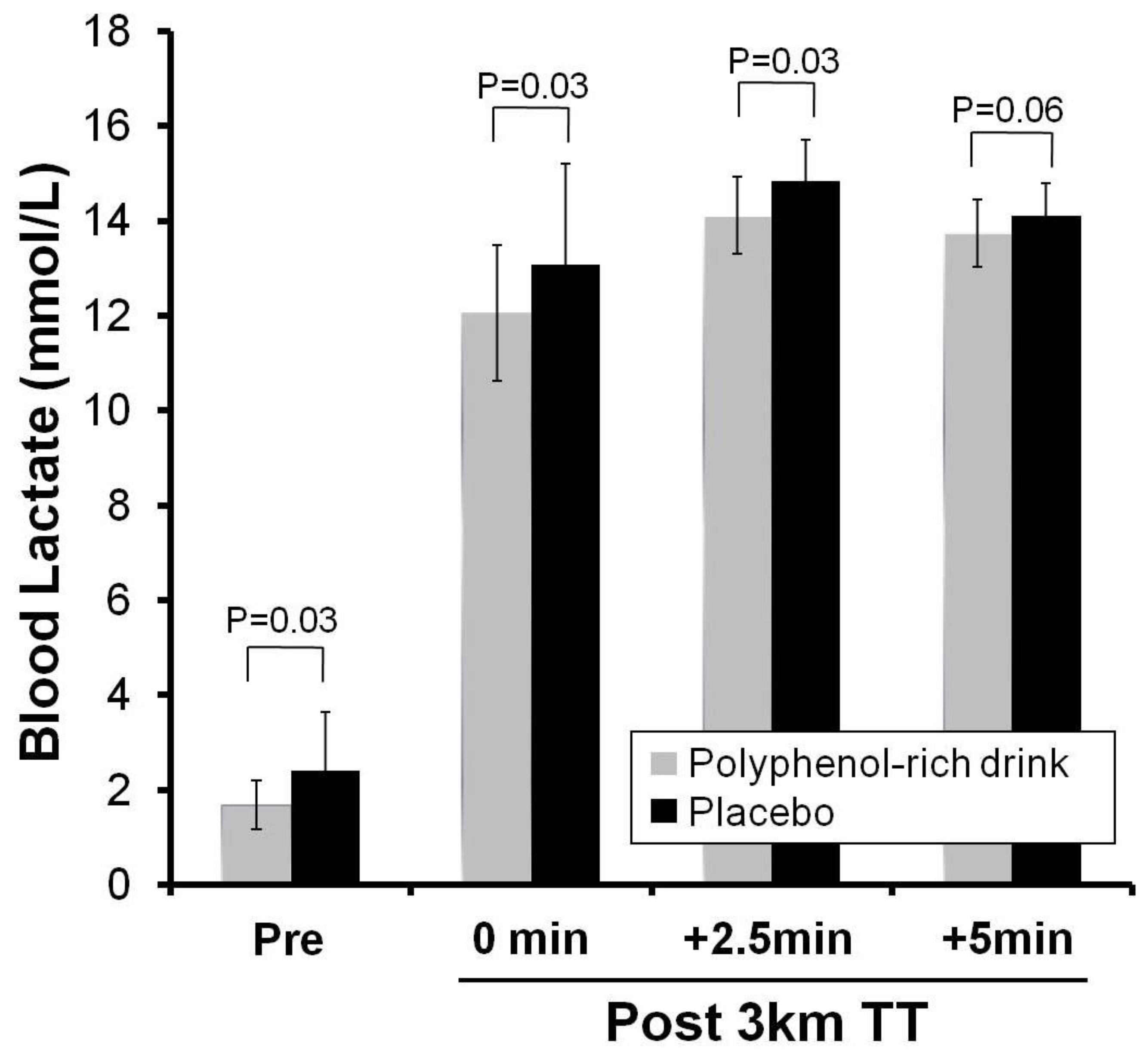
4. Discussion
4.1. Study 1- Polyphenols and FMD
4.2. Study 2- Polyphenols and Performance
5. Conclusion
Acknowledgments
Author’s contribution
Conflict of Interest
References
- Yoshihara, D.; Fujiwara, N.; Suzuki, K. Antioxidants: benefits and risks for long-term health. Maturitas 2010, 67, 103–107. [Google Scholar] [CrossRef]
- Ghosh, D.; Scheepens, A. Vascular action of polyphenols. Mol. Nutr. Food Res. 2009, 53, 322–331. [Google Scholar] [CrossRef]
- Gonzalez, R.; Ballester, I.; Lopez-Posadas, R.; Suarez, M.D.; Zarzuelo, A.; Martinez-Augustin, O.; Sanchez de Medina, F. Effects of flavonoids and other polyphenols on inflammation. Crit. Rev. Food Sci. Nutr. 2011, 51, 331–362. [Google Scholar] [CrossRef]
- Jackson, M.J. Free radicals generated by contracting muscle: By-products of metabolism or key regulators of muscle function? Free Radic. Biol. Med. 2008, 44, 132–141. [Google Scholar] [CrossRef]
- Davison, G.; Gleeson, M.; Phillips, S. Antioxidant supplementation and immunoendocrine responses to prolonged exercise. Med. Sci. Sports Exerc. 2007, 39, 645–652. [Google Scholar] [CrossRef]
- Lawrence, M.E.; Kirby, D.F. Nutrition and sports supplements: Fact or fiction. J. Clin. Gastroenterol. 2002, 35, 299–306. [Google Scholar] [CrossRef]
- Atalay, M.; Lappalainen, J.; Sen, C.K. Dietary antioxidants for the athlete. Curr. Sports Med. Rep. 2006, 5, 182–186. [Google Scholar]
- Powers, S.; Nelson, W.B.; Larson-Meyer, E. Antioxidant and Vitamin D supplements for athletes: sense or nonsense? J. Sports Sci. 2011, 29 Suppl 1, S47–S55. [Google Scholar]
- Bassett, D.R., Jr.; Howley, E.T. Limiting factors for maximum oxygen uptake and determinants of endurance performance. Med. Sci. Sports Exerc. 2000, 32, 70–84. [Google Scholar]
- Foster, C.; Rundell, K.W.; Snyder, A.C.; Stray-Gundersen, J.; Kemkers, G.; Thometz, N.; Broker, J.; Knapp, E. Evidence for restricted muscle blood flow during speed skating. Med. Sci. Sports Exerc. 1999, 31, 1433–1440. [Google Scholar] [CrossRef]
- Campbell, B.I.; La Bounty, P.M.; Roberts, M. The ergogenic potential of arginine. J. Int. Soc. Sports Nutr. 2004, 1, 35–38. [Google Scholar] [CrossRef]
- Goulet, J.; Nadeau, G.; Lapointe, A.; Lamarche, B.; Lemieux, S. Validity and reproducibility of an interviewer-administered food frequency questionnaire for healthy French-Canadian men and women. Nutr. J. 2004, 3, 13. [Google Scholar] [CrossRef] [Green Version]
- Moens, A.L.; Goovaerts, I.; Claeys, M.J.; Vrints, C.J. Flow-mediated vasodilation: A diagnostic instrument, or an experimental tool? Chest 2005, 127, 2254–2263. [Google Scholar] [CrossRef]
- Thijssen, D.H.; Black, M.A.; Pyke, K.E.; Padilla, J.; Atkinson, G.; Harris, R.A.; Parker, B.; Widlansky, M.E.; Tschakovsky, M.E.; Green, D.J. Assessment of flow-mediated dilation in humans: A methodological and physiological guideline. Am. J. Physiol. Heart Circ. Physiol. 2011, 300, H2–H12. [Google Scholar] [CrossRef]
- Corretti, M.C.; Anderson, T.J.; Benjamin, E.J.; Celermajer, D.; Charbonneau, F.; Creager, M.A.; Deanfield, J.; Drexler, H.; Gerhard-Herman, M.; Herrington, D.; et al. Guidelines for the ultrasound assessment of endothelial-dependent flow-mediated vasodilation of the brachial artery: A report of the International Brachial Artery Reactivity Task Force. J. Am. College Cardiol. 2002, 39, 257–265. [Google Scholar]
- Hashimoto, M.; Kim, S.; Eto, M.; Iijima, K.; Ako, J.; Yoshizumi, M.; Akishita, M.; Kondo, K.; Itakura, H.; Hosoda, K.; et al. Effect of acute intake of red wine on flow-mediated vasodilatation of the brachial artery. Am. J. Cardiol. 2001, 88, 1457–1460. [Google Scholar] [CrossRef]
- Agewall, S.; Wright, S.; Doughty, R.N.; Whalley, G.A.; Duxbury, M.; Sharpe, N. Does a glass of red wine improve endothelial function? Eur. Heart J. 2000, 21, 74–78. [Google Scholar] [CrossRef]
- Whelan, A.P.; Sutherland, W.H.; McCormick, M.P.; Yeoman, D.J.; de Jong, S.A.; Williams, M.J. Effects of white and red wine on endothelial function in subjects with coronary artery disease. Internal Med. J. 2004, 34, 224–228. [Google Scholar] [CrossRef]
- Dohadwala, M.M.; Holbrook, M.; Hamburg, N.M.; Shenouda, S.M.; Chung, W.B.; Titas, M.; Kluge, M.A.; Wang, N.; Palmisano, J.; Milbury, P.E.; et al. Effects of cranberry juice consumption on vascular function in patients with coronary artery disease. Am. J. Clin. Nutr. 2011, 93, 934–940. [Google Scholar] [CrossRef]
- Karatzi, K.; Papamichael, C.; Aznaouridis, K.; Karatzis, E.; Lekakis, J.; Matsouka, C.; Boskou, G.; Chiou, A.; Sitara, M.; Feliou, G.; et al. Constituents of red wine other than alcohol improve endothelial function in patients with coronary artery disease. Coron. Artery Dis. 2004, 15, 485–490. [Google Scholar] [CrossRef]
- Lekakis, J.; Rallidis, L.S.; Andreadou, I.; Vamvakou, G.; Kazantzoglou, G.; Magiatis, P.; Skaltsounis, A.L.; Kremastinos, D.T. Polyphenolic compounds from red grapes acutely improve endothelial function in patients with coronary heart disease. Eur. J. Cardiovasc. Prevent. Rehabil. 2005, 12, 596–600. [Google Scholar]
- Walther, C.; Gielen, S.; Hambrecht, R. The effect of exercise training on endothelial function in cardiovascular disease in humans. Exerc. Sport Sci. Rev. 2004, 32, 129–134. [Google Scholar] [CrossRef]
- Nualnim, N.; Barnes, J.N.; Tarumi, T.; Renzi, C.P.; Tanaka, H. Comparison of central artery elasticity in swimmers, runners, and the sedentary. Am. J. Cardiol. 2011, 107, 783–787. [Google Scholar] [CrossRef]
- Green, D.J.; Rowley, N.; Spence, A.; Carter, H.; Whyte, G.; George, K.; Naylor, L.H.; Cable, N.T.; Dawson, E.A.; DH, J.T. Why isn't flow-mediated dilation enhanced in athletes? Med. Sci. Sports Exerc. 2013, 45, 75–82. [Google Scholar] [CrossRef]
- Anter, E.; Thomas, S.R.; Schulz, E.; Shapira, O.M.; Vita, J.A.; Keaney, J.F., Jr. Activation of endothelial nitric-oxide synthase by the p38 MAPK in response to black tea polyphenols. J. Biol. Chem. 2004, 279, 46637–46643. [Google Scholar]
- Rizza, S.; Muniyappa, R.; Iantorno, M.; Kim, J.A.; Chen, H.; Pullikotil, P.; Senese, N.; Tesauro, M.; Lauro, D.; Cardillo, C.; et al. Citrus polyphenol hesperidin stimulates production of nitric oxide in endothelial cells while improving endothelial function and reducing inflammatory markers in patients with metabolic syndrome. J. Clin. Endocrinol. Metabol. 2011, 96, E782–E792. [Google Scholar] [CrossRef]
- Sies, H. Polyphenols and health: Update and perspectives. Arch. Biochem. Biophys. 2010, 501, 2–5. [Google Scholar] [CrossRef]
- Grassi, D.; Desideri, G.; Necozione, S.; Lippi, C.; Casale, R.; Properzi, G.; Blumberg, J.B.; Ferri, C. Blood pressure is reduced and insulin sensitivity increased in glucose-intolerant, hypertensive subjects after 15 days of consuming high-polyphenol dark chocolate. J. Nutr. 2008, 138, 1671–1676. [Google Scholar]
- Grassi, D.; Lippi, C.; Necozione, S.; Desideri, G.; Ferri, C. Short-term administration of dark chocolate is followed by a significant increase in insulin sensitivity and a decrease in blood pressure in healthy persons. Am. J. Clin. Nutr. 2005, 81, 611–614. [Google Scholar]
- Grassi, D.; Necozione, S.; Lippi, C.; Croce, G.; Valeri, L.; Pasqualetti, P.; Desideri, G.; Blumberg, J.B.; Ferri, C. Cocoa reduces blood pressure and insulin resistance and improves endothelium-dependent vasodilation in hypertensives. Hypertension 2005, 46, 398–405. [Google Scholar] [CrossRef]
- Reinke, S.; Karhausen, T.; Doehner, W.; Taylor, W.; Hottenrott, K.; Duda, G.N.; Reinke, P.; Volk, H.D.; Anker, S.D. The influence of recovery and training phases on body composition, peripheral vascular function and immune system of professional soccer players. PLoS ONE 2009, 4, e4910. [Google Scholar] [CrossRef]
- Oh, J.K.; Shin, Y.O.; Yoon, J.H.; Kim, S.H.; Shin, H.C.; Hwang, H.J. Effect of supplementation with Ecklonia cava polyphenol on endurance performance of college students. Int. J. Sport Nutr. Exerc. Metabol. 2010, 20, 72–79. [Google Scholar]
- Ostojic, S.M.; Stojanovic, M.D.; Djordjevic, B.; Jourkesh, M.; Vasiljevic, N. The effects of a 4-week coffeeberry supplementation on antioxidant status, endurance, and anaerobic performance in college athletes. Res. Sports Med. 2008, 16, 281–294. [Google Scholar] [CrossRef]
- Close, G.L.; Ashton, T.; Cable, T.; Doran, D.; Holloway, C.; McArdle, F.; MacLaren, D.P. Ascorbic acid supplementation does not attenuate post-exercise muscle soreness following muscle-damaging exercise but may delay the recovery process. Br. J. Nutr. 2006, 95, 976–981. [Google Scholar] [CrossRef]
- Thompson, D.; Williams, C.; Garcia-Roves, P.; McGregor, S.J.; McArdle, F.; Jackson, M.J. Post-exercise vitamin C supplementation and recovery from demanding exercise. Eur. J. Appl. Physiol. 2003, 89, 393–400. [Google Scholar]
- Avery, N.G.; Kaiser, J.L.; Sharman, M.J.; Scheett, T.P.; Barnes, D.M.; Gomez, A.L.; Kraemer, W.J.; Volek, J.S. Effects of vitamin E supplementation on recovery from repeated bouts of resistance exercise. J. Strength Cond. Res. Natl. Strength Cond. Assoc. 2003, 17, 801–809. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Labonté, K.; Couillard, C.; Motard-Bélanger, A.; Paradis, M.-E.; Couture, P.; Lamarche, B. Acute Effects of Polyphenols from Cranberries and Grape Seeds on Endothelial Function and Performance in Elite Athletes. Sports 2013, 1, 55-68. https://doi.org/10.3390/sports1030055
Labonté K, Couillard C, Motard-Bélanger A, Paradis M-E, Couture P, Lamarche B. Acute Effects of Polyphenols from Cranberries and Grape Seeds on Endothelial Function and Performance in Elite Athletes. Sports. 2013; 1(3):55-68. https://doi.org/10.3390/sports1030055
Chicago/Turabian StyleLabonté, Kim, Charles Couillard, Annie Motard-Bélanger, Marie-Eve Paradis, Patrick Couture, and Benoît Lamarche. 2013. "Acute Effects of Polyphenols from Cranberries and Grape Seeds on Endothelial Function and Performance in Elite Athletes" Sports 1, no. 3: 55-68. https://doi.org/10.3390/sports1030055




