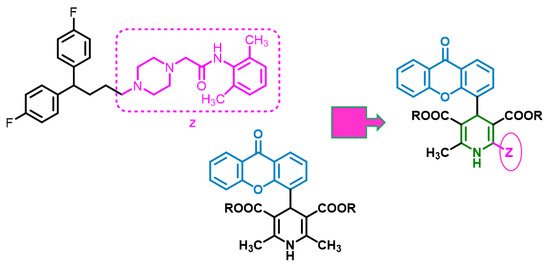
4.1. Chemistry
4.1.1. General Methods
Starting materials used, unless otherwise specified, were high-grade commercial products. Solvents were of analytical grade. Reaction progress was followed by thin layer chromatography (TLC) on precoated silica gel plates (Merck Silica Gel 60 F254, Merck, Darmstadt, Germany) and then visualized with a UV254 lamplight. Chromatographic separations were performed on silica gel columns by the flash method (Kieselgel 40, 0.040–0.063 mm, Merck, Darmstadt, Germany). Melting points were determined in open glass capillaries using a Büchi apparatus, and are uncorrected. 1H NMR and 13C NMR spectra were recorded on a Varian Gemini spectrometer at 400 MHz and 101 MHz, respectively, in CDCl3 solutions unless otherwise indicated, and chemical shifts (δ) were reported as parts per million (ppm) values relative to tetramethylsilane (TMS) as the internal standard; coupling constants (J) are reported in Hertz (Hz). Standard abbreviations indicating spin multiplicities are given as follows: s (singlet), d (doublet), t (triplet), br (broad), q (quartet), or m (multiplet). Mass spectra were recorded on a Waters ZQ 4000 apparatus operating in electrospray mode (ES). Chemical purities of tested compounds were determined by elemental analysis (C, H, N) and were within ±0.4% of the theoretical values. Compounds were named relying on the naming algorithm developed by CambridgeSoft Corporation (Waltham, MA, USA) and used in ChemDraw Professional 15.0 (Perkin Elmer INC., Waltham, MA, USA).
4.1.2. General Method I: Preparation of 4a–e
The selected 4-Xant-dihydropyridine (
3a–
e) [
12,
13] (2.7 mmol) in CH
2Cl
2 was cooled at 0 °C and treated with 0.31 mL of pyridine and 0.93 g of pyridinium bromide perbromide (2.9 mmol). The mixture was stirred at 0 °C for 40 min, diluted with CH
2Cl
2 (30 mL), and washed with HCl 2N (3 × 20 mL) and brine. The organic layer was evaporated to dryness and the residue was solubilized in THF (20 mL). A suspension of
N-(2,6-dimethyl-phenyl)-2-piperazin-1-ylacetamide (5.4 mmol) and K
2CO
3 (1 g) in DMF was then slowly added and the resulting mixture was stirred at room temperature overnight. The solvent was concentrated under reduced pressure, and the residue was diluted with CH
2Cl
2 (30 mL), washed with H
2O (3 × 30 mL), dried over sodium sulfate, and the solvent evaporated to dryness.
Dimethyl 2-((4-(2-((2,6-Dimethylphenyl)amino)-2-oxoethyl)piperazin-1-yl)methyl)-6-methyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (4a). The title compound was obtained starting from 3a (30%) and was purified by flash column chromatography (toluene/acetone 3/2) and crystallization from toluene; mp 110–112 °C. 1H NMR δ: 2.23 (s, 6H), 2.44 (s, 3H), 2.68 (broad, 4H), 2.81 (broad, 4H), 3.28 (s, 2H), 3.52 (s, 3H), 3.53 (s, 3H), 3.74–3.91 (m, 2H), 5.70 (s, 1H), 7.10–7.12 (m, 3H arom), 7.39 (t, J = 8.0 Hz, 1H arom), 7.52 (d, J = 8.0, 1H arom), 7.68–7.74 (m, 2H arom), 7.99 (s, 1H, NH DHP), 8.20 (dd, J = 8.0 and 1.6 Hz, 1H arom), 8.35 (dd, J = 8.0 and 1.6 Hz, 1H arom), 8.54 (s, 1H, CONH). 13C NMR δ: 18.6, 19.7, 35.8, 35.9, 50.9, 53.5, 53.9, 56.6, 61.7, 102.2, 102.9, 111.2, 117.5, 121.8, 123.4, 123.7, 125.2, 126.9, 127.3, 128.3, 129.0, 131.5, 133.4, 134.6, 134.8, 135.5, 136.5, 144.7, 153.4, 155.3, 156.1, 164.0, 166.7, 167.5, 167.9, 168.3, 177.5. ES-MS: m/z 661 [M + H].
Diethyl 2-((4-(2-((2,6-Dimethylphenyl)amino)-2-oxoethyl)piperazin-1-yl)methyl)-6-methyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (4b). The title compound was obtained starting from 3b (33%) and was purified by flash column chromatography (toluene/acetone 3/2) and crystallization from toluene; mp 234–236 °C. 1H NMR δ: 0.97–1.01 (m, 6H), 2.23 (s, 6H), 2.43 (s, 3H), 2.69 (broad, 4H), 2.81 (broad, 4H), 3.28 (s, 2H), 3.76–3.99 (m, 6H), 5.70 (s, 1H), 7.08–7.12 (m, 3H arom), 7.28–7.30 (m, 1H arom), 7.38 (t, J = 8.0 Hz, 1H arom), 7.50 (d, J = 8.0, 1H arom), 7.71–7.75 (m, 2H arom), 7.99 (s, 1H, NH DHP), 8.21 (dd, J = 8.0 and 1.6 Hz, 1H arom), 8.36 (dd, J = 8.0 and 1.6 Hz, 1H arom), 8.55 (s, 1H, CONH). 13C NMR δ: 15.2, 18.6, 19.7, 35.8, 35.9, 50.9, 53.5, 53.9, 56.5, 61.7, 102.2, 102.9, 111.2, 117.5, 121.8, 123.4, 123.7, 125.2, 126.9, 127.3, 128.3, 129.0, 131.5, 133.4, 134.5, 134.8, 135.4, 136.5, 144.7, 153.4, 155.3, 156.2, 164.1, 166.7, 167.5, 168.0, 168.4, 177.5. ES-MS: m/z 693 [M + H].
Diisopropyl 2-((4-(2-((2,6-Dimethylphenyl)amino)-2-oxoethyl)piperazin-1-yl)methyl)-6-methyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (4c). The title compound was obtained starting from 3c (28%) and was purified by flash column chromatography (toluene/acetone 3/2) and crystallization from toluene; mp 134–136 °C. 1H NMR δ: 0.68–0.72 (m, 6H), 1.12–1.18 (m, 6H), 2.24 (s, 6H), 2.42 (s, 3H), 2.69 (broad, 4H), 2.81 (broad, 4H), 3.27 (s, 2H), 3.75–3.96 (m, 2H), 4.81–4.93 (m, 2H), 5.68 (s, 1H), 7.11–7.15 (m, 3H arom), 7.28–7.30 (m, 1H arom), 7.38 (t, J = 8.0 Hz, 1H arom), 7.50 (d, J = 8.0, 1H arom), 7.73–7.77 (m, 2H arom), 7.97 (s, 1H, NH DHP), 8.20 (dd, J = 8.0 and 1.6 Hz, 1H arom), 8.37 (dd, J = 8.0 and 1.6 Hz, 1H arom), 8.58 (s, 1H, CONH). 13C NMR δ: 18.6, 19.7, 21.7, 36.0, 53.5, 54.0, 56.5, 61.7, 68.8, 102.3, 102.9, 111.2, 117.5, 121.8, 123.4, 123.7, 125.2, 126.9, 127.2, 128.3, 129.0, 131.5, 133.4, 134.5, 134.9, 135.4, 136.5, 144.7, 153.4, 155.3, 156.2, 164.1, 166.7, 167.5, 168.0, 168.3, 177.5. ES-MS: m/z 721 [M + H].
Diallyl 2-((4-(2-((2,6-Dimethylphenyl)amino)-2-oxoethyl)piperazin-1-yl)methyl)-6-methyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (4d). The title compound was obtained starting from 3d (28%) and was purified by flash column chromatography (toluene/acetone 3/2) and crystallization from toluene; mp 112–115 °C. 1H NMR δ 2.23 (s, 6H), 2.44 (s, 3H), 2.69 (broad, 4H), 2.82 (broad, 4H), 3.28 (s, 2H), 3.76–3.94 (m, 2H), 4.38–4.41 (m, 4H), 4.86–4.96 (m, 4H), 5.54–5.60 (m, 2H), 5.77 (s, 1H), 7.09–7.12 (m, 3H arom), 7.29–7.31 (m, 1H arom), 7.38 (t, J = 8.0 Hz, 1H arom),7.47 (d, J = 8.0, 1H arom), 7.70–7.72 (m, 2H arom), 8.00 (s, 1H, NH DHP), 8.18 (dd, J = 8.0 and 1.6 Hz, 1H arom), 8.34 (dd, J = 8.0 and 1.6 Hz, 1H arom), 8.55 (s, 1H, CONH). 13C NMR δ: 18.6, 19.8, 35.3, 53.5, 53.9, 56.6, 61.7, 64.5, 64.6, 102.47, 103.2, 117.7, 117,9, 121.5, 121.6, 123.4, 123.6, 125.2, 126.7, 127.3, 128.3, 132.3, 132.4, 133.4, 134.3, 134.8, 136.4, 137.3, 144.9, 145.1, 153.1, 156.1, 166.7, 166.9, 168.0, 177.6. ES-MS: m/z 718 [M + H].
Di(prop-2-yn-1-yl) 2-((4-(2-((2,6-Dimethylphenyl)amino)-2-oxoethyl)piperazin-1-yl)methyl)-6-methyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (4e). The title compound was obtained starting from 3e (28%) and was purified by flash column chromatography (toluene/acetone 3/2) and crystallization from toluene; mp 171–174 °C. 1H NMR δ: 2.02 (t, J = 2.4 Hz, 2H), 2.23 (s, 6H), 2.45 (s, 3H), 2.70 (broad, 4H), 2.82 (broad, 4H), 3.28 (s, 2H), 3.76–3.93 (m, 2H), 4.89–4.51 (m, 4H), 5.78 (s, 1H), 7.08–7.12 (m, 3H arom), 7.29–7.31 (m, 1H arom), 7.37 (t, J = 7.6 Hz, 1H arom),7.56 (d, J = 8.8, 1H arom), 7.70–7.74 (m, 2H arom), 8.11 (s, 1H, NH DHP), 8.21 (dd, J = 8.4 and 1.6 Hz, 1H arom), 8.35 (dd, J = 8.0 and 1.6 Hz, 1H arom), 8.54 (s, 1H, CONH). 13C NMR δ: 18.6, 19.8, 35.0, 51.2, 53.5, 53.9, 56.6, 61.7, 74.1, 74.2, 102.9, 118.2, 121.6, 121.7, 123.5, 125.3, 126.6, 127.3, 128.3, 133.4, 134.3, 134.8, 136.3, 136.9, 145.5, 145.7, 156.3, 166.1, 166.3, 168.0, 177.6. ES-MS: m/z 713 [M + H].
4.1.3. General Method II: Preparation of 6a–e
A solution of 9-oxo-9H-xanthene-4-carbaldehyde (1 eq), 2-cyanoethyl 3-oxobutanoate (1 eq), and the selected aminocrotonate (1 eq) in isopropanol was heated under reflux for 20 h. The solvent was removed under reduced pressure and the residue was purified by flash column chromatography (toluene/ethyl acetate 9/1).
3-(2-Cyanoethyl) 5-methyl 2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (6a). The compound (50%) was obtained starting from methyl 3-aminocrotonate. Mp 212–214 °C. 1H NMR δ: 2.40 (s, 3H), 2.43 (s, 3H), 2.46–2.59 (m, 2H), 3.55 (s, 3H), 4.08–4.28 (m, 2H), 5.67 (s, 1H), 5.94 (s, 1H, NH DHP), 7.31 (t, J = 7.2 Hz, 1H arom), 7.42 (t, J = 7.2 Hz, 1H arom), 7.63 (d, J = 8.0 Hz, 1H arom), 7.75–7.79 (m, 2H arom), 8.26 (d, J = 7.4 Hz, 1H arom), 8.39 (d, J = 7.4 Hz, 1H arom).
3-(2-cyanoethyl) 5-ethyl 2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (6b). The compound (60%) was obtained starting from ethyl 3-aminocrotonate. Mp 204–206 °C. 1H NMR δ: 0.99 (t, J = 8.0 Hz, 3H), 2.37 (s, 3H), 2.39 (s, 3H), 2.46–2.55 (m, 2H), 3.92–3.96 (m, 2H), 4.08–4.29 (m, 2H), 5.67 (s, 1H), 6.20 (s, 1H, NH DHP), 7.29 (t, J = 7.2 Hz, 1H arom), 7.41 (t, J = 7.2 Hz, 1H arom), 7.62 (d, J = 7.6 Hz, 1H arom), 7.74–7.77 (m, 2H arom), 8.23 (d, J = 7.2 Hz, 1H arom), 8.33 (d, J = 7.2 Hz,1H arom).
3-(2-Cyanoethyl) 5-isopropyl 2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (
6c). The compound (55%) was obtained starting from isopropyl 3-aminocrotonate [
22]. Mp 186–188 °C.
1H NMR δ: 0.78 (d,
J = 6.0 Hz, 3H), 1.17 (d,
J = 6.0 Hz, 3H), 2.40 (s, 3H), 2.42 (s, 3H), 2.50–2.54 (m, 2H), 4.08–4.28 (m, 2H), 4.83–4.90 (m, 1H), 5.68 (s, 1H), 6.08 (s, 1H, NH DHP), 7.31–7.42 (m, 2H arom), 7.64 (d,
J = 8.0 Hz, 1H arom), 7.76–7.80 (m, 2H arom), 8.26 (d,
J = 7.4 Hz, 1H arom), 8.38 (d,
J = 7.4 Hz, 1H arom).
3-(2-Cyanoethyl) 5-allyl 2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (
6d). The compound (58%) was obtained starting from allyl 3-aminocrotonate [
23]. Mp 190–192 °C.
1H NMR δ: 2.39 (s, 3H), 2.41 (s, 3H), 2.44–2.53 (m, 2H), 3.98–4.28 (m, 2H), 4.40 (d,
J = 6.0 Hz, 2H), 4.88–4.96 (m, 2H), 5.52–5.66 (m, 1H), 5.70 (s, 1H), 6.05 (s, 1H, NH DHP), 7.29–7.42 (m, 2H arom), 7.67 (d,
J = 8.0 Hz, 1H arom), 7.76–7.78 (m, 2H arom), 8.23 (d,
J = 7.4 Hz, 1H arom), 8.39 (d,
J = 7.4 Hz, 1H arom).
3-(2-Cyanoethyl) 5-propargyl 2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (
6e). The compound (62%) was obtained starting from propargyl 3-aminocrotonate [
22]. Mp 272–275 °C.
1H NMR δ: 2.01 (t,
J = 3.0 Hz, 1H), 2.39 (s, 3H), 2.42 (s, 3H), 2.46–2.51 (m, 2H), 3.98–4.28 (m, 2H), 4.50 (s, 2H), 5.70 (s, 1H), 6.28 (s, 1H, NH DHP), 7.28–7.45 (m, 2H arom), 7.67 (d,
J = 8.0 Hz, 1H arom), 7.73–7.77 (m, 2H arom), 8.23 (d,
J = 7.4 Hz, 1H arom), 8.39 (d,
J = 7.4 Hz, 1H arom).
4.1.4. General Method III: Preparation of Monoesters 7a–e
Dihydropyridine derivatives 6a–e (2.8 mmol) were dissolved in ethanol and LiOH (5.0 mmol) was added. The solution was stirred at rt for 16 h, the solvent was removed in vacuo, and the residue was kept in H2O, filtered, and acidified with diluted (1:1) HCl. Derivatives 7a–e were obtained by filtration and used in the subsequent step without further purification.
5-(Methoxycarbonyl)-2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3-carboxylic acid (7a). The compound was obtained starting from 6a (72%). Mp 197–199 °C. 1H NMR δ: 2.40 (s, 3H), 2.43 (s, 3H), 3.55 (s, 3H), 5.69 (s, 1H), 5.98 (s, 1H, NH DHP), 7.30 (t, J = 7.2 Hz, 1H arom), 7.42 (t, J = 7.2 Hz, 1H arom), 7.65 (d, J = 8.0 Hz, 1H arom), 7.75–7.80 (m, 2H arom), 8.27 (d, J = 7.4 Hz, 1H arom), 8.39 (d, J = 7.4 Hz, 1H arom).
5-(Ethoxycarbonyl)-2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3-carboxylic acid (7b). The compound was obtained starting from 6b (70%). Mp 190–193 °C. 1H NMR (DMSO) δ: 0.98 (t, J = 8.0 Hz, 3H), 2.27 (s, 3H), 2.29 (s, 3H), 3.80 (q, J = 8.0 Hz, 2H), 5.48 (s, 1H), 7.31 (t, J = 7.2 Hz, 1H arom), 7.48 (t, J = 7.2 Hz, 1H arom), 7.68–7.72 (m, 2H arom), 7.81–7.92 (m, 1H arom), 8.01 (d, J = 7.2 Hz, 1H arom), 8.18 (d, J = 7.2 Hz, 1H arom), 9.02 (s, 1H, NH DHP).
5-(Isopropoxycarbonyl)-2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3-carboxylic acid (7c). The compound was obtained starting from 6c (65%). Mp 157–160 °C. 1H NMR δ: 0.81 (d, J = 6.0 Hz, 3H), 1.14 (d, J = 6.0 Hz, 3H), 2.30 (s, 3H), 2.36 (s, 3H), 4.83–4.91 (m, 1H), 5.55 (s, 1H), 6.08 (s, 1H, NH DHP), 7.20–7.38 (m, 2H arom), 7.48 (d, J = 8.0 Hz, 1H arom), 7.65–7.76 (m, 2H arom), 8.12 (d, J = 7.4 Hz, 1H arom), 8.28 (d, J = 7.4 Hz, 1H arom).
5-((Allyloxy)carbonyl)-2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3-carboxylic acid (7d). The compound was obtained starting from 6d (69%). Mp 177–179 °C. 1H NMR (DMSO) δ: 2.329 (s, 3H), 2.32 (s, 3H), 4.34 (d, J = 6.0 Hz, 2H), 4.82–4.93 (m, 2H), 5.41 (s, 1H), 5.52–5.68 (m, 1H), 7.28–7.42 (m, 2H arom), 7.63–7.69 (m, 2H arom), 7.76–7.80 (m, 2H arom), 8.16 (d, J = 7.4 Hz, 1H arom), 9.05 (s, 1H, NH DHP).
2,6-Dimethyl-4-(9-oxo-9H-xanthen-4-yl)-5-((prop-2-yn-1-yloxy)carbonyl)-1,4-dihydropyridine-3-carboxylic acid (7e). The compound was obtained starting from 6e (60%). Mp 139–141 °C. 1H NMR δ: 2.00 (t, J = 3.0 Hz, 1H), 2.39 (s, 3H), 2.42 (s, 3H), 4.53 (s, 2H), 5.60 (s, 1H), 6.28 (s, 1H, NH DHP), 7.28–7.45 (m, 2H arom), 7.68 (d, J = 8.0 Hz, 1H arom), 7.73–7.79 (m, 2H arom), 8.19 (d, J = 7.4 Hz, 1H arom), 8.27 (d, J = 7.4 Hz, 1H arom).
4.1.5. General Method IV: Preparation of Compounds 5a–e
To a solution of 1.77 mmol of the previously obtained monoester in THF (7.5 mL), 0.5 g of CDI (3.5 mmol) were added and the slurry was stirred for 1 h at rt. The solvent was removed under reduced pressure, N-(2,6-dimethylphenyl)-2-(4-(2-hydroxyethyl)piperazin-1-yl)acetamide (1.75 mmol) was added, and the mixture was heated at 120 °C for 50 min. After cooling, the mixture was purified by flash column chromatography (toluene/acetone 1/1).
3-(2-(4-(2-((2,6-Dimethylphenyl)amino)-2-oxoethyl)piperazin-1-yl)ethyl) 5-methyl 2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (5a). The compound was obtained starting from 7a (40%). Mp 254–256 °C (toluene). 1H NMR δ: 2.25 (s, 6H), 2.27–2.34 (m, 6H), 2.36 (s, 3H), 2.39 (s, 3H), 2.53 (s, 4H), 3.12 (s, 2H), 3.51 (s, 3H), 3.97–4.12 (m, 2H), 5.69 (s, 1H), 5.83 (s, 1H, NH DHP), 7.10–7.12 (m, 3H arom), 7.28–7.30 (m, 1H arom), 7.36 (t, J = 7.2 Hz, 1H arom), 7.57 (d, J = 7.6, 1H, arom), 7.72–7.76 (m, 2H arom), 8.18 (dd J = 8.4 and 1.6 Hz, 1H arom), 8.33 (d, J = 6.4 Hz, 1H arom), 8.57 (s, 1H, NHCO). 13C NMR δ: 18.6, 19.5, 19.6, 35.0, 50.9, 53.1, 53.6, 56.3, 60.8, 61.6, 102.9, 103.2, 117.7, 121.6, 121.6, 123.5, 123.7, 125.1, 126.8, 127.2, 128.2, 133.6, 134.7, 134.9, 136.2, 137.1, 144.3, 145.0, 153.1, 156.1, 167.1, 167.8, 168.4, 177.5. ES-MS: m/z 679 [M + H].
3-(2-(4-(2-((2,6-Dimethylphenyl)amino)-2-oxoethyl)piperazin-1-yl)ethyl) 5-ethyl 2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (5b). The compound was obtained starting from 7b (35%). Mp 206–210 °C (toluene). 1H NMR δ: 1.10 (t, J = 8.0 Hz, 3H), 2.25 (s, 6H), 2.27–2.34 (m, 6H), 2.36 (s, 3H), 2.39 (s, 3H), 2.57 (s, 4H), 3.15 (s, 2H), 3.51 (s, 3H), 3.92–4.22 (m, 4H), 5.68 (s, 1H), 5.90 (s, 1H, NH DHP), 7.12–7.15 (m, 3H arom), 7.28–7.31 (m, 1H arom), 7.36 (t, J = 7.2 Hz, 1H arom), 7.56 (d, J = 7.6, 1H, arom), 7.73–7.78 (m, 2H arom), 8.18 (dd J = 8.4 and 1.6 Hz, 1H arom), 8.32 (d, J = 6.4 Hz, 1H arom), 8.57 (s, 1H, NHCO). 13C NMR δ: 15.3, 18.6, 19.6, 19.6, 35.1, 50.9, 53.6, 56.3, 60.8, 61.6, 63.4, 103.0, 103.3, 117.7, 121.6, 121.6, 123.5, 123.7, 125.1, 126.9, 127.1, 128.2, 133.6, 134.7, 135.0, 136.2, 137.1, 144.3, 145.0, 153.1, 156.1, 167.1, 167.8, 168.4, 177.5. ES-MS: m/z 693 [M + H].
3-(2-(4-(2-((2,6-Dimethylphenyl)amino)-2-oxoethyl)piperazin-1-yl)ethyl) 5-isopropyl 2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (5c). The compound was obtained starting from 7c (40%). Mp 206–210 °C (toluene). 1H NMR δ: 0.76 (d, J = 6.0 Hz, 3H), 1.10 (d, J = 6.0 Hz, 3H), 2.23 (s, 6H), 2.24–2.29 (m, 4H), 2.32–2.35 (m, 2H), 2.38 (s, 6H), 2.55 (broad, 4H), 3.13 (s, 2H), 3.96–4.14 (m, 2H), 4.86–4.91 (m, 1H), 5.65 (s, 1H), 5.71 (s, 1H, NH DHP), 7.08–7.11 (m, 3H arom), 7.28–7.30 (m, 1H arom), 7.37 (t, J = 7.2 Hz, 1H arom), 7.57 (d, J = 8.4 Hz, 1H, arom), 7.73–7.78 (m, 2H arom), 8.17 (dd J = 8.0 and 1.6 Hz, 1H arom), 8.32 (d, J = 6.8 Hz, 1H arom), 8.57 (s, 1H, NHCO). 13C NMR δ: 18.7, 19.5, 19.6, 21.4, 35.1, 53.1, 53.6, 56.3, 60.8, 61.6, 65.4, 103.0, 103.2, 117.7, 121.6, 121.6, 123.5, 123.6, 125.1, 126.8, 127.1, 128.2, 133.6, 134.7, 134.9, 136.2, 137.1, 144.3, 145.0, 153.1, 156.2, 167.1, 167.8, 168.4, 177.5. ES-MS: m/z 707 [M + H].
3-Allyl 5-(2-(4-(2-((2,6-dimethylphenyl)amino)-2-oxoethyl)piperazin-1-yl)ethyl) 2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (5d). The compound was obtained starting from 7d (30%). Mp 82–85 °C (toluene). 1H NMR δ: 2.22 (s, 6H), 2.23–2.28 (m, 6H), 2.36 (s, 3H), 2.39 (s, 3H), 2.53 (broad, 4H), 3.12 (s, 2H), 3.97–4.12 (m, 2H), 4.40 (d, J = 6.0 Hz, 2H), 4.90–4.96 (m, 2H), 5.57–5.64 (m, 1H), 5.70 (s, 1H), 6.52 (broad, 1H, NH DHP), 7.10–7.14 (m, 3H arom), 7.28–7.30 (m, 1H arom), 7.35 (t, J = 7.2 Hz, 1H arom), 7.56 (d, J = 7.6, 1H, arom), 7.68–7.73 (m, 2H arom), 8.17 (dd J = 8.0 and 1.6 Hz, 1H arom), 8.31 (d, J = 6.8 Hz, 1H arom), 8.61 (s, 1H, NHCO). 13C NMR δ: 18.5, 19.3, 19.4, 35.2, 53.0, 53.6, 56.3, 60.8, 61.5, 64.5, 102.6, 102.6, 117.7, 118.0, 121.5, 121.6, 121.8, 123.4, 123.6, 124.9, 126.6, 127.4, 128.3, 132.5, 133.4, 134.6, 134.9, 135.0, 136.6, 137.4, 145.4, 145.6, 153.2, 156.1, 167.1, 167.3, 168.9, 177.7. ES-MS: m/z 705 [M + H].
3-(2-(4-(2-((2,6-Dimethylphenyl)amino)-2-oxoethyl)piperazin-1-yl)ethyl) 5-(prop-2-yn-1-yl) 2,6-dimethyl-4-(9-oxo-9H-xanthen-4-yl)-1,4-dihydropyridine-3,5-dicarboxylate (5e). The compound was obtained starting from 7e (30%). Mp 127–129 °C (toluene/petroleum ether 3/2). 1H NMR δ: 2.01 (t, J = 3.0 Hz, 1H), 2.24 (s, 6H), 2.27–2.34 (m, 6H), 2.36 (s, 3H), 2.38 (s, 3H), 2.53 (s, 4H), 3.12 (s, 2H), 3.98–4.13 (m, 2H), 4.46 (s, 2H), 5.62 (s, 1H), 5.80 (broad, 1H, NH DHP), 7.11–7.13 (m, 3H arom), 7.28–7.30 (m, 1H arom), 7.37 (t, J = 7.2 Hz, 1H arom), 7.58 (d, J = 7.6, 1H, arom), 7.72–7.76 (m, 2H arom), 8.17 (dd J = 8.0 and 1.6 Hz, 1H arom), 8.34 (d, J = 6.8 Hz, 1H arom), 8.57 (s, 1H, NHCO). 13C NMR δ: 18.6, 19.6, 19.7, 35.0, 51.4, 53.1, 53.6, 56.3, 60.7, 61.6, 72.4, 72.7, 103.0, 103.2, 117.8, 121.6, 121.6, 123.5, 123.7, 125.0, 126.8, 127.1, 128.2, 133.6, 134.7, 134.9, 136.2, 137.1, 144.3, 145.0, 153.1, 156.1, 167.1, 167.8, 168.4, 177.5. ES-MS: m/z 703 [M + H].