New Anti-Inflammatory Aporphine and Lignan Derivatives from the Root Wood of Hernandia nymphaeifolia
Abstract
:1. Introduction
2. Results and Discussion
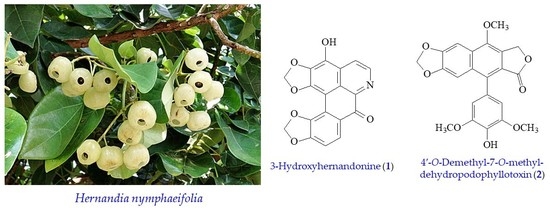
2.1. Isolation and Structural Elucidation
2.2. Structure Identification of the Known Isolates
2.3. Biological Studies
3. Experimental Section
3.1. General Procedures
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Biological Assay
3.4.1. Mensuration of Human Neutrophils
3.4.2. Mensuration of Superoxide Anion (O2•−) Generation
3.4.3. Mensuration of Elastase Release
3.4.4. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Yang, Y.P.; Lu, S.Y. Hernandiaceae. In Flora of Taiwan, 2nd ed.; Editorial Committee of the Flora of Taiwan: Taipei, Taiwan, 1996; Volume 2, pp. 500–503. ISBN 957-9019-52-5. [Google Scholar]
- Kan, W.S. Manual of Medicinal Plants in Taiwan; National Research Institute of Chinese Medicine: Taipei, Taiwan, 1970; pp. 178–179. [Google Scholar]
- Chen, J.J.; Tsai, I.L.; Ishikawa, T.; Wang, C.J.; Chen, I.S. Alkaloids from trunk bark of Hernandia nymphaeifolia. Phytochemistry 1996, 42, 1479–1484. [Google Scholar] [CrossRef]
- Chen, J.J.; Ishikawa, T.; Duh, C.Y.; Tsai, I.L.; Chen, I.S. New dimeric aporphine alkaloids and cytotoxic constituents of Hernandia nymphaeifolia. Planta Med. 1996, 62, 528–533. [Google Scholar] [CrossRef] [PubMed]
- Chen, I.S.; Chen, J.J.; Duh, C.Y.; Tsai, I.L.; Chang, C.T. New aporphine alkaloids and cytotoxic constituents of Hernandia nymphaeifolia. Planta Med. 1997, 63, 154–157. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Chang, Y.L.; Teng, C.M.; Chen, I.S. Vasorelaxing and antioxidant constituents from Hernandia nymphaeifolia. Planta Med. 2001, 67, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Chang, Y.L.; Teng, C.M.; Chen, I.S. Anti-platelet aggregation alkaloids and lignans from Hernandia nymphaeifolia. Planta Med. 2000, 66, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Chen, I.S.; Chen, J.J.; Duh, C.Y.; Tsai, I.L. New cytotoxic lignans from Formosan Hernandia nymphaeifolia. Phytochemistry 1997, 45, 991–996. [Google Scholar] [CrossRef]
- Ennis, M. Neutrophils in asthma pathophysiology. Curr. Allergy Asthma Rep. 2003, 3, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Borregaard, N. The human neutrophil. Function and dysfunction. Eur. J. Haematol. 1998, 41, 401–413. [Google Scholar] [CrossRef]
- Witko-Sarsat, V.; Rieu, P.; Descamps-Latscha, B.; Lesavre, P.; Halbwachs-Mecarelli, L. Neutrophils: Molecules, functions and pathophysiological aspects. Lab. Inverstig. 2000, 80, 617–653. [Google Scholar] [CrossRef]
- Roos, D.; van Bruggen, R.; Meischl, C. Oxidative killing of microbes by neutrophils. Microbes Infect. 2003, 5, 1307–1315. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, H.; Ueda, F.; Ito, M.; Ishii, H.; Hagniwa, J. Alkaloids of Hernandia ovigera. IV. Constituents of Hernandia ovigera collected in the Bonin Islands. Yakugaku Zasshi 1972, 92, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.H.; Liu, S.C.; Lin, T.S.; Yang, L.M. Studies on the constituents of the root-bark of Hernandia ovigera L. III. J. Chin. Chem. Soc. 1976, 23, 29–34. [Google Scholar] [CrossRef]
- Atta-ur-Rahman; Ashraf, M.; Choudhary, M.I.; Habib-ur-Rehman; Kazmi, M.H. Antifungal aryltetralin lignans from leaves of Podophyllum hexandrum. Phytochemistry 1995, 40, 427–431. [Google Scholar] [CrossRef]
- Orito, K.; Uchiito, S.; Satoh, Y.; Tatsuzawa, T.; Harada, R.; Tokuda, M. Aryl radical cyclizations of 1-(2′-bromobenzyl) isoquinolines with AIBN-Bu3SnH: Formation of aporphines and indolo[2,1-a]isoquino-lines. Org. Lett. 2000, 2, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Tsai, I.L.; Chen, I.S. New oxoaporphine alkaloids from Hernandia nymphaeifolia. J. Nat. Prod. 1996, 59, 156–158. [Google Scholar] [CrossRef]
- Kametani, T.; Nitadori, R.; Terasawa, H.; Takahashi, K.; Ihara, M.; Fukumoto, K. Studies on the syntheses of heterocyclic compounds–DCXCIII: A total synthesis of atheroline by photolysis. Tetrahedron 1977, 33, 1069–1071. [Google Scholar] [CrossRef]
- Yamaguchi, H.; Arimoto, M.; Yamamoto, K.; Numata, A. Studies on the constituents of the seeds of Hernandia ovigera L. Yakugaku Zasshi 1979, 99, 674–677. [Google Scholar] [CrossRef] [PubMed]
- Tanoguchi, M.; Arimoto, M.; Saika, H.; Yamaguchi, H. Studies on the constituents of the seeds of Hernandia ovigera L. VI. Isolation and structural determination of three lignans. Chem. Pharm. Bull. 1987, 35, 4162–4168. [Google Scholar] [CrossRef]
- Ito, C.; Matsui, T.; Wu, T.S.; Furukawa, H. Isolation of 6,7-demethyl-enedesoxypodophyllotoxin from Hernandia ovigera. Chem. Pharm. Bull. 1992, 40, 1318–1321. [Google Scholar] [CrossRef]
- Chen, C.Y.; Wang, Y.D.; Wang, H.M. Chemical constituents from the roots of Synsepalum dulcificum. Chem. Nat. Compd. 2010, 46, 46–448. [Google Scholar] [CrossRef]
- Chen, C.Y.; Chang, F.R.; Wu, Y.C. The constituents from the stems of Annona cherimola. J. Chin. Chem. Soc. 1997, 44, 313–319. [Google Scholar] [CrossRef]
- Sun, X.B.; Zhao, P.H.; Xu, Y.J.; Sun, L.M.; Cao, M.A.; Yuan, C.S. Chemical constituents from the roots of Polygonum bistorta. Chem. Nat. Compd. 2007, 43, 563–566. [Google Scholar] [CrossRef]
- Ayyad, S.N. A new cytotoxic stigmastane steroid from Pistia stratiotes. Pharmazie 2002, 57, 212–214. [Google Scholar] [PubMed]
- Chen, C.H.; Hwang, T.L.; Chen, L.C.; Chang, T.H.; Wei, C.S.; Chen, J.J. Isoflavones and anti-inflammatory constituents from the fruits of Psoralea corylifolia. Phytochemistry 2017, 143, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Boyum, A. Isolation of mononuclear cells and granulocytes from human blood. Isolation of monuclear cells by one centrifugation, and of granulocytes by combining centrifugation and sedimentation at 1 g. Scand. J. Clin. Lab. Inverstig. 1968, 97, 77–89. [Google Scholar]
- Jauregui, H.O.; Hayner, N.T.; Driscoll, J.L.; Williams-Holland, R.; Lipsky, M.H.; Galletti, P.M. Trypan blue dye uptake and lactate dehydrogenase in adult rat hepatocytes-freshly isolated cells, cell suspensions, and primary monolayer cultures. In Vitro 1981, 17, 1100–1110. [Google Scholar] [CrossRef] [PubMed]
- Babior, B.M.; Kipnes, R.S.; Curnutte, J.T. Biological defense mechanisms. The production by leukocytes of superoxide, a potential bactericidal agent. J. Clin. Inverstig. 1973, 52, 741–744. [Google Scholar] [CrossRef] [PubMed]
- Hwang, T.L.; Leu, Y.L.; Kao, S.H.; Tang, M.C.; Chang, H.L. Viscolin, a new chalcone from Viscum coloratum, inhibits human neutrophil superoxide anion and elastase release via a cAMP-dependent pathway. Free Radic. Biol. Med. 2006, 41, 1433–1441. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.J.; Ting, C.W.; Wu, Y.C.; Hwang, T.L.; Cheng, M.J.; Sung, P.J.; Wang, T.C.; Chen, J.F. New labdane-type diterpenoids and anti-inflammatory constituents from Hedychium coronarium. Int. J. Mol. Sci. 2013, 14, 13063–13077. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |


 ) and HMBC (
) and HMBC (  ) correlations of 1.
) correlations of 1.
 ) and HMBC (
) and HMBC (  ) correlations of 2.
) correlations of 2.
| Compounds | Superoxide anion | Elastase |
|---|---|---|
| IC50 [µg/mL] b or (Inh %) c | ||
| 3-Hydroxyhernandonine (1) | 4.09 ± 0.44 *** | 3.93 ± 0.48 *** |
| 4′-O-Demethyl-7-O- methyldehydro-podophyllotoxin (2) | 5.72 ± 0.42 *** | 5.40 ± 0.40 *** |
| 2-O-Methyl-7-oxolaetine (3) | 7.37 ± 0.46 *** | 6.82 ± 0.09 *** |
| Hernandonine (4) | 4.41 ± 0.76 *** | (45.76 ± 6.92) *** |
| Oxohernangerine (5) | 2.65 ± 0.97 *** | 4.82 ± 0.39 *** |
| Oxohernagine (6) | 2.86 ± 0.85 *** | 4.87 ± 0.27 *** |
| 7-Oxonorisocorydine (7) | 6.62 ± 0.28 *** | 6.58 ± 0.08 *** |
| (–)-Deoxypodophyllotoxin (8) | (38.95 ± 4.83) ** | (33.76 ± 3.82) |
| Dehydropodophyllotoxin (9) | (43.91 ± 3.86) *** | 9.53 ± 0.84 *** |
| (–)-Yatein (10) | (42.36 ± 3.41) * | (36.74 ± 3.05) ** |
| N-trans-Feruloylmethoxytyramine (11) | 6.26 ± 0.65 *** | 7.03 ± 0.21 *** |
| Mixture of β-sitostenone (12) and stigmasta-4,22-dien-3-one (13) | (24.71 ± 2.67) | (29.15 ± 2.89) |
| Mixture of 6β-hydroxystigmast-4-en-3-one (14) and 6β-hydroxystigmasta-4,22-dien-3-one (15) | (16.74 ± 2.66) ** | 7.91 ± 1.20 ** |
| Diphenyleneiodonium d | 0.55 ± 0.22 *** | – |
| Phenylmethylsulfonyl fluoride d | – | 34.5 ± 5.3 *** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, C.-Y.; Wang, S.-W.; Ye, J.-W.; Hwang, T.-L.; Cheng, M.-J.; Sung, P.-J.; Chang, T.-H.; Chen, J.-J. New Anti-Inflammatory Aporphine and Lignan Derivatives from the Root Wood of Hernandia nymphaeifolia. Molecules 2018, 23, 2286. https://doi.org/10.3390/molecules23092286
Wei C-Y, Wang S-W, Ye J-W, Hwang T-L, Cheng M-J, Sung P-J, Chang T-H, Chen J-J. New Anti-Inflammatory Aporphine and Lignan Derivatives from the Root Wood of Hernandia nymphaeifolia. Molecules. 2018; 23(9):2286. https://doi.org/10.3390/molecules23092286
Chicago/Turabian StyleWei, Chuan-Yen, Shih-Wei Wang, Jin-Wang Ye, Tsong-Long Hwang, Ming-Jen Cheng, Ping-Jyun Sung, Tsung-Hsien Chang, and Jih-Jung Chen. 2018. "New Anti-Inflammatory Aporphine and Lignan Derivatives from the Root Wood of Hernandia nymphaeifolia" Molecules 23, no. 9: 2286. https://doi.org/10.3390/molecules23092286