Prognostic Significance of the Royal Marsden Hospital (RMH) Score in Patients with Cancer: A Systematic Review and Meta-Analysis
Abstract
:Simple Summary
Abstract
1. Introduction
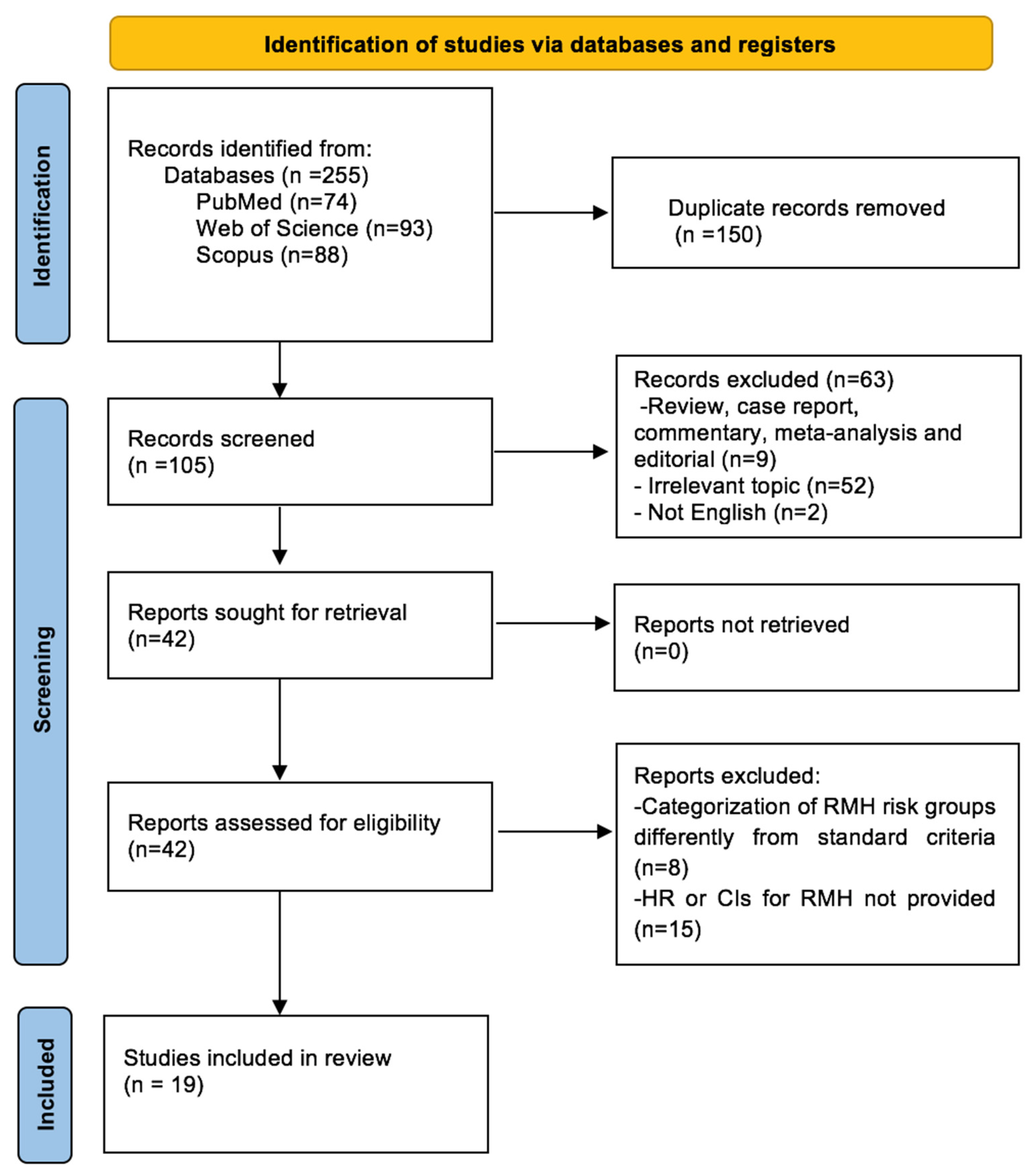
2. Materials and Methods
2.1. Literature Search
2.2. Inclusion and Exclusion Criteria
2.3. Data Extraction and Study Selection
2.4. Meta-Analysis
3. Results
3.1. Study Characteristics
3.2. Association between RMH Score and OS
3.3. Association between RMH Score and PFS
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2024. [Google Scholar] [CrossRef] [PubMed]
- Storme, G. Are We Losing the Final Fight against Cancer? Cancers 2024, 16, 421. [Google Scholar] [CrossRef]
- Scotté, F.; Taylor, A.; Davies, A. Supportive Care: The “Keystone” of Modern Oncology Practice. Cancers 2023, 15, 3860. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wang, Z.; Liu, G.; Liu, Z.; Lu, H.; Ji, S. Assessment of Naples prognostic score in predicting survival for small cell lung cancer patients treated with chemoradiotherapy. Ann. Med. 2023, 55, 2242254. [Google Scholar] [CrossRef] [PubMed]
- Guven, D.C.; Aktepe, O.H.; Taban, H.; Aktas, B.Y.; Guner, G.; Yildirim, H.C.; Sahin, T.K.; Aksun, M.S.; Dizdar, O.; Aksoy, S.; et al. Lower prognostic nutritional index is associated with poorer survival in patients receiving immune checkpoint inhibitors. Biomark. Med. 2021, 15, 1123–1130. [Google Scholar] [CrossRef] [PubMed]
- Guven, D.C.; Sahin, T.K.; Erul, E.; Kilickap, S.; Gambichler, T.; Aksoy, S. The Association between the Pan-Immune-Inflammation Value and Cancer Prognosis: A Systematic Review and Meta-Analysis. Cancers 2022, 14, 2675. [Google Scholar] [CrossRef] [PubMed]
- Arkenau, H.T.; Barriuso, J.; Olmos, D.; Ang, J.E.; de Bono, J.; Judson, I.; Kaye, S. Prospective validation of a prognostic score to improve patient selection for oncology phase I trials. J. Clin. Oncol. 2009, 27, 2692–2696. [Google Scholar] [CrossRef] [PubMed]
- Minami, S.; Ihara, S.; Ikuta, S.; Komuta, K. Gustave Roussy Immune Score and Royal Marsden Hospital Prognostic Score Are Biomarkers of Immune-Checkpoint Inhibitor for Non-Small Cell Lung Cancer. World J. Oncol. 2019, 10, 90–100. [Google Scholar] [CrossRef]
- Minami, S.; Ihara, S.; Komuta, K. Gustave Roussy Immune Score and Royal Marsden Hospital Prognostic Score Are Prognostic Markers for Extensive Disease of Small Cell Lung Cancer. World J. Oncol. 2020, 11, 98–105. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Brooke, B.S.; Schwartz, T.A.; Pawlik, T.M. MOOSE Reporting Guidelines for Meta-analyses of Observational Studies. JAMA Surg. 2021, 156, 787–788. [Google Scholar] [CrossRef]
- Chau, N.G.; Florescu, A.; Chan, K.K.; Wang, L.; Chen, E.X.; Bedard, P.; Oza, A.M.; Siu, L.L. Early mortality and overall survival in oncology phase I trial participants: Can we improve patient selection? BMC Cancer 2011, 11, 426. [Google Scholar] [CrossRef] [PubMed]
- Garrido-Laguna, I.; Janku, F.; Vaklavas, C.; Falchook, G.S.; Fu, S.; Hong, D.S.; Naing, A.; Tsimberidou, A.M.; Wen, S.; Kurzrock, R. Validation of the Royal Marsden Hospital prognostic score in patients treated in the Phase I Clinical Trials Program at the MD Anderson Cancer Center. Cancer 2012, 118, 1422–1428. [Google Scholar] [CrossRef]
- Hong, D.S.; Patel, J.C.; Wheler, J.; Naing, A.; Garrido-Laguna, I.; Falchook, G.; Fu, S.; Tsimberidou, A.M.; Kopetz, S.; Win, S.; et al. Outcomes in 144 patients with colorectal cancer treated in a phase I clinic: The MD Anderson Cancer Center experience. Clin. Color. Cancer 2012, 11, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Wheler, J.; Tsimberidou, A.M.; Hong, D.; Naing, A.; Falchook, G.; Piha-Paul, S.; Fu, S.; Moulder, S.; Stephen, B.; Wen, S.; et al. Survival of 1,181 patients in a phase I clinic: The MD Anderson Clinical Center for targeted therapy experience. Clin. Cancer Res. 2012, 18, 2922–2929. [Google Scholar] [CrossRef]
- Ferté, C.; Fernandez, M.; Hollebecque, A.; Koscielny, S.; Levy, A.; Massard, C.; Balheda, R.; Bot, B.; Gomez-Roca, C.; Dromain, C.; et al. Tumor growth rate is an early indicator of antitumor drug activity in phase I clinical trials. Clin. Cancer Res. 2014, 20, 246–252. [Google Scholar] [CrossRef]
- Khan, K.H.; Yap, T.A.; Ring, A.; Molife, L.R.; Bodla, S.; Thomas, K.; Zivi, A.; Smith, A.; Judson, I.; Banerji, U.; et al. Phase I trial outcomes in older patients with advanced solid tumours. Br. J. Cancer 2016, 114, 262–268. [Google Scholar] [CrossRef]
- Livingston, J.A.; Hess, K.R.; Naing, A.; Hong, D.S.; Patel, S.; Benjamin, R.S.; Ludwig, J.A.; Conley, A.; Herzog, C.E.; Anderson, P.; et al. Validation of prognostic scoring and assessment of clinical benefit for patients with bone sarcomas enrolled in phase I clinical trials. Oncotarget 2016, 7, 64421–64430. [Google Scholar] [CrossRef] [PubMed]
- Bando, H.; Rubinstein, L.; Harris, P.; Yoshino, T.; Doi, T.; Ohtsu, A.; Welch, J.; Takebe, N. Analysis of esophagogastric cancer patients enrolled in the National Cancer Institute Cancer Therapy Evaluation Program sponsored phase 1 trials. Gastric Cancer 2017, 20, 481–488. [Google Scholar] [CrossRef]
- Sun, R.; Champiat, S.; Dercle, L.; Aspeslagh, S.; Castanon, E.; Limkin, E.J.; Baldini, C.; Postel-Vinay, S.; Hollebecque, A.; Massard, C.; et al. Baseline lymphopenia should not be used as exclusion criteria in early clinical trials investigating immune checkpoint blockers (PD-1/PD-L1 inhibitors). Eur. J. Cancer 2017, 84, 202–211. [Google Scholar] [CrossRef]
- Maymani, H.; Hess, K.; Groisberg, R.; Hong, D.S.; Naing, A.; Piha-Paul, S.; Janku, F.; Fu, S.; Tsimberidou, A.M.; Pant, S.; et al. Predicting outcomes in patients with advanced non-small cell lung cancer enrolled in early phase immunotherapy trials. Lung Cancer 2018, 120, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Al Darazi, G.; Martin, E.; Delord, J.P.; Korakis, I.; Betrian, S.; Estrabaut, M.; Poublanc, M.; Gomez-Roca, C.; Filleron, T. Improving patient selection for immuno-oncology phase 1 trials: External validation of six prognostic scores in a French Cancer Center. Int. J. Cancer 2021, 148, 2502–2511. [Google Scholar] [CrossRef] [PubMed]
- Becker, T.; Weberpals, J.; Jegg, A.M.; So, W.V.; Fischer, A.; Weisser, M.; Schmich, F.; Rüttinger, D.; Bauer-Mehren, A. An enhanced prognostic score for overall survival of patients with cancer derived from a large real-world cohort. Ann. Oncol. 2020, 31, 1561–1568. [Google Scholar] [CrossRef] [PubMed]
- Kitadai, R.; Okuma, Y.; Hakozaki, T.; Hosomi, Y. The efficacy of immune checkpoint inhibitors in advanced non-small-cell lung cancer with liver metastases. J. Cancer Res. Clin. Oncol. 2020, 146, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Nassif, E.F.; Blay, J.Y.; Massard, C.; Dufresne, A.; Brahmi, M.; Cassier, P.; Ray-Coquard, I.; Pautier, P.; Leary, A.; Sunyach, M.P.; et al. Early phase trials in soft-tissue sarcomas: Clinical benefit of inclusion in early lines of treatment, molecular screening, and histology-driven trials. ESMO Open 2022, 7, 100425. [Google Scholar] [CrossRef] [PubMed]
- Loh, J.; Wu, J.; Chieng, J.; Chan, A.; Yong, W.P.; Sundar, R.; Lee, S.C.; Wong, A.; Lim, J.S.J.; Tan, D.S.P.; et al. Clinical outcome and prognostic factors for Asian patients in Phase I clinical trials. Br. J. Cancer 2023, 128, 1514–1520. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.X.; Espin-Garcia, O.; Bach, Y.; Aoyama, H.; Allen, M.J.; Wang, X.; Darling, G.E.; Yeung, J.; Swallow, C.J.; Brar, S.; et al. Comparison of Four Clinical Prognostic Scores in Patients with Advanced Gastric and Esophageal Cancer. Oncologist 2023, 28, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Greten, F.R.; Grivennikov, S.I. Inflammation and Cancer: Triggers, Mechanisms, and Consequences. Immunity 2019, 51, 27–41. [Google Scholar] [CrossRef] [PubMed]
- Arihara, Y.; Takada, K.; Murase, K.; Kawamura, K.; Kakiuchi, A.; Kurose, M.; Sasaki, T.; Ogi, K.; Yamazaki, M.; Miyazaki, A.; et al. Inflammation and malnutrition as markers of poor outcomes in head and neck cancer patients treated with nivolumab. Acta Otolaryngol. 2023, 143, 714–720. [Google Scholar] [CrossRef]
- Huai, Q.; Luo, C.; Song, P.; Bie, F.; Bai, G.; Li, Y.; Liu, Y.; Chen, X.; Zhou, B.; Sun, X.; et al. Peripheral blood inflammatory biomarkers dynamics reflect treatment response and predict prognosis in non-small cell lung cancer patients with neoadjuvant immunotherapy. Cancer Sci. 2023, 114, 4484–4498. [Google Scholar] [CrossRef]
- Pu, D.; Xu, Q.; Zhou, L.Y.; Zhou, Y.W.; Liu, J.Y.; Ma, X.L. Inflammation-nutritional markers of peripheral blood could predict survival in advanced non-small-cell lung cancer patients treated with PD-1 inhibitors. Thorac. Cancer 2021, 12, 2914–2923. [Google Scholar] [CrossRef]
- Arkenau, H.T.; Olmos, D.; Ang, J.E.; de Bono, J.; Judson, I.; Kaye, S. Clinical outcome and prognostic factors for patients treated within the context of a phase I study: The Royal Marsden Hospital experience. Br. J. Cancer 2008, 98, 1029–1033. [Google Scholar] [CrossRef] [PubMed]
- Bigot, F.; Castanon, E.; Baldini, C.; Hollebecque, A.; Carmona, A.; Postel-Vinay, S.; Angevin, E.; Armand, J.P.; Ribrag, V.; Aspeslagh, S.; et al. Prospective validation of a prognostic score for patients in immunotherapy phase I trials: The Gustave Roussy Immune Score (GRIm-Score). Eur. J. Cancer 2017, 84, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, E.C.; Resano, L.; Barba, L.; Vidal, C.; Arraez, A.S.; Martin, A.G.; Ponz-Sarvisé, M. Comparison of the predictive power of survival of the Royal Marsden Score (RMS), the Gustave Rousy Immunescore (GRIM score), the MDACC score (MDAS) and the LIPI score in phase I trials patients. J. Clin. Oncol. 2020, 38, e14094. [Google Scholar] [CrossRef]
- Veasey Rodrigues, H.; Baracos, V.E.; Wheler, J.J.; Parsons, H.A.; Hong, D.S.; Naing, A.; Fu, S.; Falchoock, G.; Tsimberidou, A.M.; Piha-Paul, S.; et al. Body composition and survival in the early clinical trials setting. Eur. J. Cancer 2013, 49, 3068–3075. [Google Scholar] [CrossRef] [PubMed]
- Sen, S.; Hess, K.; Hong, D.S.; Naing, A.; Piha-Paul, S.; Janku, F.; Fu, S.; Subbiah, I.M.; Liu, H.; Khanji, R.; et al. Development of a prognostic scoring system for patients with advanced cancer enrolled in immune checkpoint inhibitor phase 1 clinical trials. Br. J. Cancer 2018, 118, 763–769. [Google Scholar] [CrossRef]
- Guven, D.C.; Sahin, T.K.; Erul, E.; Cakir, I.Y.; Ucgul, E.; Yildirim, H.C.; Aktepe, O.H.; Erman, M.; Kilickap, S.; Aksoy, S.; et al. The Association between Early Changes in Neutrophil-Lymphocyte Ratio and Survival in Patients Treated with Immunotherapy. J. Clin. Med. 2022, 11, 4523. [Google Scholar] [CrossRef]




| Study, Year | Country | Study Population | Cancer Type | Study Design | Sample Size (RMH Score Low–High) (n) | Primary Treatment | TNM Stage | Multivariate Analysis | Survival Outcomes | Median Follow Up |
|---|---|---|---|---|---|---|---|---|---|---|
| Arkenau, 2009 [7] | UK | Phase 1 Trials | Mixed | P;S | 78 (43–35) | Mixed | Locoregional (%10) Metastatic (%90) | Multivariate | OS | 27.3 weeks |
| Chau, 2011 [12] | Canada | Phase 1 Trials | Mixed | R;S | 233 (191–42) | Mixed | Metastatic | Univariate | OS | 224 days |
| Garrido-Laguna, 2011 [13] | USA | Phase 1 Trials | Mixed | R;S | 229 (181–48) | Mixed | Metastatic | Multivariate | OS | N/A |
| Hong, 2012 [14] | USA | Phase 1 Trials | CRC | R;S | 144 (82–62) | Mixed | Metastatic | Multivariate | OS | N/A |
| Wheler, 2012 [15] | USA | Phase 1 Trials | Mixed | R;S | 1169 (908–261) | Mixed | Mixed | Multivariate | OS | 8.13 months |
| Ferte, 2014 [16] | France | Phase 1 Trials | Mixed | P;M | 188 (123–65) | Mixed | Metastatic | Multivariate | OS, PFS | N/A |
| Khan, 2016 [17] | UK | Phase 1 Trials | Mixed | R;S | 315 (N/A) | Mixed | Metastatic | Multivariate | OS, PFS | N/A |
| Livingston, 2016 [18] | USA | Phase 1 Trials | Sarcomas | R;S | 91 (74–17) | Mixed | Metastatic | Univariate | OS | N/A |
| Bando, 2017 [19] | USA | Phase 1 Trials | Esophagogastric | R;M | 115 (86–29) | Mixed | Metastatic | Multivariate | OS | N/A |
| Sun, 2017 [20] | France | Phase 1 Trials | Mixed | R;S | 167 (102–65) | ICI | Metastatic | Multivariate | OS | 12.9 months |
| Maymani, 2018 [21] | USA | Phase 1 Trials | NSCLC | R;S | 73 (67–6) | ICI | Metastatic | Univariate | OS | 12.3 months |
| Minami, 2019 [8] | Japan | Real-World Cohort | NSCLC | R;S | 74 (44–32) | ICI | Metastatic | Multivariate | OS, PFS | N/A |
| Minami, 2020 [9] | Japan | Real-World Cohort | SCLC | R;S | 128 (54–74) | ICI | Metastatic | Multivariate | OS, PFS | N/A |
| Al Darazi, 2020 [22] | France | Phase 1 Trials | Mixed | R;S | 259 (153–106) | ICI | Metastatic | Univariate | OS | 15 months |
| Becker, 2020 [23] | USA | Real-World Cohort | Mixed | R;M | 122,694 (112,365–10,339) | Mixed | Mixed | Univariate | OS | N/A |
| Kitadai, 2020 [24] | Japan | Real-World Cohort | NSCLC | R;S | 215 (N/A) | ICI | Metastatic | Univariate | OS, PFS | N/A |
| Nassif, 2022 [25] | France | Phase 1 Trials | Sarcomas | R;M | 194 (132–62) | Mixed | Mixed | Multivariate | PFS | 35 months |
| Loh, 2023 [26] | Singapore | Phase 1 Trials | Mixed | R;S | 413 (307–106) | Mixed | Metastatic | Univariate | OS | 2.3 years |
| Ma, 2023 [27] | Canada | Real-World Cohort | Gastric-Esophageal | R;S | 451 (338–113) | Mixed | Metastatic | Multivariate | OS | 9 months |
| Author, Year | Selection | Comparability | Exposure/Outcome | NOS Score |
|---|---|---|---|---|
| Arkenau, 2009 [7] | **** | ** | *** | 9 |
| Chau, 2011 [12] | *** | ** | ** | 7 |
| Garrido-Laguna, 2011 [13] | *** | ** | *** | 8 |
| Hong, 2012 [14] | *** | ** | *** | 8 |
| Wherler, 2012 [15] | *** | ** | *** | 8 |
| Ferte, 2014 [16] | *** | ** | *** | 8 |
| Khan, 2016 [17] | **** | ** | *** | 9 |
| Livingston, 2016 [18] | *** | ** | *** | 8 |
| Bando, 2017 [19] | *** | ** | ** | 7 |
| Sun, 2017 [20] | **** | ** | ** | 8 |
| Maymani, 2018 [21] | **** | ** | ** | 8 |
| Minami, 2019 [8] | *** | * | ** | 6 |
| Minami, 2020 [9] | *** | * | ** | 6 |
| Al Darazi, 2020 [22] | **** | ** | *** | 9 |
| Becker, 2020 [23] | **** | ** | *** | 9 |
| Kitadai, 2020 [24] | **** | ** | ** | 8 |
| Nassif, 2022 [25] | **** | ** | ** | 8 |
| Loh, 2023 [26] | **** | ** | *** | 9 |
| Ma, 2023 [27] | **** | ** | ** | 8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sahin, T.K.; Rizzo, A.; Aksoy, S.; Guven, D.C. Prognostic Significance of the Royal Marsden Hospital (RMH) Score in Patients with Cancer: A Systematic Review and Meta-Analysis. Cancers 2024, 16, 1835. https://doi.org/10.3390/cancers16101835
Sahin TK, Rizzo A, Aksoy S, Guven DC. Prognostic Significance of the Royal Marsden Hospital (RMH) Score in Patients with Cancer: A Systematic Review and Meta-Analysis. Cancers. 2024; 16(10):1835. https://doi.org/10.3390/cancers16101835
Chicago/Turabian StyleSahin, Taha Koray, Alessandro Rizzo, Sercan Aksoy, and Deniz Can Guven. 2024. "Prognostic Significance of the Royal Marsden Hospital (RMH) Score in Patients with Cancer: A Systematic Review and Meta-Analysis" Cancers 16, no. 10: 1835. https://doi.org/10.3390/cancers16101835





