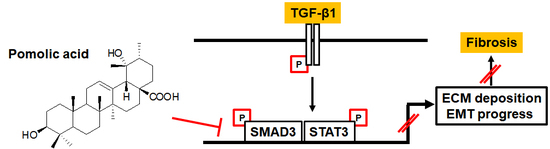
Pomolic Acid Ameliorates Fibroblast Activation and Renal Interstitial Fibrosis through Inhibition of SMAD-STAT Signaling Pathways
Abstract
:1. Introduction
2. Results
2.1. PA Improves Histopathological Changes in UUO Mice
2.2. PA Attenuates UUO-Induced EMT Progression
2.3. PA Inhibits TGF-β1 Stimulated Fibroblast Activation
2.4. PA Inhibits TGF-β1 Induced Canonical and Non-Canonical Signaling
3. Discussion
4. Materials and Methods
4.1. Cell Cultures and Reagents
4.2. Cytotoxicity Assay
4.3. Induction of UUO Injury
4.4. Pathology
4.5. Immunoblot Analysis
4.6. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| TGF-β1 | Transforming growth factor β1 |
| SMAD3 | Mothers against decapentaplegic homolog 3 |
| STAT3 | Signal transducer and activator of transcription 3 |
| PA | Pomolic acid |
| ECM | Extracellular matrix |
| EMT | Epithelial-mesenchymal transition |
| α-SMA | α-Smooth muscle actin |
| mTOR | Mammalian target of rapamycin |
| DMSO | Dimethylsulfoxide |
References
- An, H.J.; Kim, J.Y.; Kim, W.H.; Han, S.M.; Park, K.K. The Protective Effect of Melittin on Renal Fibrosis in an Animal Model of Unilateral Ureteral Obstruction. Molecules 2016, 21, 1137. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xu, X.; Yang, R.; Chen, J.; Wang, S.; Yang, J.; Xiang, X.; He, Z.; Zhao, Y.; Dong, Z.; et al. Paclitaxel attenuates renal interstitial fibroblast activation and interstitial fibrosis by inhibiting STAT3 signaling. Drug Des. Dev. Ther. 2015, 9, 2139–2148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Jia, L.; Hu, Z.; Entman, M.L.; Mitch, W.E.; Wang, Y. AMP-activated protein kinase/myocardin-related transcription factor-A signaling regulates fibroblast activation and renal fibrosis. Kidney Int. 2018, 93, 81–94. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.S.; Kim, J.H.; Jang, H.N.; Lee, T.W.; Jung, M.H.; Kim, T.H.; Chang, S.H.; Park, D.J. Alpha-lipoic acid ameliorates the epithelial mesenchymal transition induced by unilateral ureteral obstruction in mice. Sci. Rep. 2017, 7, 46065. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.S.; Kim, I.J.; Bae, E.H.; Ma, S.K.; Lee, J.; Kim, S.W. Angiotensin-(1-7) Attenuates Kidney Injury Due to Obstructive Nephropathy in Rats. PLoS ONE 2015, 10, e0142664. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Xia, Y.; Lin, X.; Feng, X.H.; Wang, Y. Smad3 signaling activates bone marrow-derived fibroblasts in renal fibrosis. Lab. Investig. 2014, 94, 545–556. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castellone, M.D.; Laukkanen, M.O. TGF-beta1, WNT, and SHH signaling in tumor progression and in fibrotic diseases. Front. Biosci. (Schol. Ed.) 2017, 9, 31–45. [Google Scholar] [PubMed]
- Lopez-Hernandez, F.J.; Lopez-Novoa, J.M. Role of TGF-beta in chronic kidney disease: An integration of tubular, glomerular and vascular effects. Cell Tissue Res. 2012, 347, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Xu, M.; Wang, W.; Zhang, J. Lefty1 alleviates renal tubulointerstitial injury in mice with unilateral ureteral obstruction. Mol. Med. Rep. 2016, 13, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.R.; Chung, A.C.; Yang, F.; Yue, W.; Deng, C.; Lau, C.P.; Tse, H.F.; Lan, H.Y. Smad3 mediates cardiac inflammation and fibrosis in angiotensin II-induced hypertensive cardiac remodeling. Hypertension 2010, 55, 1165–1171. [Google Scholar] [CrossRef] [PubMed]
- Kuratsune, M.; Masaki, T.; Hirai, T.; Kiribayashi, K.; Yokoyama, Y.; Arakawa, T.; Yorioka, N.; Kohno, N. Signal transducer and activator of transcription 3 involvement in the development of renal interstitial fibrosis after unilateral ureteral obstruction. Nephrology (Carlton) 2007, 12, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Youn, S.H.; Lee, J.S.; Lee, M.S.; Cha, E.Y.; Thuong, P.T.; Kim, J.R.; Chang, E.S. Anticancer properties of pomolic acid-induced AMP-activated protein kinase activation in MCF7 human breast cancer cells. Biol. Pharm. Bull. 2012, 35, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.K.; Lee, K.Y.; Jeon, H.Y.; Sung, S.H.; Kim, Y.C. Antifibrotic activity of triterpenoids from the aerial parts of Euscaphis japonica on hepatic stellate cells. J. Enzyme Inhib. Med. Chem. 2009, 24, 1276–1279. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; Fuchigami, M.; Nagao, T.; Okabe, H.; Matsunaga, K.; Takata, J.; Karube, Y.; Tsuchihashi, R.; Kinjo, J.; Mihashi, K.; et al. Antiproliferative constituents from Umbelliferae plants VII. Active triterpenes and rosmarinic acid from Centella asiatica. Biol. Pharm. Bull. 2005, 28, 173–175. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Cho, Y.Y.; Yoon, S.W.; Park, B. Suppression of MMP-9 and FAK expression by pomolic acid via blocking of NF-kappaB/ERK/mTOR signaling pathways in growth factor-stimulated human breast cancer cells. Int. J. Oncol. 2016, 49, 1230–1240. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Yoon, J.; Park, B. Pomolic acid suppresses HIF1alpha/VEGF-mediated angiogenesis by targeting p38-MAPK and mTOR signaling cascades. Phytomedicine 2016, 23, 1716–1726. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Park, B.; Park, K.K. Suppression of Hepatic Epithelial-to-Mesenchymal Transition by Melittin via Blocking of TGFbeta/Smad and MAPK-JNK Signaling Pathways. Toxins 2017, 9, 138. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Lee, M.K.; Yoon, J. Gamma-linolenic acid inhibits hepatic PAI-1 expression by inhibiting p38 MAPK-dependent activator protein and mitochondria-mediated apoptosis pathway. Apoptosis 2015, 20, 336–347. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Yoon, J.; Lee, K.Y.; Park, B. Effects of geniposide on hepatocytes undergoing epithelial-mesenchymal transition in hepatic fibrosis by targeting TGFbeta/Smad and ERK-MAPK signaling pathways. Biochimie 2015, 113, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Pang, M.; Ma, L.; Gong, R.; Tolbert, E.; Mao, H.; Ponnusamy, M.; Chin, Y.E.; Yan, H.; Dworkin, L.D.; Zhuang, S. A novel STAT3 inhibitor, S3I-201, attenuates renal interstitial fibroblast activation and interstitial fibrosis in obstructive nephropathy. Kidney Int. 2010, 78, 257–268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okada, H.; Kalluri, R. Cellular and molecular pathways that lead to progression and regression of renal fibrogenesis. Curr. Mol. Med. 2005, 5, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.C.; Zuo, Y.; Fogo, A.B. Models of chronic kidney disease. Drug Discov. Today Dis. Models 2010, 7, 13–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, K.H.; Park, J.H.; Lee, W.R.; Park, J.S.; Kim, H.C.; Park, K.K. The inhibitory effect of chimeric decoy oligodeoxynucleotide against NF-kappaB and Sp1 in renal interstitial fibrosis. J. Mol. Med. 2013, 91, 573–586. [Google Scholar] [CrossRef] [PubMed]
- Schinella, G.; Aquila, S.; Dade, M.; Giner, R.; Recio Mdel, C.; Spegazzini, E.; de Buschiazzo, P.; Tournier, H.; Rios, J.L. Anti-inflammatory and apoptotic activities of pomolic acid isolated from Cecropia pachystachya. Planta Med. 2008, 74, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, J.; Weinlich, R.; Castilho, R.O.; Amarante-Mendes, G.P.; Gattass, C.R. Pomolic acid may overcome multidrug resistance mediated by overexpression of anti-apoptotic Bcl-2 proteins. Cancer Lett. 2007, 245, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, F.C.; Gattass, C.R.; Rumjanek, V.M.; Maia, R.C. Pomolic acid-induced apoptosis in cells from patients with chronic myeloid leukemia exhibiting different drug resistance profile. Investig. New Drugs 2007, 25, 525–533. [Google Scholar] [CrossRef] [PubMed]
- Fagone, P.; Mangano, K.; Pesce, A.; Portale, T.R.; Puleo, S.; Nicoletti, F. Emerging therapeutic targets for the treatment of hepatic fibrosis. Drug Discov. Today 2016, 21, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, F.; Amiri, P.; Sayahpour, F.A.; Pirmoradi, S.; Abolhalaj, M.; Larijani, B.; Bazzaz, J.T.; Amoli, M.M. TGF-beta and IL-23 gene expression in unstimulated PBMCs of patients with diabetes. Endocrine 2012, 41, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Ayyaz, A.; Attisano, L.; Wrana, J.L. Recent advances in understanding contextual TGFbeta signaling. F1000Res. 2017, 6, 749. [Google Scholar] [CrossRef] [PubMed]
- Nicoletti, F.; Di Marco, R.; Patti, F.; Reggio, E.; Nicoletti, A.; Zaccone, P.; Stivala, F.; Meroni, P.; Reggio, A. Blood levels of transforming growth factor-beta 1 (TGF-β1) are elevated in both relapsing remitting and chronic progressive multiple sclerosis (MS) patients and are further augmented by treatment with interferon-beta 1b (IFN-β1b). J. Clin. Exp. Immunol. 1998, 113, 96–99. [Google Scholar] [CrossRef]
- Choi, H.S.; Song, J.H.; Kim, I.J.; Joo, S.Y.; Eom, G.H.; Kim, I.; Cha, H.; Cho, J.M.; Ma, S.K.; Kim, S.W.; et al. Histone deacetylase inhibitor, CG200745 attenuates renal fibrosis in obstructive kidney disease. Sci. Rep. 2018, 8, 11546. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Solbes, A.S.; Youker, K. Epithelial to Mesenchymal Transition (EMT) and Endothelial to Mesenchymal Transition (EndMT): Role and Implications in Kidney Fibrosis. Results Probl. Cell Differ. 2017, 60, 345–372. [Google Scholar] [PubMed]
- Boutet, A.; De Frutos, C.A.; Maxwell, P.H.; Mayol, M.J.; Romero, J.; Nieto, M.A. Snail activation disrupts tissue homeostasis and induces fibrosis in the adult kidney. EMBO J. 2006, 25, 5603–5613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lan, H.Y. Diverse roles of TGF-beta/Smads in renal fibrosis and inflammation. Int. J. Biol. Sci. 2011, 7, 1056–1067. [Google Scholar] [CrossRef] [PubMed]
- An, H.J.; Kim, K.H.; Lee, W.R.; Kim, J.Y.; Lee, S.J.; Pak, S.C.; Han, S.M.; Park, K.K. Anti-fibrotic effect of natural toxin bee venom on animal model of unilateral ureteral obstruction. Toxins 2015, 7, 1917–1928. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |









© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, J.-H.; Jang, K.M.; An, H.J.; Kim, J.-Y.; Gwon, M.-G.; Gu, H.; Park, B.; Park, K.-K. Pomolic Acid Ameliorates Fibroblast Activation and Renal Interstitial Fibrosis through Inhibition of SMAD-STAT Signaling Pathways. Molecules 2018, 23, 2236. https://doi.org/10.3390/molecules23092236
Park J-H, Jang KM, An HJ, Kim J-Y, Gwon M-G, Gu H, Park B, Park K-K. Pomolic Acid Ameliorates Fibroblast Activation and Renal Interstitial Fibrosis through Inhibition of SMAD-STAT Signaling Pathways. Molecules. 2018; 23(9):2236. https://doi.org/10.3390/molecules23092236
Chicago/Turabian StylePark, Ji-Hyun, Kyung Mi Jang, Hyun Jin An, Jung-Yeon Kim, Mi-Gyeong Gwon, Hyemin Gu, Byoungduck Park, and Kwan-Kyu Park. 2018. "Pomolic Acid Ameliorates Fibroblast Activation and Renal Interstitial Fibrosis through Inhibition of SMAD-STAT Signaling Pathways" Molecules 23, no. 9: 2236. https://doi.org/10.3390/molecules23092236