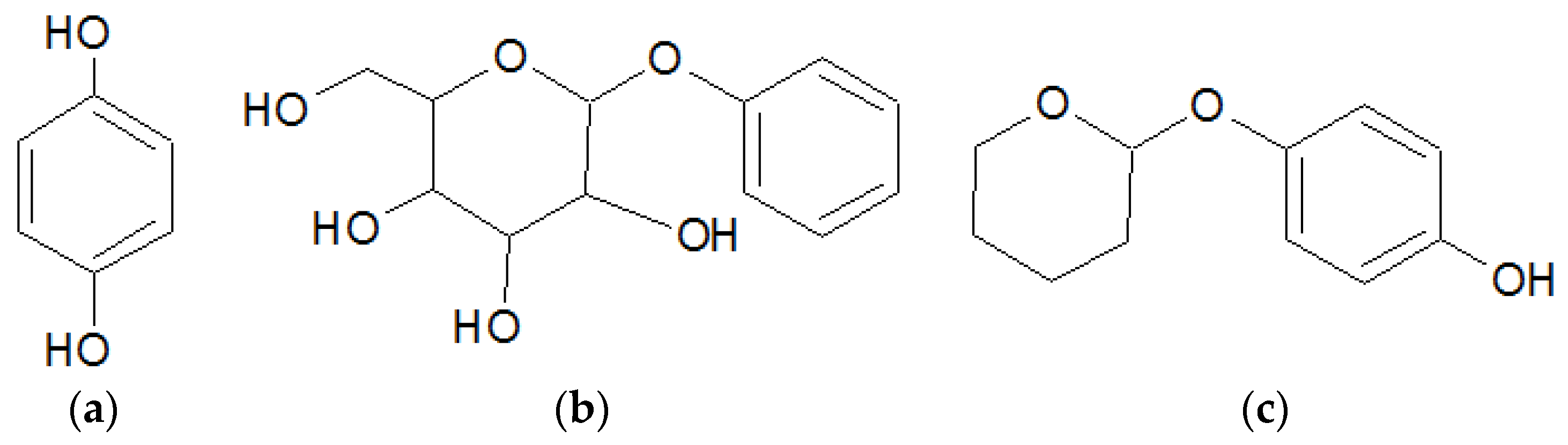
3.1. Screening and Formulation of NLCs
The solid lipids CP, MM, and P8B showed melting points of 51.37 ± 1.06 °C, 42.57 ± 0.85 °C, and 69.80 ± 1.10 °C, respectively. The solubility of dArb in each of the solid lipids was 81.42 ± 0.58 mg/g (at 55 °C), 85.36 ± 3.31 mg/g (at 47 °C), and 149.71 ± 3.28 mg/g (at 75 °C), respectively. In liquid lipids, dArb showed an increased solubility of 105.37 ± 2.94 mg/g in Myr compared to 71.64 ± 2.63 mg/g in CC. Therefore, Myr was chosen as the liquid lipid. CP, the solid lipid with the highest solubility, was selected to maximise loading. The moderate melting point of CP was a positive factor. At room temperature, the lipid nanoparticles stay in a solid state, whereas during and after penetration across the stratum corneum, the CP matrix releases dArb from the NLCs with a good profile.
dArb solubility was also evaluated in two surfactants, P-188 and PEG-40 hydrogenated castor oil (Cremophor RH-40: C-40). The values were 1.41 ± 0.04 mg/g in 1% P-188 aqueous solution and 1.58 ± 0.08 mg/g in 1% C-40 aqueous solution. When choosing the appropriate surfactant, the one with lower solubility is preferred because the surfactant should act as a physicochemical barrier between the lipid matrix containing dArb and the outer water phase. The high solubility of dArb within the surfactant will stimulate the partitioning of dArb from the lipid matrix, resulting in some of the dArb solubilised in the surfactant. Thus, the main purpose for including dArb within the lipid matrix was not achieved.
The solid and liquid lipids chosen (CP and Myr) were physically mixed in several different compositions and were evaluated for their melting points and miscibilities. Visually, all physical mixtures demonstrated good miscibility of the two lipids. The melting points of the four mixtures containing more CP than Myr were only slightly different from the melting point of CP alone, ranging from 50.0–51.8 °C as shown in
Table 2, whereas the mixture that contained more Myr than CP was semisolid or waxy at room temperature. When combined with dArb, the data (
Table 3) indicated that a ratio of CP:Myr 8:2 exhibited ideal solubility at 150.23 ± 2.34 mg/g and used a small amount of liquid lipid. These results agree with Attama et al., who found that lipid matrices with a certain degree of “imperfection“ were ideal for high-loading NLC formulations [
8].
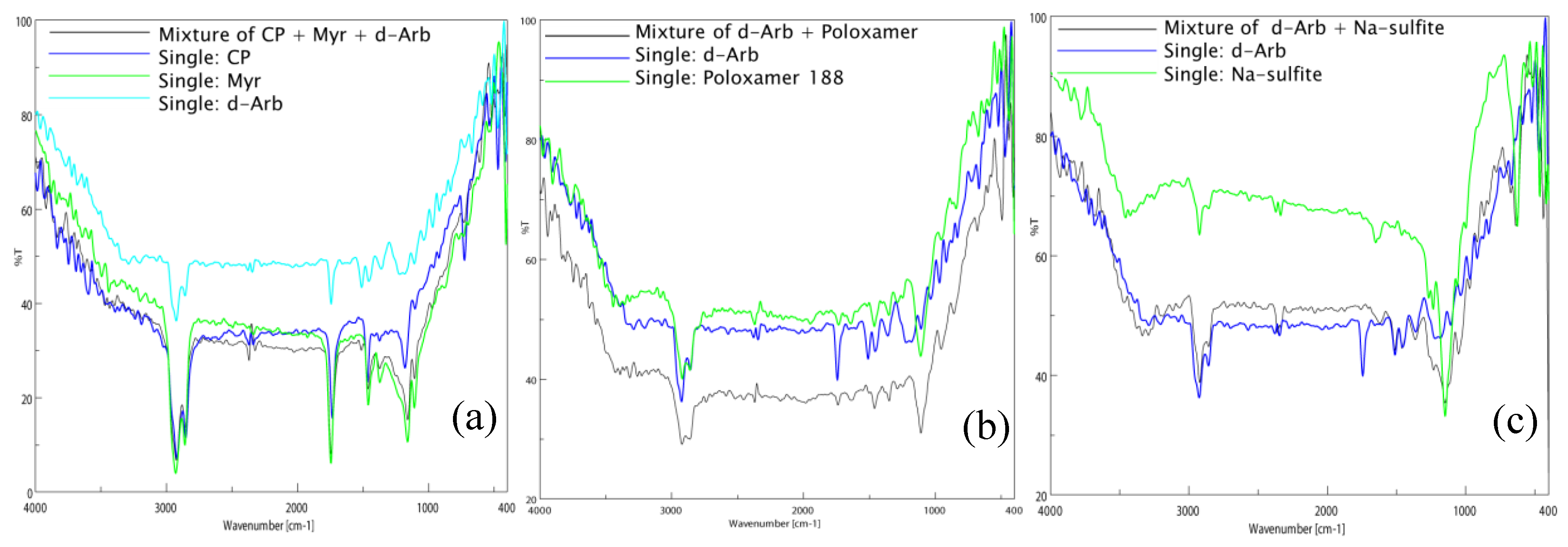
The FTIR spectra of single substances and their physical mixtures confirmed that CP, Myr, and dArb were compatible and that dArb was compatible with the surfactant (P-188) and the antioxidant (sodium sulfite), as shown in
Figure 2. There were no new peaks observed in the spectra of any of the mixtures, indicating that there were no molecular interactions between the substances.
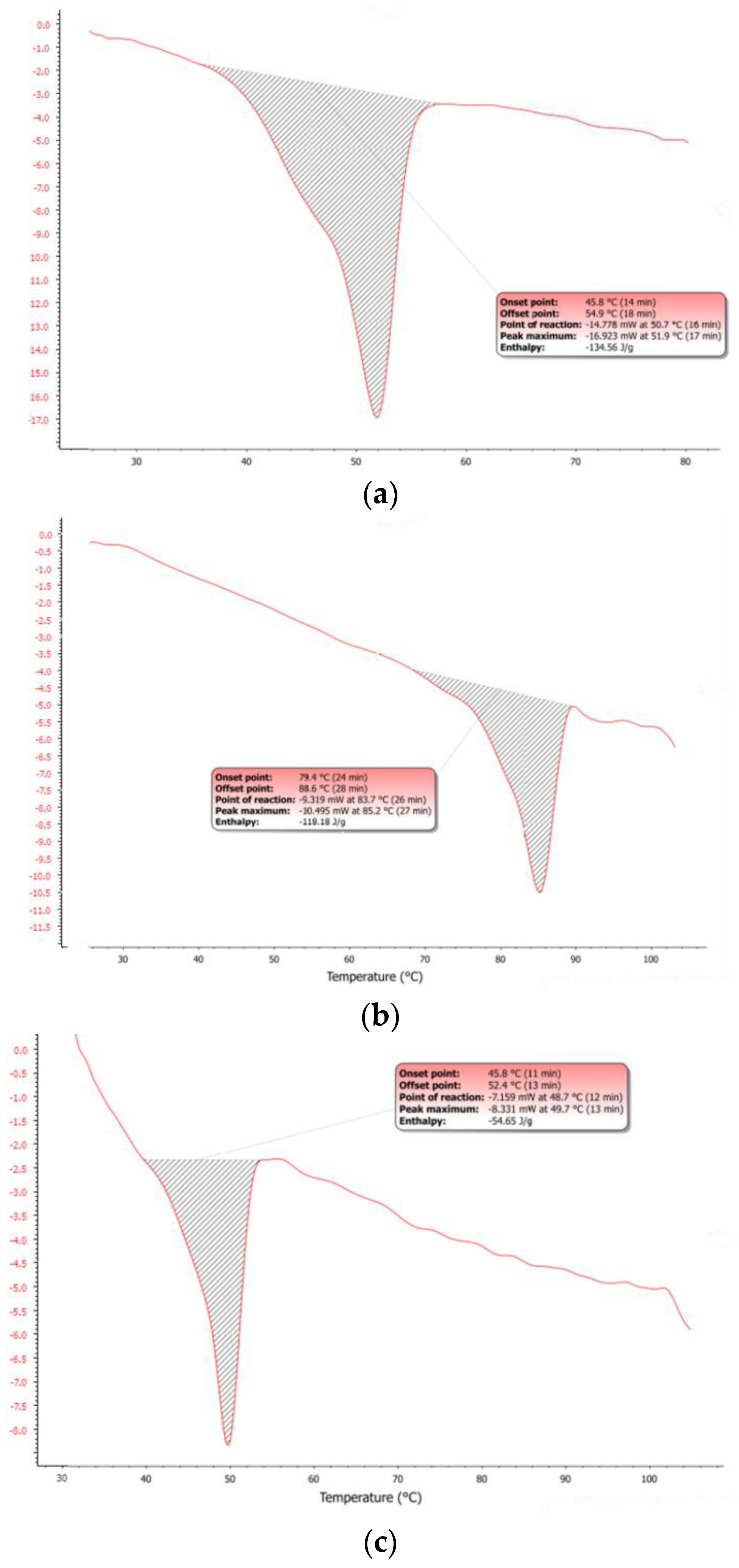
The observed dArb-lipid compatibilities and the inclusion of dArb within lipid matrices were explained by the differential scanning calorimetry (DSC) thermograms shown in
Figure 3a–c. The DSC for bulk CP showed an endothermic curve ranging from 45.8–54.9 °C, with a peak melting point of 51.9 °C. However, the dArb thermogram began to form an endothermic curve at 79.4 °C that ended at 88.6 °C and peaked at 85.2 °C. After dArb was solubilised in a mixture of lipids (CP and Myr) with the ratio indicated in
Table 4, the DSC thermogram showed an endothermic curve only at 45.8–52.4 °C and did not exhibit a curve near the dArb melting point (80–90 °C). This result indicated that dArb was completely dissolved in the lipid mixture and that the inclusion of dArb within the lipid matrix was possible.
The mixture of solid lipid (CP), liquid lipid (Myr), surfactant (P-188), co-surfactant (PEG 400), antioxidant (sodium sulfite), and dArb was optimised. The optimised formula for 1% dArb NLCs is shown in
Table 4 and describes a stable smallest particle size NLC (434.9 ± 21.48 nm) with a polydispersity index of 0.219 ± 0.004. Optimisation of the preparation technique yielded a simple production method using high shear mixing (Ultrathurrax T25, Ika, Germany) at 10,000 rpm for 5 min and 60 °C followed by size reduction using sonophoresis (Misonix Sonicator, Farmingdale, NY, USA) at 6500 J for 3 min 30 s.
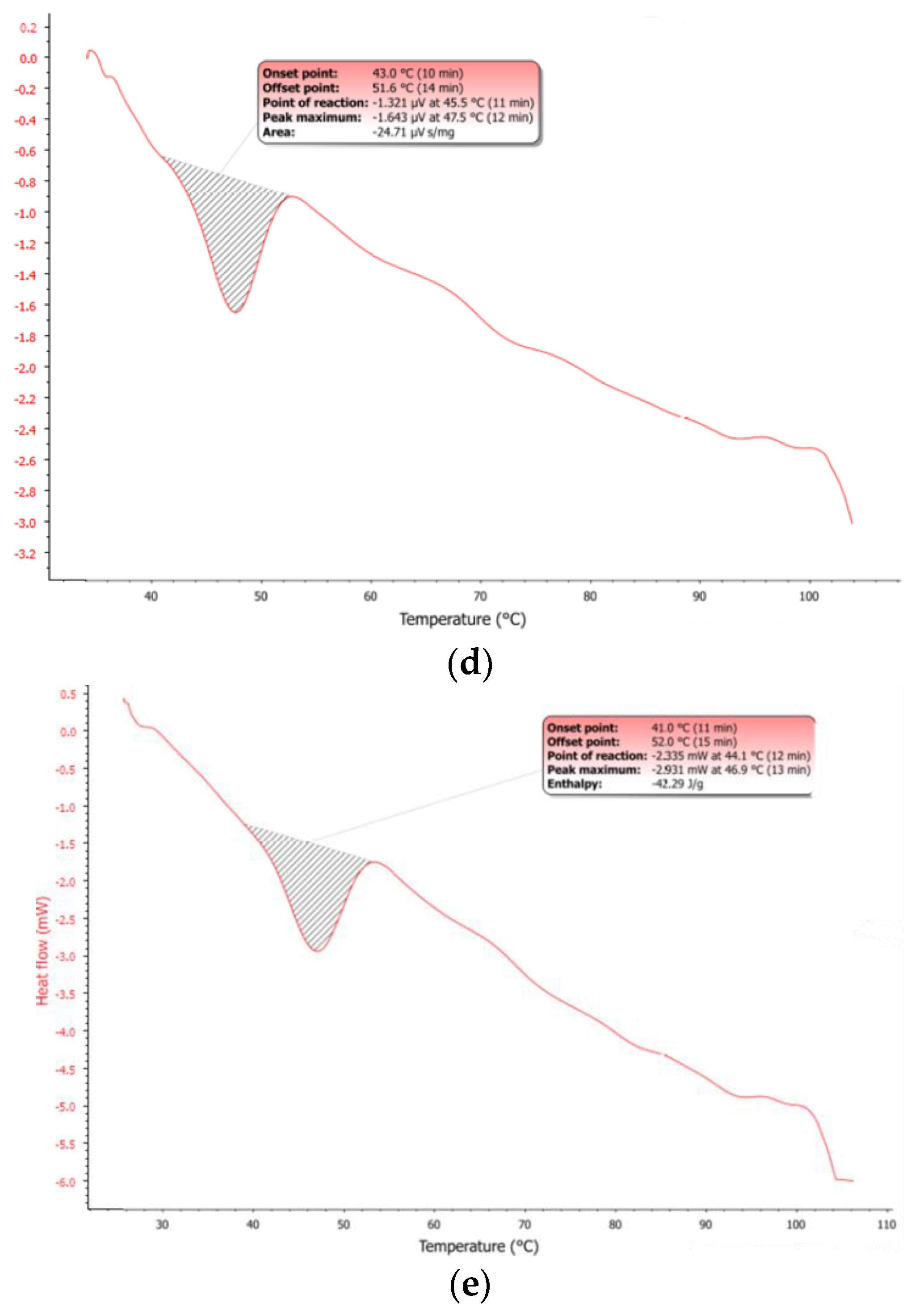
A mixture of dArb and the lipid phase was heated to 60 °C. The solution was clear, and no crystals were detected, indicating complete dArb solubilisation in the lipid matrix and agreeing with the DSC thermograms in
Figure 3. A two-step NLC production method using high shear mixing coupled with ultrasonication was expected to result in increased homogeneity and reduced nanoparticle dispersion [
9]. These advantages were clear from the polydispersity index of less than 0.25, which indicates uniformity of the hydrodynamic lipid particle diameter and a minimal aggregation tendency [
10]. Particle sizes of less than 500 nm were successfully obtained, which is an ideal value for z [
11].
Figure 3d,e shows that the DSC dArb-NLC thermogram exhibited a similar profile to blank NLCs and did not have a peak at the dArb melting point. This finding confirmed that dArb was successfully included in the lipid matrix. Further analysis comparing the physical mixture of CP-dArb-Myr (
Figure 3c) and dArb NLC (
Figure 3e), presented in
Table 5, showed that the NLC endotherm curve broadened (11.0 °C breadth) compared to the physical mixture (6.8 °C breadth), and shifted to a slightly lower temperature, from 49.7 °C to 46.9 °C. Other than a possible effect from adsorbed emulsifier molecules, this phenomenon also conformed to the expected behaviour of smaller particle sized nanodispersed materials [
12] according to the Gibbs-Thomson equation [
13]:
in which
T is the melting temperature of a particle with radius
r,
T0 is the melting temperature of the bulk material at the same external pressure,
γsl is the interfacial tension at the solid-liquid interface,
Vs is the specific volume of the solid, and
ΔHfus is the specific heat of fusion.
The equation demonstrates that smaller nanoparticles would have a lower melting temperature. The endothermic curve broadening observed was due to the polydispersity of nanoparticles, suggesting the presence of different sizes of nanoparticles that melted at different temperatures [
8]. Decreased NLC enthalpy indicated a decrease in the degree of crystallinity, which was an expected advantage of the NLC system [
14].
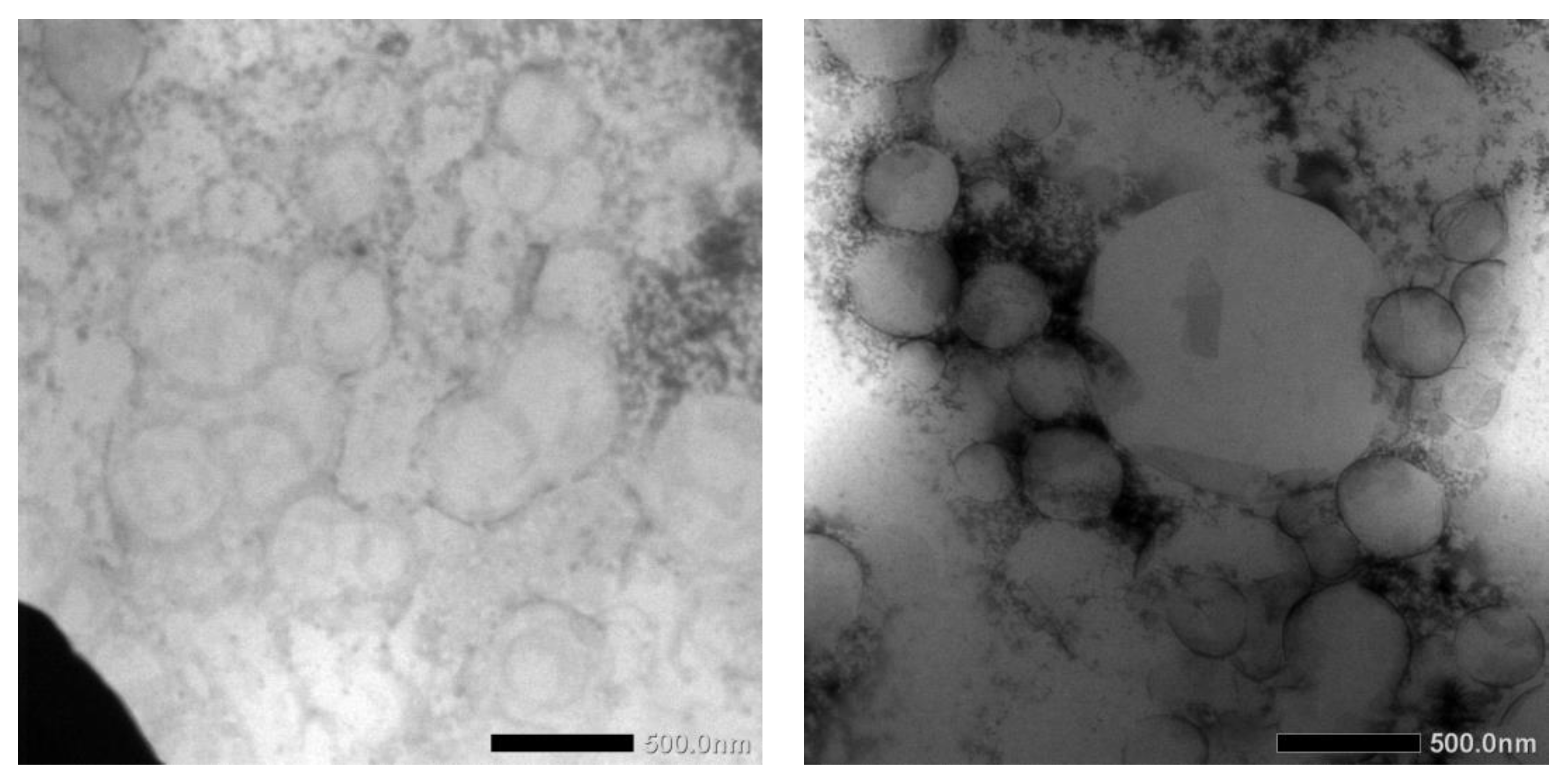
TEM images of blank NLCs and dArb-NLCs are shown in
Figure 4 and show spherically shaped particles with sizes of approximately 400–500 nm, which generally agrees with previously reported photon correlation spectroscopy (PCS) results. The differences observed between the images were primarily caused by different positions and amplification factors. Moreover, different sample preparations and the different principles behind PCS and TEM might introduce slight variations. PCS detects light scattering effects that are used to calculate average particle size, whereas TEM can “visually” measure the particle dimension and allow observation of their actual shapes [
15].
Triplicate measurements of EE resulted in a convincing 99.821% ± 0.004%. This result was possibly due to only 1% of the dArb being loaded into a total of 12% lipids, while the maximum solubility of dArb was approximately 1 in 8 parts of mixed lipids. An ultrafiltration method was used to evaluate the EE of dArb in the NLC and was able to quickly determine the amount of unentrapped dArb. A smaller cut-off molecular weight (MW) value of the filter provided more accurate results because the filtrate separated by centrifugation was limited to only the soluble dArb in the water + surfactant system, as long as the MW of the substance was much smaller than the cut-off value.
3.2. In Vitro Skin Penetration Studies
Table 6 shows the formula used for skin penetration studies. All formulations contained 1% dArb with the same percentage of lipid phase and surfactant. Such formulas were designed to minimise the influence of ingredient composition, thereby evaluating only responses due to differences in the delivery system.
This study compared dArb-NLC in its original liquid form and a hydroxypropyl methylcellulose (HPMC)-thickened formulation. Higher topical formulation viscosity might form a film layer; however, due to the resulting occlusive effect, it enhances the dermal penetration of the active substance. Skin penetration profiles for a dArb nanoemulsion (NE) and a conventional 1% dArb cream emulsion (CR) were evaluated against NLCs. NE is a colloidal dispersion containing only liquid lipid (Myr) as the lipid phase, and CR is a semisolid emulsion containing micron-sized globules of dArb-loaded lipid and wax.
The membrane used to represent the penetration barrier of skin was a synthetic sebum membrane consisting of Whatman paper impregnated with a mixture of several lipids modelled after the semi-polar lipids found in the epidermis extracellular matrix and known as Spangler’s synthetic sebum [
16]. Spangler’s sebum was chosen instead of the mixture proposed by Wertz [
17] because Spangler suggested a more complex lipid/oil composition that better mimics actual physiologic sebum. Spangler’s membrane (SP) represents the barrier properties of the stratum corneum without the appendages.
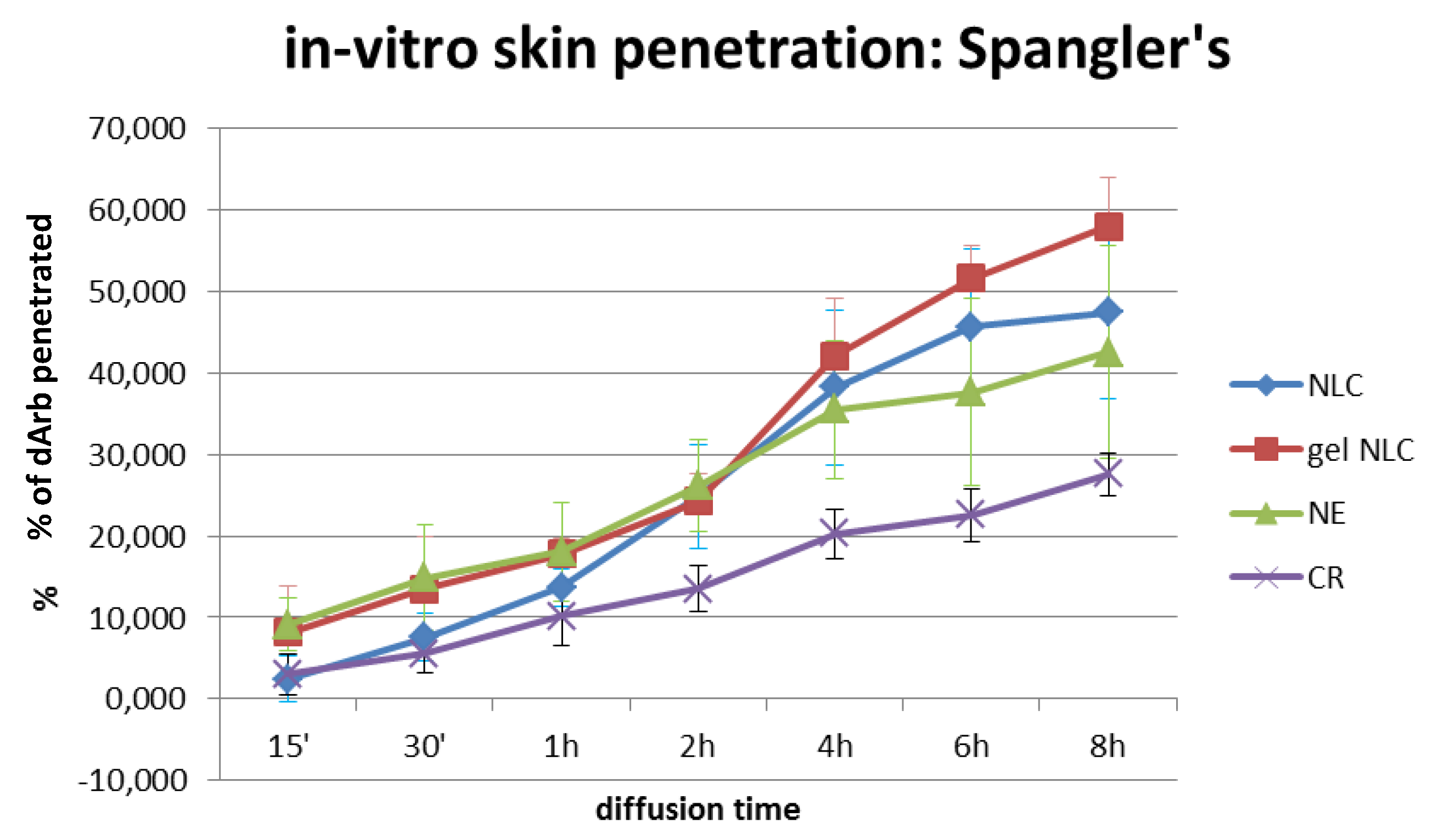
As expected, the dArb penetration through the SP for different delivery systems (
Figure 5) showed that gel-NLCs generated the highest dArb penetration (57.97% ± 6.08%) after 8 h of topical application, followed by NLCs (47.39% ± 10.59%), NE (42.49% ± 13.07%), and CR (27.58% ± 2.59%). The in vitro diffusion profiles demonstrated that both NLCs and gel-NLCs facilitated gradual and improved permeation of skin lipids and confirming the effectiveness of these systems at increasing the intercellular permeation of dArb.
A two-way ANOVA of the data showed a
p value of 2.2 × 10
−10 (
Table 7), indicating a significant difference in dArb penetration between the four delivery systems. Further analysis with the least significant difference (LSD) method was performed to understand which delivery system showed a significant difference in dArb penetration compared to gel-NLCs. The LSD value was 6.705, and the absolute values of the mean differences of the two groups were calculated. They were 5.084, 4.503 and 16.095 for gel-NLCs vs. NLCs, gel-NLCs vs. NE, and gel-NLCs vs. CR, respectively. This result suggested that there was a significant difference between gel-NLCs and CR in terms of dArb penetration. However, although NLCs and NE did not show significant differences in the amount of dArb penetration, their penetration profiles over time were observably different.
The permeation profiles of gel-NLCs were slightly higher than that of NLCs and might indicate that the gelling agent HPMC played a role enhancing penetration. Cellulose derivatives are known to have film-forming properties that reduce trans epidermal water loss (TEWL) and cause skin hydration, increasing the percutaneous absorption of lipid nanoparticles [
18].
The permeation of NE across the SP before 2 h of application was similar to gel-NLCs and was improved over liquid NLCs because the liquid oil NE globules were more fluid than the solidified lipid matrices of the NLCs. The fluidity, and thus elasticity, of the nanoglobules resulted in a quicker permeation rate using an intercellular route across the brick-and-mortar-like model of the stratum corneum represented by the SP. However, after the 2nd hour, the “imperfect” lipid matrices of the NLCs started to release more dArb to the receptor solution, surpassing NE. The solid nature of the NLC lipid matrix caused the entrapped dArb to be released more slowly from the matrix and into the receptor fluid than the solubilised dArb within the liquid oil NE globules.
As predicted, the CR showed the least dArb permeation into the receptor medium. A conventional cream formulation generally has much larger globule sizes and is less permeable across the stratum corneum lipids, consistently showing the lowest concentration of dArb in the receptor medium.