Preparation of Progesterone Co-Crystals Based on Crystal Engineering Strategies
Abstract
:1. Introduction
2. Results and Discussion
2.1. Preparation of Co-Crystals
2.2. Crystal Structure Analysis
2.2.1. Structure of the PROG-CNA Co-Crystal
2.2.2. Structure of the PROG-DHB Co-Crystal
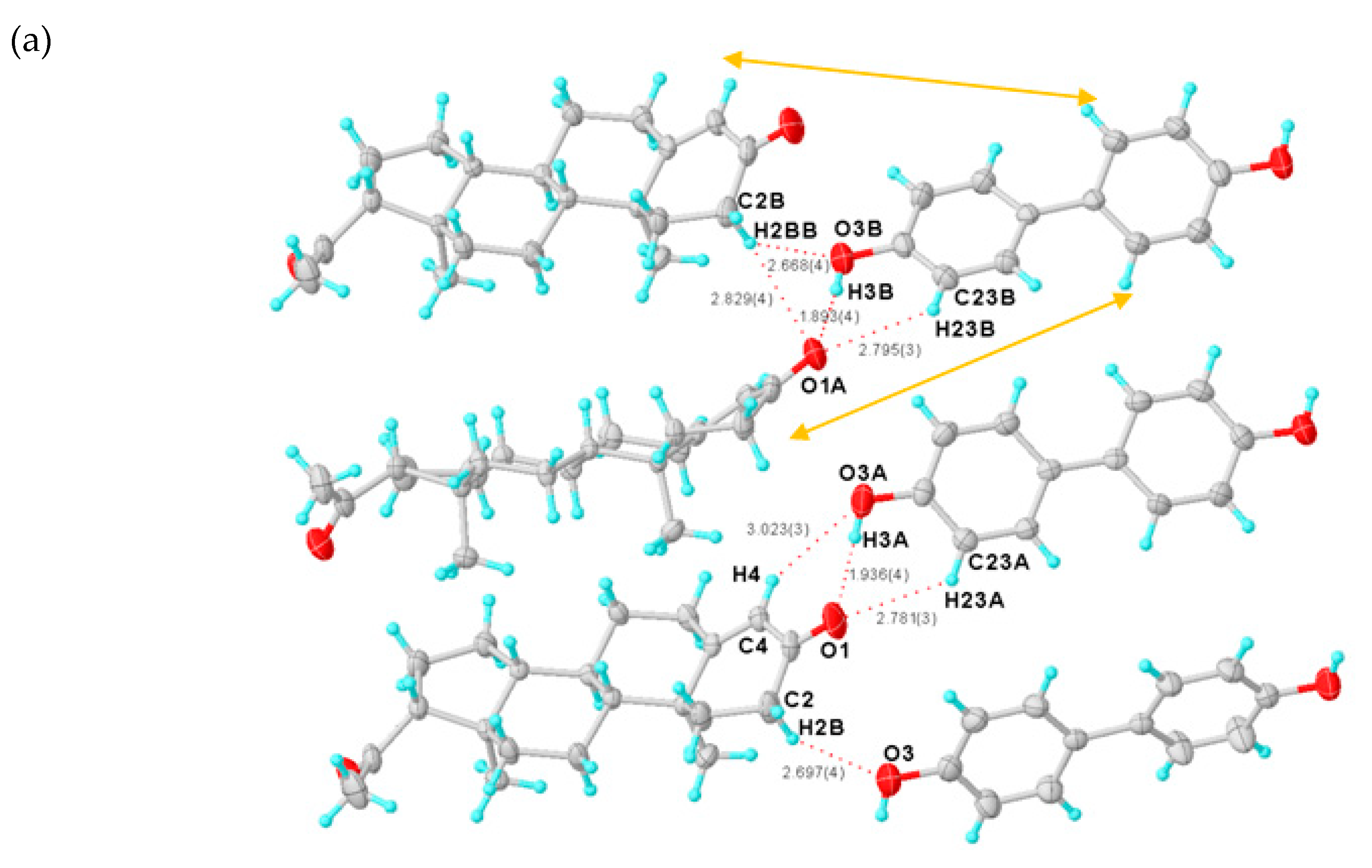
2.2.3. Structure of the PROG-DOD Co-Crystal
2.3. IR Spectroscopy
2.4. DSC and PXRD Analysis
2.5. In Vitro Dissolution
3. Materials and Methods
3.1. Materials and Reagents
3.2. Synthesis and Crystallization
3.3. Differential Scanning Calorimetry
3.4. IR Spectroscopy
3.5. Powder X-Ray Diffraction
3.6. In Vitro Dissolution
3.7. X-Ray Crystallography
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Remenar, J.F.; Morissette, S.L.; Peterson, M.L.; Moulton, B.; Macphee, J.M.; Guzmán, H.R.; Almarsson, O. Crystal engineering of novel cocrystals of a triazole drug with 1,4-dicarboxylic acids. J. Am. Chem. Soc. 2003, 125, 8456–8457. [Google Scholar] [CrossRef] [PubMed]
- Bhogala, B.R.; Basavoju, S.; Nangia, A. Tape and layer structures in cocrystals of some di- and tricarboxylic acids with 4,4′-bipyridines and isonicotinamide from binary to ternary cocrystals. Cryst. Eng. Comm. 2005, 7, 551. [Google Scholar] [CrossRef]
- Grobelny, P.; Desiraju, G.R.; Mukherjee, A. Drug-drug co-crystals: Temperature-dependent proton mobility in the molecular complex of isoniazid with 4-aminosalicylic acid. Cryst. Eng. Comm. 2011, 13, 4358–4364. [Google Scholar] [CrossRef]
- Cheney, M.L.; Weyna, D.R.; Shan, N.; Hanna, M.; Wojtas, L.; Zaworotko, M.J. Coformer selection in pharmaceutical cocrystal development: A case study of a meloxicam aspirin cocrystal that exhibits enhanced solubility and pharmacokinetics. J. Pharm. Sci. 2011, 100, 2172–2181. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.J.; Wang, Y.; Yang, F.X.; Zhang, X.T.; Hu, W.P. Cocrystal Engineering: A Collaborative Strategy toward Functional Materials. Adv. Mater. 2019, 31, 1902328. [Google Scholar] [CrossRef] [PubMed]
- Linda, L.; Stefan, H.; Gabriele, S. Predicting the solubility of pharmaceutical cocrystals in solvent/anti-solvent mixtures. Molecules 2016, 21, 593–610. [Google Scholar]
- Van Beek, E.R.; Lowik, C.W.; Ebetino, F.H.; Papapoulos, S.E. Binding and antiresorptive properties of heterocycle-containing bisphosphonate analogs: Structure-activity relationships. Bone 1998, 23, 437–442. [Google Scholar] [CrossRef]
- Samipillai, M.; Rohani, S. The role of higher coformer stoichiometry ratio in pharmaceutical cocrystals for improving their solid-state properties: The cocrystals of progesterone and 4-hydroxybenzoic acid. J. Cryst. Growth. 2019, 507, 270−282. [Google Scholar] [CrossRef]
- Takata, N.; Shiraki, K.; Takano, R.; Hayashi, Y.; Terada, K. Cocrystal screening of stanolone and mestanolone using slurry crystallization. Cryst. Growth Des. 2008, 8, 3032–3037. [Google Scholar] [CrossRef]
- Friščić, T.; Lancaster, R.W.; Fábián, L.; Atwood, K.J.L. Tunable recognition of the steroid α-face by adjacent π-electron density. Proc. Natl. Acad. Sci. USA 2010, 107, 13216–13221. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, R.W.; Karamertzanis, P.G.; Hulme, A.T.; Tocher, D.A.; Lewis, T.C.; Price, S.L. The polymorphism of progesterone: Stabilization of a ‘disappearing’ polymorph by co-crystallization. J. Pharm. Sci. 2010, 96, 3419–3431. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Ragab, D.; Rohani, S. Polymorphism of progesterone: A new approach for the formation of form ii and the relative stabilities of form i and form ii. Cryst. Growth Des. 2014, 14, 4574–4582. [Google Scholar] [CrossRef]
- Stahly, G.P. Diversity in Single- and Multiple-Component Crystals. The Search for and Prevalence of Polymorphs and Cocrystals. Cryst. Growth Des. 7 2007, 6, 1007–1026. [Google Scholar] [CrossRef]
- Patrycja, G.; Marek, W. DSC, FTIR and raman spectroscopy coupled with multivariate analysis in a study of co-crystals of pharmaceutical interest. Molecules 2018, 23, 2136–2153. [Google Scholar]
- Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer17; University of Western Australia: Crawley, WA, Australia, 2017. [Google Scholar]
- Liao, X.; Gautam, M.; Grill, A.; Zhu, H.J. Effect of position isomerism on the formation and physicochemical properties of pharmaceutical co-crystals. J. Pharm. Sci. 2010, 99, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Stilinović, V.; Kaitner, B. Salts and co-crystals of gentisic acid with pyridine derivatives: The effect of proton transfer on the crystal packing (and vice versa). Cryst. Growth Des. 2012, 12, 5763–5772. [Google Scholar] [CrossRef]
- Weyna, D.R.; Shattock, T.; Vishweshwar, P.; Zaworotko, M.J. Synthesis and structural characterization of cocrystals and pharmaceutical cocrystals: Mechanochemistry vs slow evaporation from solution. Cryst. Growth Des. 2009, 9, 1106–1123. [Google Scholar] [CrossRef]
- Bruker. SMART, SAINT and SADABS. Bruker AXS Inc.: Madison, Wisconsin, USA, 2004. Available online: https://www.bruker.com/products/x-ray-diffraction-and-elemental-analysis/single-crystal-x-ray-diffraction.html (accessed on 31 October 2019).
- Sheldrick, G.M. Shelxt-integrated space-group and crystal-structure determination. Acta. Cryst. 2015, A71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. Olex2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds of PROG-CNA, PROG-DHB, PROG-DOD are available from the authors. |













| Co-Crystal | X-H···Cg | H···Cg (Å) | X-H···Cg (°) | X···Cg (Å) |
|---|---|---|---|---|
| PROG-CNA | C12-H12A···Cg1 | 2.8276 (8) | 178.9 | 3.797 |
| PROG-DHB | C14-H14···Cg1 a | 2.8527 | 171.21 | 3.824 |
| C21-C21C···Cg1 b | 3.0350 | 166.4 | 3.975 |
| Co-Crystal | Donor-H···Acceptor | D-H (Å) | H···A (Å) | D···A (Å) | D-H···A (°) |
|---|---|---|---|---|---|
| PROG-CNA | C1-H1B···O4 | 0.970 | 2.964 | 3.767 | 143.0 |
| C6-H6B···O1 a | 0.970 | 2.901 | 3.758 | 147.92 | |
| C7-H7B···O3 b | 0.970 | 2.909 | 3.669 | 136.0 | |
| C7-H7B···O4 b | 0.970 | 3.088 | 3.794 | 130.8 | |
| C8-H8···O2 c | 0.980 | 2.799 | 3.769 | 170.30 | |
| C14-H14···O3 b | 0.980 | 2.917 | 3.613 | 128.9 | |
| C15-H15A···O3 b | 0.970 | 2.845 | 3.554 | 130.6 | |
| C18-H18B···N1 c | 0.960 | 3.130 | 3.777 | 126.2 | |
| C19-H19H···O2 c | 0.960 | 2.621 | 3.554 | 164.3 | |
| C26-H26···O4 d | 0.930 | 2.442 | 3.262 | 147.0 | |
| C27-H27···O3 b | 0.930 | 2.516 | 3.353 | 149.8 | |
| PROG-DHB | C6-H6B···O4 d | 0.970 | 3.199 | 3.789 | 120.91 |
| C11-H11A···O3 e | 0.970 | 2.629 | 3.459 | 143.7 | |
| C19-H19C···O5 f | 0.960 | 3.008 | 3.770 | 137.3 | |
| C21-H21A···O4 | 0.960 | 2.724 | 3.643 | 160.4 | |
| C25-H25···O6 g | 0.930 | 2.488 | 3.281 | 143.3 | |
| PROG-DOD | C2-H2B···O3 | 0.970 | 2.697 | 3.592 | 153.6 |
| C4-H4···O3A | 0.930 | 3.023 | 3.634 | 124.7 | |
| C6-H6A···O1A h | 0.970 | 2.806 | 3.620 | 142.0 | |
| C6-H6B···O3A h | 0.970 | 2.903 | 3.661 | 135.7 | |
| C21-H21A···O4B i | 0.960 | 2.880 | 3.581 | 130.7 | |
| C21-H21B···O2B j | 0.960 | 2.712 | 3.459 | 135.1 | |
| C21-H21B···O4 k | 0.960 | 2.978 | 3.565 | 120.7 | |
| C6A-H6AB···O1 h | 0.970 | 2.674 | 3.542 | 149.2 | |
| C21A-H21D···O4A k | 0.960 | 2.990 | 3.630 | 125.2 | |
| C2B-H2BB···O1A | 0.970 | 2.829 | 3.703 | 150.5 | |
| C2B-H2BB···O3B | 0.970 | 2.668 | 3.498 | 143.7 | |
| C16B-H16E···O4B k | 0.970 | 2.820 | 3.454 | 123.7 | |
| C21B-H21G···O4A k | 0.960 | 2.925 | 3.585 | 126.9 | |
| C21B-H21H···O2 j | 0.960 | 2.989 | 3.602 | 122.8 | |
| C32-H32···O2A l | 0.930 | 2.831 | 3.477 | 127.6 | |
| C23A-H23A···O1 | 0.930 | 2.781 | 3.438 | 128.5 | |
| C30A-H30A···O2A l | 0.930 | 2.815 | 3.692 | 157.5 | |
| C32A-H32A···O2B l | 0.930 | 2.962 | 3.577 | 125.0 | |
| C23B-H23B···O1A | 0.930 | 2.795 | 3.444 | 127.8 | |
| C32B-H32B···O2 m | 0.930 | 3.170 | 3.751 | 122.4 |
| Co-Crystal | Donor-H···Acceptor | D-H (Å) | H···A (Å) | D···A (Å) | D−H···A (°) |
|---|---|---|---|---|---|
| PROG-CNA | N1-H1C···O1 a | 0.869 (3) | 2.109 | 2.952 | 163.6 |
| N1-H1D···O2 b | 0.868 | 2.401 | 3.031 | 129.8 | |
| PROG-DHB | O3-H3···O4 | 0.820 | 1.893 | 2.612 | 145.76 |
| O5-H5···O2 | 0.820 | 1.833 | 2.620 | 160.33 | |
| O6-H6···O1 a | 0.820 | 1.972 | 2.774 | 165.9 | |
| PROG-DOD | O3-H3···O1B c | 0.820 | 1.862 | 2.649 | 160.3 |
| O4-H4C···O2A d | 0.820 | 1.972 | 2.786 | 171.9 | |
| O3A-H3A···O1 | 0.820 | 1.936 | 2.747 | 169.9 | |
| O4A-H4AA···O2B d | 0.820 | 1.951 | 2.758 | 168.2 | |
| O3B-H3B···O1A | 0.820 | 1.893 | 2.706 | 171.2 | |
| O4B-H4BA···O2 e | 0.820 | 1.996 | 2.782 | 160.5 |
| PROG·CNA | PROG·DHB | PROG·DOD | |
|---|---|---|---|
| Crystal data | |||
| Chemical formula | C21H30O2·C6H5ClN2O2 | C21H30O2·C7H6O4 | C21H30O2·C12H10O2 |
| Mr | 487.02 | 468.57 | 500.65 |
| Crystal system, space group | Orthorhombic, P212121 | Orthorhombic, P212121 | Monoclinic, P21 |
| Temperature (K) | 296 | 296 | 296 |
| a, b, c (Å) | 7.721, 11.783, 27.762 | 7.328, 14.173, 23.417 | 17.4955, 10.4030, 22.825 |
| α, β, γ (°) | 90, 90, 90 | 90, 90, 90 | 90, 97.252, 90 |
| V (Å3) | 2525.8 | 2432.1 | 4121.1 |
| Z | 4 | 4 | 6 |
| Dx (Mg·m−3) | 1.281 | 1.280 | 1.210 |
| Radiation type | Mo Kα | Mo Kα | Mo Kα |
| μ (mm−1) | 0.187 | 0.089 | 0.078 |
| Crystal size (mm) | 0.05 × 0.04 × 0.03 | 0.06 × 0.05 × 0.04 | 0.07 × 0.06 × 0.05 |
| Data collection | |||
| Diffractometer | CCD area detector | ||
| Absorption correction | Multi-scan (SADABS; Bruker, 2004) | ||
| Tmin, Tmax | 0.991, 0.994 | 0.995, 0.996 | 0.995, 0.996 |
| No. of measured, independent, and observed [I > 2σ(I)] reflections | 11115, 3736, 3019 | 17382, 5635, 3715 | 80611, 18695, 11453 |
| Rint | 0.0385 | 0.0582 | 0.0797 |
| (sin θ/λ)max (Å−1) | 0.562 | 0.652 | 0.647 |
| Refinement | |||
| R[F2 > 2σ(F2)], wR(F2), S(GooF) | 0.0375, 0.0910, 1.029 | 0.0502, 0.1159, 1.007 | 0.0616, 0.1691, 1.008 |
| No. of reflections | 3736 | 5635 | 18695 |
| No. of parameters | 311 | 313 | 1015 |
| No. of restraints | 0 | 0 | 1 |
| H-atom treatment | H atoms treated by a mixture of independent and constrained refinement | ||
| Δρmax, Δρmin (e Å−3) | 0.117, −0.139 | 0.161, −0.182 | 0.176, −0.254 |
| Absolute structure | Flack x determined using 1054 quotients [(I+) − (I−)]/[(I+)+(I−)] | Flack x determined using 1184 quotients [(I+) − (I−)]/[(I+) + (I−)] | Flack x determined using 4022 quotients [(I+) − (I−)]/[(I+) + (I−)] |
| Absolute structure parameter | −0.08 | −0.2 | 0.4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeng, H.; Xiong, J.; Zhao, Z.; Qiao, J.; Xu, D.; Miao, M.; He, L.; Wu, X. Preparation of Progesterone Co-Crystals Based on Crystal Engineering Strategies. Molecules 2019, 24, 3936. https://doi.org/10.3390/molecules24213936
Zeng H, Xiong J, Zhao Z, Qiao J, Xu D, Miao M, He L, Wu X. Preparation of Progesterone Co-Crystals Based on Crystal Engineering Strategies. Molecules. 2019; 24(21):3936. https://doi.org/10.3390/molecules24213936
Chicago/Turabian StyleZeng, Huahui, Jing Xiong, Zhuang Zhao, Jingyi Qiao, Duanjie Xu, Mingsan Miao, Lan He, and Xiangxiang Wu. 2019. "Preparation of Progesterone Co-Crystals Based on Crystal Engineering Strategies" Molecules 24, no. 21: 3936. https://doi.org/10.3390/molecules24213936





