Discovery of Novel Triazole-Containing Pyrazole Ester Derivatives as Potential Antibacterial Agents
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
2.2. Antibacterial Activity and Structure-Activity Relationships (SAR) Discussion
2.3. Inhibitory Effects against DNA Gyrase and Topoisomerase IV
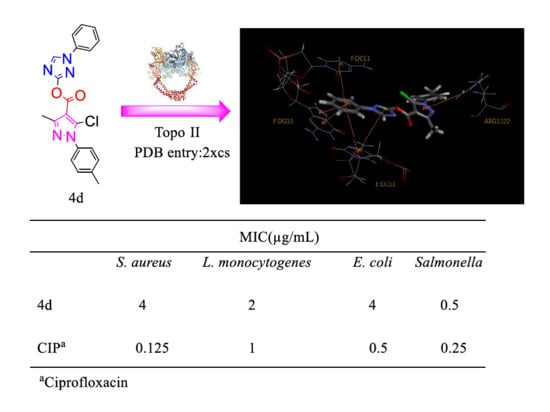
2.4. Docking Analysis
3. Experimental Section
3.1. Materials and Methods
3.1.1. General Procedure for the Synthesis of 5-Chloro-1-aryl-3-methyl-1H-pyrazole-4-carboxylic Acids 2a–d
3.1.2. General Pathway for Prepare of 1-phenyl-1H-1,2,4-triazol-3-ol 3a–d
3.1.3. General Procedure for Synthesis of 1-phenyl-1H-1,2,4-triazol-3-yl 5-chloro-3-methyl-1-phenyl-1H-pyrazole-4-carboxylate 4a–m
3.1.4. 1-phenyl-1H-1,2,4-triazol-3-yl5-chloro-3-methyl-1-phenyl-1H-pyrazole-4-carboxylate (4a)
3.1.5. 1-phenyl-1H-1,2,4-triazol-3-yl5-chloro-1-(4-fluorophenyl)-3-methyl-1H-pyrazole-4-carboxylate (4b)
3.1.6. 1-phenyl-1H-1,2,4-triazol-3-yl5-chloro-1-(4-chlorophenyl)-3-methyl-1H-pyrazole-4-carboxylate (4c)
3.1.7. 1-phenyl-1H-1,2,4-triazol-3-yl5-chloro-3-methyl-1-(p-tolyl)-1H-pyrazole-4-carboxylate (4d)
3.1.8. 1-(4-fluorophenyl)-1H-1,2,4-triazol-3-yl5-chloro-3-methyl-1-phenyl-1H-pyrazole-4-carboxylate (4e)
3.1.9. 1-(4-fluorophenyl)-1H-1,2,4-triazol-3-yl5-chloro-1-(4-fluorophenyl)-3-methyl-1H-pyrazole-4-carboxylate (4f)
3.1.10. 1-(4-fluorophenyl)-1H-1,2,4-triazol-3-yl5-chloro-1-(4-chlorophenyl)-3-methyl-1H-pyrazole-4-carboxylate (4g)
3.1.11. 1-(4-fluorophenyl)-1H-1,2,4-triazol-3-yl5-chloro-3-methyl-1-(p-tolyl)-1H-pyrazole-4-carboxylate (4h)
3.1.12. 1-(4-chlorophenyl)-1H-1,2,4-triazol-3-yl5-chloro-1-(4-fluorophenyl)-3-methyl-1H-pyrazole-4-carboxylate (4i)
3.1.13. 1-(4-chlorophenyl)-1H-1,2,4-triazol-3-yl5-chloro-1-(4-chlorophenyl)-3-methyl-1H-pyrazole-4-carboxylate (4j)
3.1.14. 1-(p-tolyl)-1H-1,2,4-triazol-3-yl-5-chloro-3-methyl-1-phenyl-1H-pyrazole-4-carboxylate (4k)
3.1.15. 1-(p-tolyl)-1H-1,2,4-triazol-3-yl5-chloro-1-(4-fluorophenyl)-3-methyl-1H-pyrazole-4-carboxylate (4l)
3.1.16. 1-(p-tolyl)-1H-1,2,4-triazol-3-yl5-chloro-1-(4-chlorophenyl)-3-methyl-1H-pyrazole-4-carboxylate (4m)
3.2. In Vitro Antibacterial Activity
3.2.1. Medium
3.2.2. Minimum Inhibitory Concentration (MIC)
3.3. Enzyme Inhibition Experimental
3.4. Molecular Docking
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Naeem, A.; Badshah, S.L.; Muska, M.; Ahmad, N.; Khan, K. The Current Case of Quinolones: Synthetic Approaches and Antibacterial Activity. Molecules 2016, 21, 268. [Google Scholar] [CrossRef]
- Esteves-Souza, A.; Rodrigues-Santos, C.E.; Del Cistia Cde, N.; Silva, D.R.; Sant’anna, C.M.; Echevarria, A. Solvent-free synthesis, DNA-topoisomerase II activity and molecular docking study of new asymmetrically N,N’-substituted ureas. Molecules 2012, 17, 12882–12894. [Google Scholar] [CrossRef] [PubMed]
- Neumann, W.; Sassone-Corsi, M.; Raffatellu, M.; Nolan, E.M. Esterase-Catalyzed Siderophore Hydrolysis Activates an Enterobactin-Ciprofloxacin Conjugate and Confers Targeted Antibacterial Activity. J. Am. Chem. Soc. 2018, 140, 5193–5201. [Google Scholar] [CrossRef] [PubMed]
- Gellerman, G. Recent Developments in the Synthesis and Applications of Anticancer Amonafide Derivatives. A Mini Review. Lett. Drug Des. Discov. 2016, 13, 47–63. [Google Scholar] [CrossRef]
- Mcgarry, D.H.; Cooper, I.R.; Walker, R.; Warrilow, C.E.; Pichowicz, M.; Ratcliffe, A.J.; Salisbury, A.M.; Savage, V.J.; Moyo, E.; Maclean, J. Design, synthesis and antibacterial properties of pyrimido[4,5-b]indol-8-amine inhibitors of DNA gyrase. Bioorg. Med. Chem. Lett. 2018, 28, 2998–3003. [Google Scholar] [CrossRef]
- Kamal, A.; Satyanarayanaa, M.; Devaiaha, V.V.; Yadava, J.S.; Mullickb, B.; Nagarajab, V. Synthesis and biological evaluation of coumarin linked fluoroquinolones, phthalimides and naphthalimides as potential DNA gyrase inhibitors. Lett. Drug Des. Discov. 2006, 3, 494–502. [Google Scholar] [CrossRef]
- Xie, Z.; Cheng, D.; Luo, L.; Shen, G.; Pan, S.; Pan, Y.; Chen, B.; Wang, X.; Liu, Z.; Zhang, Y.; et al. Design, synthesis and biological evaluation of 4-bromo-N-(3,5-dimethoxyphenyl)benzamide derivatives as novel FGFR1 inhibitors for treatment of non-small cell lung cancer. J. Enzym. Inhib. Med. Chem. 2018, 33, 905–919. [Google Scholar] [CrossRef]
- Liu, H.; Ren, Z.L.; Wang, W.; Gong, J.X.; Chu, M.J.; Ma, Q.W.; Wang, J.C.; Lv, X.H. Novel coumarin-pyrazole carboxamide derivatives as potential topoisomerase II inhibitors: Design, synthesis and antibacterial activity. Eur. J. Med. Chem. 2018, 157, 81–87. [Google Scholar] [CrossRef]
- Nastasa, C.; Vodnar, D.C.; Ionut, I.; Stana, A.; Benedec, D.; Tamaian, R.; Oniga, O.; Tiperciuc, B. Antibacterial Evaluation and Virtual Screening of New Thiazolyl-Triazole Schiff Bases as Potential DNA-Gyrase Inhibitors. Int. J. Mol. Sci. 2018, 19, 222. [Google Scholar] [CrossRef]
- Calver, A.D.; Falmer, A.A.; Murray, M.; Strauss, O.J.; Streicher, E.M.; Hanekom, M.; Liversage, T.; Masibi, M.; van Helden, P.D.; Warren, R.M. Emergence of increased resistance and extensively drug-resistant tuberculosis despite treatment adherence, South Africa. Emerg. Infect. Dis. 2010, 16, 264–271. [Google Scholar] [CrossRef]
- Singh, H.; Nand, B.; Sindhu, J.; Khurana, J.M.; Sharma, C.; Aneja, K.R. Efficient One Pot Synthesis of Xanthene-Triazole-Quinoline/Phenyl Conjugates and Evaluation of their Antimicrobial Activity. J. Braz. Chem. Soc. 2014, 7, 1178–1193. [Google Scholar] [CrossRef]
- Akhtar, M.J.; Yar, M.S.; Khan, A.A.; Ali, Z.; Haider, M.R. Recent advances in the synthesis and anticancer activity of molecules other than nitrogen containing heterocyclic moeities. Mini-Rev. Med. Chem. 2016, 17, 1602–1632. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Chen, W.; Jia, Y.; Tian, Y.; Zhao, Y.; Long, F.; Rui, Y.; Jiang, X. N-Heterocyclic molecule-capped gold nanoparticles as effective antibiotics against multi-drug resistant bacteria. Nanoscale 2016, 8, 13223–13227. [Google Scholar] [CrossRef] [PubMed]
- Fauser, B.C.; Mannaerts, B.M.; Devroey, P.; Leader, A.; Boime, I.; Baird, D.T. Advances in recombinant DNA technology: Corifollitropin alfa, a hybrid molecule with sustained follicle-stimulating activity and reduced injection frequency. Hum. Reprod. Update 2009, 15, 309–321. [Google Scholar] [CrossRef]
- Tanitame, A.; Oyamada, Y.; Ofuji, K.; Fujimoto, M.; Suzuki, K.; Ueda, T.; Terauchi, H.; Kawasaki, M.; Nagai, K.; Wachi, M.; et al. Synthesis and antibacterial activity of novel and potent DNA gyrase inhibitors with azole ring. Bioorg. Med. Chem. 2004, 12, 5515–5524. [Google Scholar] [CrossRef]
- Azimvand, J. Synthesis of new triazole and oxadiazole containing compound in the azide reaction as an antibacterial drug. Nature 2012, 410, 231–235. [Google Scholar]
- Efthimiadou, E.K.; Thomadaki, H.; Sanakis, Y.; Raptopoulou, C.P.; Katsaros, N.; Scorilas, A.; Karaliota, A.; Psomas, G. Structure and biological properties of the copper(II) complex with the quinolone antibacterial drug N-propyl-norfloxacin and 2,2′-bipyridine. J. Inorg. Biochem. 2007, 101, 64–73. [Google Scholar] [CrossRef]
- El Shehry, M.F.; Ghorab, M.M.; Abbas, S.Y.; Fayed, E.A.; Shedid, S.A.; Ammar, Y.A. Quinoline derivatives bearing pyrazole moiety: Synthesis and biological evaluation as possible antibacterial and antifungal agents. Eur. J. Med. Chem. 2017, 143, 1463–1473. [Google Scholar] [CrossRef]
- Lv, X.H.; Ren, Z.L.; Liu, H.; Li, H.D.; Li, Q.S.; Wang, L.; Zhang, L.S.; Yao, X.K.; Cao, H.Q. Design, Synthesis and Biological Evaluation of Novel Pyrazole Sulfonamide Derivatives as Potential AHAS Inhibitors. Chem. Pharm. Bull. 2018, 66, 358–362. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.H.; Zhao, W.; Shen, Z.H.; Xing, J.H.; Xu, T.M.; Peng, W.L. Synthesis, nematocidal activity and SAR study of novel difluoromethylpyrazole carboxamide derivatives containing flexible alkyl chain moieties. Eur. J. Med. Chem. 2017, 125, 881–889. [Google Scholar] [CrossRef] [PubMed]
- Plech, T.; Kaproń, B.; Paneth, A.; Kosikowska, U.; Malm, A.; Strzelczyk, A.; Stączek, P.; Świątek, Ś.; Rajtar, B.; Polz-Dacewicz, M. Determination of the primary molecular target of 1,2,4-triazole-ciprofloxacin hybrids. Molecules 2015, 20, 6254–6272. [Google Scholar] [CrossRef]
- Lal, K.; Yadav, P.; Kumar, A.; Kumar, A.; Paul, A.K. Design, synthesis, characterization, antimicrobial evaluation and molecular modeling studies of some dehydroacetic acid-chalcone-1,2,3-triazole hybrids. Bioorg. Chem. 2018, 236–244. [Google Scholar] [CrossRef]
- Broussard, G.; Bramanti, O.; Marchese, F.M. [Environmental study on toxicity of water resulting from pesticide and biocide pollution]. Giornale Italiano Di Medicina Del Lavoro Ed Ergonomia 2003, 25, 149. [Google Scholar] [PubMed]
- Ory, J.; Bricheux, G.; Togola, A.; Bonnet, J.L.; Donnadieu-Bernard, F.; Nakusi, L.; Forestier, C.; Traore, O. Ciprofloxacin residue and antibiotic-resistant biofilm bacteria in hospital effluent. Environ. Pollut. 2016, 214, 635–645. [Google Scholar] [CrossRef]
- Haldar, J.; Kondaiah, P.; Bhattacharya, S. Synthesis and antibacterial properties of novel hydrolyzable cationic amphiphiles. Incorporation of multiple head groups leads to impressive antibacterial activity. J. Med. Chem. 2005, 48, 3823–3831. [Google Scholar] [CrossRef]
- Gomez, L.; Hack, M.D.; Wu, J.; Wiener, J.J.; Venkatesan, H.; Santillan, A., Jr.; Pippel, D.J.; Mani, N.; Morrow, B.J.; Motley, S.T.; et al. Novel pyrazole derivatives as potent inhibitors of type II topoisomerases. Part 1: Synthesis and preliminary SAR analysis. Bioorg. Med. Chem. Lett. 2007, 17, 2723–2727. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.H.; Cao, H.Q.; Ren, Z.L.; Chu, M.J. From Faming Zhuanli Shenqing. Patent CN 107141283A, 8 September 2017. [Google Scholar]
- Kim, D.K.; Lee, N.; Kim, Y.W.; Chang, K.; Im, G.J.; Choi, W.S.; Kim, K.H. Synthesis and evaluation of amino acid esters of 6-deoxypenciclovir as potential prodrugs of penciclovir. Bioorg. Med. Chem. 1999, 7, 419–424. [Google Scholar] [CrossRef]
- Wu, G.; Robertson, D.H.; Brooks, R.C.; Vieth, M. Detailed analysis of grid-based molecular docking: A case study of CDOCKER-A CHARMm-based MD docking algorithm. J. Comput. Chem. 2003, 24, 1549–1562. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.J.; Liao, M.; Chu, M.J.; Ren, Z.L.; Zhang, X.; Lv, X.H.; Cao, H.Q. Design, synthesis and anti-tobacco mosaic virus (TMV) activity of 5-chloro-N-(4-cyano-1-aryl-1H-pyrazol-5-yl)-1-aryl-3-methyl-1H-pyrazole-4-carboxamide derivatives. Molecules 2015, 20, 807–821. [Google Scholar] [CrossRef]
- Sun, B.; Liu, K.; Han, J.; Zhao, L.Y.; Su, X.; Lin, B.; Zhao, D.M.; Cheng, M.S. Design, synthesis, and biological evaluation of amide imidazole derivatives as novel metabolic enzyme CYP26A1 inhibitors. Bioorg. Med. Chem. 2015, 23, 6763–6773. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, P.F.; Murray, P.R.; Baron, E.J.; Pfaller, M.A.; Tenover, F.C.; Yolken, R.H. (Eds.) Manual of Clinical Microbiology, 7th ed.; American Society of Microbiology: Washington, DC, USA, 1999; Volume 146, pp. 107–108. [Google Scholar]
- Jorgensen, J.H. NCCLS Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, Approved Standard. Infect. Dis. Clin. N. Am. 1993, 7, 393–409. [Google Scholar]
- Aragade, P.; Maddi, V.; Khode, S.; Palkar, M.; Ronad, P.; Mamledesai, S.; Satyanarayana, D. Synthesis and Antibacterial Activity of a New Series of 3-3-(Substituted Phenyl)-1-Isonicotinoyl-1H-Pyrazol-5-yl-2H-Chromen-2-one Derivatives. Arch. Der Pharm. 2009, 342, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Inoue, Y.; Fujii, T.; Aoyama, H.; Inoue, M.; Mitsuhashi, S. Purification and properties of DNA gyrase from a fluoroquinolone-resistant strain of Escherichia coli. Antimicrob. Agents Chemother. 1986, 30, 777–780. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Marians, K.J. Escherichia coli topoisomerase IV. Purification, characterization, subunit structure, and subunit interactions. J. Biol. Chem. 1993, 268, 24481–24490. [Google Scholar] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |




| Compounds | MIC (µg/mL) | |||||
|---|---|---|---|---|---|---|
| R1 | R2 | S.aa | L.ma | E. ca | S.ea | |
| 2a | H | 64 | 32 | 32 | 64 | |
| 2b | F | 64 | >128 | >128 | 64 | |
| 2c | Cl | 64 | 64 | >128 | 64 | |
| 2d | CH3 | 16 | 32 | 16 | 4 | |
| 3a | H | 32 | 32 | 16 | 16 | |
| 3b | F | >128 | 32 | 64 | 64 | |
| 3c | Cl | 64 | >128 | 64 | 64 | |
| 3d | CH3 | 32 | 64 | 16 | 32 | |
| 4a | H | H | 16 | 16 | 8 | 16 |
| 4b | F | H | >128 | >128 | 64 | 64 |
| 4c | Cl | H | 64 | >128 | 64 | 64 |
| 4d | CH3 | H | 4 | 2 | 4 | 0.5 |
| 4e | H | F | 64 | 32 | 32 | 32 |
| 4f | F | F | >128 | >128 | >128 | >128 |
| 4g | Cl | F | 64 | 64 | 64 | 64 |
| 4h | CH3 | F | 8 | 16 | 32 | 32 |
| 4i | F | Cl | >128 | 64 | >128 | 64 |
| 4j | Cl | Cl | 64 | 64 | 64 | 32 |
| 4k | H | CH3 | 4 | 8 | 4 | 4 |
| 4l | F | CH3 | 32 | 64 | 32 | 32 |
| 4m DMSO | Cl | CH3 | 64 >128 | 32 >128 | 64 >128 | 32 >128 |
| CIP b | 0.125 | 1 | 0.5 | 0.25 | ||
| Compd. | IC50 (µg/mL) | |
|---|---|---|
| Gyrase a | Topo IV b | |
| 4d 4k | 13.5 25.7 | 24.2 33.1 |
| CIP c | 0.25 | 6.9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chu, M.-J.; Wang, W.; Ren, Z.-L.; Liu, H.; Cheng, X.; Mo, K.; Wang, L.; Tang, F.; Lv, X.-H. Discovery of Novel Triazole-Containing Pyrazole Ester Derivatives as Potential Antibacterial Agents. Molecules 2019, 24, 1311. https://doi.org/10.3390/molecules24071311
Chu M-J, Wang W, Ren Z-L, Liu H, Cheng X, Mo K, Wang L, Tang F, Lv X-H. Discovery of Novel Triazole-Containing Pyrazole Ester Derivatives as Potential Antibacterial Agents. Molecules. 2019; 24(7):1311. https://doi.org/10.3390/molecules24071311
Chicago/Turabian StyleChu, Ming-Jie, Wei Wang, Zi-Li Ren, Hao Liu, Xiang Cheng, Kai Mo, Li Wang, Feng Tang, and Xian-Hai Lv. 2019. "Discovery of Novel Triazole-Containing Pyrazole Ester Derivatives as Potential Antibacterial Agents" Molecules 24, no. 7: 1311. https://doi.org/10.3390/molecules24071311