Journal Description
Antioxidants
Antioxidants
is an international, peer-reviewed, open access journal, published monthly online by MDPI. The International Coenzyme Q10 Association (ICQ10A), Israel Society for Oxygen and Free Radical Research (ISOFRR) and European Academy for Molecular Hydrogen Research (EAMHR) are affiliated with Antioxidants and their members receive discounts on the article processing charge.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, FSTA, PubAg, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q1 (Food Science & Technology) / CiteScore - Q1 (Food Science)
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 13.9 days after submission; acceptance to publication is undertaken in 2.6 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Testimonials: See what our editors and authors say about Antioxidants.
- Companion journal: Oxygen.
Impact Factor:
7.0 (2022);
5-Year Impact Factor:
7.3 (2022)
Latest Articles
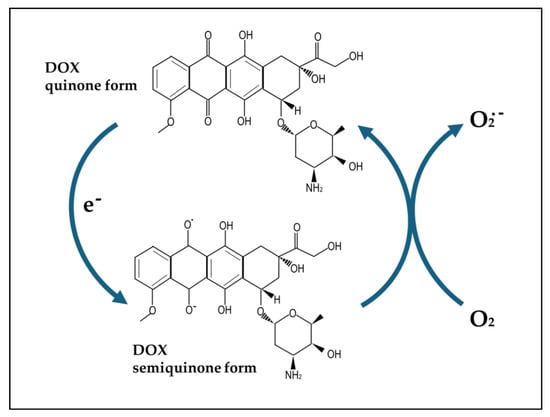
Potential of Natural Phenolic Compounds against Doxorubicin-Induced Chemobrain: Biological and Molecular Mechanisms Involved
Antioxidants 2024, 13(4), 486; https://doi.org/10.3390/antiox13040486 - 18 Apr 2024
Abstract
Chemotherapy-induced cognitive impairment or “chemobrain” is a prevalent long-term complication of chemotherapy and one of the more devastating. Most of the studies performed so far to identify the cognitive dysfunctions induced by antineoplastic chemotherapies have been focused on treatment with anthracyclines, frequently administered
[...] Read more.
Chemotherapy-induced cognitive impairment or “chemobrain” is a prevalent long-term complication of chemotherapy and one of the more devastating. Most of the studies performed so far to identify the cognitive dysfunctions induced by antineoplastic chemotherapies have been focused on treatment with anthracyclines, frequently administered to breast cancer patients, a population that, after treatment, shows a high possibility of long survival and, consequently, of chemobrain development. In the last few years, different possible strategies have been explored to prevent or reduce chemobrain induced by the anthracycline doxorubicin (DOX), known to promote oxidative stress and inflammation, which have been strongly implicated in the development of this brain dysfunction. Here, we have critically analyzed the results of the preclinical studies from the last few years that have evaluated the potential of phenolic compounds (PheCs), a large class of natural products able to exert powerful antioxidant and anti-inflammatory activities, in inhibiting DOX-induced chemobrain. Several PheCs belonging to different classes have been shown to be able to revert DOX-induced brain morphological damages and deficits associated with learning, memory, and exploratory behavior. We have analyzed the biological and molecular mechanisms implicated and suggested possible future perspectives in this research area.
Full article
(This article belongs to the Special Issue Oxidative Stress and the Central Nervous System)
►
Show Figures
Open AccessReview
Unlocking the Therapeutic Potential of Ellagic Acid for Non-Alcoholic Fatty Liver Disease and Non-Alcoholic Steatohepatitis
by
Tharani Senavirathna, Armaghan Shafaei, Ricky Lareu and Lois Balmer
Antioxidants 2024, 13(4), 485; https://doi.org/10.3390/antiox13040485 - 18 Apr 2024
Abstract
Obesity is in epidemic proportions in many parts of the world, contributing to increasing rates of non-alcoholic fatty liver disease (NAFLD). NAFLD represents a range of conditions from the initial stage of fatty liver to non-alcoholic steatohepatitis (NASH), which can progress to severe
[...] Read more.
Obesity is in epidemic proportions in many parts of the world, contributing to increasing rates of non-alcoholic fatty liver disease (NAFLD). NAFLD represents a range of conditions from the initial stage of fatty liver to non-alcoholic steatohepatitis (NASH), which can progress to severe fibrosis, through to hepatocellular carcinoma. There currently exists no treatment for the long-term management of NAFLD/NASH, however, dietary interventions have been investigated for the treatment of NASH, including several polyphenolic compounds. Ellagic acid is one such polyphenolic compound. Nutraceutical food abundant in ellagic acid undergoes initial hydrolysis to free ellagic acid within the stomach and small intestine. The proposed mechanism of action of ellagic acid extends beyond its initial therapeutic potential, as it is further broken down by the gut microbiome into urolithin. Both ellagic acid and urolithin have been found to alleviate oxidative stress, inflammation, and fibrosis, which are associated with NAFLD/NASH. While progress has been made in understanding the pharmacological and biological activity of ellagic acid and its involvement in NAFLD/NASH, it has yet to be fully elucidated. Thus, the aim of this review is to summarise the currently available literature elucidating the therapeutic potential of ellagic acid and its microbial-derived metabolite urolithin in NAFLD/NASH.
Full article
(This article belongs to the Section Health Outcomes of Antioxidants and Oxidative Stress)
Open AccessReview
Hormetic Nutrition and Redox Regulation in Gut–Brain Axis Disorders
by
Maria Scuto, Francesco Rampulla, Giuseppe Maria Reali, Sestina Maria Spanò, Angela Trovato Salinaro and Vittorio Calabrese
Antioxidants 2024, 13(4), 484; https://doi.org/10.3390/antiox13040484 - 18 Apr 2024
Abstract
The antioxidant and anti-inflammatory effects of hormetic nutrition for enhancing stress resilience and overall human health have received much attention. Recently, the gut–brain axis has attracted prominent interest for preventing and therapeutically impacting neuropathologies and gastrointestinal diseases. Polyphenols and polyphenol-combined nanoparticles in synergy
[...] Read more.
The antioxidant and anti-inflammatory effects of hormetic nutrition for enhancing stress resilience and overall human health have received much attention. Recently, the gut–brain axis has attracted prominent interest for preventing and therapeutically impacting neuropathologies and gastrointestinal diseases. Polyphenols and polyphenol-combined nanoparticles in synergy with probiotics have shown to improve gut bioavailability and blood–brain barrier (BBB) permeability, thus inhibiting the oxidative stress, metabolic dysfunction and inflammation linked to gut dysbiosis and ultimately the onset and progression of central nervous system (CNS) disorders. In accordance with hormesis, polyphenols display biphasic dose–response effects by activating at a low dose the Nrf2 pathway resulting in the upregulation of antioxidant vitagenes, as in the case of heme oxygenase-1 upregulated by hidrox® or curcumin and sirtuin-1 activated by resveratrol to inhibit reactive oxygen species (ROS) overproduction, microbiota dysfunction and neurotoxic damage. Importantly, modulation of the composition and function of the gut microbiota through polyphenols and/or probiotics enhances the abundance of beneficial bacteria and can prevent and treat Alzheimer’s disease and other neurological disorders. Interestingly, dysregulation of the Nrf2 pathway in the gut and the brain can exacerbate selective susceptibility under neuroinflammatory conditions to CNS disorders due to the high vulnerability of vagal sensory neurons to oxidative stress. Herein, we aimed to discuss hormetic nutrients, including polyphenols and/or probiotics, targeting the Nrf2 pathway and vitagenes for the development of promising neuroprotective and therapeutic strategies to suppress oxidative stress, inflammation and microbiota deregulation, and consequently improve cognitive performance and brain health. In this review, we also explore interactions of the gut–brain axis based on sophisticated and cutting-edge technologies for novel anti-neuroinflammatory approaches and personalized nutritional therapies.
Full article
(This article belongs to the Special Issue The Interplay of Oxidative Stress, the Gut–Brain Axis, and Neurodegenerative Diseases)
►▼
Show Figures
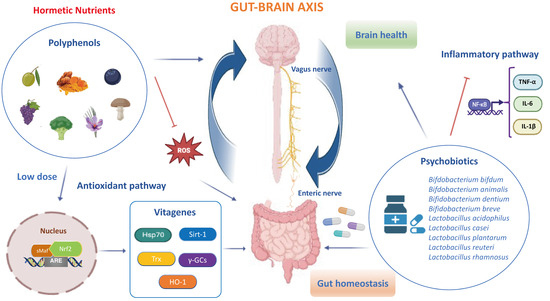
Figure 1
Open AccessArticle
Comparison of the Antioxidant Potency of Four Triterpenes of Centella asiatica against Oxidative Stress
by
Jinyeong Lim, Hana Lee, Seonghwa Hong, Junsoo Lee and Younghwa Kim
Antioxidants 2024, 13(4), 483; https://doi.org/10.3390/antiox13040483 - 18 Apr 2024
Abstract
We comparatively evaluated the antioxidant properties of key triterpenes from Centella asiatica, including asiatic acid (AA), asiaticoside, madecassic acid, and madecassoside, in several cell types, including skin fibroblasts, macrophages, hepatocytes, and endothelial cells, under conditions promoting oxidative stress. AA conferred the highest
[...] Read more.
We comparatively evaluated the antioxidant properties of key triterpenes from Centella asiatica, including asiatic acid (AA), asiaticoside, madecassic acid, and madecassoside, in several cell types, including skin fibroblasts, macrophages, hepatocytes, and endothelial cells, under conditions promoting oxidative stress. AA conferred the highest viability on Hs68 cells exposed to ultraviolet B (UVB) irradiation. Triterpene pretreatment attenuated the UVB-induced generation of reactive oxygen species (ROS) and malondialdehyde (MDA), as well as the UVB-induced depletion of glutathione (GSH) in skin fibroblasts. AA most potently inhibited UVB-induced MMP generation, resulting in increased intracellular collagen levels. Pretreatment with triterpenes, particularly AA, significantly improved cell viability and attenuated TBHP-induced levels of ROS, alanine aminotransferase, and aspartate aminotransferase in HepG2 cells. Triterpenes attenuated ROS levels and reduced MDA and GSH expression in EA.hy926 cells. In RAW264.7 macrophages, production of nitric oxide, tumor necrosis factor-α, and interleukin-6 (indicators of LPS-induced oxidative damage) was significantly reduced by treatment with any of the triterpenes. Statistical analyses of triterpene biological activities using principal component analysis and hierarchical clustering revealed that AA exerted the greatest overall influence and showed remarkable activity in Hs68 and HepG2 cells.
Full article
(This article belongs to the Special Issue Antioxidant Capacity of Natural Products)
►▼
Show Figures
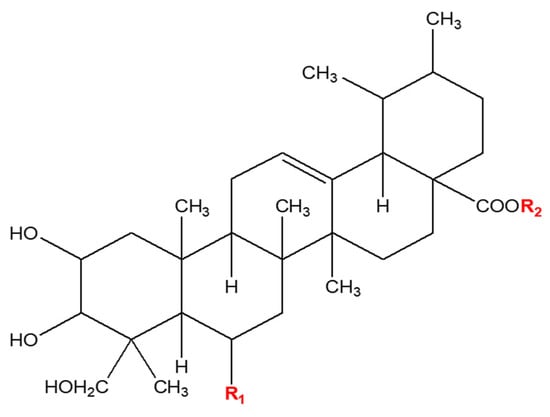
Figure 1
Open AccessArticle
Chemical Characterization and Biological Properties of Leguminous Honey
by
Florinda Fratianni, Giuseppe Amato, Maria Neve Ombra, Vincenzo De Feo, Filomena Nazzaro and Beatrice De Giulio
Antioxidants 2024, 13(4), 482; https://doi.org/10.3390/antiox13040482 - 18 Apr 2024
Abstract
Honey can beneficially act against different human diseases, helping our body to improve its health. The aim of the present study was first to increase knowledge of some biochemical characteristics (amount and composition of polyphenols and volatile organic compounds, vitamin C content) of
[...] Read more.
Honey can beneficially act against different human diseases, helping our body to improve its health. The aim of the present study was first to increase knowledge of some biochemical characteristics (amount and composition of polyphenols and volatile organic compounds, vitamin C content) of five Italian legume honeys (alfalfa, astragalus, carob, indigo, and sainfoin). Furthermore, we evaluated their potential health properties by studying their antioxidant and in vitro anti-inflammatory activities and in vitro inhibitory effects on three enzymes involved in neurodegenerative diseases (acetylcholinesterase, butyrylcholinesterase, and tyrosinase). Alfalfa honey showed the highest total polyphenol content (TPC) (408 μg g−1 of product). Indigo honey showed the lowest TPC (110 μg g−1 of product). The antioxidant activity was noteworthy, especially in the case of sainfoin honey (IC50 = 6.08 mg), which also exhibited excellent inhibitory action against butyrylcholinesterase (74%). Finally, the correlation between the biochemical and functional results allowed us to identify classes of molecules, or even single molecules, present in these five honeys, which are capable of influencing the properties indicated above.
Full article
(This article belongs to the Special Issue Bee Products as a Source of Natural Antioxidants: Second Edition)
►▼
Show Figures
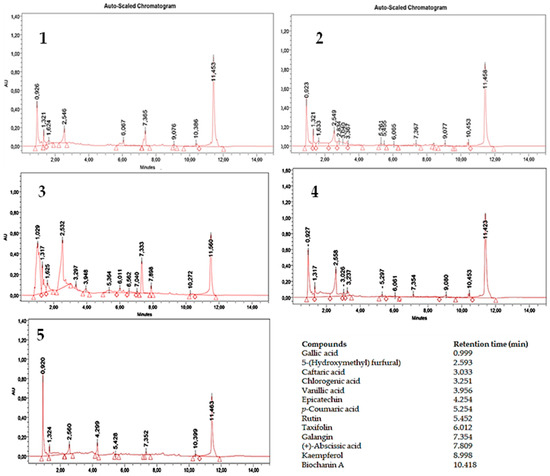
Figure 1
Open AccessArticle
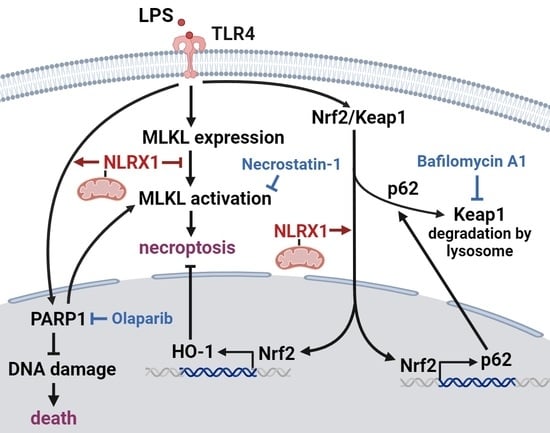
NLRX1 Inhibits LPS-Induced Microglial Death via Inducing p62-Dependent HO-1 Expression, Inhibiting MLKL and Activating PARP-1
by
Yu-Ling Huang, Duen-Yi Huang, Vladlen Klochkov, Chi-Ming Chan, Yuan-Shen Chen and Wan-Wan Lin
Antioxidants 2024, 13(4), 481; https://doi.org/10.3390/antiox13040481 - 17 Apr 2024
Abstract
The activation of microglia and the production of cytokines are key factors contributing to progressive neurodegeneration. Despite the well-recognized neuronal programmed cell death regulated by microglial activation, the death of microglia themselves is less investigated. Nucleotide-binding oligomerization domain, leucine-rich repeat-containing X1 (NLRX1) functions
[...] Read more.
The activation of microglia and the production of cytokines are key factors contributing to progressive neurodegeneration. Despite the well-recognized neuronal programmed cell death regulated by microglial activation, the death of microglia themselves is less investigated. Nucleotide-binding oligomerization domain, leucine-rich repeat-containing X1 (NLRX1) functions as a scaffolding protein and is involved in various central nervous system diseases. In this study, we used the SM826 microglial cells to understand the role of NLRX1 in lipopolysaccharide (LPS)-induced cell death. We found LPS-induced cell death is blocked by necrostatin-1 and zVAD. Meanwhile, LPS can activate poly (ADP-ribose) polymerase-1 (PARP-1) to reduce DNA damage and induce heme oxygenase (HO)-1 expression to counteract cell death. NLRX1 silencing and PARP-1 inhibition by olaparib enhance LPS-induced SM826 microglial cell death in an additive manner. Less PARylation and higher DNA damage are observed in NLRX1-silencing cells. Moreover, LPS-induced HO-1 gene and protein expression through the p62-Keap1-Nrf2 axis are attenuated by NLRX1 silencing. In addition, the Nrf2-mediated positive feedback regulation of p62 is accordingly reduced by NLRX1 silencing. Of note, NLRX1 silencing does not affect LPS-induced cellular reactive oxygen species (ROS) production but increases mixed lineage kinase domain-like pseudokinase (MLKL) activation and cell necroptosis. In addition, NLRX1 silencing blocks bafilomycin A1-induced PARP-1 activation. Taken together, for the first time, we demonstrate the role of NLRX1 in protecting microglia from LPS-induced cell death. The underlying protective mechanisms of NLRX1 include upregulating LPS-induced HO-1 expression via Nrf2-dependent p62 expression and downstream Keap1-Nrf2 axis, mediating PARP-1 activation for DNA repair via ROS- and autophagy-independent pathway, and reducing MLKL activation.
Full article
(This article belongs to the Special Issue Oxidative Stress and Nrf2-Mediated Cellular Inflammation)
►▼
Show Figures
Graphical abstract
Open AccessArticle
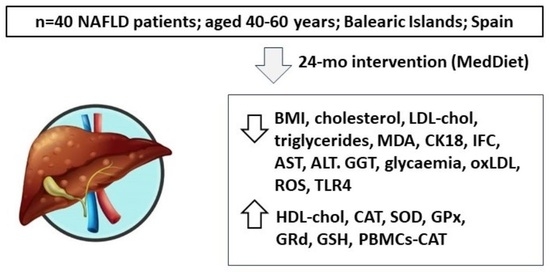
Impact of Adherence to the Mediterranean Diet on Antioxidant Status and Metabolic Parameters in NAFLD Patients: A 24-Month Lifestyle Intervention Study
by
Maria Magdalena Quetglas-Llabrés, Margalida Monserrat-Mesquida, Cristina Bouzas, Silvia García, Emma Argelich, Miguel Casares, Lucía Ugarriza, Isabel Llompart, Josep A. Tur and Antoni Sureda
Antioxidants 2024, 13(4), 480; https://doi.org/10.3390/antiox13040480 - 17 Apr 2024
Abstract
Background: The Mediterranean Diet (MedDiet) is recognized as a healthy dietary pattern. Non-alcoholic fatty liver disease (NAFLD) is characterized by the excessive accumulation of fat in the liver. Objectives: To assess the antioxidant status in erythrocytes, plasma, and peripheral blood mononuclear cells (PBMCs)
[...] Read more.
Background: The Mediterranean Diet (MedDiet) is recognized as a healthy dietary pattern. Non-alcoholic fatty liver disease (NAFLD) is characterized by the excessive accumulation of fat in the liver. Objectives: To assess the antioxidant status in erythrocytes, plasma, and peripheral blood mononuclear cells (PBMCs) of NAFLD patients following a 24-month lifestyle intervention based on the MedDiet. Adult patients (n = 40; aged 40–60 years) diagnosed with NAFLD by magnetic resonance imaging were divided into two groups based on their adherence to the MedDiet. Consumption was assessed using a validated 143-item semiquantitative Food Frequency Questionnaire. Anthropometrics, biochemistry parameters, intrahepatic fat contents (IFC), antioxidants, and inflammatory biomarkers were measured in plasma and erythrocytes before and after the intervention. Results: After the intervention, body mass index (BMI) and plasma levels of total cholesterol, low-density lipoprotein cholesterol (LDL-chol), triglycerides, malondialdehyde (MDA), and cytokeratin-18 (CK18) decreased, and high-density lipoprotein cholesterol (HDL-chol) increased. Participants with high adherence to MedDiet showed lower IFC, hepatic enzyme (AST, ALT, and GGT), glycemia, oxidase LDL (oxLDL) plasma levels, and erythrocyte MDA levels. Higher antioxidant activity (erythrocyte catalase-CAT, superoxide dismutase-SOD, glutathione peroxidase-GPx, glutathione reductase-GRd, and total glutathione-GSH as well as PBMCs-CAT gene expression) was observed in these patients, along with a reduction of PBMCs reactive oxygen species production and Toll-like receptor 4 (TLR4) expression. Inverse associations were observed between adherence to the MedDiet and BMI, glycemia, AST, IFC, and CK18 plasma levels and oxLDL, CAT, SOD, and GRd activities in erythrocytes. A significant linear regression was observed between adherence to the MedDiet and antioxidant score. Conclusions: Adherence to the MedDiet is associated with improved plasma and PBMC antioxidant and inflammatory biomarker profiles and high antioxidant defences in erythrocytes.
Full article
(This article belongs to the Special Issue The Role of Oxidative Stress in Non-Alcoholic Fatty Liver Disease)
►▼
Show Figures
Graphical abstract
Open AccessReview
Advances in Research on the Activity Evaluation, Mechanism and Structure-Activity Relationships of Natural Antioxidant Peptides
by
Baoting Xu, Qin Dong, Changxia Yu, Hongyu Chen, Yan Zhao, Baosheng Zhang, Panling Yu and Mingjie Chen
Antioxidants 2024, 13(4), 479; https://doi.org/10.3390/antiox13040479 - 17 Apr 2024
Abstract
Antioxidant peptides are a class of biologically active peptides with low molecular weights and stable antioxidant properties that are isolated from proteins. In this review, the progress in research on the activity evaluation, action mechanism, and structure-activity relationships of natural antioxidant peptides are
[...] Read more.
Antioxidant peptides are a class of biologically active peptides with low molecular weights and stable antioxidant properties that are isolated from proteins. In this review, the progress in research on the activity evaluation, action mechanism, and structure-activity relationships of natural antioxidant peptides are summarized. The methods used to evaluate antioxidant activity are mainly classified into three categories: in vitro chemical, in vitro cellular, and in vivo animal methods. Also, the biological effects produced by these three methods are listed: the scavenging of free radicals, chelation of metal ions, inhibition of lipid peroxidation, inhibition of oxidative enzyme activities, and activation of antioxidant enzymes and non-enzymatic systems. The antioxidant effects of natural peptides primarily consist of the regulation of redox signaling pathways, which includes activation of the Nrf2 pathway and the inhibition of the NF-κB pathway. The structure-activity relationships of the antioxidant peptides are investigated, including the effects of peptide molecular weight, amino acid composition and sequence, and secondary structure on antioxidant activity. In addition, four computer-assisted methods (molecular docking, molecular dynamics simulation, quantum chemical calculations, and the determination of quantitative structure-activity relationships) for analyzing the structure-activity effects of natural peptides are summarized. Thus, this review lays a theoretical foundation for the development of new antioxidants, nutraceuticals, and cosmetics.
Full article
(This article belongs to the Special Issue Dietary Antioxidants and Cosmetics—2nd Edition)
►▼
Show Figures
Figure 1
Open AccessArticle
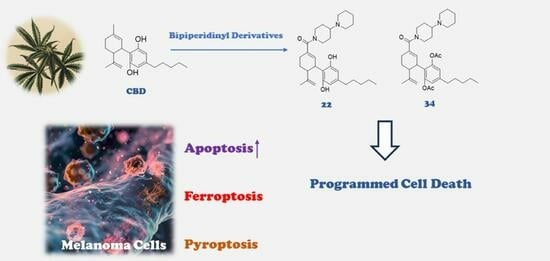
Bipiperidinyl Derivatives of Cannabidiol Enhance Its Antiproliferative Effects in Melanoma Cells
by
Peihong Lyu, Huifang Li, Junzhao Wan, Ying Chen, Zhen Zhang, Panpan Wu, Yinsheng Wan, Navindra P. Seeram, Jean Christopher Chamcheu, Chang Liu and Hang Ma
Antioxidants 2024, 13(4), 478; https://doi.org/10.3390/antiox13040478 - 17 Apr 2024
Abstract
Cannabis and its major cannabinoid cannabidiol (CBD) are reported to exhibit anticancer activity against skin tumors. However, the cytotoxic effects of other minor cannabinoids and synthetic CBD derivatives in melanoma are not fully elucidated. Herein, the antiproliferative activity of a panel of phytocannabinoids
[...] Read more.
Cannabis and its major cannabinoid cannabidiol (CBD) are reported to exhibit anticancer activity against skin tumors. However, the cytotoxic effects of other minor cannabinoids and synthetic CBD derivatives in melanoma are not fully elucidated. Herein, the antiproliferative activity of a panel of phytocannabinoids was screened against murine (B16F10) and human (A375) melanoma cells. CBD was the most cytotoxic natural cannabinoid with respective IC50 of 28.6 and 51.6 μM. Further assessment of the cytotoxicity of synthetic CBD derivatives in B16F10 cells identified two bipiperidinyl group-bearing derivatives (22 and 34) with enhanced cytotoxicity (IC50 = 3.1 and 8.5 μM, respectively). Furthermore, several cell death assays including flow cytometric (for apoptosis and ferroptosis) and lactate dehydrogenase (for pyroptosis) assays were used to characterize the antiproliferative activity of CBD and its bipiperidinyl derivatives. The augmented cytotoxicity of 22 and 34 in B16F10 cells was attributed to their capacity to promote apoptosis (as evidenced by increased apoptotic population). Taken together, this study supports the notion that CBD and its derivatives are promising lead compounds for cannabinoid-based interventions for melanoma management.
Full article
(This article belongs to the Special Issue Natural Antioxidants: Multiple Mechanisms for Skin Protection)
►▼
Show Figures
Graphical abstract
Open AccessArticle
Single-Cell Profiling Reveals Immune-Based Mechanisms Underlying Tumor Radiosensitization by a Novel Mn Porphyrin Clinical Candidate, MnTnBuOE-2-PyP5+ (BMX-001)
by
Sun Up Noh, Jinyeong Lim, Sung-Won Shin, Yeeun Kim, Woong-Yang Park, Ines Batinic-Haberle, Changhoon Choi and Won Park
Antioxidants 2024, 13(4), 477; https://doi.org/10.3390/antiox13040477 - 17 Apr 2024
Abstract
Manganese porphyrins reportedly exhibit synergic effects when combined with irradiation. However, an in-depth understanding of intratumoral heterogeneity and immune pathways, as affected by Mn porphyrins, remains limited. Here, we explored the mechanisms underlying immunomodulation of a clinical candidate, MnTnBuOE-2-PyP5+ (BMX-001, MnBuOE), using
[...] Read more.
Manganese porphyrins reportedly exhibit synergic effects when combined with irradiation. However, an in-depth understanding of intratumoral heterogeneity and immune pathways, as affected by Mn porphyrins, remains limited. Here, we explored the mechanisms underlying immunomodulation of a clinical candidate, MnTnBuOE-2-PyP5+ (BMX-001, MnBuOE), using single-cell analysis in a murine carcinoma model. Mice bearing 4T1 tumors were divided into four groups: control, MnBuOE, radiotherapy (RT), and combined MnBuOE and radiotherapy (MnBuOE/RT). In epithelial cells, the epithelial–mesenchymal transition, TNF-α signaling via NF-кB, angiogenesis, and hypoxia-related genes were significantly downregulated in the MnBuOE/RT group compared with the RT group. All subtypes of cancer-associated fibroblasts (CAFs) were clearly reduced in MnBuOE and MnBuOE/RT. Inhibitory receptor–ligand interactions, in which epithelial cells and CAFs interacted with CD8+ T cells, were significantly lower in the MnBuOE/RT group than in the RT group. Trajectory analysis showed that dendritic cells maturation-associated markers were increased in MnBuOE/RT. M1 macrophages were significantly increased in the MnBuOE/RT group compared with the RT group, whereas myeloid-derived suppressor cells were decreased. CellChat analysis showed that the number of cell–cell communications was the lowest in the MnBuOE/RT group. Our study is the first to provide evidence for the combined radiotherapy with a novel Mn porphyrin clinical candidate, BMX-001, from the perspective of each cell type within the tumor microenvironment.
Full article
(This article belongs to the Section Natural and Synthetic Antioxidants)
►▼
Show Figures
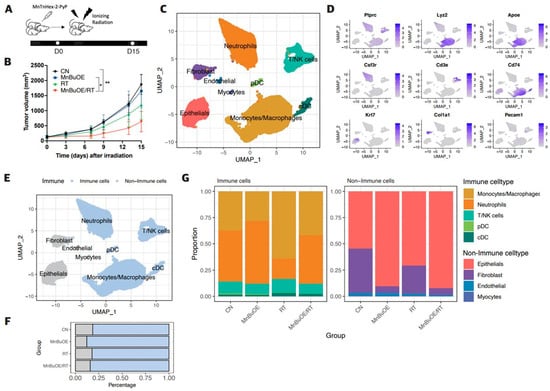
Figure 1
Open AccessArticle
Porcine Brain Enzyme Hydrolysate Enhances Immune Function and Antioxidant Defense via Modulation of Gut Microbiota in a Cyclophosphamide-Induced Immunodeficiency Model
by
Yu Yue, Hye Jeong Yang, Ting Zhang, Chen Li, Min Jung Kim, Keun-Nam Kim and Sunmin Park
Antioxidants 2024, 13(4), 476; https://doi.org/10.3390/antiox13040476 - 17 Apr 2024
Abstract
►▼
Show Figures
This study examined how consuming porcine brain enzyme hydrolysate (PBEH) affects the immune function and composition of the gut microbiota in an immunodeficient animal model. Male Wistar rats aged 6 weeks were fed casein (control), 100 mg/kg body weight (BW), red ginseng extract
[...] Read more.
This study examined how consuming porcine brain enzyme hydrolysate (PBEH) affects the immune function and composition of the gut microbiota in an immunodeficient animal model. Male Wistar rats aged 6 weeks were fed casein (control), 100 mg/kg body weight (BW), red ginseng extract (positive-control), and 6, 13, and 26 mg PBEH per kg BW (PBEH-L, PBEH-M, and PBEH-H, respectively) daily for 4 weeks. At 30 min after consuming assigned compounds, they were orally administered cyclophosphamide (CTX; 5 mg/kg BW), an immunosuppressive agent, to suppress the immune system by inhibiting the proliferation of lymphocytes. The normal-control rats were fed casein and water instead of CTX. Natural killer cell activity and splenocyte proliferation induced by 1 μg/mL lipopolysaccharide were lower in the control group than the normal-control group, and they significantly increased with PBEH consumption, particularly at high doses. The PBEH consumption increased dose-dependently in the Th1/Th2 ratio compared to the control. The lipid peroxide contents were lower in the PBEH group than in the control group. Moreover, PBEH m and PBEH-H consumption mitigated white pulp cell damage, reduced red pulp congestion, and increased spleen mast cells in the histological analysis. Intestinal microbiota composition demonstrated differences between the groups at the genus levels, with Akkermansia being more abundant in the control group than the normal-control group and the PBEH-H group showing a decrease. However, Bifidobacterium decreased in the control group but increased in the PBEH-H group. The β-diversity revealed distinct microbial communities of PBEH and positive-control groups compared to the control group (p < 0.05). The metagenome predictions revealed that PBEH-H influenced amino acid metabolism, antioxidant defense, insulin sensitivity, and longevity pathways. In conclusion, PBEH-H intake boosted immune responses and reduced lipid peroxides by modulating gut microbiota composition. These findings suggest that PBEH-H has the potential as a dietary supplement for improving immune function and gut health in individuals with immunodeficiency.
Full article
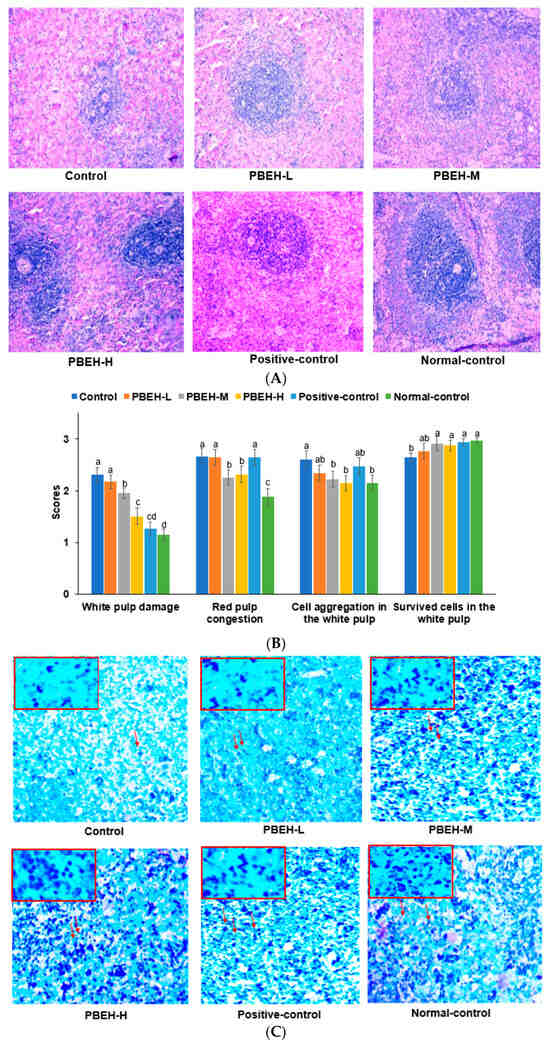
Figure 1
Open AccessReview
Potential New Therapies “ROS-Based” in CLL: An Innovative Paradigm in the Induction of Tumor Cell Apoptosis
by
Raffaele Sciaccotta, Sebastiano Gangemi, Giuseppa Penna, Laura Giordano, Giovanni Pioggia and Alessandro Allegra
Antioxidants 2024, 13(4), 475; https://doi.org/10.3390/antiox13040475 - 17 Apr 2024
Abstract
►▼
Show Figures
Chronic lymphocytic leukemia, in spite of recent advancements, is still an incurable disease; the majority of patients eventually acquire resistance to treatment through relapses. In all subtypes of chronic lymphocytic leukemia, the disruption of normal B-cell homeostasis is thought to be mostly caused
[...] Read more.
Chronic lymphocytic leukemia, in spite of recent advancements, is still an incurable disease; the majority of patients eventually acquire resistance to treatment through relapses. In all subtypes of chronic lymphocytic leukemia, the disruption of normal B-cell homeostasis is thought to be mostly caused by the absence of apoptosis. Consequently, apoptosis induction is crucial to the management of this illness. Damaged biological components can accumulate as a result of the oxidation of intracellular lipids, proteins, and DNA by reactive oxygen species. It is possible that cancer cells are more susceptible to apoptosis because of their increased production of reactive oxygen species. An excess of reactive oxygen species can lead to oxidative stress, which can harm biological elements like DNA and trigger apoptotic pathways that cause planned cell death. In order to upset the balance of oxidative stress in cells, recent therapeutic treatments in chronic lymphocytic leukemia have focused on either producing reactive oxygen species or inhibiting it. Examples include targets created in the field of nanomedicine, natural extracts and nutraceuticals, tailored therapy using biomarkers, and metabolic targets. Current developments in the complex connection between apoptosis, particularly ferroptosis and its involvement in epigenomics and alterations, have created a new paradigm.
Full article
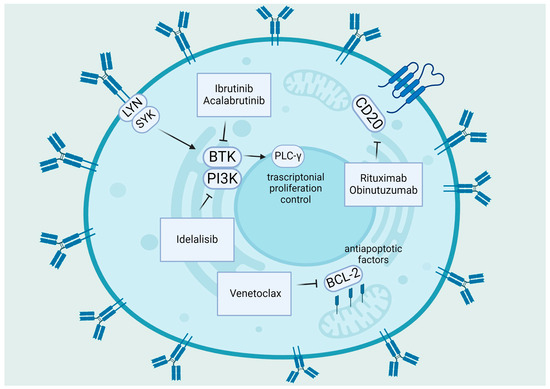
Figure 1
Open AccessArticle
S-Allylmercapto-N-Acetylcysteine (ASSNAC) Attenuates Osteoporosis in Ovariectomized (OVX) Mice
by
Itay Bleichman, Sahar Hiram-Bab, Yankel Gabet and Naphtali Savion
Antioxidants 2024, 13(4), 474; https://doi.org/10.3390/antiox13040474 - 17 Apr 2024
Abstract
Osteoporosis is a bone-debilitating disease, demonstrating a higher prevalence in post-menopausal women due to estrogen deprivation. One of the main mechanisms underlying menopause-related bone loss is oxidative stress. S-allylmercapto-N-acetylcysteine (ASSNAC) is a nuclear factor erythroid 2-related factor 2 (Nrf2) activator
[...] Read more.
Osteoporosis is a bone-debilitating disease, demonstrating a higher prevalence in post-menopausal women due to estrogen deprivation. One of the main mechanisms underlying menopause-related bone loss is oxidative stress. S-allylmercapto-N-acetylcysteine (ASSNAC) is a nuclear factor erythroid 2-related factor 2 (Nrf2) activator and cysteine supplier, previously shown to have anti-oxidation protective effects in cultured cells and animal models. Here, we studied the therapeutic potential of ASSNAC with and without Alendronate in ovariectomized (OVX) female mice. The experimental outcome included (i) femur and L3 lumbar vertebra morphometry via Micro-Computed Tomography (μCT); (ii) bone remodeling (formation vs. resorption); and (iii) oxidative stress markers in bone marrow (BM) cells. Four weeks after OVX, there was a significant bone loss that remained evident after 8 weeks, as demonstrated via µCT in the femur (cortical and trabecular bone compartments) and vertebra (trabecular bone). ASSNAC at a dose of 50 mg/Kg/day prevented bone loss after the four-week treatment but had no significant effect after 8 weeks, while ASSNAC at a dose of 20 mg/Kg/day significantly protected against bone loss after 8 weeks of treatment. Alendronate prevented ovariectomy-induced bone loss, and combining it with ASSNAC further augmented this effect. OVX mice demonstrated high serum levels of both C-terminal cross-linked telopeptides of type I collagen (CTX) (bone resorption) and procollagen I N-terminal propeptide (P1NP) (bone formation) after 2 weeks, and these returned to control levels after 8 weeks. Alendronate, ASSNAC and their combination decreased CTX and increased P1NP. Alendronate induced oxidative stress as reflected by decreased glutathione and increased malondialdehyde (MDA) levels, and combining it with ASSNAC partially attenuated these changes. These results portray the therapeutic potential of ASSNAC for the management of post-menopausal osteoporosis. Furthermore, ASSNAC ameliorates the Alendronate-associated oxidative stress, suggesting its potential to prevent Alendronate side effects as well as improve its bone-protective effect.
Full article
(This article belongs to the Special Issue Free Radicals, Antioxidants and Oxidative Stress in Aging and Age-Related Diseases)
►▼
Show Figures
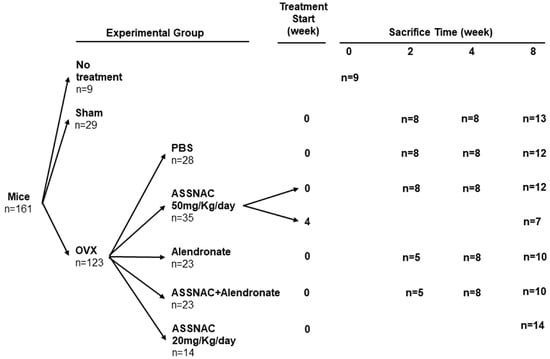
Figure 1
Open AccessCommunication
Redox Regulation of Phosphatase and Tensin Homolog by Bicarbonate and Hydrogen Peroxide: Implication of Peroxymonocarbonate in Cell Signaling
by
Vu Hoang Trinh, Jin-Myung Choi, Thang Nguyen Huu, Dhiraj Kumar Sah, Hyun-Joong Yoon, Sang-Chul Park, Yu-Seok Jung, Young-Keun Ahn, Kun-Ho Lee and Seung-Rock Lee
Antioxidants 2024, 13(4), 473; https://doi.org/10.3390/antiox13040473 - 17 Apr 2024
Abstract
Phosphatase and tensin homolog (PTEN) is a negative regulator of the phosphoinositide 3-kinases/protein kinase B (PI3K/AKT) signaling pathway. Notably, its active site contains a cysteine residue that is susceptible to oxidation by hydrogen peroxide (H2O2). This oxidation inhibits the
[...] Read more.
Phosphatase and tensin homolog (PTEN) is a negative regulator of the phosphoinositide 3-kinases/protein kinase B (PI3K/AKT) signaling pathway. Notably, its active site contains a cysteine residue that is susceptible to oxidation by hydrogen peroxide (H2O2). This oxidation inhibits the phosphatase function of PTEN, critically contributing to the activation of the PI3K/AKT pathway. Upon the stimulation of cell surface receptors, the activity of NADPH oxidase (NOX) generates a transient amount of H2O2, serving as a mediator in this pathway by oxidizing PTEN. The mechanism underlying this oxidation, occurring despite the presence of highly efficient and abundant cellular oxidant-protecting and reducing systems, continues to pose a perplexing conundrum. Here, we demonstrate that the presence of bicarbonate (HCO3−) promoted the rate of H2O2-mediated PTEN oxidation, probably through the formation of peroxymonocarbonate (HCO4−), and consequently potentiated the phosphorylation of AKT. Acetazolamide (ATZ), a carbonic anhydrase (CA) inhibitor, was shown to diminish the oxidation of PTEN. Thus, CA can also be considered as a modulator in this context. In essence, our findings consolidate the crucial role of HCO3− in the redox regulation of PTEN by H2O2, leading to the presumption that HCO4− is a signaling molecule during cellular physiological processes.
Full article
(This article belongs to the Special Issue Reactive Oxygen Species in Different Biological Processes—Second Edition)
►▼
Show Figures
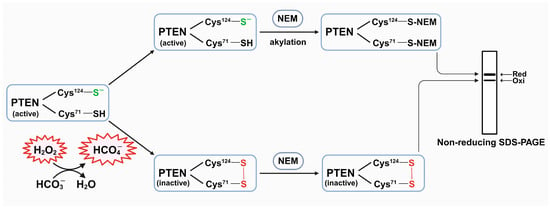
Figure 1
Open AccessArticle
Tributyrin Supplementation Rescues Chronic–Binge Ethanol-Induced Oxidative Stress in the Gut–Lung Axis in Mice
by
Anthony Santilli, David Shapiro, Yingchun Han, Naseer Sangwan and Gail A. M. Cresci
Antioxidants 2024, 13(4), 472; https://doi.org/10.3390/antiox13040472 - 17 Apr 2024
Abstract
Excessive alcohol consumption increases the severity and worsens outcomes of pulmonary infections, often due to oxidative stress and tissue damage. While the mechanism behind this relationship is multifaceted, recent evidence suggests ethanol-induced changes to the gut microbiome impact the gut–lung axis. To assess
[...] Read more.
Excessive alcohol consumption increases the severity and worsens outcomes of pulmonary infections, often due to oxidative stress and tissue damage. While the mechanism behind this relationship is multifaceted, recent evidence suggests ethanol-induced changes to the gut microbiome impact the gut–lung axis. To assess this, a chronic–binge ethanol feeding mouse model was used to determine how ethanol altered the gut microbiome, small intestinal epithelial barrier, and immune responses, as well as neutrophil abundance and oxidative stress in the lungs, and how supporting gut health with tributyrin supplementation during chronic–binge ethanol exposure affected these responses. We found that ethanol consumption altered gut bacterial taxa and metabolic processes, distorted small intestinal immune responses, and induced both bacteria and endotoxin translocation into the lymphatic and circulatory systems. These changes were associated with increased neutrophil (Ly6G) presence and markers of oxidative stress, lipocalin-2 and myeloperoxidase, in the lungs. Importantly, tributyrin supplementation during ethanol exposure rescued gut bacterial function (p < 0.05), small intestinal barrier integrity, and immune responses, as well as reducing both Ly6G mRNA (p < 0.05) and lipocalin-2 mRNA (p < 0.01) in the lungs. These data suggest ethanol-associated disruption of gut homeostasis influenced the health of the lungs, and that therapeutics supporting gut health may also support lung health.
Full article
(This article belongs to the Special Issue Alcohol-Induced Oxidative Stress in Health and Disease)
►▼
Show Figures
Figure 1
Open AccessCorrection
Correction: Abbas et al. Bioactive Compounds, Antioxidant, Anti-Inflammatory, Anti-Cancer, and Toxicity Assessment of Tribulus terrestris—In Vitro and In Vivo Studies. Antioxidants 2022, 11, 1160
by
Malik Waseem Abbas, Mazhar Hussain, Saeed Akhtar, Tariq Ismail, Muhammad Qamar, Zahid Shafiq and Tuba Esatbeyoglu
Antioxidants 2024, 13(4), 471; https://doi.org/10.3390/antiox13040471 - 17 Apr 2024
Abstract
►▼
Show Figures
In the original publication [...]
Full article
Figure 2
Open AccessReview
Tricarboxylic Acid Cycle Regulation of Metabolic Program, Redox System, and Epigenetic Remodeling for Bone Health and Disease
by
Wei-Shiung Lian, Re-Wen Wu, Yu-Han Lin, Yu-Shan Chen, Holger Jahr and Feng-Sheng Wang
Antioxidants 2024, 13(4), 470; https://doi.org/10.3390/antiox13040470 - 17 Apr 2024
Abstract
Imbalanced osteogenic cell-mediated bone gain and osteoclastic remodeling accelerates the development of osteoporosis, which is the leading risk factor of disability in the elderly. Harmonizing the metabolic actions of bone-making cells and bone resorbing cells to the mineralized matrix network is required to
[...] Read more.
Imbalanced osteogenic cell-mediated bone gain and osteoclastic remodeling accelerates the development of osteoporosis, which is the leading risk factor of disability in the elderly. Harmonizing the metabolic actions of bone-making cells and bone resorbing cells to the mineralized matrix network is required to maintain bone mass homeostasis. The tricarboxylic acid (TCA) cycle in mitochondria is a crucial process for cellular energy production and redox homeostasis. The canonical actions of TCA cycle enzymes and intermediates are indispensable in oxidative phosphorylation and adenosine triphosphate (ATP) biosynthesis for osteogenic differentiation and osteoclast formation. Knockout mouse models identify these enzymes’ roles in bone mass and microarchitecture. In the noncanonical processes, the metabolites as a co-factor or a substrate involve epigenetic modification, including histone acetyltransferases, DNA demethylases, RNA m6A demethylases, and histone demethylases, which affect genomic stability or chromatin accessibility for cell metabolism and bone formation and resorption. The genetic manipulation of these epigenetic regulators or TCA cycle intermediate supplementation compromises age, estrogen deficiency, or inflammation-induced bone mass loss and microstructure deterioration. This review sheds light on the metabolic functions of the TCA cycle in terms of bone integrity and highlights the crosstalk of the TCA cycle and redox and epigenetic pathways in skeletal tissue metabolism and the intermediates as treatment options for delaying osteoporosis.
Full article
(This article belongs to the Special Issue Genetics and Epigenetic Modifications on Metabolic Diseases Oxidative Related)
►▼
Show Figures
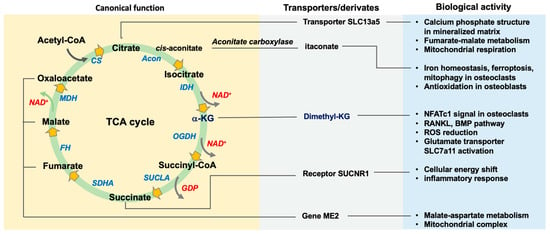
Figure 1
Open AccessArticle
Antioxidant Activity and Mechanism of Action of Amwaprin: A Protein in Honeybee (Apis mellifera) Venom
by
Bo-Yeon Kim, Kwang-Sik Lee and Byung-Rae Jin
Antioxidants 2024, 13(4), 469; https://doi.org/10.3390/antiox13040469 - 17 Apr 2024
Abstract
►▼
Show Figures
Bee venom contains several bioactive components, including enzymatic and non-enzymatic proteins. There is increasing interest in the bioactive components of bee venom since they have exhibited various pharmacological effects. Recently, Apis mellifera waprin (Amwaprin) was identified as a novel protein in Apis mellifera
[...] Read more.
Bee venom contains several bioactive components, including enzymatic and non-enzymatic proteins. There is increasing interest in the bioactive components of bee venom since they have exhibited various pharmacological effects. Recently, Apis mellifera waprin (Amwaprin) was identified as a novel protein in Apis mellifera (honeybee) venom and characterized as an antimicrobial agent. Herein, the novel biological function of Amwaprin as an antioxidant is described. In addition, the antioxidant effects of Amwaprin in mammalian cells were investigated. Amwaprin inhibited the growth of, oxidative stress-induced cytotoxicity, and inflammatory response in mammalian NIH-3T3 cells. Amwaprin decreased caspase-3 activity during oxidative stress and exhibited protective activity against oxidative stress-induced cell apoptosis in NIH-3T3 and insect Sf9 cells. The mechanism underlying the cell protective effect of Amwaprin against oxidative stress is due to its direct binding to the cell membrane. Furthermore, Amwaprin demonstrated radical-scavenging activity and protected against oxidative DNA damage. These results suggest that the antioxidant capacity of Amwaprin is attributed to the synergistic effects of its radical-scavenging action and cell shielding, indicating its novel role as an antioxidant agent.
Full article
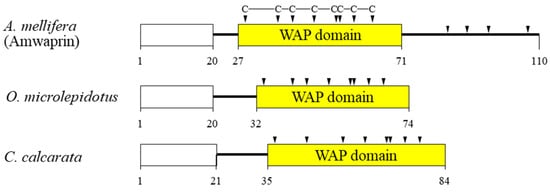
Figure 1
Open AccessArticle
18Beta-Glycyrrhetinic Acid Attenuates H2O2-Induced Oxidative Damage and Apoptosis in Intestinal Epithelial Cells via Activating the PI3K/Akt Signaling Pathway
by
Cui Ma, Fuxi Wang, Jiawei Zhu, Shiyi Wang, Yaqing Liu, Jianfang Xu, Qingyu Zhao, Yuchang Qin, Wei Si and Junmin Zhang
Antioxidants 2024, 13(4), 468; https://doi.org/10.3390/antiox13040468 - 16 Apr 2024
Abstract
Oxidative stress causes gut dysfunction and is a contributing factor in several intestinal disorders. Intestinal epithelial cell survival is essential for maintaining human and animal health under oxidative stress. 18beta-Glycyrrhetinic acid (GA) is known to have multiple beneficial effects, including antioxidant activity; however,
[...] Read more.
Oxidative stress causes gut dysfunction and is a contributing factor in several intestinal disorders. Intestinal epithelial cell survival is essential for maintaining human and animal health under oxidative stress. 18beta-Glycyrrhetinic acid (GA) is known to have multiple beneficial effects, including antioxidant activity; however, the underlying molecular mechanisms have not been well established. Thus, the present study evaluated the therapeutic effects of GA on H2O2-induced oxidative stress in intestinal porcine epithelial cells. The results showed that pretreatment with GA (100 nM for 16 h) significantly increased the levels of several antioxidant enzymes and reduced corresponding intracellular levels of reactive oxidative species and malondialdehyde. GA inhibited cell apoptosis via activating the phosphoinositide 3-kinase/protein kinase B (PI3K/Akt) signaling pathway, as confirmed by RNA sequencing. Further analyses demonstrated that GA upregulated the phosphorylation levels of PI3K and Akt and the protein level of B cell lymphoma 2, whereas it downregulated Cytochrome c and tumor suppressor protein p53 levels. Moreover, molecular docking analysis predicted the binding of GA to Vasoactive intestinal peptide receptor 1, a primary membrane receptor, to activate the PI3K/Akt signaling pathway. Collectively, these results revealed that GA protected against H2O2-induced oxidative damage and cell apoptosis via activating the PI3K/Akt signaling pathway, suggesting the potential therapeutic use of GA to alleviate oxidative stress in humans/animals.
Full article
(This article belongs to the Section Health Outcomes of Antioxidants and Oxidative Stress)
►▼
Show Figures
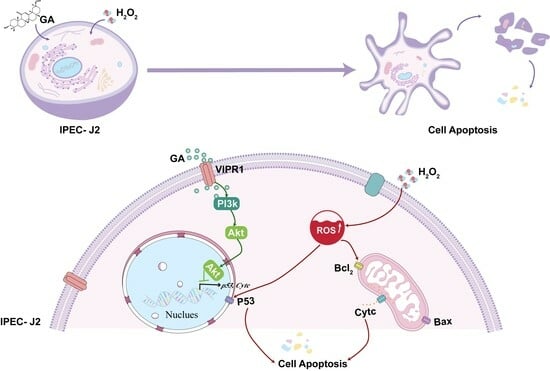
Graphical abstract
Open AccessArticle
Inflammatory Cytokines and Chemokines Are Synergistically Induced in a ROS-Dependent Manner by a Co-Culture of Corneal Epithelial Cells and Neutrophil-like Cells in the Presence of Particulate Matter
by
Zirui Zeng, Yasuhiro Yoshida, Duo Wang, Yuri Fujii, Mengyue Shen, Tatsuya Mimura and Yoshiya Tanaka
Antioxidants 2024, 13(4), 467; https://doi.org/10.3390/antiox13040467 - 16 Apr 2024
Abstract
Ocular exposure to particulate matter (PM) causes local inflammation; however, the influence of neutrophils on PM-induced ocular inflammation is still not fully understood. In this study, we constructed a system to investigate the role of PM in ocular inflammation using a co-culture of
[...] Read more.
Ocular exposure to particulate matter (PM) causes local inflammation; however, the influence of neutrophils on PM-induced ocular inflammation is still not fully understood. In this study, we constructed a system to investigate the role of PM in ocular inflammation using a co-culture of human corneal epithelial cells (HCE-T) and differentiation-induced neutrophils (dHL-60). To investigate whether HCE-T directly endocytosed PM, we performed a holographic analysis, which showed the endocytosis of PM in HCE-T. The cytokines and chemokines produced by HCE-T were measured using an ELISA. HCE-T treated with PM produced IL-6 and IL-8, which were inhibited by N-Acetyl-L-cysteine (NAC), suggesting the involvement of ROS. Their co-culture with dHL-60 enhanced their production of IL-6, IL-8, and MCP-1. This suggests an inflammatory loop involving intraocular corneal epithelial cells and neutrophils. These cytokines and chemokines are mainly regulated by NF-κB. Therefore, this co-culture system was examined in the presence of an IKK inhibitor known to downregulate NF-κB activity. The IKK inhibitor dramatically suppressed the production of these factors in co-culture supernatants. The results suggest that the inflammatory loop observed in the co-culture is mediated through ROS and the transcription factor NF-κB. Thus, the co-culture system is considered a valuable tool for analyzing complex inflammations.
Full article
(This article belongs to the Special Issue Oxidative Stress Induced by Air Pollution)
►▼
Show Figures
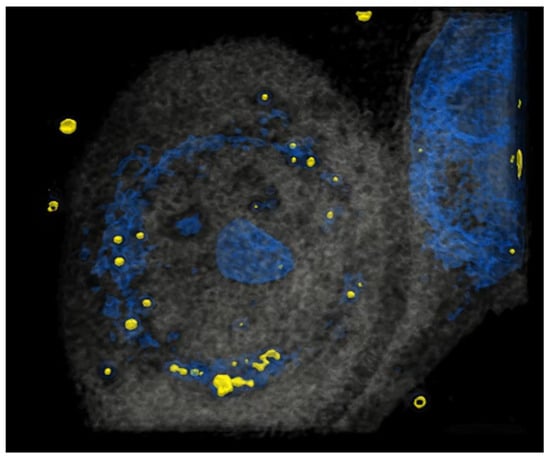
Figure 1

Journal Menu
► ▼ Journal Menu-
- Antioxidants Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Antioxidants, IJPB, Molecules, Pharmaceuticals, Plants
Plants Volatile Compounds
Topic Editors: Dario Kremer, Igor Jerković, Valerija DunkićDeadline: 30 April 2024
Topic in
IJMS, Molecules, Antioxidants, CIMB, BioChem, Foods
Biological Activities and Chemical Composition of Bee Products and Derivatives
Topic Editors: Maria da Graça Costa G. Miguel, Ofelia AnjosDeadline: 30 June 2024
Topic in
Analytica, Antioxidants, Biomedicines, Nutrients, Separations
Discovery of Bioactive Compounds from Natural Organisms and Their Molecular Mechanisms against Diseases
Topic Editors: Jun Dang, Tengfei Ji, Xinxin ZhangDeadline: 31 July 2024
Topic in
Analytica, Antioxidants, Applied Sciences, Molecules, Separations
New Analytical Methods in Plant Active Components Analysis
Topic Editors: Filomena Lelario, Giuliana Bianco, Radosław KowalskiDeadline: 31 October 2024

Conferences
Special Issues
Special Issue in
Antioxidants
Oxidative Stress Markers and Endothelial Dysfunction in Pre- and Perinatal Period
Guest Editors: Endre Sulyok, József BódisDeadline: 20 April 2024
Special Issue in
Antioxidants
Advances for the NO/NOS System
Guest Editors: Adriana Bulboaca, Sorana D. BolboacăDeadline: 30 April 2024
Special Issue in
Antioxidants
Oxidative Stress and NRF2 in Health and Disease
Guest Editor: Lidija MilkovićDeadline: 20 May 2024
Special Issue in
Antioxidants
Antioxidant Activity of Fermented Foods and Food Microorganisms
Guest Editor: Myung-Ji SeoDeadline: 31 May 2024
Topical Collections
Topical Collection in
Antioxidants
Advances in Antioxidant Ingredients from Natural Products
Collection Editors: Carla Pereira, Lillian Barros