Detection of 17 β-Estradiol in Environmental Samples and for Health Care Using a Single-Use, Cost-Effective Biosensor Based on Differential Pulse Voltammetry (DPV)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Apparatus and Reagents
2.2. Biosensor Fabrication
2.3. Chemical Modification of the Biosensor
2.3.1. Pretreatment of Gold Electrode (AuE)
2.3.2. Chemical Immobilization Steps on the Gold Electrode (AuE)
2.4. Characterization of the Biosensor
3. Results and Discussion
3.1. Preparation of Different Concentrations of 17 β-Estradiol Testing Solution
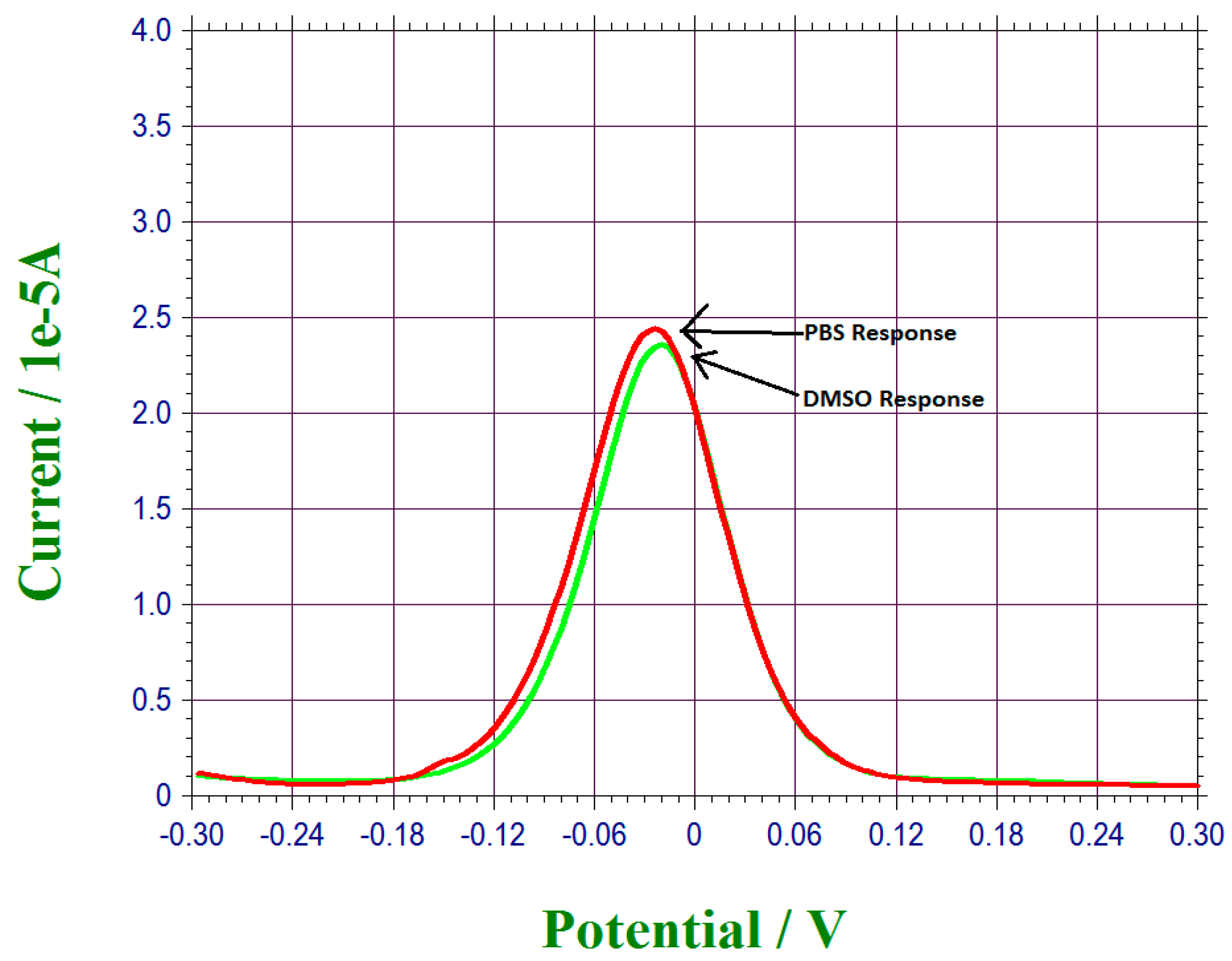
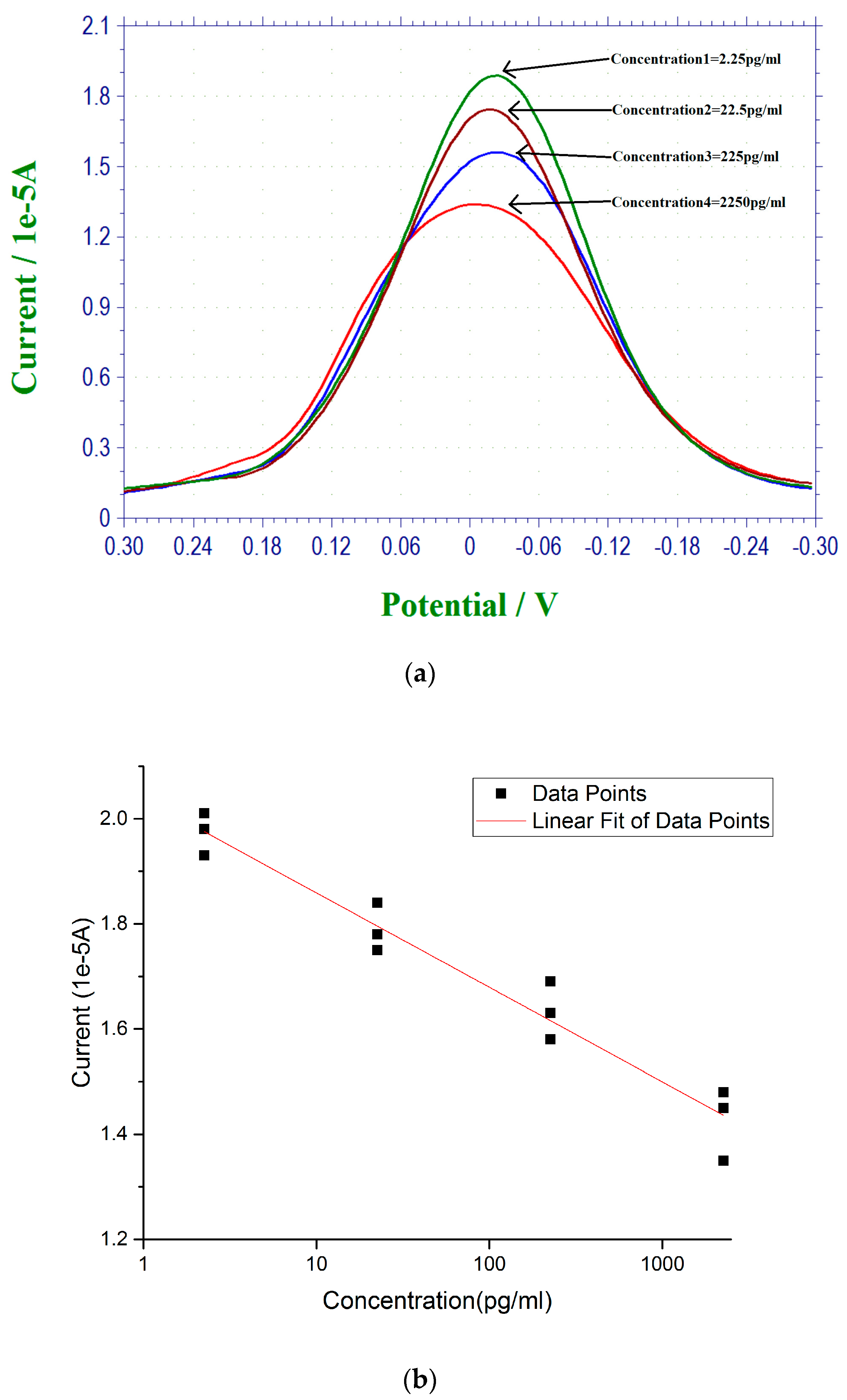
3.2. 17 β-Estradiol Detection in 0.1 M PBS
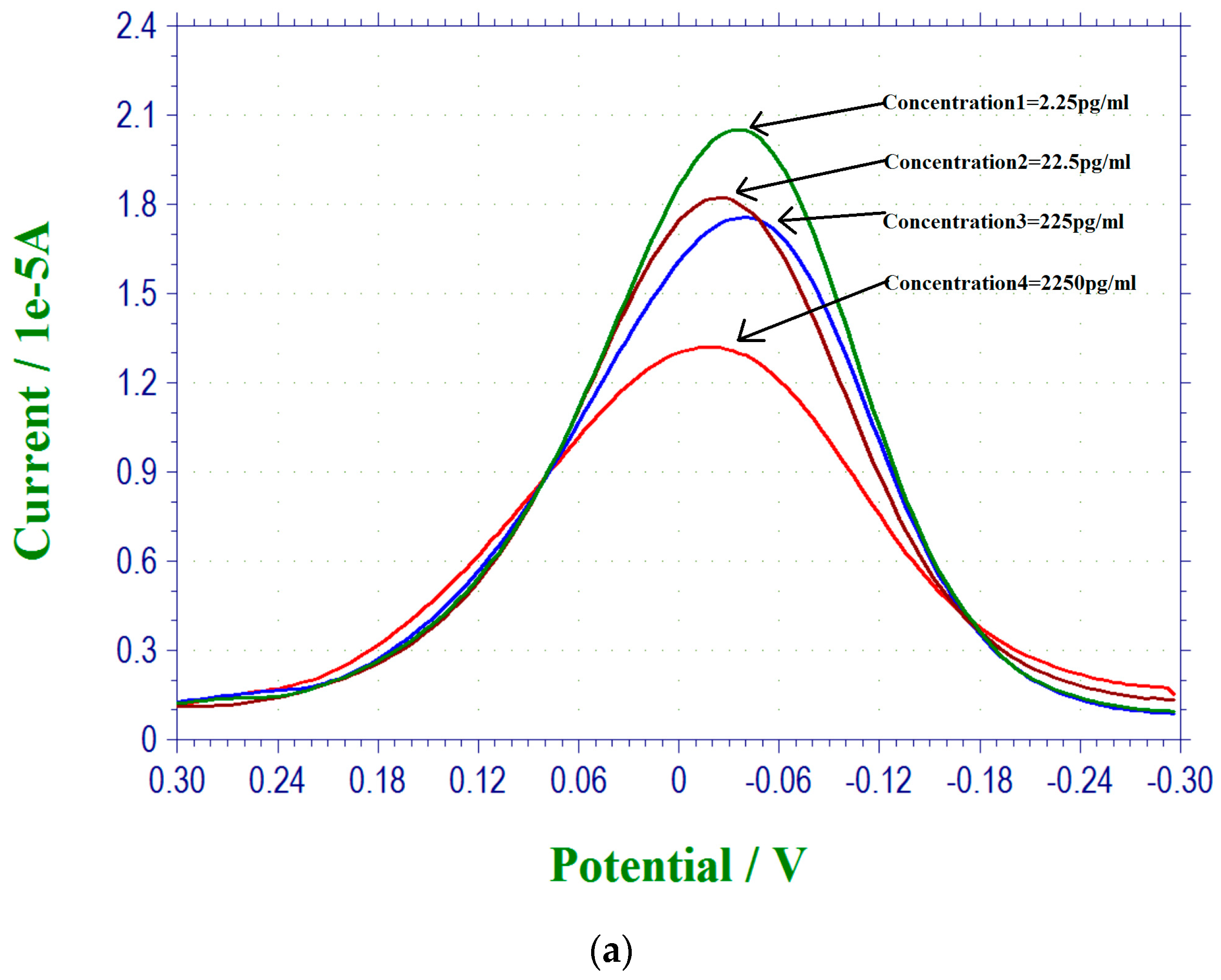
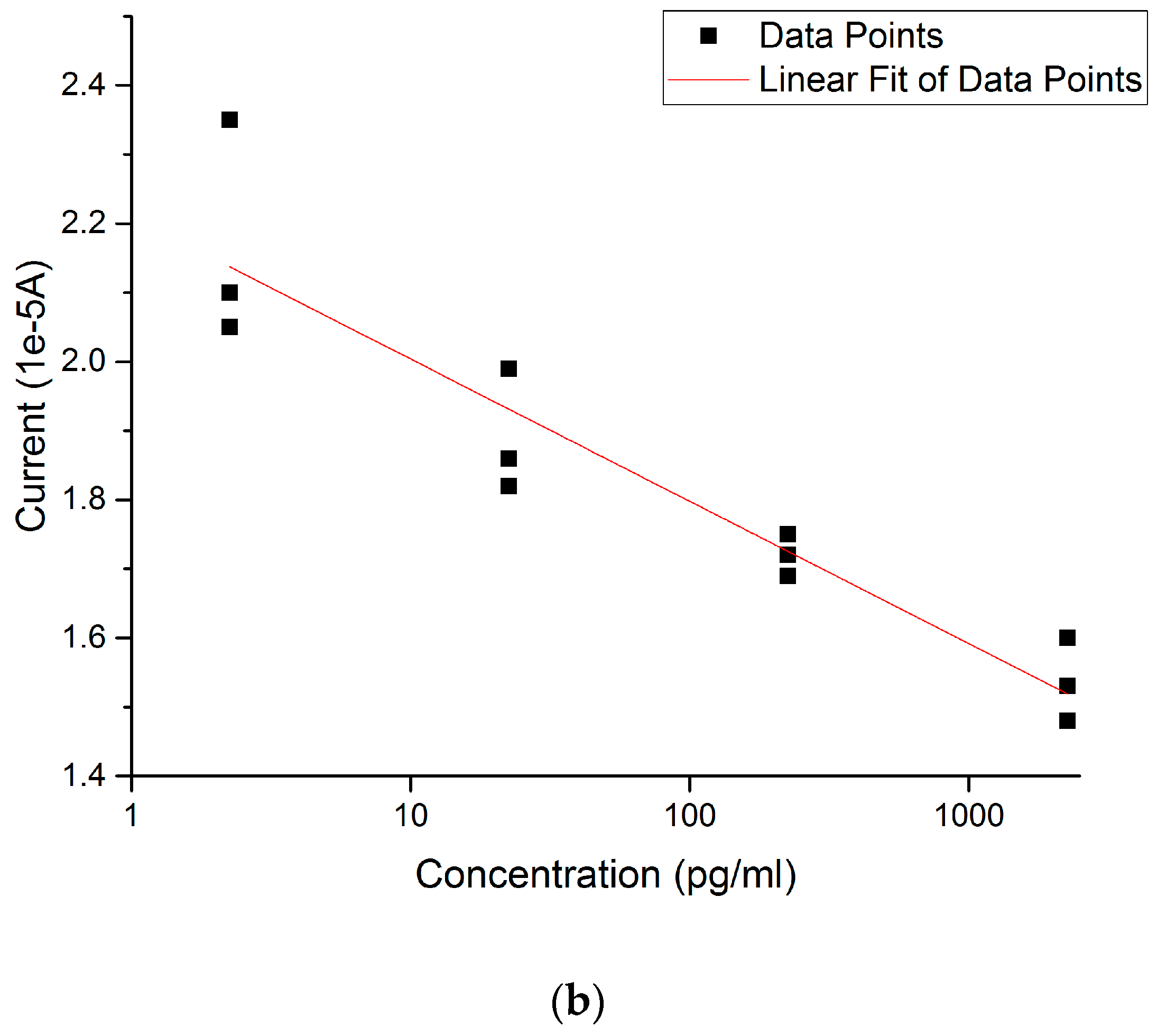
3.3. 17 β-Estradiol Detection in Tap Water from Cleveland, Ohio Regional Water District
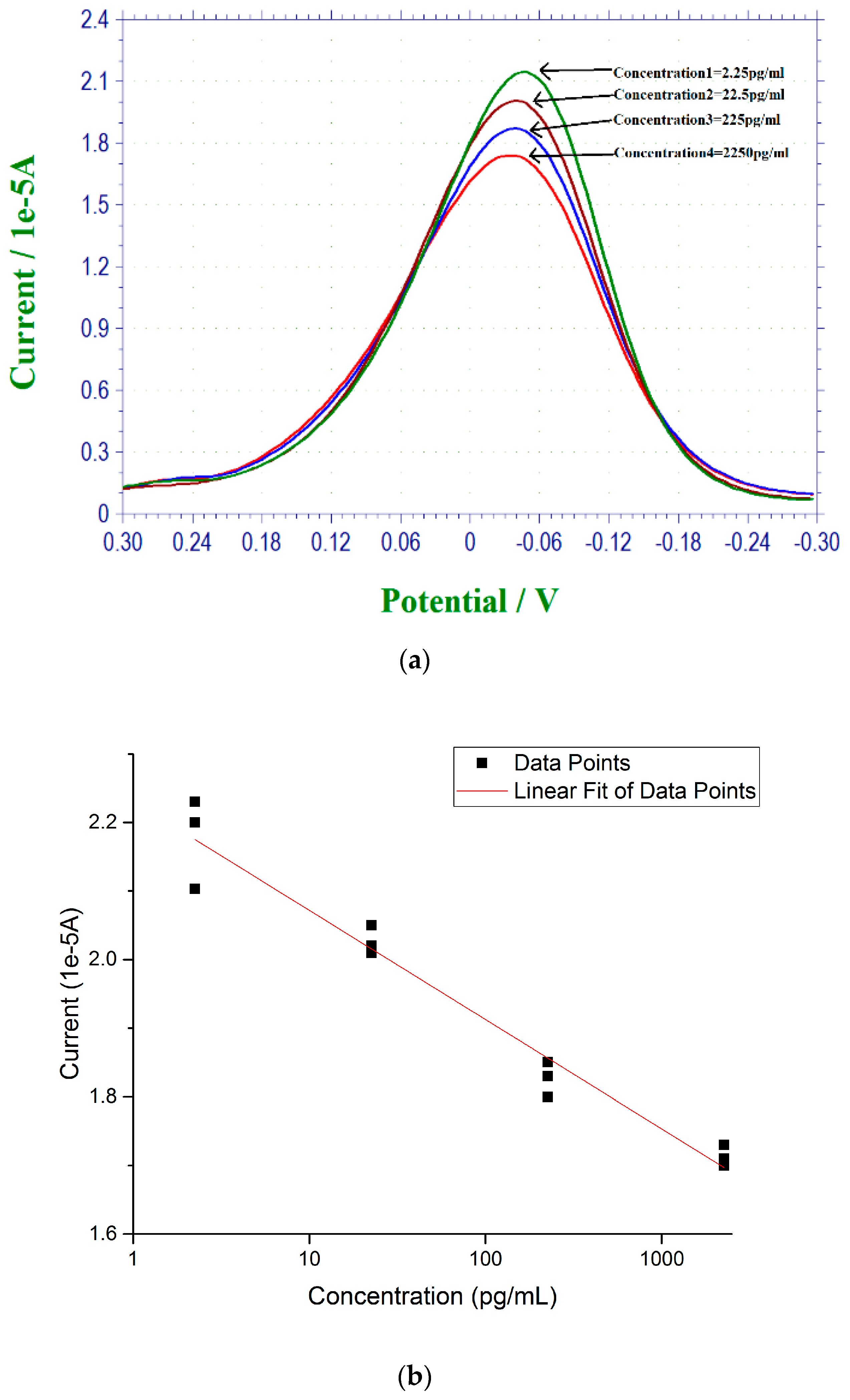
3.4. 17 β-Estradiol Detection in Simulated Urine Test Sample
3.5. Interference Study of This 17 β-Estradiol Biosensor
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lin, X.; Li, Y. A sensitive determination of estrogens with a Pt nano-clusters/multiwalled carbon nanotubes modified glassy carbon electrode. Biosens. Bioelectron. 2006, 22, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Dijksma, M.; Kamp, B.; Hoogvliet, J.C.; van Bennekom, W.P. Development of an electrochemical immunosensor for direct detection of interferon-y at the attomolar level. Anal. Chem. 2001, 71, 901–907. [Google Scholar] [CrossRef]
- Byfield, M.P.; Abuknesha, R.A. Biochemical aspects of biosensors. Biosens. Bioelectron. 1994, 9, 373–399. [Google Scholar] [CrossRef]
- Zhang, X.; Tworoger, S.S.; Eliassen, A.H.; Hankinson, S.E. Postmenopausal plasma sex hormone levels and breast cancer risk over 20 years of follow-up. Breast Cancer Res. Treat. 2013, 137, 883–892. [Google Scholar] [CrossRef] [PubMed]
- Fuhrman, B.J.; Schairer, C.; Gail, M.H.; Boyd-Morin, J.; Xu, X.; Sue, L.Y.; Buys, S.S.; Isaacs, C.; Keefer, L.K.; Veenstra, T.D; et al. Estrogen metabolism and risk of breast cancer in postmenopausal women. J. Natl. Cancer Inst. 2012, 104, 326–339. [Google Scholar] [CrossRef] [PubMed]
- Falk, R.T.; Brinton, L.A.; Dorgen, J.F.; Fuhrman, B.J.; Veenstra, T.D.; Xu, X.; Gierach, G.L. Relationship of serum estrogens and estrogen metabolites to postmenopausal breast cancer risk: a nested case-control study. Breast Cancer Res. 2013, 15, R34. [Google Scholar] [CrossRef] [PubMed]
- Fourkala, E.O.; Zaikin, A.; Burnell, M.; Gentry-Maharaj, A.; Ford, J.; Gunu, R.; Soromani, C.; Hasenbrink, G.; Jacobs, I.; Dawnay, A.; et al. Association of serum sex steroid receptor bioactivity and sex steroid hormones with breast cancer risk in postmenopausal women. Endocr. Relat. Cancer 2012, 19, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Ganti, A.K.; Marr, A.; Batra, S.K. Lung cancer in women: Role of estrogens. Expert Rev. Respir. Med. 2010, 4, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Allen, N.E.; Key, T.J.; Dossus, L.; Rinaldi, S.; Cust, A.; Lukanova, A.; Peeters, P.H.; Onland-Moret, N.C.; Lahmann, P.H.; Berrino, F.; et al. Endogenous sex hormones and endometrial cancer risk in women in the European prospective investigation into cancer and nutrition (EPIC). Endocr. Relat. Cancer 2008, 15, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Kaaks, R.; Lukanova, A.; Kurzer, M.S. Obesity, endogenous hormones, and endometrial cancer risk: A synthetic review, Cancer Epidemislogy. Biomark. Prev. 2002, 11, 1532–1543. [Google Scholar]
- Sumpter, A.P.; Jobling, S. Vitellogenesis as a biomarker for estrogenic contamination of the aquatic environment. Environ. Health Perspect. 1995, 103, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Hanselman, T.A.; Graetz, D.A.; Wilkie, A.C. Manure-borne estrogens as potential environmental contaminants: A review. Environ. Sci. Technol. 2003, 37, 5471–5478. [Google Scholar] [CrossRef] [PubMed]
- Vethaak, A.D.; Lahr, J.; Schrap, S.M.; Belfroid, A.C.; Rijs, G.B.; Gerritsen, A.; de Boer, J.; Bulder, A.S.; Grinwis, G.C.; Kuiper, R.V.; et al. An integrated assessment of estrogenic contamination and biological effects in the aquatic environment of the Netherlands. Chemsphere 2005, 59, 511–524. [Google Scholar] [CrossRef] [PubMed]
- Dmowski, W.P.; Ann, S.; John, W.; Fred, A.; Antonio, S. Effect of intrautenne estrol on reproductive function in the rabbit. Fertil. Steril. 1977, 28, 262–268. [Google Scholar] [CrossRef]
- Bauld, W.S.; Morris, L.G.; Ian, G.M. Abnormality of estrogen metabolism in human subjects with myocardial infraction, Canadian. J. Biochem. Physiol. 1957, 35, 1277–1288. [Google Scholar] [CrossRef]
- Introduction to EU Water Framework Directive. Available online: http://ec.europa.eu/environment/water/water-framework/info/intro_en.htm (accessed on 6 August 2016).
- Guo, Z.; Zine, N.; Balaguer, P.; Zhang, A.; Namour, P.; Lagarde, F.; Jaffrezic-Renault, N. Electrochemical estrogen receptor α based biosensor for label-free detection of estradiol. Electroanalysis 2013, 25, 1765–1772. [Google Scholar] [CrossRef]
- Liscio, C.; Magi, E.; Di Carro, M.; Suter, M.J.; Vermeirssen, E.L. Combining passive samplers and biomonitors to evaluate endocrine disrupting compounds in a wastewater treatment plant by LC/MS/MS and bioassay analyses. Environ. Pollut. 2009, 157, 2716–2721. [Google Scholar] [CrossRef] [PubMed]
- Jordan, V.J.; Mittal, S.; Gosden, B.; Koch, R.; Lieberman, M.E. Structure-activity relationship of estrogen Environment Health Perspectives. Environ. Health Perspect. 1985, 61, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Rossouw, L.E.; Anderson, G.L.; Prentice, R.L.; LaCroix, A.Z.; Kooperberg, C.; Stefanick, M.L.; Jackson, R.D.; Beresford, S.A.A.; Howard, B.V.; Johnson, K.C.; et al. Risks and benefits of estrogens plus progestin I healthy postmenopausal women: principal results from the women’s health initiative randomized controlled trial. JAMA 2002, 288, 321–333. [Google Scholar] [PubMed]
- Dickson, R.B.; Huff, K.K.; Spencer, E.M.; Lippman, M.E. Induction of epidermal growth factor-related polypeptides by 17 β-estradiol in MCF-7 human breast cancer cells. Endocrinology 1985, 118, 138–142. [Google Scholar] [CrossRef] [PubMed]
- Barreiros, L.; Queiroz, J.F.; Magahaes, L.M.; Silva, A.M.T.; Segundo, M.A. Analysis of 17 β-estradiol and 17-α-ethinylestradiol in biological and environmental matrices-a review. Microchem. J. 2016, 126, 243–262. [Google Scholar] [CrossRef]
- Kim, Y.S.; Jung, H.S.; Matsuura, T.; Lee, H.Y.; Kawai, T. GU MB electrochemical detection of 17 β- using DNA aptamer immobilized gold electrode chip. Biosens. Bioelectron. 2007, 22, 2525–2531. [Google Scholar] [CrossRef] [PubMed]
- Im, J.E.; Han, J.A.; Kim, B.K.; Han, J.H.; Park, T.S.; Hwang, S.; Cho, S.I.; Lee, W.Y.; Kim, Y.P. Electrochemical detection of estrogen hormone by immobilized estrogen receptor on Au electrode. Surf. Coat. Technol. 2010, 205, S275–S278. [Google Scholar] [CrossRef]
- Florea, A.; Cristea, C.; Vocanson, F.; Sandulescu, R.; Jaffrezic-Renault, N. Electrochemical sensor for the detection of estradiol based on electropolymerized molecularly imprinted polythioaniline film with signal amplification using gold nanoparticles. Electrochem. Commun. 2015, 50, 36–39. [Google Scholar] [CrossRef]
- Olowu, R.A.; Arotiba, O.; Mailu, S.N.; Waryo, T.T.; Baker, P.; Iwuoha, E. Electrochemical aptasensor for endocrine disrupting 17 β-estradiol based on a poly(3,4-ethylenedioxylthiopene)-gold nanocomposite platform. Sensors 2010, 10, 9872–9890. [Google Scholar] [CrossRef] [PubMed]
- He, Q.; Yuan, S.; Chen, C.; Hu, S. Electrochemical properties of estradiol at glassy carbon electrode modified with nano-Al2O3 film. Mater. Sci. Eng. C 2003, 23, 621–625. [Google Scholar] [CrossRef]
- Liu, X.; Wong, D.K.Y. Electrocatalytic detection of estradiol at a carbon nanotube-Ni(Cyclam) composite electrode fabricated based on a two-factorial design. Anal. Chim. Acta 2007, 594, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Yang, J.; Hu, X. Electrochemical determination of estradiol using a poly(L-serine)film-modified electrode. J. Appl. Electrochem. 2008, 38, 833–836. [Google Scholar] [CrossRef]
- Haddour, N.; Cosnier, S.; Gondran, C. Electrogeneration of a poly9pyrrole)-NTA chelator film for a reversible oriented immobilization of histidine-tagged proteins. J. Am. Chem. Soc. 2005, 127, 5752–5753. [Google Scholar] [CrossRef] [PubMed]
- Janyasupab, M.; Lee, Y.H.; Zhang, Y.; Liu, C.W.; Cai, J.; Popa, A.; Samia, A.C.; Wang, K.W.; Xu, J.; Hu, C.C.; et al. Detection of lysyl oxidase-like 2 (LOXL2), a biomarker of metastasis from breast cancers using human blood samples. Recent Pat. Biomark. 2015, 5, 93–100. [Google Scholar] [CrossRef]
- Molazemhosseini, A.; Magagnin, L.; Vena, P.; Liu, C.C. Single-use disposable electrochemical label-free immunosensor for detection of glycated hemoglobin (HbA1c) using differential pulse voltammetry (DPV). Sensors 2016, 16, 1024. [Google Scholar] [CrossRef] [PubMed]
- Chebil, S.; Hafaiedh, I.; Sauriat-dorizon, H.; Jaffrezic-Resnault, N.; Errachid, A.; Ali, Z.; Korr-Youssoufi, H. Electrochemical detection of D-dimer as deep vein thrombosis marker using single-chain D-dimer antibody immobilized on functionalized polypyrrole. Biosens. Bioelectron. 2010, 26, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Molazemhosseini, A.; Liu, C.C. A single-use, in vitro biosensor for the detection of T-tau protein, a biomarker of neuro-degenerative disorders, in PBS and human serum using differential pulse voltammetry (DPV). Biosensors 2017, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- Willner, I.; Riklin, A. Electrical communication between electrode and NAD(P)+-dependent enzymes using pyrroloquiuolinequinone-electrodes in a self-assembled monolayer configuration: Design of a new class of amperometric biosensors. Anal. Chem. 1994, 66, 1535–1539. [Google Scholar] [CrossRef]
- Campuzano, S.; Glavez, R.; Dedrero, M.; DeVillena, F.J.M.; Pingarron, J.M. Preparation, characterization and application of alkanethiol self-assembled monolayers modified with tetrathiafulvalene and glucose oxidase at a gold disk electrode. J. Electroanal. Chem. 2002, 526, 92–100. [Google Scholar] [CrossRef]
- Abcam Catalog for 17 β-Estradiol (ab 120657); Abcam: Cambridge, MA, USA, 2017.
- Aoki, K.; Tokuda, K.; Matsuda, H. Theory of differential pulse voltammetry at stationary planar electrodes. J. Electroanal. Chem. 1984, 175, 1–13. [Google Scholar] [CrossRef]
- Chopra, A.; Rawat, S.; Bhalla, V.; Suri, C.R. Point-of-care amperometric testing of diabetic marker (HbA1c) using specific electroactive antibodies. Electroanalysis 2014, 26, 469–472. [Google Scholar] [CrossRef]
- Hoberman, J.M. The histroy of synthetic testosterone. Sci. Am. 1995, 272, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Park, J.Y.; Chang, B.Y.; Nam, H.; Park, S.M. Selective electrochemical sensing of glycated hemoglobin (HbA(1c)) on thiophene-3-boronic acid self-assembled monolayer covered gold electrodes. Anal. Chem. 2008, 80, 8035–8044. [Google Scholar] [CrossRef] [PubMed]
- Kuhl, H. Phamacology of estregens and progestgens:influence of different route of adminstration. Climateric 2005, 8 (Suppl. 1), 3–63. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, Y.; Liu, C.C. Detection of 17 β-Estradiol in Environmental Samples and for Health Care Using a Single-Use, Cost-Effective Biosensor Based on Differential Pulse Voltammetry (DPV). Biosensors 2017, 7, 15. https://doi.org/10.3390/bios7020015
Dai Y, Liu CC. Detection of 17 β-Estradiol in Environmental Samples and for Health Care Using a Single-Use, Cost-Effective Biosensor Based on Differential Pulse Voltammetry (DPV). Biosensors. 2017; 7(2):15. https://doi.org/10.3390/bios7020015
Chicago/Turabian StyleDai, Yifan, and Chung Chiun Liu. 2017. "Detection of 17 β-Estradiol in Environmental Samples and for Health Care Using a Single-Use, Cost-Effective Biosensor Based on Differential Pulse Voltammetry (DPV)" Biosensors 7, no. 2: 15. https://doi.org/10.3390/bios7020015





