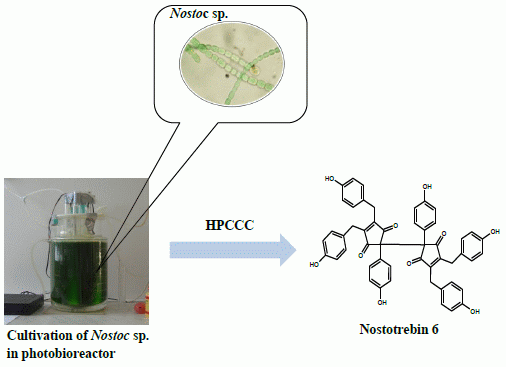
Two-Step Separation of Nostotrebin 6 from Cultivated Soil Cyanobacterium (Nostoc sp.) by High Performance Countercurrent Chromatography
Abstract
:1. Introduction

2. Results and Discussion
2.1. HPLC/DAD-ESI-HRMS Analysis of the Crude Extract
2.2. Optimization of HPCCC Conditions

| System no. | Composition | Relative Proportions of Solvents (v/v/v/v) | Phase Volume Ratio | Settling Time (s) | Partition Coefficient (K) |
|---|---|---|---|---|---|
| 1 | n-hexane–ethyl acetate–MeOH–water | 6:4:5:5 | 0.72 | 18 | 0.10 |
| 2 | n-hexane–ethyl acetate–MeOH–water | 5:5:5:5 | 0.67 | 19 | 0.21 |
| 3 | n-hexane–ethyl acetate–MeOH–water | 4:5:4:5 | 0.70 | 17 | 0.85 |
| 4 | n-hexane–ethyl acetate–MeOH–water | 3:5:3:5 | 0.82 | 23 | 1.47 |
| 5 | n-hexane–ethyl acetate–MeOH–water | 2:5:2:5 | 0.94 | 24 | 1.62 |
| 6 | n-hexane–ethyl acetate–MeOH–water | 1:5:1:5 | 1.00 | 23 | 3.53 |
| 7 | EtOAc–water | 5:5 | 0.86 | 20 | 12.40 |
| 8 | EtOAc–n-BuOH–water | 4:1:5 | 0.91 | 60 | 23.71 |
| 9 | EtOAc–n-BuOH–water | 3:2:5 | 0.99 | 38 | 16.57 |
| 10 | EtOAc–n-BuOH–water | 2:3:5 | 1.09 | 55 | 17.53 |

| System no. | Partition coefficient (K) | Separation factor (α) | Density difference (LP−UP g/mL) | |
|---|---|---|---|---|
| K a | K b | |||
| 3 | 0.85 | 0.90 | 1.06 | 0.09 |
| 3 c | 0.51 | 0.80 | 1.57 | 0.12 |
2.3. Separation of Nostotrebin-6 by HPCCC and Structural Identification


3. Experimental
3.1. Reagents
3.2. Cultivation of Nostoc sp. str. Lukešová 27/97
3.3. Preparation of the Crude Extract and Sample Solution
3.4. Apparatus
3.4.1. Countercurrent Chromatography
3.4.2. HPLC/DAD-ESI-HRMS
3.5. Selection of the Two-Phase Solvent Systems for HPCCC
3.6. Preparation of the Two-Phase Solvent System and Sample Solution for HPCCC Separation
3.7. HPCCC Separation Procedure

3.8. HPLC/DAD-ESI-HRMS Analysis and Identification of HPCCC Peak Fractions
3.9. Structural Identification
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chlipala, G.E.; Mo, S.; Orjala, J. Chemodiversity in freshwater and terrestrial cyanobacteria-a source for drug discovery. Curr. Drug Targets 2011, 12, 1654–1673. [Google Scholar] [CrossRef]
- Grond, S.; Meurer, G. Exploiting green treasures. Chem. Biol. 2007, 14, 469–471. [Google Scholar] [CrossRef]
- Rastogi, R.P.; Sinha, R.P. Biotechnological and industrial significance of cyanobacterial secondary metabolites. Biotechnol. Adv. 2009, 27, 521–539. [Google Scholar]
- Singh, R.K.; Tiwari, S.P.; Rai, A.K.; Mohapatra, T.M. Cyanobacteria: An emerging source for drug discovery. J. Antibiot. 2011, 64, 401–412. [Google Scholar] [CrossRef]
- Mundt, S.; Kreitlow, S.; Nowotny, A.; Effmert, U. Biochemical and pharmacological investigations of selected cyanobacteria. Int. J. Hyg. Environ. Health 2001, 203, 327–334. [Google Scholar] [CrossRef]
- Plaza, M.; Santoyo, S.; Jaime, L.; Reina, G.G.B.; Herrero, M.; Senorans, F.J.; Ibanez, E. Screening for bioactive compounds from algae. J. Pharm. Biomed. 2010, 51, 450–455. [Google Scholar] [CrossRef]
- Klejdus, B.; Kopecký, J.; Benesova, L.; Vacek, J. Solid-phase/supercritical-fluid extraction for liquid chromatography of phenolic compounds in freshwater microalgae and selected cyanobacterial species. J. Chromatogr. A 2009, 1216, 763–771. [Google Scholar] [CrossRef]
- Onofrejova, L.; Vasickova, J.; Klejdus, B.; Stratil, P.; Misurcova, L.; Kracmar, S.; Kopecký, J.; Vacek, J. Bioactive phenols in algae: The application of pressurized-liquid and solid-phase extraction techniques. J. Pharm. Biomed. 2010, 51, 464–470. [Google Scholar] [CrossRef]
- Ragan, M.A.; Glombitza, K.W. Phlorotannins, brown algal polyphenols. In Progress in Phycological Research; Round, F.E., Chapman, D.J., Eds.; Biopress: Bristol, UK, 1986; pp. 129–241. [Google Scholar]
- La Barre, S.; Potin, P.; Leblanc, C.; Delage, L. The halogenated metabolism of brown algae (phaeophyta), its biological importance and its environmental significance. Mar. Drugs 2010, 8, 988–1010. [Google Scholar] [CrossRef]
- Vacek, J.; Snoblova, M.; Klejdus, B. A short introduction to algal and cyanobacterial constituents-the occurrence of phenolic metabolites (in czech). Čes. Slov. Farm. 2009, 58, 103–108. [Google Scholar]
- Zelik, P.; Lukesova, A.; Voloshko, L.; Stys, D.; Kopecký, J. Screening for acetylcholinesterase inhibitory activity in cyanobacteria of the genus Nostoc. J. Enzym. Inhib. Med. Chem. 2009, 24, 531–536. [Google Scholar] [CrossRef]
- Zelik, P.; Lukesova, A.; Cejka, J.; Budesinsky, M.; Havlicek, V.; Cegan, A.; Kopecký, J. Nostotrebin 6, a bis(cyclopentenedione) with cholinesterase inhibitory activity isolated from Nostoc sp str. Lukesova 27/97. J. Enzym. Inhib. Med. Chem. 2010, 25, 414–420. [Google Scholar] [CrossRef]
- Massoud, F.; Gauthier, S. Update on the pharmacological treatment of Alzheimer's disease. Curr. Neuropharmacol. 2010, 8, 69–80. [Google Scholar] [CrossRef]
- Greig, N.H.; Utsuki, T.; Yu, Q.; Zhu, X.; Holloway, H.W.; Perry, T.; Lee, B.; Ingram, D.K.; Lahiri, D.K. A new therapeutic target in Alzheimer's disease treatment: Attention to butyrylcholinesterase. Curr. Med. Res. Opin. 2001, 17, 159–165. [Google Scholar]
- Ito, Y. Golden rules and pitfalls in selecting optimum conditions for high-speed counter-current chromatography. J. Chromatogr. A 2005, 1065, 145–168. [Google Scholar] [CrossRef]
- Cheel, J.; Theoduloz, C.; Rodríguez, J.; Schmeda-Hirschmann, G. Free radical scavengers and antioxidants from Lemongrass (Cymbopogon citratus (DC.) Stapf.). J. Agric. Food Chem. 2005, 53, 2511–2517. [Google Scholar] [CrossRef]
- Tapia, A.; Cheel, J.; Theoduloz, C.; Rodríguez, J.; Schmeda-Hirschmann, G.; Gerth, A.; Wilken, D.; Jordan, M.; Jiménez-González, E.; Gomez-Kosky, R.; et al. Free radical scavengers from Cymbopogon citratus (DC.) Stapf. plants cultivated in bioreactors by the temporary immersion (TIS)-principle. Z. Naturforsch. C 2007, 62, 447–457. [Google Scholar]
- Spórna-Kucab, A.; Ignatova, S.; Garrard, I.; Wybraniec, S. Versatile solvent systems for the separation of betalains from processed Beta vulgaris L. juice using counter-current chromatography. J. Chromatogr. B 2013, 941, 54–61. [Google Scholar] [CrossRef]
- Costa, Fd.; Garrard, I.; da Silva, A.J.; Leitão, G.G. Changes in the mobile phase composition on a stepwise counter-current chromatography elution for the isolation of flavonoids from Siparuna glycycarpa. J. Sep. Sci. 2013, 36, 2253–2259. [Google Scholar] [CrossRef]
- Du, Q.Z.; Jerz, G.; Waibel, R.; Winterhalter, P. Isolation of dammarane saponins from Panax notoginseng by high-speed counter-current chromatography. J. Chromatogr. A 2003, 1008, 173–180. [Google Scholar] [CrossRef]
- Niu, L.; Xie, Z.; Cai, T.; Wu, P.; Xue, P.; Chen, X.; Wu, Z.; Ito, Y.; Li, F.; Yang, F. Preparative isolation of alkaloids from Corydalis bungeana Turcz. by high-speed counter-current chromatography using stepwise elution. J. Sep. Sci. 2011, 34, 987–994. [Google Scholar] [CrossRef]
- Chen, F.; Li, H.B.; Wong, R.N.; Ji, B.; Jiang, Y. Isolation and purification of the bioactive carotenoid zeaxanthin from the microalga Microcystis aeruginosa by high-speed counter-current chromatography. J. Chromatogr. A 2005, 1064, 183–186. [Google Scholar]
- Li, H.B.; Chen, F. Preparative isolation and purification of astaxanthin from the microalga Chlorococcum sp. by high-speed counter-current chromatography. J. Chromatogr. A 2001, 925, 133–137. [Google Scholar] [CrossRef]
- Li, H.B.; Fan, K.W.; Chen, F. Isolation and purification of canthaxanthin from the microalga Chlorella zofingiensis by high-speed counter-current chromatography. J. Sep. Sci. 2006, 29, 699–703. [Google Scholar] [CrossRef]
- Lu, H.T.; Jiang, Y.; Chen, F. Preparative separation and purification of squalene from the microalga Thraustochytrium ATCC 26185 by high-speed counter-current chromatography. J. Chromatogr. A 2003, 994, 37–43. [Google Scholar] [CrossRef]
- Ito, Y.; Conway, W.D. Experimental observations of the hydrodynamic behavior of solvent systems in high-speed counter-current chromatography. III. Effects of physical properties of the solvent systems and operating temperature on the distribution of two-phase solvent systems. J. Chromatogr. A 1984, 301, 405–414. [Google Scholar] [CrossRef]
- Berthod, A.; Maryutina, T.; Spivakov, B.; Shpigun, O.; Sutherland, I.A. Countercurrent chromatography in analytical chemistry. Pure Appl. Chem. 2009, 81, 355–387. [Google Scholar]
- Oka, F.; Oka, H.; Ito, Y. Systematic search for suitable two-phase solvent systems for high-speed counter-current chromatography. J. Chromatogr. A 1991, 538, 99–108. [Google Scholar] [CrossRef]
- Antonopoulou, S.; Nomikos, T.; Oikonomou, A.; Kyriacou, A.; Andriotis, M.; Fragopoulou, E.; Pantazidou, A. Characterization of bioactive glycolipids from Scytonema julianum (cyanobacteria). Comp. Biochem. Physiol. B-Biochem. Mol. Biol. 2005, 140, 219–231. [Google Scholar] [CrossRef]
- Wada, H.; Murata, N. Membrane lipids in cyanobacteria. In Lipids in Photosynthesis: Structure, Function and Genetics; Siegenthaler, P.A., Murata, N., Eds.; Springer: Dordrecht, The Netherlands, 1998; volume 6, pp. 65–81. [Google Scholar]
- Pacwa-Płociniczak, M.; Płaza, G.A.; Piotrowska-Seget, Z.; Cameotra, S.S. Environmental applications of biosurfactants: Recent advances. Int. J. Mol. Sci. 2011, 12, 633–654. [Google Scholar] [CrossRef]
- Kiełbowicz, G.; Gładkowski, W.; Chojnacka, A.; Wawrzeńczyk, C. A simple method for positional analysis of phosphatidylcholine. Food Chem. 2012, 135, 2542–2548. [Google Scholar] [CrossRef]
- Liu, R.; Li, A.; Sun, A. Preparative isolation and purification of hydroxyanthraquinones and cinnamic acid from the chinese medicinal herb Rheum officinale Baill. by high-speed counter-current chromatography. J. Chromatogr. A 2004, 1052, 217–221. [Google Scholar] [CrossRef]
- Wang, T.; Jiang, X.; Yang, L.; Wu, S. pH-gradient counter-current chromatography isolation of natural antioxidant chlorogenic acid from Lonicera japonica Thumb. using an upright coil planet centrifuge with three multi-layer coils connected in series. J. Chromatogr. A 2008, 1180, 53–58. [Google Scholar] [CrossRef]
- Sample Availability: Samples of nostotrebin-6 and crude extract are available from the authors.
© 2014 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheel, J.; Kučerová, P.; Garrard, I.; Ignatova, S.; Hrouzek, P.; Kopecký, J. Two-Step Separation of Nostotrebin 6 from Cultivated Soil Cyanobacterium (Nostoc sp.) by High Performance Countercurrent Chromatography. Molecules 2014, 19, 8773-8787. https://doi.org/10.3390/molecules19078773
Cheel J, Kučerová P, Garrard I, Ignatova S, Hrouzek P, Kopecký J. Two-Step Separation of Nostotrebin 6 from Cultivated Soil Cyanobacterium (Nostoc sp.) by High Performance Countercurrent Chromatography. Molecules. 2014; 19(7):8773-8787. https://doi.org/10.3390/molecules19078773
Chicago/Turabian StyleCheel, José, Petra Kučerová, Ian Garrard, Svetlana Ignatova, Pavel Hrouzek, and Jiří Kopecký. 2014. "Two-Step Separation of Nostotrebin 6 from Cultivated Soil Cyanobacterium (Nostoc sp.) by High Performance Countercurrent Chromatography" Molecules 19, no. 7: 8773-8787. https://doi.org/10.3390/molecules19078773