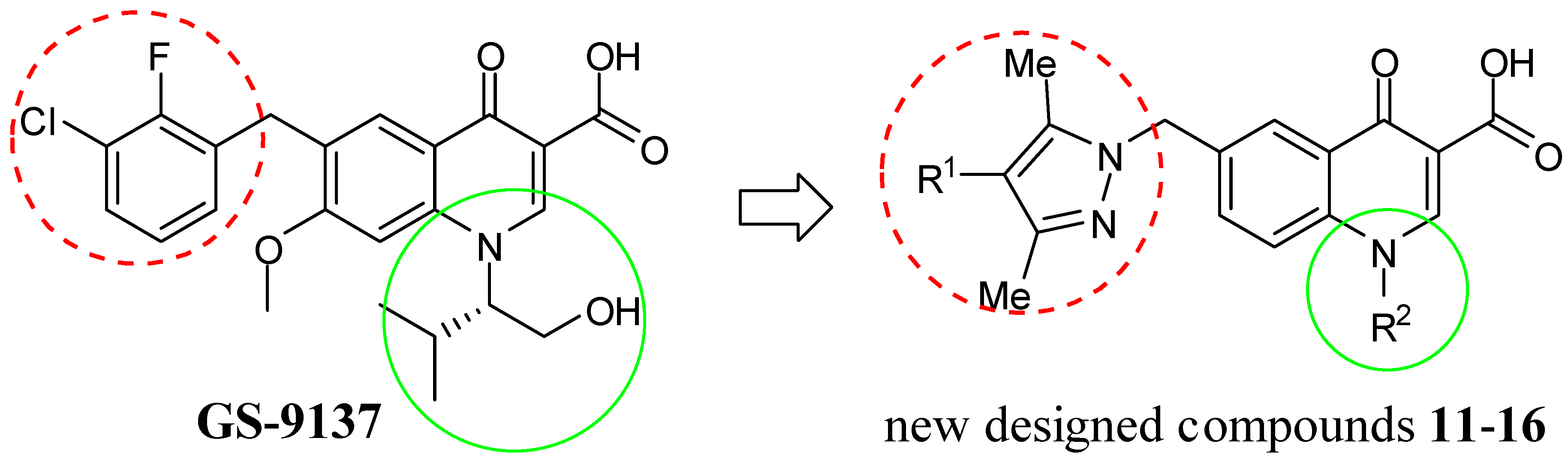
3.2. General Procedure for the Synthesis of Ethyl 1-Substitued-6-(pyrazolylmethyl)-4-oxo-4H-quinoline-3-carboxylates 6–10
A mixture of the appropriate quinolone derivative 5 (2 mmol), the appropriate alkyl halide or benzyl bromide (5.6 mmol), and solid K2CO3 (772 mg, 5.6 mmol) in dry DMF (5 mL) was heated under Ar atmosphere at 60 °C for 12 h and then poured into an ice-water mixture, extracted with DCM and purified by flash chromatography on silica gel eluting with DCM/MeOH (40:1–20:1).
Ethyl6-((3,5-Dimethyl-1H-pyrazol-1-yl)methyl)-1-(oxiran-2-ylmethyl)-4-ox-1,4-dihydroquinoline-3-carboxylate (6a). Yield: 33%; mp: 162–165 °C; 1H-NMR (CDCl3, δ ppm) 1.43 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.16 (s, 3H, CH3-pyrazole), 2.24 (s, 3H, CH3-pyrazole), 2.59 (q, 1H, J = 4.4 Hz, CH2OCHCH2-), 2.92 (t, 1H, J = 4.4 Hz, CH2OCHCH2-), 3.33–3.36 (m, 1H, CH2OCHCH2-), 4.20 (dd, 1H, J = 5.6 and 15.6 Hz, CH2OCHCH2-), 4.42 (q, 2H, J = 7.2 Hz, -CH2CH3), 4.55 (dd, 1H, J = 2.0 and 15.6 Hz, CH2OCHCH2-), 5.32 (s, 2H, -CH2-pyrazole), 5.86 (s, 1H, H-pyrazole), 7.44 (dd, 1H, J = 2.4 and 8.8 Hz, H7), 7.50 (d, 1H, J = 8.8 Hz, H8), 8.32 (d, 1H, J = 2.0 Hz, H5), 8.43 (s, 1H, H2).
Ethyl 6-((4-Chloro-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-1-(oxiran-2-ylmethyl)-4-oxo-1,4-Dihydro- quinoline-3-carboxylate (6b). Yield: 33%; mp: 200–202 °C; 1H-NMR (CDCl3, δ ppm) 1.42 (t, 3H, J = 6.8 Hz, -CH2CH3), 2.15 (s, 3H, CH3-pyrazole), 2.23 (s, 3H, CH3-pyrazole), 2.59 (q, 1H, J = 4.8 Hz, CH2OCHCH2-), 2.92 (t, 1H, J = 4.4 Hz, CH2OCHCH2-), 3.32–3.36 (m, 1H, CH2OCHCH2-), 4.19 (dd, 1H, J = 6.0 and 15.6 Hz, CH2OCHCH2-), 4.42 (q, 2H, J = 7.0 Hz, -CH2CH3), 4.56 (dd, 1H, J = 2.0 and 15.6 Hz, CH2OCHCH2-), 5.31 (s, 2H, -CH2-pyrazole), 7.44 (dd, 1H, J = 2.4 and 8.8 Hz, H7), 7.51 (d, 1H, J = 8.8 Hz, H8), 8.33 (d, 1H, J = 2.0 Hz, H5), 8.43 (s, 1H, H2).
Ethyl 6-((4-Bromo-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-1-(oxiran-2-ylmethyl)-4-oxo-1,4-dihydro-quinoline-3-carboxylate (6c). Yield: 47%; mp: 199–201 °C; 1H-NMR (CDCl3, δ ppm) 1.42 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.16 (s, 3H, CH3-pyrazole), 2.23 (s, 3H, CH3-pyrazole), 2.59 (q, 1H, J = 4.4 Hz, CH2OCHCH2-), 2.92 (t, 1H, J = 4.4 Hz, CH2OCHCH2-), 3.33–3.36 (m, 1H, CH2OCHCH2-), 4.18 (dd, 1H, J = 6.0 and 15.6 Hz, CH2OCHCH2-), 4.41 (q, 2H, J = 7.2 Hz, -CH2CH3), 4.56 (dd, 1H, J = 2.4 and 15.6 Hz, CH2OCHCH2-), 5.33 (s, 2H, -CH2-pyrazole), 7.43 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 7.50 (d, 1H, J = 8.8 Hz, H8), 8.31 (d, 1H, J = 2.0 Hz, H5), 8.42 (s, 1H, H2); 13C-NMR (CDCl3, 100 MHz, δ ppm) 10.4, 12.3, 14.4, 45.2, 49.6, 53.1, 54.8, 60.9, 94.8, 111.6, 116.7, 125.9, 128.8, 131.5, 133.9, 137.4, 138.8, 146.6, 149.4, 165.4, 173.8; ESI-MS: m/z 459.9, 461.9 [M+H]+.
Ethyl 6-((3,5-Dimethyl-1H-pyrazol-1-yl)methyl)-1-(2-methoxy-2-oxoethyl)-4-oxo-1,4-dihydroquino-line-3-carboxylate (7a). Yield: 53%; mp: 225–228 °C; 1H-NMR (CDCl3, δ ppm) 1.41 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.16 (s, 3H, CH3-pyrazole), 2.24 (s, 3H, CH3-pyrazole), 3.79 (s, 3H, -OCH3), 4.42 (q, 2H, J = 7.0 Hz, -CH2CH3), 4.84 (s, 2H, -COCH2-), 5.31 (s, 2H, -CH2-pyrazole), 5.86 (s, 1H, H-pyrazole), 7.15 (d, 1H, J = 8.8 Hz, H8), 7.40 (dd, 1H, J = 2.0 and 8.4 Hz, H7), 8.28 (d, 1H, J = 2.0 Hz, H5), 8.39 (s, 1H, H2).
Ethyl 6-((4-Chloro-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-1-(2-methoxy-2-oxoethyl)-4-oxo-1,4-dihydro quinoline-3-carboxylate (7b). Yield: 43%; mp: 235–237 °C; 1H-NMR (CDCl3, δ ppm) 1.43 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.14 (s, 3H, CH3-pyrazole), 2.26 (s, 3H, CH3-pyrazole), 3.80 (s, 3H, -OCH3), 4.42 (q, 2H, J = 7.2 Hz, -CH2CH3), 4.84 (s, 2H, -COCH2-), 5.31 (s, 2H, -CH2-pyrazole), 7.16 (d, 1H, J = 8.4 Hz, H8), 7.40 (dd, 1H, J = 2.4 and 8.8 Hz, H7), 8.32 (d, 1H, J = 2.0 Hz, H5), 8.39 (s, 1H, H2).
Ethyl 6-((4-Bromo-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-1-(2-methoxy-2-oxoethyl)-4-oxo-1,4-dihydro quinoline-3-carboxylate (7c). Yield: 55%; mp: 239–242 °C; 1H-NMR (CDCl3, δ ppm) 1.43 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.16 (s, 3H, CH3-pyrazole), 2.26 (s, 3H, CH3-pyrazole), 3.80 (s, 3H, -OCH3), 4.42 (q, 2H, J = 7.2 Hz, -CH2CH3), 4.84 (s, 2H, -COCH2-), 5.34 (s, 2H, -CH2-pyrazole), 7.17 (d, 1H, J = 8.8 Hz, H8), 7.40 (dd, 1H, J = 2.0 and 8.4 Hz, H7), 8.33 (d, 1H, J = 2.0 Hz, H5), 8.41 (s, 1H, H2); ESI-MS: m/z 476, 478 [M+H]+, 498, 500 [M+Na]+, 514, 516 [M+K]+.
Ethyl1-(3-(Acetylthio)propyl)-6-((3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydroquinoline-3-carboxylate (8a). Yield: 49%; mp: 125–128 °C; 1H-NMR (CDCl3, δ ppm) 1.43 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.11–2.14 (m, 2H, -CH2CH2CH2S-), 2.16 (s, 3H, CH3-pyrazole), 2.24 (s, 3H, CH3-pyrazole), 2.38 (s, 3H, -COCH3), 2.95 (t, 2H, J = 6.4 Hz, -CH2CH2CH2S-), 4.22 (t, 2H, J = 7.6 Hz, -CH2CH2CH2S-), 4.42 (q, 2H, J = 7.0 Hz, -CH2CH3), 5.31 (s, 2H, -CH2-pyrazole), 5.85 (s, 1H, H-pyrazole), 7.37–7.44 (m, 2H, H7 H8), 8.32 (s, 1H, H5), 8.47 (d, 1H, J = 8.4 Hz, H2); 13C-NMR (CDCl3, δ ppm) 11.1, 13.5, 14.4, 25.7, 28.9, 30.6, 51.9, 52.4, 60.9, 105.8, 111.4, 116.2, 126.1, 129.2, 131.5, 134.7, 138.1, 139.3, 147.9, 148.8, 165.7, 173.9, 194.9; ESI-MS: m/z 441.9 [M+H]+, 463.9 [M+Na]+, 479.8 [M+K]+.
Ethyl 1-(3-(Acetylthio)propyl)-6-((4-chloro-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydro-quinoline-3-carboxylate (8b). Yield: 51%; mp: 122–125 °C; 1H-NMR (CDCl3, δ ppm) 1.43 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.13–2.17 (m, 2H, -CH2CH2CH2S-), 2.19 (s, 3H, CH3-pyrazole), 2.21 (s, 3H, CH3-pyrazole), 2.32 (s, 3H, -COCH3), 2.96 (t, 2H, J = 6.8 Hz, -CH2CH2CH2S-), 4.22 (t, 2H, J = 7.6 Hz, -CH2CH2CH2S-), 4.44 (q, 2H, J = 7.2 Hz, -CH2CH3), 5.32 (s, 2H, -CH2-pyrazole), 7.39–7.46 (m, 2H, H7 H8), 8.35 (d, 1H, J = 2.0 Hz, H5), 8.45 (s, 1H, H2).
Ethyl 1-(3-(Acetylthio)propyl)-6-((4-bromo-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydro- quinoline-3-carboxylate (8c). Yield: 46%; mp: 141–143 °C; 1H-NMR (CDCl3, δ ppm) 1.43 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.16 (s, 3H, CH3-pyrazole), 2.17–2.18 (m, 2H, -CH2CH2CH2S-), 2.24 (s, 3H, CH3-pyrazole), 2.38 (s, 3H, -COCH3), 2.96 (t, 2H, J = 6.8 Hz, -CH2CH2CH2S-), 4.22 (t, 2H, J = 7.6 Hz, -CH2CH2CH2S-), 4.43 (q, 2H, J = 7.2 Hz, -CH2CH3), 5.34 (s, 2H, -CH2-pyrazole), 7.38–7.42 (m, 2H, H7 H8), 8.35 (d, 1H, J = 1.6 Hz, H5), 8.46 (s, 1H, H2).
Ethyl 6-((3,5-Dimethyl-1H-pyrazol-1-yl)methyl)-1-(4-fluorobenzyl)-4-oxo-1,4-dihydroquinoline-3-carboxylate (9a). Yield: 50%; mp: 209–211 °C; 1H-NMR (CDCl3, δ ppm) 1.43 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.14 (s, 3H, CH3-pyrazole), 2.21 (s, 3H, CH3-pyrazole), 4.42 (q, 2H, J = 7.0 Hz, -CH2CH3), 5.28 (s, 2H, -CH2-pyrazole), 5.33 (s, 2H, -CH2-Ar), 5.83 (s, 1H, H-pyrazole), 7.05 (t, 2H, J = 6.4 Hz, Ar-H), 7.13 (q, 2H, J = 8.8 Hz, Ar-H), 7.24 (d, 1H, J = 8.8 Hz, H8), 7.29 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 8.30 (d, 1H, J = 2.0 Hz, H5), 8.57 (s, 1H, H2); ESI-MS: m/z 433.9 [M+H]+, 455.9 [M+Na]+, 471.8 [M+K]+.
Ethyl 6-((4-Chloro-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-1-(4-fluorobenzyl)-4-oxo-1,4-dihydroquinoline-3-carboxylate (9b). Yield: 51%; mp: 226–228 °C; 1H-NMR (CDCl3, δ ppm) 1.44 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.13 (s, 3H, CH3-pyrazole), 2.20 (s, 3H, CH3-pyrazole), 4.42 (q, 2H, J = 7.2 Hz, -CH2CH3), 5.28 (s, 2H, -CH2-pyrazole), 5.33 (s, 2H, -CH2-Ar), 7.05 (t, 2H, J = 6.8 Hz, Ar-H), 7.07 (q, 2H, J = 8.0 Hz, Ar-H), 7.27 (d, 1H, J = 8.8 Hz, H8), 7.30 (dd, 1H, J = 2.4 and 8.8 Hz, H7), 8.32 (d, 1H, J = 1.6 Hz, H5), 8.58 (s, 1H, H2); ESI-MS: m/z 467.9 [M+H]+.
Ethyl 6-((4-Bromo-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-1-(4-fluorobenzyl)-4-oxo-1,4-dihydroquinoline-3-carboxylate (9c). Yield: 55%; mp: 219–221 °C; 1H-NMR (CDCl3, δ ppm) 1.44 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.16 (s, 3H, CH3-pyrazole), 2.22 (s, 3H, CH3-pyrazole), 4.42 (q, 2H, J = 7.0 Hz, -CH2CH3), 5.32 (s, 2H, -CH2-pyrazole), 5.35 (s, 2H, -CH2-Ar), 7.06 (t, 2H, J = 8.4 Hz, Ar-H), 7.16 (q, 2H, J = 9.2 Hz, Ar-H), 7.27 (d, 1H, J = 8.8 Hz, H8), 7.31 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 8.30 (d, 1H,J = 1.2 Hz, H5), 8.59 (s, 1H, H2); ESI-MS: m/z 521, 514 [M+H]+, 534, 536 [M+Na]+, 550, 552 [M+K]+.
Ethyl 6-((3,5-Dimethyl-1H-pyrazol-1-yl)methyl)-1-(4-nitrobenzyl)-4-oxo-1,4-dihydroquinoline-3-carboxylate (10a). Yield: 49%; mp: 194–196 °C; 1H-NMR (CDCl3, δ ppm) 1.43 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.15 (s, 3H, CH3-pyrazole), 2.19 (s, 3H, CH3-pyrazole), 4.42 (q, 2H, J = 7.2 Hz, -CH2CH3), 5.27 (s, 2H, -CH2-pyrazole), 5.47 (s, 2H, -CH2-Ar), 5.83 (s, 1H, H-pyrazole), 7.10 (d, 1H, J = 8.8 Hz, H8), 7.24 (dd, 1H, J = 2.4 and 8.8 Hz, H7), 7.31 (d, 2H, J = 8.8 Hz, Ar-H), 8.22 (d, 2H, J = 6.0 Hz, Ar-H), 8.27 (d, 1H, J = 2.0 Hz, H5), 8.56 (s, 1H, H2).
Ethyl 6-((4-Chloro-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-1-(4-nitrobenzyl)-4-oxo-1,4-dihydroquinoline-3-carboxylate (10b). Yield: 39%; mp: 232–235 °C; 1H-NMR (CDCl3, δ ppm) 1.44 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.13 (s, 3H, CH3-pyrazole), 2.19 (s, 3H, CH3-pyrazole), 4.42 (q, 2H, J = 7.0 Hz, -CH2CH3), 5.27 (s, 2H, -CH2-pyrazole), 5.47 (s, 2H, -CH2-Ar), 7.12 (d, 1H, J = 8.4 Hz, H8), 7.28 (dd, 1H, J = 2.0 and 8.4 Hz, H7), 7.32 (d, 2H, J = 8.8 Hz, Ar-H), 8.23 (d, 2H, J = 6.8 Hz, Ar-H), 8.33 (d, 1H, J = 2.0 Hz, H5), 8.58 (s, 1H, H2); ESI-MS: m/z 516.8 [M+Na]+, 493.0 [M-H]−.
Ethyl 6-((4-Bromo-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-1-(4-nitrobenzyl)-4-oxo-1,4-dihydroquinoline-3-carboxylate (10c). Yield: 40%; mp: 240–242 °C; 1H-NMR (CDCl3, δ ppm) 1.43 (t, 3H, J = 7.2 Hz, -CH2CH3), 2.15 (s, 3H, CH3-pyrazole), 2.19 (s, 3H, CH3-pyrazole), 4.42 (q, 2H, J = 7.0 Hz, -CH2CH3), 5.30 (s, 2H, -CH2-pyrazole), 5.47 (s, 2H, -CH2-Ar), 7.12 (d, 1H, J = 8.8 Hz, H8), 7.28 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 7.32 (d, 2H, J = 8.8 Hz, Ar-H), 8.23 (d, 2H, J = 6.4 Hz, Ar-H), 8.33 (d, 1H, J = 1.6 Hz, H5), 8.58 (s, 1H, H2).
3.3. General Procedure for the Synthesis of 1-Substitued-6-(pyrazolylmethyl)-4-oxo-4H-quinoline-3-carboxylic Acids 11–16
A suspension of the appropriate ester 5–8 (0.5 mmol) in 10% aq. NaOH (10 mL) was refluxed for 2 h. After cooling at room temperature, the reaction mixture was acidified using conc. HCl. The resulting precipitate was filtered and washed with water and DCM to give the corresponding 4-quinolone-3-carboxylic acids 11–14. Alternatively, a suspension of the appropriate ester 9–10 (0.5 mmol) in 10% aq. HCl (10 mL) was refluxed for 2 h. After cooling at room temperature, the resulting precipitate was filtered and washed with water and DCM to give the corresponding 4-quinolone-3-carboxylic acids 15–16.
6-((3,5-Dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (11a). Yield: 63%; mp: 254–256 °C; 1H-NMR (DMSO-d6, δ ppm) 2.11 (s, 3H, CH3-pyrazole), 2.18 (s, 3H, CH3-pyrazole), 5.38 (s, 2H, -CH2-pyrazole), 5.89 (s, 1H, H-pyrazole), 7.64 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 7.81 (d, 1H, J = 8.4 Hz, H8), 7.99 (d, 1H, J = 1.6 Hz, H5), 8.84 (s, 1H, H2), 13.36 (s, 1H, -NH-), 15.29 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 11.1, 13.8, 51.5, 105.7, 108.2, 120.9, 123.2, 124.8, 133.2, 136.6, 139.4, 139.5, 145.8, 146.9, 166.9, 178.4; HRMS: m/z calcd for C16H16N3O3: 298.1186; found: 298.1188; HPLC purity 98.36%.
6-((4-Chloro-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (11b). Yield: 47%; mp: 263–265 °C; 1H-NMR (DMSO-d6, δ ppm) 2.09 (s, 3H, CH3-pyrazole), 2.19 (s, 3H, CH3-pyrazole), 5.45 (s, 2H, -CH2-pyrazole), 7.66 (dd, 1H, J = 2.0 and 8.4 Hz, H7), 7.82 (d, 1H, J = 8.4 Hz, H8), 8.04 (d, 1H, J = 1.6 Hz, H5), 8.88 (s, 1H, H2), 13.51 (s, 1H, -NH-), 15.29 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 9.5, 11.5, 52.7, 107.0, 108.18, 120.9, 123.5, 124.8, 133.3, 135.7, 136.1, 139.4, 144.0, 145.7, 166.8, 178.5; HRMS: m/z calcd for C16H15ClN3O3: 332.0796; found: 332.0799; HPLC purity 99.40%.
6-((4-Bromo-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (11c). Yield: 81%; mp: 253–256 °C; 1H-NMR (DMSO-d6, δ ppm) 2.09 (s, 3H, CH3-pyrazole), 2.17 (s, 3H, CH3-pyrazole), 5.44 (s, 2H, -CH2-pyrazole), 7.64 (dd, 1H, J = 2.0 and 8.4 Hz, H7), 7.79 (d, 1H, J = 8.4 Hz, H8), 8.01 (d, 1H, J = 1.6 Hz, H5), 8.84 (s, 1H, H2), 13.36 (s, 1H, -NH-), 15.20 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 10.4, 12.5, 52.8, 93.8, 108.2, 120.8, 123.6, 124.8, 133.4, 135.8, 137.8, 139.3, 145.5, 145.6, 166.7, 178.5; HRMS: m/z calcd for C16H15BrN3O3: 376.0291, 378.0276; found: 376.0296, 378.0278; HPLC purity 97.17%.
1-(2,3-Dihydroxypropyl)-6-((3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid(12a). Yield: 52%; mp: 187–189 °C; 1H-NMR (DMSO-d6, δ ppm) 2.19 (s, 3H, CH3-pyrazole), 2.27 (s, 3H, CH3-pyrazole), 3.40–3.55 (m, 2H, HOCH2CHOHCH2-), 3.81–3.83 (m, 1H, HOCH2CHOHCH2-), 4.28 (q, 1H, J = 14.4 Hz, HOCH2CHOHCH2-), 4.82 (dd, 1H, J = 2.4 and 10.4 Hz, HOCH2CHOHCH2-), 5.53 (s, 2H, -CH2-pyrazole), 6.08 (s, 1H, H-pyrazole), 6.31 (br, 2H, HOCH2CHOHCH2-), 7.75 (dd, 1H, J = 2.0 and 9.2 Hz, H7), 8.02 (d, 1H, J = 9.2 Hz, H8), 8.12 (d, 1H, J = 2.0 Hz, H5), 8.86 (s, 1H, H2), 15.01 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 11.1, 13.2, 51.1, 57.4, 63.7, 69.5, 106.5, 107.7, 119.3, 124.3, 125.9, 133.4, 135.6, 139.5, 141.3, 146.8, 151.0, 166.5, 177.9; HRMS: m/z calcd for C19H22N3O5: 372.1554; found: 372.1556; HPLC purity 98.10%.
1-(2,3-Dihydroxypropyl)-6-((4-chloro-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydro-quinoline-3-carboxylic acid (12b). Yield: 63%; mp: 278–280 °C; 1H-NMR (DMSO-d6, δ ppm) 2.12 (s, 3H, CH3-pyrazole), 2.20 (s, 3H, CH3-pyrazole), 3.39–3.55 (m, 2H, HOCH2CHOHCH2-), 3.82 (m, 1H, HOCH2CHOHCH2-), 4.27 (q, 1H, J = 14.4 Hz, HOCH2CHOHCH2-), 4.80 (dd, 1H, J = 2.0 and 14.4 Hz, HOCH2CHOHCH2-), 4.96 (t, 1H, J = 5.6 Hz, HOCH2CHOHCH2-), 5.22 (d, 1H, J = 5.6 Hz, HOCH2CHOHCH2-), 5.48 (s, 2H, -CH2-pyrazole), 7.73 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 8.00 (d, 1H, J = 9.2 Hz, H8), 8.13 (d, 1H, J = 1.6 Hz, H5), 8.86 (s, 1H, H2), 15.13 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 9.5, 11.5, 52.4, 57.5, 63.8, 69.4, 107.5, 107.7, 119.2, 124.4, 125.9, 133.4, 135.8, 136.1, 139.5, 144.1, 151.0, 166.5, 178.0; HRMS: m/z calcd for C19H21ClN3O5: 406.1164; found: 406.1167; HPLC purity 97.%.
1-(2,3-Dihydroxypropyl)-6-((4-bromo-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydroquino-line-3-carboxylic acid (12c). Yield: 62%; mp: 267–269 °C; 1H-NMR (DMSO-d6, δ ppm) 2.19 (s, 3H, CH3-pyrazole), 2.27 (s, 3H, CH3-pyrazole), 3.39–3.53 (m, 2H, HOCH2CHOHCH2-), 3.81 (m, 1H, HOCH2CHOHCH2-), 4.27 (q, 1H, J = 10.8 Hz, HOCH2CHOHCH2-), 4.80 (d, 1H, J = 12.8 Hz, HOCH2CHOHCH2-), 4.98 (t, 1H, J = 5.6 Hz, HOCH2CHOHCH2-), 5.22 (d, 1H, J = 5.6 Hz, HOCH2CHOHCH2-), 5.51 (s, 2H, -CH2-pyrazole), 7.73 (dd, 1H, J = 1.6 and 8.8 Hz, H7), 8.00 (d, 1H, J = 4.8 Hz, H8), 8.13 (s, 1H, H5), 8.86 (s, 1H, H2), 15.15 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 10.4, 12.5, 52.5, 57.4, 63.7, 69.4, 93.8, 107.7, 119.2, 124.4, 125.9, 133.4, 135.8, 137.8, 139.5, 145.6, 151.0, 166.5, 178.0; HRMS: m/z calcd for C19H21BrN3O5: 450.0659, 452.0644; found: 450.0664, 452.0651; HPLC purity 97.73%.
1-(Carboxymethyl)-6-((3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (13a). Yield: 78%; mp > 300 °C; 1H-NMR (DMSO-d6, δ ppm) 2.09 (s, 3H, CH3-pyrazole), 2.17 (s, 3H, CH3-pyrazole), 5.38 (s, 2H, -CH2-pyrazole), 5.42 (s, 2H, -CH2COOH), 5.88 (s, 1H, H-pyrazole), 7.68 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 7.79 (d, 1H, J = 8.8 Hz, H8), 8.05 (d, 1H, J = 1.6 Hz, H5), 9.02 (s, 1H, H2), 13.63 (s, 1H, -NH-), 14.97 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 11.1, 13.8, 51.2, 54.7, 105.7, 108.3, 118.8, 124.0, 125.5, 133.6, 136.9, 139.4, 139.7, 147.0, 151.1, 166.2, 169.2, 178.2; HRMS: m/z calcd for C18H18N3O5: 356.1241; found: 356.1242; HPLC purity 97.80%.
1-(Carboxymethyl)-6-((4-chloro-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (13b). Yield: 58%; mp: 292–295 °C; 1H-NMR (DMSO-d6, δ ppm) 2.12 (s, 3H, CH3-pyrazole), 2.20 (s, 3H, CH3-pyrazole), 5.35 (s, 2H, -CH2-pyrazole), 5.46 (s, 2H, -CH2COOH), 7.71 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 7.79 (d, 1H, J = 9.2 Hz, H8), 8.12 (d, 1H, J = 1.6 Hz, H5), 9.07 (s, 1H, H2), 15.01 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 9.5, 11.5, 52.3, 55.4, 107.0, 108.2, 119.1, 124.2, 125.5, 133.7, 135.9, 136.2, 139.9, 144.1, 151.1, 166.3, 169.1, 178.1; HRMS: m/z calcd for C18H17ClN3O5: 390.0851; found: 390.0855; HPLC purity 99.15%.
1-(Carboxymethyl)-6-((4-bromo-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydro-quinoline-3-carboxylic acid (13c). Yield: 72%; mp: 278–280 °C; 1H-NMR (DMSO-d6, δ ppm) 2.12 (s, 3H, CH3-pyrazole), 2.21 (s, 3H, CH3-pyrazole), 5.36 (s, 2H, -CH2-pyrazole), 5.48 (s, 2H, -CH2COOH), 7.71 (dd, 1H, J = 2.0 and 9.2 Hz, H7), 7.79 (d, 1H, J = 9.2 Hz, H8), 8.12 (d, 1H, J = 2.0 Hz, H5), 9.07 (s, 1H, H2), 14.99 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 10.4, 12.5, 52.5, 55.3, 93.8, 108.3, 119.0, 124.3, 125.6, 133.7, 135.9, 137.8, 139.9, 145.6, 151.1, 166.2, 169.1, 178.2; HRMS: m/z calcd for C18H17BrN3O5: 434.0346, 436.0331; found: 434.0350, 436.0329; HPLC purity 99.23%.
1-(3-Mercaptopropyl)-6-((3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (14a). Yield: 65%; mp: 225–226 °C; 1H-NMR (DMSO-d6, δ ppm) 2.08 (m, 2H, -CH2CH2CH2SH), 2.12 (s, 3H, CH3-pyrazole), 2.19 (s, 3H, CH3-pyrazole), 2.54–2.58 (m, 2H, -CH2CH2CH2SH), 4.62 (t, 2H, J = 7.2 Hz, -CH2CH2CH2SH), 5.41 (s, 2H, -CH2-pyrazole), 5.89 (s, 1H, H-pyrazole), 7.71 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 8.05 (d, 1H, J = 8.8 Hz, H8), 8.08 (d, 1H, J = 2.0 Hz, H5), 9.02 (s, 1H, H2), 15.13 (s, 1H, -COOH); 13C-NMR (DMSO-d6, 100 MHz, δ ppm) 11.1, 13.8, 21.1, 28.5, 34.3, 51.2, 105.7, 108.2, 118.9, 124.2, 126.1, 133.5, 136.8, 138.9, 139.5, 147.0, 149.9, 166.5, 178.0; HRMS: m/z calcd for C19H22N3O3S: 372.1376; found: 372.1380; HPLC purity 97.50%.
1-(3-Mercaptopropyl)-6-((4-chloro-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydro-quinoline-3-carboxylic acid (14b). Yield: 54%; mp: 238–240 °C; 1H-NMR (DMSO-d6, δ ppm) 2.08–2.09 (m, 2H, -CH2CH2CH2SH), 2.11 (s, 3H, CH3-pyrazole), 2.20 (s, 3H, CH3-pyrazole), 2.56–2.58 (m, 2H, -CH2CH2CH2SH), 4.62 (t, 2H, J = 7.2 Hz, -CH2CH2CH2SH), 5.45 (s, 2H, -CH2-pyrazole), 7.73 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 8.06 (d, 1H, J = 8.4 Hz, H8), 8.14 (d, 1H, J = 2.0 Hz, H5), 9.03 (s, 1H, H2), 15.09 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 9.5, 11.5, 21.1, 28.6, 34.3, 52.4, 107.1, 108.3, 119.0, 124.5, 126.1, 133.6, 135.9, 136.1, 139.1, 144.1, 149.9, 166.4, 178.0; HRMS: m/z calcd for C19H21ClN3O3S: 406.0986; found: 406.0989; HPLC purity 96.68%.
6-((4-Bromo-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-1-(3-mercaptopropyl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (14c). Yield: 42%; mp: 231–233 °C; 1H-NMR (DMSO-d6, δ ppm) 2.08–2.11 (m, 2H, -CH2CH2CH2SH), 2.12 (s, 3H, CH3-pyrazole), 2.21 (s, 3H, CH3-pyrazole), 2.55–2.58 (m, 2H, -CH2CH2CH2SH), 4.62 (t, 2H, J = 7.2 Hz, -CH2CH2CH2SH), 5.51 (s, 2H, -CH2-pyrazole), 7.73 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 8.06 (d, 1H, J = 8.8 Hz, H8), 8.14 (d, 1H, J = 2.0 Hz, H5), 9.03 (s, 1H, H2), 15.09 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 10.4, 12.5, 21.1, 28.4, 34.3, 52.5, 93.9, 108.3, 119.1, 124.5, 126.1, 133.6, 135.9, 137.8, 139.0, 145.6, 149.9, 166.4, 178.0; HRMS: m/z calcd for C19H21BrN3O3S: 450.0481, 452.0466; found: 450.0489, 452.0467; HPLC purity 96.90%.
1-(4-Fluorobenzyl)-6-((3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydro-quinoline-3-carboxylic acid(15a). Yield: 63%; mp: 271–273 °C; 1H-NMR (DMSO-d6, δ ppm) 2.09 (s, 3H, CH3-pyrazole), 2.16 (s, 3H, CH3-pyrazole), 5.36 (s, 2H, -CH2-pyrazole), 5.82 (s, 2H, -CH2-Ar), 5.87 (s, 1H, H-pyrazole), 7.19 (t, 2H, J = 8.8 Hz, Ar-H), 7.37 (q, 2H, J = 8.4 Hz, Ar-H), 7.60 (dd, 1H, J = 2.0 and 9.2 Hz, H7), 7.87 (d, 1H, J = 8.8 Hz, H8), 8.05 (d, 1H, J = 1.6 Hz, H5), 9.26 (s, 1H, H2), 15.06 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 11.1, 13.8, 51.1, 56.2, 105.7, 108.5, 116.1, 116.4, 119.5, 124.1, 126.2, 129.5, 129.5, 133.4, 136.9, 139.1, 139.4, 147.0, 150.5, 166.4, 178.2; HRMS: m/z calcd for C23H21FN3O3: 406.1561; found: 406.1569; HPLC purity 99.70%.
1-(4-Fluorobenzyl)-6-((4-chloro-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydro-quinoline-3-carboxylic acid (15b). Yield: 75%; mp: 239–241 °C; 1H-NMR (DMSO-d6, δ ppm) 2.17 (s, 3H, CH3-pyrazole), 2.22 (s, 3H, CH3-pyrazole), 5.34 (s, 2H, -CH2-pyrazole), 5.49 (s, 2H, -CH2-Ar), 7.10 (t, 2H, J = 8.0 Hz, Ar-H), 7.14–7.18 (m, 2H, Ar-H), 7.42–7.49 (m, 2H, H7 H8), 8.35 (s, 1H, H5), 8.89 (s, 1H, H2), 14.68 (s, 1H, -COOH); 13C-NMR (DMSO-d6, 100 MHz, δ ppm) 9.5, 11.4, 52.7, 57.6, 108.7, 109.4, 116.6, 116.8, 118.1, 125.1, 126.7, 128.0, 128.1, 132.8, 135.4, 135.6, 139.0, 145.4, 149.1, 166.6, 178.3; HRMS: m/z calcd for C23H20ClFN3O3: 440.1171; found: 440.1169; HPLC purity 99.55%.
1-(4-Fluorobenzyl)-6-((4-bromo-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydro-quinoline-3-carboxylic acid (15c). Yield: 73%; mp: 255–257 °C; 1H-NMR (DMSO-d6, δ ppm) 2.17 (s, 3H, CH3-pyrazole), 2.21 (s, 3H, CH3-pyrazole), 5.35 (s, 2H, -CH2-pyrazole), 5.48 (s, 2H, -CH2-Ar), 7.08 (t, 2H, J = 7.6 Hz, Ar-H), 7.14 (s, 2H, Ar-H), 7.43 (s, 2H, H7 H8), 8.35 (s, 1H, H5), 8.88 (s, 1H, H2), 15.06 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 10.4, 12.31, 52.8, 57.6, 109.4, 116.6, 116.8, 118.0, 125.2, 126.7, 128.0, 128.1, 132.8, 135.4, 137.4, 139.1, 139.0, 146.9, 149.1, 166.5, 178.3; HRMS: m/z calcd for C23H20BrFN3O3: 484.0666, 486.0651; found: 484.0665, 486.0646; HPLC purity 99.57%.
1-(4-Nitrobenzyl)-6-((3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydro-quinoline-3-carboxylic acid (16a). Yield: 56%; mp: 274–276 °C; 1H-NMR (DMSO-d6, δ ppm) 2.08 (s, 3H, CH3-pyrazole), 2.16 (s, 3H, CH3-pyrazole), 5.36 (s, 2H, -CH2-pyrazole), 5.87 (s, 1H, H-pyrazole), 6.01 (s, 2H, -CH2-Ar), 7.52 (d, 2H, J = 8.8 Hz, Ar-H), 7.58 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 7.75 (d, 1H, J = 8.8 Hz, H8), 8.07 (d, 1H, J = 2.0 Hz, H5), 8.18 (d, 2H, J = 8.8 Hz, Ar-H), 9.32 (s, 1H, H2), 15.01 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 11.1, 13.8, 51.1, 56.3, 105.7, 108.8, 119.3, 124.2, 124.4, 126.2, 128.3, 133.6, 137.1, 139.0, 139.4, 143.5, 147.0, 147.6, 150.9, 166.3, 178.4; HRMS: m/z calcd for C23H21N4O5: 433.1506; found: 433.1508; HPLC purity 99.80%.
1-(4-Nitrobenzyl)-6-((4-chloro-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4-dihydroquinoline-3-carboxylic acid (16b). Yield: 55%; mp: 282–284 °C; 1H-NMR (DMSO-d6, δ ppm) 2.09 (s, 3H, CH3-pyrazole), 2.17 (s, 3H, CH3-pyrazole), 5.42 (s, 2H, -CH2-pyrazole), 6.01 (s, 2H, -CH2-Ar), 7.53 (d, 2H, J = 8.8 Hz, Ar-H), 7.60 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 7.75 (d, 1H, J = 6.8 Hz, H8), 8.13 (d, 1H, J = 2.0 Hz, H5), 8.19 (d, 2H, J = 8.8 Hz, Ar-H), 9.33 (s, 1H, H2), 14.98 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 9.5, 11.5, 52.2, 56.3, 107.7, 108.8, 119.5, 124.4, 124.5, 126.2, 128.3, 133.7, 136.2, 139.2, 143.5, 144.1, 147.6, 151.0, 166.3, 178.3; HRMS: m/z calcd for C23H20ClN4O5: 467.1116; found: 467.1124; HPLC purity 97.32%.
1-(4-Nitrobenzyl)-6-((4-bromo-3,5-dimethyl-1H-pyrazol-1-yl)methyl)-4-oxo-1,4dihydroquinoline-3-carboxylic acid (16c). Yield: 69%; mp: 266–268 °C; 1H-NMR (DMSO-d6, δ ppm) 2.09 (s, 3H, CH3-pyrazole), 2.18 (s, 3H, CH3-pyrazole), 5.45 (s, 2H, -CH2-pyrazole), 6.01 (s, 2H, -CH2-Ar), 7.53 (d, 2H, J = 8.8 Hz, Ar-H), 7.60 (dd, 1H, J = 2.0 and 8.8 Hz, H7), 7.75 (d, 1H, J = 8.8 Hz, H8), 8.13 (d, 1H, J = 1.6 Hz, H5), 8.19 (d, 2H, J = 8.8 Hz, Ar-H), 9.33 (s, 1H, H2), 14.99 (s, 1H, -COOH); 13C-NMR (DMSO-d6, δ ppm) 10.4, 12.5, 52.3, 56.4, 93.8, 108.8, 119.5, 124.4, 124.5, 126.6, 128.3, 133.7, 136.2, 137.9, 139.2, 143.5, 145.7, 147.6, 150.9, 166.3, 178.3; HRMS: m/z calcd for C23H20BrN4O5: 511.0611, 513.0596; found: 511.0616, 513.0599; HPLC purity 97.64%.