Study of Cyclic Quaternary Ammonium Bromides by B3LYP Calculations, NMR and FTIR Spectroscopies
Abstract
:1. Introduction
2. Results and Discusion
2.1. Synthesis
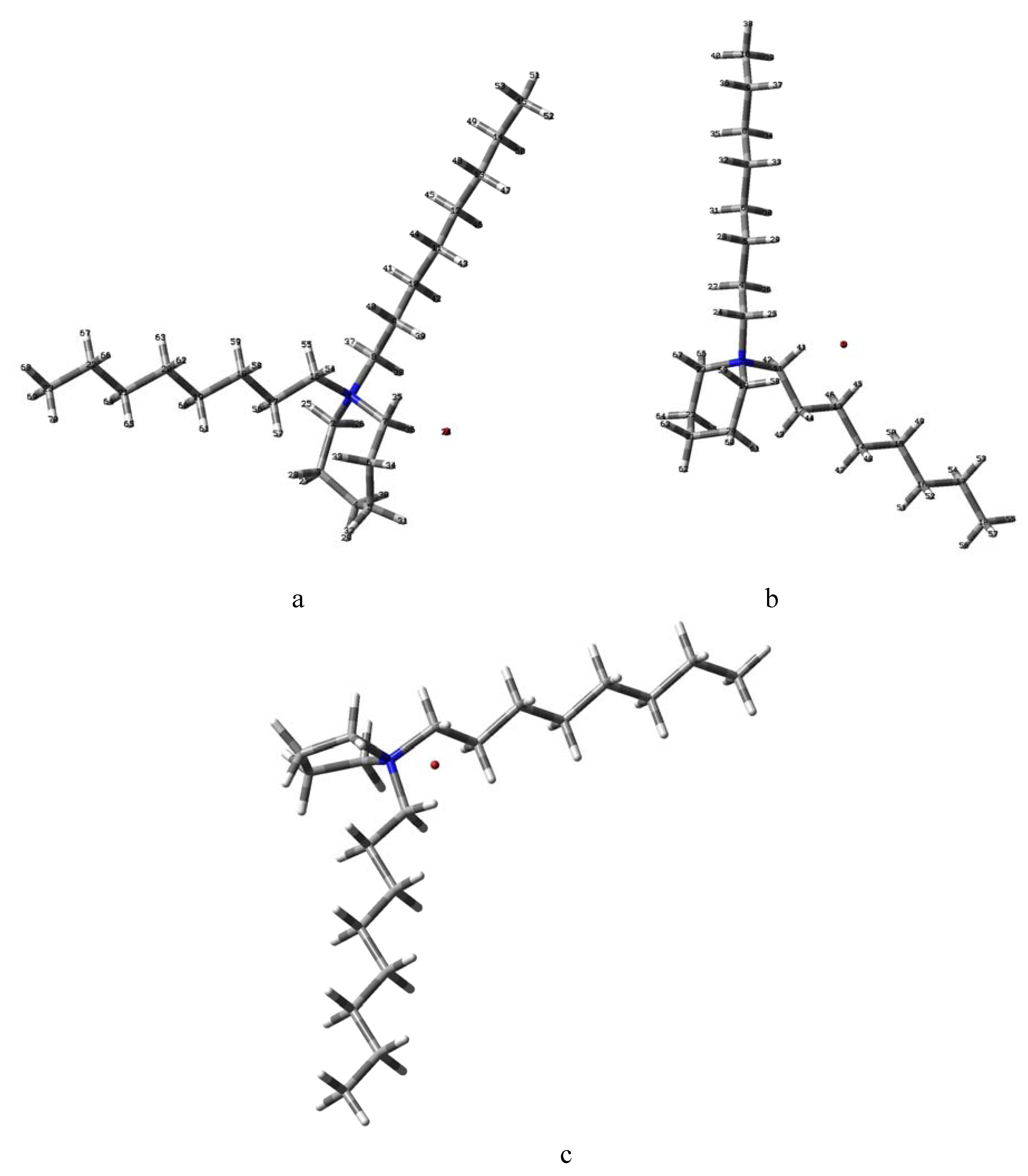
2.2. B3LYP Calculations

| Parameters | 1 | 2 | 3 |
|---|---|---|---|
| Energy (a.u) | -3495.20808 | -3453.27811 | -3413.96044 |
| Dipol moment (Debye) | 13.4951 | 11.4097 | 11.4657 |
| Bond length (Å) | |||
| N+…Br- | 3.888 | 3.709 | 3.674 |
| C(1)-H…Br- | 3.636 | 3.536 | 3.486 |
| C(1’)-H…Br- | 3.686 | ||
| C(4)-H…Br- | 3.551 | 3.570 | 3.616 |
| C(4’)-H…Br- | 3.360 | 3.346 | |
| N-C(1) | 1.535 | 1.538 | 1.532 |
| N-C(1’) | 1.533 | 1.514 | 1.513 |
| N-C(4) | 1.548 | 1.542 | 1.529 |
| N-C(4’) | 1.531 | 1.551 | 1.543 |
| Bond angle (o) | |||
| N-C(1)-C(2) | 119.5 | 115.3 | 106.2 |
| N-C(1’)-C(2’) | 116.9 | 114.2 | 106.2 |
| N-C(4)-C(5) | 117.9 | 116.3 | 115.6 |
| N-C(4’)-C(5’) | 120.2 | 119.9 | 118.6 |
| Dihedral angle (o) | |||
| N-C(1)-C(2)-C(3) | -70.3 | -49.5 | |
| N-C(1’)-C(2’)-C(3’) | 88.6 | ||
| N-C(1’)-C(2’)-C(3) | 57.8 | ||
| N-C(1)-C(2)-C(2’) | -18.2 | ||
| N-C(1’)-C(2’)-C(2) | 25.2 | ||
| N-C(4)-C(5)-C(6) | -176.8 | -177.4 | -176.9 |
| N-C(4’)-C(5’)-C(6’) | -176.5 | -172.3 | -170.0 |
2.3. FTIR and Raman Spectra Study

| Raman | IR | IR(calc.) | INT | Proposed assignment |
|---|---|---|---|---|
| 3437w | νOH | |||
| 2973m | 2956s | 3016 | 43.7 | νCH2 |
| 3013 | 62.3 | νCH2 | ||
| 3011 | 64.9 | νCH2 | ||
| 2999 | 23.3 | νCH2 | ||
| 2987 | 63.2 | νCH2 | ||
| 2974 | 18.6 | νCH2 | ||
| 2943 | 112 | νCH2 | ||
| 2926s | 2925s | 2934 | 61.4 | νCH2 |
| 2919 | 6.4 | νCH2 | ||
| 2864s | 2856s | 2914 | 200 | νCH2 |
| 2781vw | νCH2 | |||
| 2727vw | νCH2 | |||
| 2709vw | 2696vw | νCH2 | ||
| 2669vw | 2670vw | νCH2 | ||
| 1490vw | 1485m | 1501 | 21.9 | νCC |
| 1481 | 4.7 | |||
| 1448w | 1468m | 1467 | 8.0 | νCC |
| 1456 | 7.9 | |||
| 1452 | 2.3 | |||
| 1392w | 1396 | 1.5 | νCN | |
| 1377w | 1376 | 3.5 | νCN | |
| 1372 | 1.6 | |||
| 1358vw | 1360w | 1354 | 4.9 | νCC, βCH2 |
| 1349vw | 1338w | 1344 | 1.4 | βCH2 |
| 1321 | 2.8 | |||
| 1313vw | 1310w | 1308 | 2.7 | βCH2 |
| 1295 | 1.6 | |||
| 1280vw | 1277w | 1281 | 3.4 | νCC |
| 1263vw | 1251vw | 1264 | 2.8 | νCC |
| 1245 | 0,81 | |||
| 1217vw | 1218vw | 1205 | 0.63 | νCC |
| 1186 | 1.3 | |||
| 1141vw | 1141vw | 1169 | 3.3 | νCN |
| 1115vw | 1115vw | 1115 | 2.4 | νCN |
| 1087vw | 1088w | 1075 | 15.3 | γCH2 |
| 1069vw | 1068vw | 1055 | 1.6 | γCH2 |
| 1048vw | 1047vw | 1029 | 3.8 | βCH2 |
| 1014vw | 1007w | 1014 | 2.9 | βCCC |
| 960vw | 962w | 997 | 2.6 | βCCC |
| 930vw | 930vw | 944 | 2.0 | βCCC |
| 933 | 9.7 | |||
| 865vw | 875w | 878 | 13.7 | βCCC |
| 846vw | 847vw | 853 | 4.6 | βCCC |
| 831vw | 832w | βCCC | ||
| 803vw | 800vw | 788 | 2.5 | βCCC |
| 767vw | 765vw | 742 | 19.5 | βCCC |
| 741vw | 738w | βCCC | ||
| 723w | 714 | 4.0 | βCCC | |
| 706vw | βCCC | |||
| 659vw | 651vw | 616 | 1.8 | βCNC |
| 580vw | 578vw | βNCC | ||
| 542vw | 538vw | βCCC | ||
| 498vw | 499vw | 499 | 3.8 | γCCC |
| 403vw | 403vw | 439 | 1.5 | γCCC |
| 375vw | 346 | 1.3 | Lattice mode | |
| 360vw | Lattice mode | |||
| 330vw | Lattice mode | |||
| 303vw | Lattice mode | |||
| 288vw | 224 | 1.2 | Lattice mode | |
| 201vw | 123 | 4.1 | Lattice mode | |
| 86vw | 91 | 0.59 | Lattice mode | |
| 51 | 2.5 |
2.4. 1H-NMR and 13C-NMR Spectra

| δ exp | δcalc | σcalc | δexp | δcalc | σcalc | ||
|---|---|---|---|---|---|---|---|
| C(1) | 63.1 | 57.4 | 118.0 | H(1) | 3.70 | 3.85 | 27.44 |
| C(2) | 22.2 | 23.3 | 155.4 | H(2) | 2.01 | 1.62 | 30.27 |
| C(3) | 27.3 | 21.7 | 157.1 | H(3) | 1.79 | 2.06 | 29.72 |
| C(4) | 61.3 | 64.6 | 110.1 | H(4) | 3.45 | 3.25 | 28.20 |
| C(5) | 22.6 | 25.7 | 152.7 | H(5) | 1.71 | 1.59 | 30.32 |
| C(6) | 26.4 | 27.1 | 151.2 | H(6) | 1.27 | 1.23 | 30.77 |
| C(7) | 29.1 | 30.3 | 147.7 | H(7) | 1.27 | 1.33 | 30.65 |
| C(8) | 29.0 | 30.3 | 147.7 | H(8) | 1.27 | 1.31 | 30.67 |
| C(9) | 31.6 | 32.2 | 145.6 | H(9) | 1.27 | 1.27 | 30.72 |
| C(10) | 22.6 | 24.1 | 154.5 | H(10) | 1.27 | 1.34 | 30.63 |
| C(11) | 14.0 | 12.4 | 167.3 | H(11) | 0.88 | 1.05 | 31.00 |
| a | -0.9113 | a | -0.7865 | ||||
| b | 164.9046 | b | 25.4318 | ||||
| r2 | 0.9622 | r2 | 0.9609 |
| δ exp | δcalc | σcalc | δexp | δcalc | σcalc | ||
|---|---|---|---|---|---|---|---|
| C(1) | 58.9 | 54.8 | 129.6 | H(1) | 3.78 | 3.28 | 27.90 |
| C(2) | 20.0 | 20.5 | 167.8 | H(2) | 1.90 | 1.67 | 29.95 |
| C(3) | 26.4 | 20.5 | 167.8 | H(3) | 1.90 | 1.47 | 30.23 |
| C(4) | 58.1 | 61.0 | 122.7 | H(4) | 3.46 | 3.88 | 27.12 |
| C(5) | 21.7 | 23.2 | 161.8 | H(5) | 1.65 | 1.77 | 29.84 |
| C(6) | 22.5 | 25.7 | 162.1 | H(6) | 1.27 | 1.42 | 30.30 |
| C(7) | 29.0 | 29.2 | 158.1 | H(7) | 1.27 | 1.33 | 30.41 |
| C(8) | 28.9 | 29.2 | 158.1 | H(8) | 1.27 | 1.37 | 30.36 |
| C(9) | 31.6 | 30.7 | 156.5 | H(9) | 1.27 | 1.30 | 30.45 |
| C(10) | 20.6 | 23.1 | 164.9 | H(10) | 1.27 | 1.39 | 30.33 |
| C(11) | 14.0 | 13.6 | 175.5 | H(11) | 0.88 | 1.02 | 30.81 |
| a | 170.9303 | a | 24.0232 | ||||
| b | -0.8962 | b | -0.7758 | ||||
| r2 | 0.9640 | r2 | 0.9168 |
| δ exp | δcalc | σcalc | δexp | δcalc | σcalc | ||
| C(1) | 62.9 | 61.9 | 126.0 | H(1) | 3.85 | 3.69 | 27.57 |
| C(2) | 21.8 | 18.6 | 169.5 | H(2) | 2.31 | 1.77 | 29.81 |
| C(4) | 59.4 | 59.7 | 128.2 | H(4) | 3.43 | 3.32 | 28.00 |
| C(5) | 23.4 | 24.2 | 163.9 | H(5) | 1.70 | 2.43 | 29.04 |
| C(6) | 26.3 | 27.5 | 160.6 | H(6) | 1.27 | 1.30 | 30.49 |
| C(7) | 29.0 | 29.2 | 158.9 | H(7) | 1.27 | 1.26 | 30.24 |
| C(8) | 28.9 | 30.2 | 157.8 | H(8) | 1.27 | 1.25 | 30.41 |
| C(9) | 31.5 | 31.1 | 156.9 | H(9) | 1.27 | 1.40 | 30.40 |
| C(10) | 22.5 | 23.3 | 164.8 | H(10) | 1.27 | 1.18 | 30.35 |
| C(11) | 14.0 | 12.5 | 175.6 | H(11) | 0.88 | 0.91 | 30.80 |
| a | 187.2433 | a | 27.4355 | ||||
| b | -0.9949 | b | -0.8611 | ||||
| r2 | 0.9920 | r2 | 0.9049 |
3. Conclusions
4. Experimental
4.1. General
4.2. Computational Details
4.3. General procedure for the synthesis of N,N-dioctylcycloalkylammonium salts 1-3
Acknowledgments
References
- Domagk, G. A new class of disinfectants. Dtsch. Med. Wochenscher 1935, 61, 829–832. [Google Scholar]
- Block, S.S. Disinfection, Sterilization, and Preservation, 5th ed; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2001. [Google Scholar]
- Dega-Szafran, Z.; Dulewicz, E.; Brycki, B. Synthesis and characterization of 1-carbalkoxymethyl-4-hydroxy-1-methylpiperidinium chlorides. ARKIVOC 2007, VI, 90–102. [Google Scholar]
- Erickson, J.G.; Keps, J.S. Reactions of Long-chain Amines. IV. Preparation of N-Alkylpyrrolidines, N,N-Dialkylpyrrolidinium Chlorides and N,N-Dialkylpiperidinium Chlorides. J. Am. Chem. Soc. 1955, 77, 485–486. [Google Scholar] [CrossRef]
- Ringdahl, B.; Roch, M.; Jenden, D.J. Tertiary 3- and 4-Haloalkylamine Analogues of Oxotremorine as Prodrugs of Potent Muscarinic Agonists. J. Med. Chem. 1988, 31, 160–164. [Google Scholar]
- Chukhadzhyan, É.O.; Chukhadzhyan, Él.O.; Shakhatuni, K.G.; Gevorkyan, N.T. Synthesis of Chlorine-Substituted Naphto- and Benzisoindolinium Salts by Base-catalyzed Intramolecular Cyclization of Ammonium Salts. Chem. Heterocyclic Comp. 1998, 34, 912–915. [Google Scholar] [CrossRef]
- Schmitt, K.D. Surfactant-Mediated Phase Transfer as an Alternative to Propanesultone Alkylation. Formation of a New Class of Zwitterionic Surfactants. J. Org. Chem. 1995, 60, 5474–5479. [Google Scholar] [CrossRef]
- Venkataramu, S.D.; Macdonell, G.D.; Purdum, W.R.; Dilbeck, G.A.; Berlin, K.D. Polyphosphoric Acid Catalyzed Cyclization of Aralkenyl-Substituted Quaternary Ammonium Salts. J. Org. Chem. 1977, 42, 2195–2200. [Google Scholar]
- Walker, M.A. An Unusual Tandem Cyclization-Stevens Rearrangement Mediated by Ph3P/DEAD or Bu3P/ADDP. Tetrahedron Lett. 1996, 37, 8133–8136. [Google Scholar]
- Glaeske, K.W.; West, F.G. Chirality Transfer from Carbon to Nitrogen to Carbon via Cyclic Ammonium Ylides. Org. Lett. 1999, 1, 31–33. [Google Scholar] [CrossRef]
- Manivannan, G. Disinfection and Decontamination; Principles, Applications and Related Issues; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2008. [Google Scholar]
- Menger, F.M.; Keiper, J.S. Gemini surfactants. Angew. Chem. Int. Ed. 2000, 39, 1906–1920. [Google Scholar] [CrossRef]
- Paulus, W. Directory of Microbiocides for the Protection of Materials; A. Handbook, Springer: Dordrecht, The Netherlands, 2005. [Google Scholar]
- Zana, R.; Xia, J. Gemini Surfactants. Synthesis, Interfacial and Solution PhaseBehavior, and Applications; Marcel Dekker: New York, NY, USA, 2004. [Google Scholar]
- Fraise, A.P.; Lambert, P.A.; Maillard, J-Y. Russell, Hugo & Ayliffe’s Principles and Practice of Disinfection, Preservation and Sterilization, 4th ed; Blackwell Publishing: Malden, MA, USA, 2004. [Google Scholar]
- Park, E.J.; Kim, M.H.; Kim, D.Y. Enantioselective Alkylation of â-Keto Esters by Phase-Transfer Catalysis Using Chiral Quaternary Ammonium Salts. J. Org. Chem. 2004, 69, 6897–6899. [Google Scholar] [CrossRef]
- Kim, D.Y.; Huh, S.Ch.; Kim, S.M. Enantioselective Michael reaction of malonates and chalcones by phase-transfer catalysis using chiral quaternary ammonium salt. Tetrahedron Lett. 2001, 42, 6299–6301. [Google Scholar]
- Kim, D.Y.; Park, E.J. Catalytic Enantioselective Fluorination of α-Keto Esters by Phase-Transfer Catalysis Using Chiral Quaternary Ammonium Salts. Org. Lett. 2002, 4, 545–547. [Google Scholar] [CrossRef]
- Niess, B.; Jorgensen, K.A. The asymmetric vinylogous Mannich reaction of dicyanoalkylidenes with α-amido sulfones under phase-transfer conditions. Chem. Commun. 2007, 1620–1622. [Google Scholar]
- Orglmeister, E.; Mallat, T.; Baiker, A. Quaternary ammonium derivatives of cinchonidine as new chiral modifiers for platinum. J. Catal. 2005, 233, 333–341. [Google Scholar] [CrossRef]
- Breistein, P.; Karlsson, S.; Hendenström, E. Chiral pyrrolidinium salts as organocatalysts in the stereoselective 1,4-conjugate addition of N-methylpyrrole to cyclopent-1-ene carbaldehyde. Tetrahedron: Asymmetry 2006, 17, 107–111. [Google Scholar] [CrossRef]
- Fuijmori, T.; Fujii, K.; Kanzaki, R.; Chiba, K.; Yamamoto, H.; Umebayashi, Y.; Ishiguro, S. Conformational structure of room temperature ionic liquid N-butyl-N-methyl-pyrrolidinium bis(trifluoromethanesulfonyl) imide—Raman spectroscopic study and DFT calculations. J. Mol. Liquids 2007, 131-132, 216–224. [Google Scholar] [CrossRef]
- Yoshinawa-Fujita, M.; Johansson, K.; Newman, P.; MacFarlane, D.R.; Forsyth, M. Novel Lewis-base ionic liquids replacing typical anions. Tetrahedron Lett. 20 0647, 2755–2758. [Google Scholar]
- Henderson, W.A.; Passerini, S. Phase Behavior of Ionic Liquid-LiX Mixtures: Pyrrolidinium Cations and TFSI- Anions. Chem. Matter. 2004, 16, 2881–2885. [Google Scholar] [CrossRef]
- Sun, J.; MacFarlane, D.R.; Forsyth, M. A new family of ionic liquids based on the 1-alkyl-2-methyl pyrrolinium cation. Electrochim. Acta 2003, 48, 1707–1711. [Google Scholar] [CrossRef]
- MacFarlane, D.R.; Forsyth, S.A.; Golding, J.; Deacon, G.B. Ionic liquids based on imidazolium, ammonium and pyrrolidinium salts of the dicyanamide anion. Green Chem. 2002, 4, 444–448. [Google Scholar] [CrossRef]
- Sakaaebe, H.; Matsumoto, H. N-Methyl-N-propylpiperidinium bis(trifluoromethanesulfonyl) imide (PP13–TFSI) – novel electrolyte base for Li battery. Electrochem. Communi. 2003, 5, 594–598. [Google Scholar] [CrossRef]
- Tigelaar, D.M.; Meador, M.A.B.; Bennett, W.R. Composite Electrolytes for Lithium Batteries: Ionic Liquids in APTES Cross-Linked Polymers. Macromolecules 2007, 40, 4159–4164. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Montgomery, J.A.; Vreven, T., Jr.; Kudin, K.N.; Burant, J.C.; Millam, J.M.; Iyengar, S.S.; Tomasi, J.; Barone, V.; Mennucci, B.; Cossi, M.; Scalmani, G.; Rega, N.; Petersson, G.A.; Nakatsuji, H.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Klene, M.X.Li.; Knox, J.E.; Hratchian, H.P.; Cross, J.B.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R.E.; Yazyev, O.; Austin, A.J.; Cammi, R.; Pomelli, C.; Ochterski, J.W.; Ayala, P.Y.; Morokuma, K.; Voth, G.A.; Salvador, P.; Dannenberg, J.J.; Zakrzewski, V.G.; Dapprich, S.; Daniels, A.D.; Strain, M.C.; Farkas, O.; Malick, D.K.; Rabuck, A.D.; Raghavachari, K.; Foresman, J.B.; Ortiz, J.V.; Cui, Q.; Baboul, A.G.; Clifford, S.; Cioslowski, J.; Stefanov, B.B.; Liu, G., Liashenko; Piskorz, P.; Komaromi, I.; Martin, R.L.; Fox, D.J.; Keith, T.; Al-Laham, M.A.; Peng, C.Y.; Nanayakkara, A.; Challacombe, M.; Gill, P.M.W.; Johnson, B.; Chen, W.; Wong, M.W.; Gonzalez, C.; Pople, J.A. Gaussian 03, Revision C.01; Gaussian, Inc.: Wallingford, CT, USA, 2004. [Google Scholar]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional thermochemistry. V. Systematic optimization of exchange-correlation functionals. J. Chem. Phys. 1997, 107, 8554–8560. [Google Scholar] [CrossRef]
- Hehre, W.J.; Random, L.; Schleyer, P.v.R.; Pople, J.A. Ab Initio Molecular Orbital Theory; Wiley: New York, NY, USA, 1986. [Google Scholar]
- Wolinski, K.; Hilton, J.F.; Pulay, P. Efficient implementation of the gauge-independent atomic orbital method for NMR chemical shift calculations. J. Am. Chem. Soc. 1990, 112, 8251–8260. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Brycki, B.; Szulc, A.; Kowalczyk, I. Study of Cyclic Quaternary Ammonium Bromides by B3LYP Calculations, NMR and FTIR Spectroscopies. Molecules 2010, 15, 5644-5657. https://doi.org/10.3390/molecules15085644
Brycki B, Szulc A, Kowalczyk I. Study of Cyclic Quaternary Ammonium Bromides by B3LYP Calculations, NMR and FTIR Spectroscopies. Molecules. 2010; 15(8):5644-5657. https://doi.org/10.3390/molecules15085644
Chicago/Turabian StyleBrycki, Bogumił, Adrianna Szulc, and Iwona Kowalczyk. 2010. "Study of Cyclic Quaternary Ammonium Bromides by B3LYP Calculations, NMR and FTIR Spectroscopies" Molecules 15, no. 8: 5644-5657. https://doi.org/10.3390/molecules15085644





